Effect on Chest Compression Fraction of Continuous Manual Compressions with Asynchronous Ventilations Using an i-gel® versus 30:2 Approach during Simulated Out-of-Hospital Cardiac Arrest: Protocol for a Manikin Multicenter Randomized Controlled Trial
Abstract
1. Introduction
1.1. Background
1.2. Objectives
2. Materials and Methods
2.1. Study Design
2.2. Setting
2.3. Trial Centers
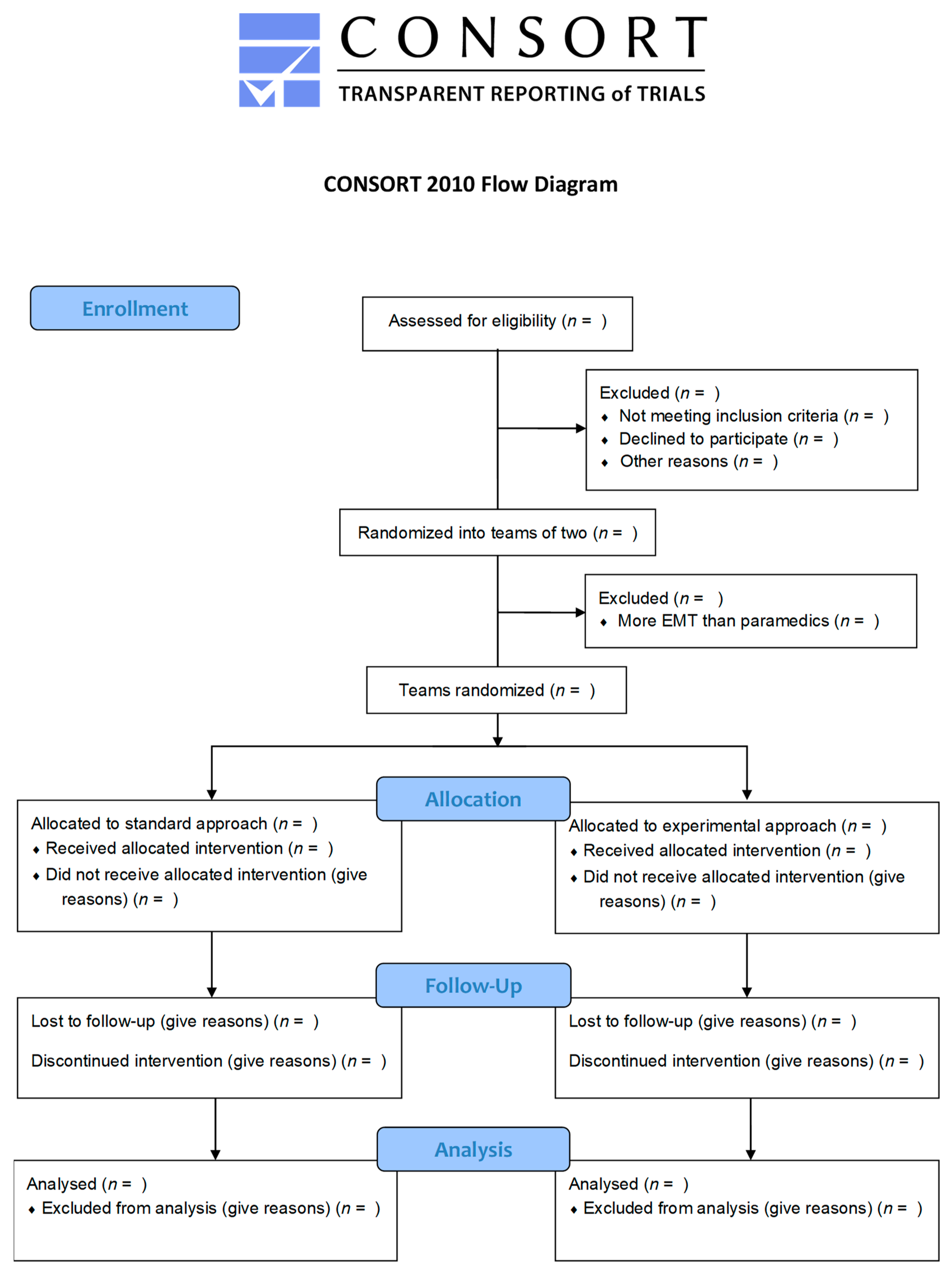
2.4. Participants, Study Flow and Recruitment
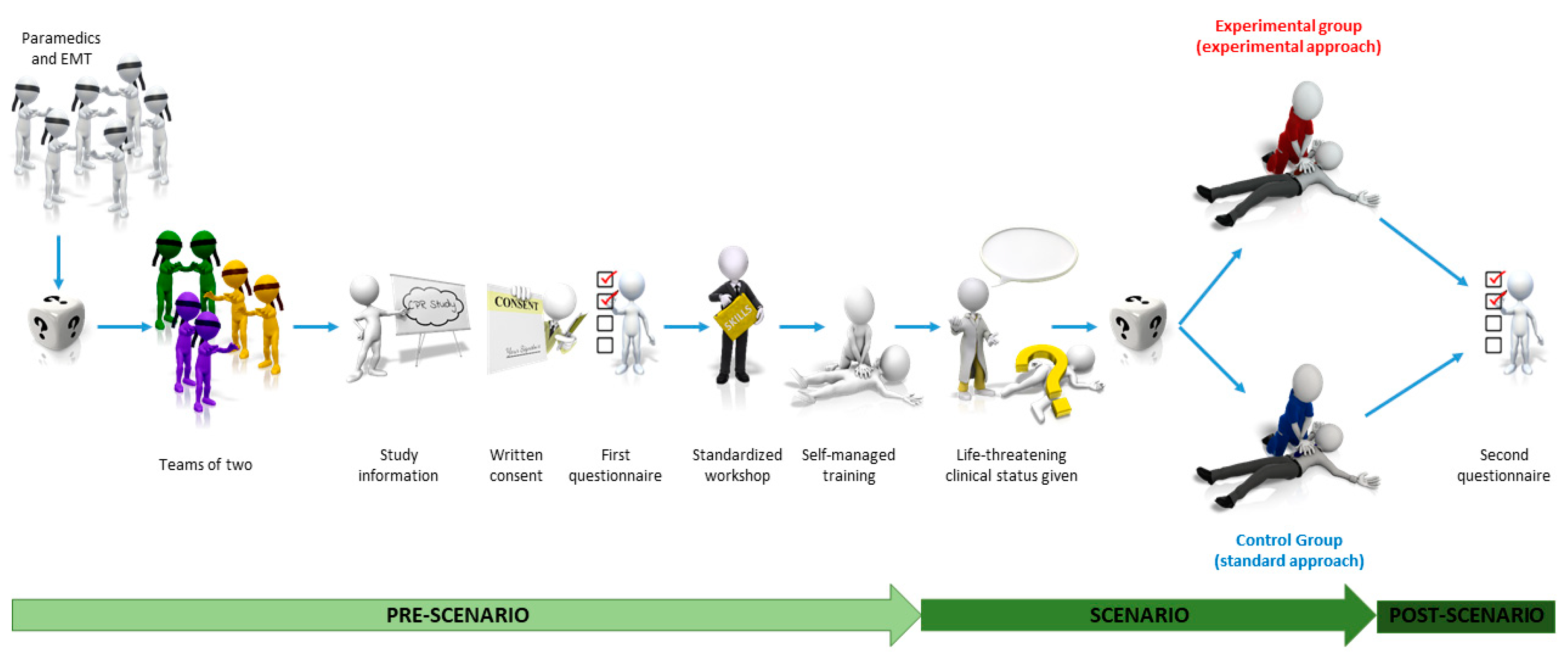
2.5. Study Sequence
2.5.1. Randomization
2.5.2. Pre-Scenario Standardized Workshop
2.5.3. Self-Managed Training Session
2.5.4. Resuscitation Scenario
2.5.5. Study Groups
2.5.6. Data Collection
2.6. Equipment
2.7. Outcomes
2.8. Blinding and Bias Minimization
2.9. Sample Size Calculation, Data Extraction and Statistical Analysis
3. Results, Ethics and Dissemination
3.1. Research Ethics Approval
3.2. Protocol Version
3.3. Trial Registration
3.4. Data Curation and Availability
3.5. Consent or Assent
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Panchal Ashish, R.; Bartos Jason, A.; Cabañas José, G.; Donnino Michael, W.; Drennan Ian, R.; Hirsch Karen, G.; Kudenchuk Peter, J.; Kurz Michael, C.; Lavonas Eric, J.; Morley Peter, T.; et al. Part 3: Adult Basic and Advanced Life Support: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2020, 142, S366–S468. [Google Scholar] [CrossRef]
- Soar, J.; Böttiger, B.W.; Carli, P.; Couper, K.; Deakin, C.D.; Djärv, T.; Lott, C.; Olasveengen, T.; Paal, P.; Pellis, T.; et al. European Resuscitation Council 2020—Draft Guidelines for Public Comment. p. 107. Available online: https://cprguidelines.eu/sites/573c777f5e61585a053d7ba5/content_entry5f8e9d3b4c848637d1e4d1a5/5f9023ed4c848608eee4d21c/files/Draft_ALS_for_Public_Comment.pdf? (accessed on 8 January 2021).
- Kern Karl, B.; Hilwig Ronald, W.; Berg Robert, A.; Sanders Arthur, B.; Ewy Gordon, A. Importance of Continuous Chest Compressions During Cardiopulmonary Resuscitation. Circulation 2002, 105, 645–649. [Google Scholar] [CrossRef]
- Ewy, G.A.; Zuercher, M.; Hilwig, R.W.; Sanders, A.B.; Berg, R.A.; Otto, C.W.; Hayes, M.M.; Kern, K.B. Improved Neurological Outcome with Continuous Chest Compressions Compared with 30:2 Compressions-to-Ventilations Cardiopulmonary Resuscitation in a Realistic Swine Model of out-of-Hospital Cardiac Arrest. Circulation 2007, 116, 2525–2530. [Google Scholar] [CrossRef] [PubMed]
- Rea, T.; Olsufka, M.; Yin, L.; Maynard, C.; Cobb, L. The Relationship between Chest Compression Fraction and Outcome from Ventricular Fibrillation Arrests in Prolonged Resuscitations. Resuscitation 2014, 85, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Vaillancourt, C.; Everson-Stewart, S.; Christenson, J.; Andrusiek, D.; Powell, J.; Nichol, G.; Cheskes, S.; Aufderheide, T.P.; Berg, R.; Stiell, I.G. The Impact of Increased Chest Compression Fraction on Return of Spontaneous Circulation for Out-of-Hospital Cardiac Arrest Patients Not in Ventricular Fibrillation. Resuscitation 2011, 82, 1501–1507. [Google Scholar] [CrossRef] [PubMed]
- Uppiretla, A.K.; Gangalal, G.M.; Rao, S.; Don Bosco, D.; Shareef, S.M.; Sampath, V. Effects of Chest Compression Fraction on Return of Spontaneous Circulation in Patients with Cardiac Arrest; A Brief Report. Adv. J. Emerg. Med. 2019, 4. [Google Scholar] [CrossRef]
- Christenson, J.; Andrusiek, D.; Everson-Stewart, S.; Kudenchuk, P.; Hostler, D.; Powell, J.; Callaway, C.W.; Bishop, D.; Vaillancourt, C.; Davis, D.; et al. Chest Compression Fraction Determines Survival in Patients with Out-of-Hospital Ventricular Fibrillation. Circulation 2009, 120, 1241–1247. [Google Scholar] [CrossRef]
- Vaillancourt, C.; Petersen, A.; Meier, E.N.; Christenson, J.; Menegazzi, J.J.; Aufderheide, T.P.; Nichol, G.; Berg, R.; Callaway, C.W.; Idris, A.H.; et al. The Impact of Increased Chest Compression Fraction on Survival for Out-of-Hospital Cardiac Arrest Patients with a Non-Shockable Initial Rhythm. Resuscitation 2020, 154, 93–100. [Google Scholar] [CrossRef]
- Wik, L.; Olsen, J.-A.; Persse, D.; Sterz, F.; Lozano, M.; Brouwer, M.A.; Westfall, M.; Souders, C.M.; Travis, D.T.; Herken, U.R.; et al. Why Do Some Studies Find That CPR Fraction Is Not a Predictor of Survival? Resuscitation 2016, 104, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Bobrow, B.J.; Clark, L.L.; Ewy, G.A.; Chikani, V.; Sanders, A.B.; Berg, R.A.; Richman, P.B.; Kern, K.B. Minimally Interrupted Cardiac Resuscitation by Emergency Medical Services for Out-of-Hospital Cardiac Arrest. JAMA 2008, 299, 1158–1165. [Google Scholar] [CrossRef]
- Newell, C.; Grier, S.; Soar, J. Airway and Ventilation Management during Cardiopulmonary Resuscitation and after Successful Resuscitation. Crit. Care 2018, 22, 190. [Google Scholar] [CrossRef]
- Kurz, M.C.; Prince, D.K.; Christenson, J.; Carlson, J.; Stub, D.; Cheskes, S.; Lin, S.; Aziz, M.; Austin, M.; Vaillancourt, C.; et al. Association of Advanced Airway Device with Chest Compression Fraction during Out-of-Hospital Cardiopulmonary Arrest. Resuscitation 2016, 98, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Granfeldt, A.; Avis, S.R.; Nicholson, T.C.; Holmberg, M.J.; Moskowitz, A.; Coker, A.; Berg, K.M.; Parr, M.J.; Donnino, M.W.; Soar, J.; et al. Advanced Airway Management during Adult Cardiac Arrest: A Systematic Review. Resuscitation 2019, 139, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Olasveengen, T.; Mancini, M.B.; Berg, R.A.; Brooks, S.; Castren, M.; Chung, S.P.; Considine, J.; Escalante, R.; Gazmuri, R.; Hatanaka, T.; et al. CPR: Chest Compression to Ventilation Ratio-EMS Delivered. Consensus on Science and Treatment Recommendation. Brussels, Belgium: International Liaison Committee on Resuscitation (ILCOR), Basic Life Support Task Force, 2017 July 30. CPR: Chest Compression to Ventilation Ratio—EMS Delivered (BLS): Systematic Review. Available online: https://costr.ilcor.org/document/cpr-chest-compression-to-ventilation-ratio-ems-delivered (accessed on 12 February 2021).
- Saracoglu, A.; Saracoglu, K. Advanced Airway Management in Out-of-Hospital Cardiac Arrest—To Intubate or Not to Intubate: A Narrative Review of the Existing Literature. Anaesthesiol. Intensive Ther. 2020, 52, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Buis, M.L.; Maissan, I.M.; Hoeks, S.E.; Klimek, M.; Stolker, R.J. Defining the Learning Curve for Endotracheal Intubation Using Direct Laryngoscopy: A Systematic Review. Resuscitation 2016, 99, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.; Fehlmann, C.A.; Pasquier, M.; Suppan, L.; Savoldelli, G.L. Endotracheal Intubation Success Rate in an Urban, Supervised, Resident-Staffed Emergency Mobile System: An 11-Year Retrospective Cohort Study. J. Clin. Med. 2020, 9, 238. [Google Scholar] [CrossRef]
- Stone, B.J.; Chantler, P.J.; Baskett, P.J. The Incidence of Regurgitation during Cardiopulmonary Resuscitation: A Comparison between the Bag Valve Mask and Laryngeal Mask Airway. Resuscitation 1998, 38, 3–6. [Google Scholar] [CrossRef]
- Duckett, J.; Fell, P.; Han, K.; Kimber, C.; Taylor, C. Introduction of the I-Gel Supraglottic Airway Device for Prehospital Airway Management in a UK Ambulance Service. Emerg. Med. J. 2014, 31, 505–507. [Google Scholar] [CrossRef]
- Wharton, N.M.; Gibbison, B.; Gabbott, D.A.; Haslam, G.M.; Muchatuta, N.; Cook, T.M. I-Gel Insertion by Novices in Manikins and Patients. Anaesthesia 2008, 63, 991–995. [Google Scholar] [CrossRef]
- Leventis, C.; Chalkias, A.; Sampanis, M.A.; Foulidou, X.; Xanthos, T. Emergency Airway Management by Paramedics: Comparison between Standard Endotracheal Intubation, Laryngeal Mask Airway, and I-Gel. Eur. J. Emerg. Med. 2014, 21, 371–373. [Google Scholar] [CrossRef]
- Castle, N.; Owen, R.; Hann, M.; Naidoo, R.; Reeves, D. Assessment of the Speed and Ease of Insertion of Three Supraglottic Airway Devices by Paramedics: A Manikin Study. Emerg. Med. J. 2010, 27, 860–863. [Google Scholar] [CrossRef]
- Goliasch, G.; Ruetzler, A.; Fischer, H.; Frass, M.; Sessler, D.I.; Ruetzler, K. Evaluation of Advanced Airway Management in Absolutely Inexperienced Hands: A Randomized Manikin Trial. Eur. J. Emerg. Med. 2013, 20, 310–314. [Google Scholar] [CrossRef]
- Ruetzler, K.; Roessler, B.; Potura, L.; Priemayr, A.; Robak, O.; Schuster, E.; Frass, M. Performance and Skill Retention of Intubation by Paramedics Using Seven Different Airway Devices—A Manikin Study. Resuscitation 2011, 82, 593–597. [Google Scholar] [CrossRef]
- Chauhan, G.; Nayar, P.; Seth, A.; Gupta, K.; Panwar, M.; Agrawal, N. Comparison of Clinical Performance of the I-Gel with LMA Proseal. J. Anaesthesiol. Clin. Pharmacol. 2013, 29, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Gabbott, D.A.; Beringer, R. The IGEL Supraglottic Airway: A Potential Role for Resuscitation? Resuscitation 2007, 73, 161–162. [Google Scholar] [CrossRef]
- Middleton, P.M.; Simpson, P.M.; Thomas, R.E.; Bendall, J.C. Higher Insertion Success with the I-Gel® Supraglottic Airway in out-of-Hospital Cardiac Arrest: A Randomised Controlled Trial. Resuscitation 2014, 85, 893–897. [Google Scholar] [CrossRef] [PubMed]
- Häske, D.; Gaier, G.; Heinemann, N.; Schempf, B.; Renz, J.-U. Minimal Training for First Responders with the I-GelTM Leads to Successful Use in Prehospital Cardiopulmonary Resuscitation. Resuscitation 2019, 134, 167–168. [Google Scholar] [CrossRef] [PubMed]
- Häske, D.; Schempf, B.; Gaier, G.; Niederberger, C. Performance of the I-GelTM during Pre-Hospital Cardiopulmonary Resuscitation. Resuscitation 2013, 84, 1229–1232. [Google Scholar] [CrossRef] [PubMed]
- Piegeler, T.; Roessler, B.; Goliasch, G.; Fischer, H.; Schlaepfer, M.; Lang, S.; Ruetzler, K. Evaluation of Six Different Airway Devices Regarding Regurgitation and Pulmonary Aspiration during Cardio-Pulmonary Resuscitation (CPR)—A Human Cadaver Pilot Study. Resuscitation 2016, 102, 70–74. [Google Scholar] [CrossRef]
- Benger, J.R.; Kirby, K.; Black, S.; Brett, S.J.; Clout, M.; Lazaroo, M.J.; Nolan, J.P.; Reeves, B.C.; Robinson, M.; Scott, L.J.; et al. Effect of a Strategy of a Supraglottic Airway Device vs Tracheal Intubation During Out-of-Hospital Cardiac Arrest on Functional Outcome: The AIRWAYS-2 Randomized Clinical Trial. JAMA 2018, 320, 779–791. [Google Scholar] [CrossRef]
- Soar, J.; Berg, K.M.; Andersen, L.W.; Böttiger, B.W.; Cacciola, S.; Callaway, C.W.; Couper, K.; Cronberg, T.; D’Arrigo, S.; Deakin, C.D.; et al. Adult Advanced Life Support: 2020 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations. Resuscitation 2020, 156, A80–A119. [Google Scholar] [CrossRef] [PubMed]
- Nolan Jerry, P.; Maconochie, I.; Soar, J.; Olasveengen, T.M.; Greif, R.; Wyckoff, M.H.; Singletary, E.M.; Aickin, R.; Berg, K.M.; Mancini, M.E.; et al. Executive Summary: 2020 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations. Circulation 2020, 142, S2–S27. [Google Scholar] [CrossRef]
- Chan, A.-W.; Tetzlaff, J.M.; Gøtzsche, P.C.; Altman, D.G.; Mann, H.; Berlin, J.A.; Dickersin, K.; Hróbjartsson, A.; Schulz, K.F.; Parulekar, W.R.; et al. SPIRIT 2013 Explanation and Elaboration: Guidance for Protocols of Clinical Trials. BMJ 2013, 346, e7586. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomised Trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef] [PubMed]
- Suppan, L.; Herren, T.; Taramarcaz, V.; Regard, S.; Martin-Achard, S.; Zamberg, I.; Larribau, R.; Niquille, M.; Mach, F.; Suppan, M.; et al. A Short Intervention Followed by an Interactive E-Learning Module to Motivate Medical Students to Enlist as First Responders: Protocol for a Prospective Implementation Study. JMIR Res. Protoc. 2020, 9, e24664. [Google Scholar] [CrossRef]
- Suppan, L.; Chan, M.; Gartner, B.; Regard, S.; Campana, M.; Chatellard, G.; Cottet, P.; Larribau, R.; Sarasin, F.P.; Niquille, M. Evaluation of a Prehospital Rotation by Senior Residents: A Web-Based Survey. Healthcare 2021, 9, 24. [Google Scholar] [CrossRef]
- Keamk—Create Random and Balanced Teams. Available online: https://www.keamk.com/ (accessed on 5 January 2021).
- Create a Blocked Randomisation List|Sealed Envelope. Available online: https://www.sealedenvelope.com/simple-randomiser/v1/lists (accessed on 6 December 2020).
- Peyton, J.W.R. Teaching & Learning in Medical Practice; Manticore Europe Ltd.: Heronsgate Rickmansworth, Herts, 1998; ISBN 978-1-900887-00-7. [Google Scholar]
- Stuby, L.; Currat, L.; Gartner, B.; Mayoraz, M.; Harbarth, S.; Suppan, L.; Suppan, M. Impact of Face-to-Face Teaching in Addition to Electronic Learning on Personal Protective Equipment Doffing Proficiency in Student Paramedics: Protocol for a Randomized Controlled Trial. JMIR Preprints. Available online: https://preprints.jmir.org/preprint/26927 (accessed on 6 December 2020).
- Giacomino, K.; Caliesch, R.; Sattelmayer, K.M. The Effectiveness of the Peyton’s 4-Step Teaching Approach on Skill Acquisition of Procedures in Health Professions Education: A Systematic Review and Meta-Analysis with Integrated Meta-Regression. PeerJ 2020, 8. [Google Scholar] [CrossRef]
- Algorithms. Available online: https://cpr.heart.org/en/resuscitation-science/cpr-and-ecc-guidelines/algorithms (accessed on 13 January 2021).
- Paas, F.G.W.C.; Van Merriënboer, J.J.G. The Efficiency of Instructional Conditions: An Approach to Combine Mental Effort and Performance Measures. Hum. Factors 1993, 35, 737–743. [Google Scholar] [CrossRef]
- Pawar, S.; Jacques, T.; Deshpande, K.; Pusapati, R.; Meguerdichian, M.J. Evaluation of Cognitive Load and Emotional States during Multidisciplinary Critical Care Simulation Sessions. BMJ STEL 2018, 4, 87–91. [Google Scholar] [CrossRef]
- Dörges, V.; Ocker, H.; Hagelberg, S.; Wenzel, V.; Schmucker, P. Optimisation of Tidal Volumes given with Self-Inflatable Bags without Additional Oxygen. Resuscitation 2000, 43, 195–199. [Google Scholar] [CrossRef]
- Dörges, V.; Ocker, H.; Hagelberg, S.; Wenzel, V.; Idris, A.H.; Schmucker, P. Smaller Tidal Volumes with Room-Air Are Not Sufficient to Ensure Adequate Oxygenation during Bag-Valve-Mask Ventilation. Resuscitation 2000, 44, 37–41. [Google Scholar] [CrossRef]
- Baskett, P.; Nolan, J.; Parr, M. Tidal Volumes Which Are Perceived to Be Adequate for Resuscitation. Resuscitation 1996, 31, 231–234. [Google Scholar] [CrossRef]
- Aramendi, E.; Lu, Y.; Chang, M.P.; Elola, A.; Irusta, U.; Owens, P.; Idris, A.H. A Novel Technique to Assess the Quality of Ventilation during Pre-Hospital Cardiopulmonary Resuscitation. Resuscitation 2018, 132, 41–46. [Google Scholar] [CrossRef]
- Serpa Neto, A.; Cardoso, S.O.; Manetta, J.A.; Pereira, V.G.M.; Espósito, D.C.; de Pasqualucci, M.O.P.; Damasceno, M.C.T.; Schultz, M.J. Association between Use of Lung-Protective Ventilation with Lower Tidal Volumes and Clinical Outcomes Among Patients without Acute Respiratory Distress Syndrome: A Meta-Analysis. JAMA 2012, 308, 1651. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, T.; Lauritsen, J. EpiData Software. Available online: https://www.epidata.dk/ (accessed on 18 December 2020).
- Swiss Confederation CC 810.30 Federal Act of 30 September 2011 on Research Involving Human Beings (Human Research Act, HRA). Available online: https://www.admin.ch/opc/en/classified-compilation/20061313/index.html (accessed on 24 November 2020).
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- International Conference on Harmonisation. Good Clinical Practice. Available online: https://www.ich.org/page/efficacy-guidelines (accessed on 24 November 2020).
- Mendeley Data. Available online: https://data.mendeley.com/ (accessed on 21 December 2020).
- Siebert, J.N.; Ehrler, F.; Combescure, C.; Lacroix, L.; Haddad, K.; Sanchez, O.; Gervaix, A.; Lovis, C.; Manzano, S. A Mobile Device App to Reduce Time to Drug Delivery and Medication Errors During Simulated Pediatric Cardiopulmonary Resuscitation: A Randomized Controlled Trial. J. Med. Internet Res. 2017, 19, e31. [Google Scholar] [CrossRef]
- Fernández-Ayuso, D.; Fernández-Ayuso, R.; Del-Campo-Cazallas, C.; Pérez-Olmo, J.L.; Matías-Pompa, B.; Fernández-Carnero, J.; Calvo-Lobo, C. The Modification of Vital Signs According to Nursing Students’ Experiences Undergoing Cardiopulmonary Resuscitation Training via High-Fidelity Simulation: Quasi-Experimental Study. JMIR Serious Games 2018, 6, e11061. [Google Scholar] [CrossRef]
- Sahu, S.; Lata, I. Simulation in Resuscitation Teaching and Training, an Evidence Based Practice Review. J. Emerg. Trauma Shock 2010, 3, 378–384. [Google Scholar] [CrossRef]
- Bjørshol, C.; Myklebust, H.; Nilsen, K.; Hoff, T.; Bjørkli, C.; EIllguth, E.; Søreide, E.; Sunde, K. Effect of Socioemotional Stress on the Quality of Cardiopulmonary Resuscitation during Advanced Life Support in a Randomized Manikin Study. Crit. Care Med. 2011, 39, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Judd, B.K.; Alison, J.A.; Waters, D.; Gordon, C.J. Comparison of Psychophysiological Stress in Physiotherapy Students Undertaking Simulation and Hospital-Based Clinical Education. Simul. Healthc. 2016, 11, 271–277. [Google Scholar] [CrossRef]
- Judd, B.K.; Currie, J.; Dodds, K.L.; Fethney, J.; Gordon, C.J. Registered Nurses Psychophysiological Stress and Confidence during High-Fidelity Emergency Simulation: Effects on Performance. Nurse Educ. Today 2019, 78, 44–49. [Google Scholar] [CrossRef] [PubMed]
| STUDY PERIOD | ||||||
|---|---|---|---|---|---|---|
| Enrolment | Workshop | Training Session | Allocation | Post-Allocation | Close-Out | |
| TIMEPOINT | t-3 before study day | t-2 -40 min | t-1 -20 min | t0 | t1 | t2 |
| ENROLMENT: | ||||||
| Eligibility screen | X | X | ||||
| Randomization into pairs | X | |||||
| Informed consent | X | |||||
| Standardized workshop | X | |||||
| Training session | X | |||||
| Allocation | X | |||||
| INTERVENTIONS: | ||||||
| Standard approach | X | |||||
| Experimental approach | X | |||||
| ASSESSMENTS: | ||||||
| Age, gender, years of experience, profession title, estimated insertions on manikin, estimated insertions on real patients | X | |||||
| CCF, compressions depth and rate, chest recoil, time to first shock and to first ventilation, number and volume of ventilations. | X | |||||
| User satisfaction, cognitive load | X | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stuby, L.; Jampen, L.; Sierro, J.; Paus, E.; Spichiger, T.; Suppan, L.; Thurre, D. Effect on Chest Compression Fraction of Continuous Manual Compressions with Asynchronous Ventilations Using an i-gel® versus 30:2 Approach during Simulated Out-of-Hospital Cardiac Arrest: Protocol for a Manikin Multicenter Randomized Controlled Trial. Healthcare 2021, 9, 354. https://doi.org/10.3390/healthcare9030354
Stuby L, Jampen L, Sierro J, Paus E, Spichiger T, Suppan L, Thurre D. Effect on Chest Compression Fraction of Continuous Manual Compressions with Asynchronous Ventilations Using an i-gel® versus 30:2 Approach during Simulated Out-of-Hospital Cardiac Arrest: Protocol for a Manikin Multicenter Randomized Controlled Trial. Healthcare. 2021; 9(3):354. https://doi.org/10.3390/healthcare9030354
Chicago/Turabian StyleStuby, Loric, Laurent Jampen, Julien Sierro, Erik Paus, Thierry Spichiger, Laurent Suppan, and David Thurre. 2021. "Effect on Chest Compression Fraction of Continuous Manual Compressions with Asynchronous Ventilations Using an i-gel® versus 30:2 Approach during Simulated Out-of-Hospital Cardiac Arrest: Protocol for a Manikin Multicenter Randomized Controlled Trial" Healthcare 9, no. 3: 354. https://doi.org/10.3390/healthcare9030354
APA StyleStuby, L., Jampen, L., Sierro, J., Paus, E., Spichiger, T., Suppan, L., & Thurre, D. (2021). Effect on Chest Compression Fraction of Continuous Manual Compressions with Asynchronous Ventilations Using an i-gel® versus 30:2 Approach during Simulated Out-of-Hospital Cardiac Arrest: Protocol for a Manikin Multicenter Randomized Controlled Trial. Healthcare, 9(3), 354. https://doi.org/10.3390/healthcare9030354








