Association of Maternal Observation and Motivation (MOM) Program with m-Health Support on Maternal and Newborn Health
Abstract
1. Background
1.1. Significance of the Study
1.2. Objectives
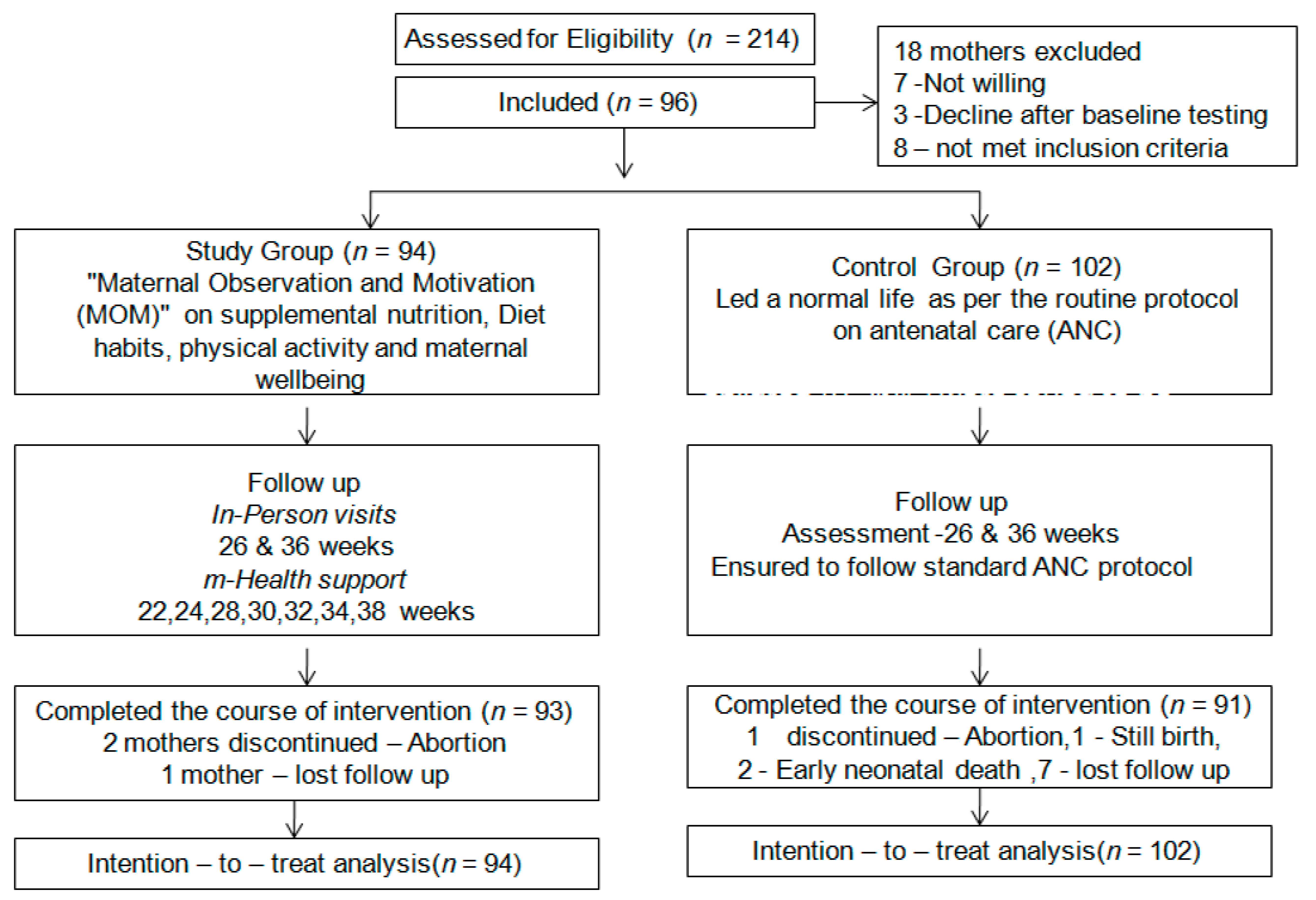
2. Methods
2.1. Design
2.2. Participants and Setting
2.3. Sample Size and Sampling Process
2.4. Data Collection Tools/Instruments
2.5. Assessment
2.6. Intervention
2.7. Ethical Consideration
2.8. Statistical Analysis
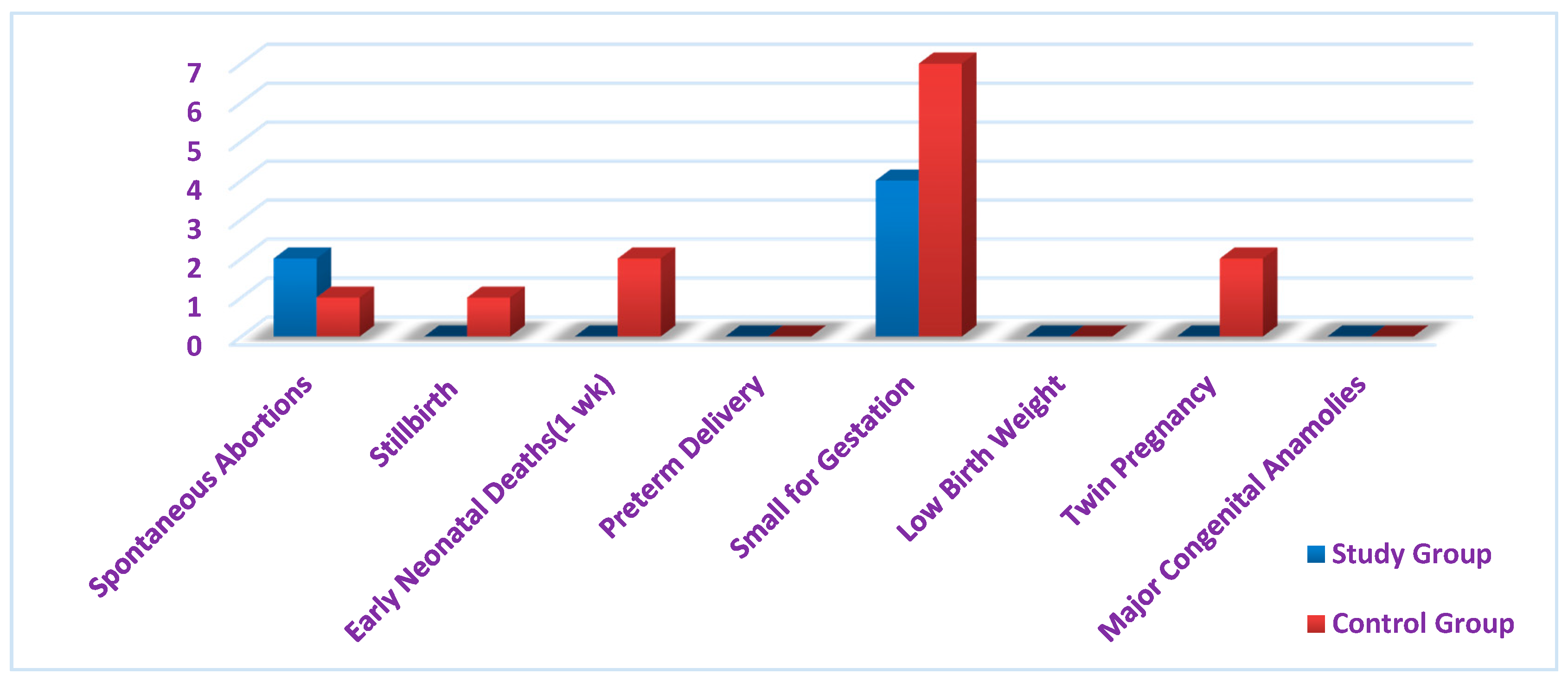
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Recommendations on Antenatal Care for a Positive Pregnancy Experience. Available online: https://www.who.int/publications/i/item/9789241549912 (accessed on 27 October 2021).
- Morrison, J.L.; Regnault, T.R.H. Nutrition in Pregnancy: Optimising Maternal Diet and Fetal Adaptations to Altered Nutrient Supply. Nutrients 2016, 8, 342. [Google Scholar] [CrossRef]
- Preconception Care: Maximizing the Gains for Maternal and Child Health. Available online: https://www.who.int/maternal_child_adolescent/documents/preconception_care_policy_brief.pdf (accessed on 27 October 2021).
- Tam, E.; Keats, E.C.; Rind, F.; Das, J.K.; Bhutta, A.Z.A. Micronutrient Supplementation and Fortification Interventions on Health and Development Outcomes among Children Under-Five in Low- and Middle-Income Countries: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 289. [Google Scholar] [CrossRef]
- Misner, B. Food alone may not provide sufficient micronutrients for preventing deficiency. J. Int. Soc. Sports Nutr. 2006, 3, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, J.; Heslehurst, N.; Hall, J.; Schoenaker, D.A.J.M.; Hutchinson, J.; Cade, J.E.; Poston, L.; Barrett, G.; Crozier, S.R.; Barker, M.; et al. Before the beginning: Nutrition and lifestyle in the preconception period and its importance for future health. Lancet 2018, 391, 1830–1841. [Google Scholar] [CrossRef]
- Tunçalp, Ö.; Were, W.M.; MacLennan, C.; Oladapo, O.T.; Gülmezoglu, A.M.; Bahl, R.; Daelmans, B.; Mathai, M.; Say, L.; Kristensen, F.; et al. Quality of care for pregnant women and newborns—The WHO vision. BJOG Int. J. Obstet. Gynaecol. 2015, 122, 1045–1049. [Google Scholar] [CrossRef]
- Campbell, O.M.R.; Graham, W.J.; The Lancet Maternal Survival Series Steering Group. Strategies for reducing maternal mortality: Getting on with what works. Lancet 2006, 368, 1284–1299. [Google Scholar] [CrossRef]
- Fisk, N.M.; McKee, M.; Atun, R. Relative and absolute addressability of global disease burden in maternal and perinatal health by investment in R&D. Trop. Med. Int. Health 2011, 16, 662–668. [Google Scholar] [PubMed]
- Alkema, L.; Chou, D.; Hogan, D.; Zhang, S.; Moller, A.-B.; Gemmill, A.; Fat, D.M.; Boerma, T.; Temmerman, M.; Mathers, C.; et al. Global, regional, and national levels and trends in maternal mortality between 1990 and 2015, with scenario-based projections to 2030: A systematic analysis by the UN Maternal Mortality Estimation Inter-Agency Group. Lancet 2016, 387, 462–474. [Google Scholar] [CrossRef]
- Maternal Mortality; Fact Sheet No. 348; World Health Organization: Geneva, Switzerland, 2014; Available online: http://www.who.int/mediacentre/factsheets/fs348/en/index.html (accessed on 22 October 2021).
- Blencowe, H.; Cousens, S.; Jassir, F.B.; Say, L.; Chou, D.; Mathers, C.; Hogan, D.; Shiekh, S.; Qureshi, Z.U.; You, D.; et al. National, regional, and worldwide estimates of stillbirth rates in 2015, with trends from 2000: A systematic analysis. Lancet 2016, 4, e98–e108. [Google Scholar] [CrossRef]
- Imdad, A.; Bhutta, Z.A. Maternal nutrition and birth outcomes: Effect of balanced protein-energy supplementation. Paediatr. Perinat. Epidemiol. 2012, 26 (Suppl. 1), 178–190. [Google Scholar] [CrossRef]
- Office of the United Nations High Commissioner for Human Rights (OHCHR). Technical Guidance on the Application of a Human Rights-Based Approach to the Implementation of Policies and Programmes to Reduce Preventable Maternal Morbidity and Mortality. In Human Rights Council, Twentieth Session; United Nations General Assembly: New York, NY, USA, 2012; Available online: http://www2.ohchr.org/english/issues/women/docs/A.HRC.21.22_en.pdf (accessed on 11 February 2021).
- Tang, A.M.; Chung, M.; Dong, K.; Terrin, N.; Edmonds, A.; Assefa, N.; Chetty, T.; Ramlal, R.; Christian, P.; West, K.; et al. Determining a Global Mid Upper Arm Circumference Cut off to Assess Malnutrition in Pregnant Women; FHI 360/Food and Nutrition Technical Assistance III Project (FANTA); Washington, DC, USA, 2016; Available online: http://www.fantaproject.org/sites/default/files/resources/FANTA-MUAC-cutoffs-pregnantwomen-June2016.pdf (accessed on 4 April 2021).
- Yu, Z.; Han, S.; Zhu, J.; Sun, X.; Ji, C.; Guo, X. Pre-pregnancy body mass index in relation to infant birth weight and offspring overweight/obesity: A systematic review and meta-analysis. PLoS ONE 2013, 8, e61627. [Google Scholar] [CrossRef]
- Han, Z.; Lutsiv, O.; Mulla, S.; Rosen, A.; Beyene, J.; McDonald, S.D.; Knowledge Synthesis Group. Low gestational weight gain and the risk of preterm birth and low birth weight: A systematic review and meta-analyses. Acta Obstet. Gynecol. Scand. 2011, 90, 935–954. [Google Scholar] [CrossRef]
- WHO; de Benoist, B.; McLean, E.; Egli, I.; Cogswell, M. (Eds.) Worldwide Prevalence of Anaemia 1993–2005. In WHO Global Database on Anaemia; World Health Organization (WHO): Geneva, Switzerland, 2008; Available online: http://apps.who.int/iris/bitstream/10665/43894/1/9789241596657_eng.pdf (accessed on 3 March 2021).
- WHO/UNICEF Discussion Paper: The Extension of the 2025 Maternal, Infant and Young Child Nutrition Targets to 2030. Available online: https://www.who.int/nutrition/global-target-2025/discussion-paper-extension-targets-2030.pdf (accessed on 18 February 2021).
- Soofi, S.; Khan, G.N.; Sadiq, K.; Ariff, S.; Habib, A.; Kureishy, S.; Hussain, I.; Umer, M.; Suhag, Z.; Rizvi, A.; et al. Prevalence and possible factors associated with anaemia, and vitamin B12 and folate deficiencies in women of reproductive age in Pakistan: Analysis of national-level secondary survey data. BMJ Open 2017, 7, e018007. [Google Scholar] [CrossRef] [PubMed]
- Guideline: Daily Iron and Folic Acid Supplementation in Pregnant Women; World Health Organization: Geneva, Switzerland, 2012. Available online: http://www.who.int/nutrition/publications/micronutrients/guidelines/daily_ifa_supp_pregnant_women/en/ (accessed on 18 March 2021).
- Levels and Trends in Child Malnutrition. UNICEF/WHO/World Bank Group Joint Child Malnutrition Estimates. Key Findings of the 2018 Edition. 2018. Available online: http://www.who.int/nutgrowthdb/2018-jme-brochure.pdf?ua=1 (accessed on 24 March 2021).
- Stevens, G.A.; Finucane, M.M.; De-Regil, L.M.; Paciorek, C.J.; Flaxman, S.R.; Branca, F.; Peña-Rosas, J.R.; Bhutta, Z.A.; Ezzati, M.; on behalf of Nutrition Impact Model Study Group (Anaemia). Global, Regional, and National Trends in Haemoglobin Concentration and Prevalence of Total and Severe Anaemia in Children and Pregnant and Non-Pregnant Women for 1995–2011: A Systematic Analysis of Population-Representative Data. Lancet Glob. Health 2013, 1. [Google Scholar] [CrossRef]
- Abu-Saad, K.; Fraser, D. Maternal Nutrition and Birth Outcomes. Epidemiol. Rev. 2010, 32, 5–25. Available online: https://academic.oup.com/epirev/article-lookup/doi/10.1093/epirev/mxq001 (accessed on 4 April 2021). [CrossRef] [PubMed]
- Akhtar, S.; Ahmed, A.; Randhawa, M.A.; Atukorala, S.; Arlappa, N.; Ismail, T.; Ali, Z. Prevalence of vitamin A deficiency in South Asia: Causes, outcomes, and possible remedies. J. Health Popul. Nutr. 2013, 31, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Christian, P.; Khatry, S.K.; Le Clerq, S.C.; Dali, S.M. Effects of prenatal micronutrient supplementation on complications of labor and delivery and puerperal morbidity in rural Nepal. Int. J. Gynaecol. Obstet. 2009, 106, 3–7. [Google Scholar] [CrossRef]
- Chappell, L.C.; Cluver, C.A.; Kingdom, J.; Tong, S. Pre-eclampsia. Lancet 2021, 398, 341–354. [Google Scholar] [CrossRef]
- Gernand, A.D.; Schulze, K.J.; Stewart, C.P.; West, K.P., Jr.; Christian, P. Micronutrient deficiencies in pregnancy worldwide: Health effects and prevention. Nat. Rev. Endocrinol. 2016, 12, 274–289. [Google Scholar] [CrossRef]
- Wessels, I.; Maywald, M.; Rink, L. Zinc as a Gatekeeper of Immune Function. Nutrients 2017, 9, 1286. [Google Scholar] [CrossRef]
- FAO; IFAD; UNICEF; WFP; WHO. The State of Food Security and Nutrition in the World 2017. Building Resilience for Peace and Food Security. 2017. Available online: http://www.fao.org/3/a-I7695e.pdf (accessed on 11 April 2021).
- Rich Mother, Poor Mother: The Social Determinants of Maternal Death and Disability. Fact Sheet. UNFPA, December 2012. Available online: https://www.unfpa.org/sites/default/files/resource-pdf/EN-SRH%20fact%20sheet-Poormother.pdf (accessed on 11 April 2021).
- Golbasi, Z.; Kelleci, M.; Kisacik, G.; Cetin, A. Prevalence and correlates of depression in pregnancy among Turkish women. Matern. Child Health J. 2010, 14, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A.; Berkley, J.A.; Bandsma, R.H.J.; Kerac, M.; Trehan, I.; Briend, A. Severe childhood malnutrition. Nat. Rev. Dis. Primers 2017, 3, 17067. [Google Scholar] [CrossRef]
- Mahmood, A.; Kedia, S.; Wyant, D.K.; Ahn, S.; Bhuyan, S.S. Use of mobile health applications for health-promoting behaviour among individuals with chronic medical conditions. Digit. Health 2019, 5, 2055207619882181. [Google Scholar] [CrossRef]
- Topp, C.W.; Østergaard, S.D.; Søndergaard, S.; Bech, P. WHO-5 Well-Being Index: A systematic review of the literature. Psychother. Psychosom. 2015, 84, 167–176. [Google Scholar] [CrossRef]
- National Institute of Health and Family Welfare, Government of India. National Nutritional Anemia Prophylaxis Programme. Available online: http://nihfw.nic.in/ndc-nihfw/html/Programmes/NationalNutritionAnemia.htm (accessed on 29 July 2013).
- Trends in Maternal Mortality: 1990 to 2015: WHO, UNICEF, UNFPA, The World Bank and the United Nations Population Division. World Health Organization. 2015. Available online: http://apps.who.int/iris/bitstream/10665/194254/1/9789241565141_eng.pdf?ua=1 (accessed on 1 April 2021).
- Rao, S.; Yajnik, C.S.; Kanade, A.; Fall, C.H.D.; Margetts, B.M.; Jackson, A.A.; Desai, B. Intake of Micronutrient-Rich Foods in Rural Indian Mothers Is Associated with the Size of Their Babies at Birth: Pune Maternal Nutrition Study. J. Nutr. 2001, 131, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Murray-Kolb, L.E.; Chen, L.; Chen, P.; Shapiro, M.; Caulfield, L. CHERG Iron Report: Maternal Mortality, Child Mortality, Perinatal Mortality, Child Cognition, and Estimates of Prevalence of Anemia Due to Iron Deficiency. Available online: cherg.org/publications/iron-report.pdf (accessed on 5 February 2014).
- Chowdhury, H.A.; Ahmed, K.R.; Jebunessa, F.; Akter, J.; Hossain, S. Factors associated with maternal anaemia among pregnant women in Dhaka city. BMC Womens Health 2015, 15, 77. [Google Scholar] [CrossRef] [PubMed]
- Folic Acid: Birth Defects Count. Available online: https://www.cdc.gov/ncbddd/birthdefectscount/basics.html (accessed on 27 October 2021).
- Persson, L.Å.; Arifeen, S.; Ekström, E.-C.; Rasmussen, K.M.; Frongillo, E.A.; Yunus, M.; for the MINIMat Study Team. Effects of Prenatal icronutrient and Early Food Supplementation on Maternal Hemoglobin, Birth Weight, and Infant Mortality among—Children in Bangladesh. JAMA 2012, 7, 2050–2059. [Google Scholar] [CrossRef]
- Potdar, R.D.; Sahariah, S.A.; Gandhi, M.; Kehoe, S.H.; Brown, N.; Sane, H.; Fall, C.H. Improving women’s diet quality preconceptionally and during gestation: Effects on birth weight and prevalence of low birth weight—A randomised controlled efficacy trial in India (Mumbai Maternal Nutrition Project). Am. J. Clin. Nutr. 2014, 100, 1257–1268. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists. Physical activity and exercise during pregnancy and the postpartum period. Committee opinion no. 650. Obstet. Gynecol 2015, 126, e135–e142. [Google Scholar] [CrossRef]
- Gülmezoglu, A.M.; Lawrie, T.A.; Hezelgrave, N.; Oladapo, O.T.; Souza, J.P.; Gielen, M.; Lawn, J.E.; Bahl, R.; Althabe, F.; Colaci, D.; et al. Interventions to Reduce Maternal and Newborn Morbidity and Mortality. In Reproductive, Maternal, Newborn, and Child Health: Disease Control Priorities, 3rd ed.; Black, R.E., Laxminarayan, R., Temmerman, M., Walker, N., Eds.; The International Bank for Reconstruction and Development/The World Bank: Washington, DC, USA, 2016; Volume 2, Chapter 7. Available online: https://www.ncbi.nlm.nih.gov/books/NBK361904/doi:10.1596/978-1-4648-0348-2_ch7 (accessed on 7 September 2021).
| Variables | Study (n = 94) (%) | Control (n = 102) (%) |
|---|---|---|
| Age (in years) Mean ± SD | 28.14.6 | 27.9 ± 4.8 |
| Parity | ||
| Primipara mothers | 46 (48.9) | 55 (53.9) |
| Multipara mothers | 48 (51.1) | 47 (46.1) |
| Height (cm) | 144.8 ± 6.1 | 147 ± 6.9 |
| Weight (kg) | 45.8 ± 4.3 | 46.1 ± 3.9 |
| BMI (kg/m2) | 21.7 ± 3.1 | 21.3 ± 2.9 |
| Educational Attainment | ||
| Primary or less | 21 (19.7) | 19 (18.6) |
| Up to secondary | 62 (66.0) | 57 (55.9) |
| Completed college education | 11 (11.7) | 26 (25.5) |
| Occupation | ||
| Semiskilled/unskilled | 16 (17.0) | 19 (18.6) |
| Skilled/self-employed | 3 (3.2) | 5 (4.9) |
| Professional | 2 (2.1) | 7 (6.7) |
| Not working | 73 (77.7) | 71 (69.1) |
| Husband’s Education | ||
| Primary or less | 17 (18.7) | 16 (15.7) |
| Secondary | 69 (73.4) | 78 (76.5) |
| Graduate | 8 (8.5) | 8 (7.8) |
| Husband’s Occupation | ||
| Semi-skilled/unskilled | 58 (61.7) | 62 (60.8) |
| Skilled/self-employed | 24 (25.5) | 27 (26.5) |
| Professional | 4 (4.3) | 6 (5.9) |
| Not working/other | 8 (8.5) | 7 (6.8) |
| Dietary Intake | ||
| Milk and Milk Products (other than in coffee/tea) | ||
| 1 time/week | 43 (45.7) | 50 (49.0) |
| 1–6 times/week | 31 (33) | 37 (36.3) |
| ≥7 times/week | 20 (21.3) | 15 (14.7) |
| Green Leafy Vegetables (GLV) | ||
| 1 time/week | 23 (24.5) | 27 (26.5) |
| 1–6 times/week | 61 (64.9) | 58 (56.9) |
| ≥7 times/week | 10 (10.6) | 17 (16.7) |
| Fruits | ||
| 1 time/week | 14 (14.9) | 19 (18.3) |
| 1–6 times/week | 59 (55.5) | 62 (60.8) |
| ≥7 times/week | 21 (22.3) | 21 (20.6) |
| Maternal Hb Level | Study Group Mean (SD) | Control Group Mean (SD) | t Value p-Value |
|---|---|---|---|
| Hb level at baseline, g/L | 110.4 (3.08) | 110.1 (2.91) | t = 0.848 p = 0.397 (N.S.) |
| Hb level at 28 week gestation, g/L | 112.7 (4.13) | 112.1 (3.99) | t = 0.199 p = 0.843 (N.S.) |
| Hb level at 36 week 113.1 gestation, g/L (3.16) | 112.5 (3.05) | t = 0.985 p = 0.21 (N.S.) | |
| Repeated ANOVA F = 19.305, p < 0.001 | F = 0.633, p = 0.532 | ||
| Maternal Outcome Variables | Study Group (n = 94) | Control Group (n = 102) | p |
|---|---|---|---|
| Anemia (Hb < 110 g/L) | |||
| At 28 weeks | 26 (27.7%) | 31 (30.4%) | 0.001 |
| At 36 weeks | 14 (15.0%) | 19 (18.6%) | |
| Weight Gain | |||
| At 28 weeks | 43.9 ± 6.2 | 41.8 ± 8.1 | |
| At 36 weeks | 47.6 ± 7.4 | 46.2 ± 6.7 | 0.001 |
| Physical Activity (min/week) | |||
| Pre-intervention | 81 ± 11 | 86 ± 16 | |
| Post-intervention | 131 ± 29 | 103 ± 17 | 0.001 |
| Maternal Well-being | |||
| Pre-intervention | 54 ± 13 | 59 ± 18 | 0.01 |
| Post-intervention | 81 ± 19 | 67 ± 11 | |
| Newborn Outcome Variables | |||
| Birth weight (kg) | 2.78 ± 0.56 | 2.56 ± 0.49 | 0.01 |
| Crown–heel length (cm) | 47.6 ± 3.0 | 46.8 ± 3.5 | 0.001 |
| Gestational age at birth (wk) | 38.9 (38.6–39.1) | 38.6 (38.4–39.0) | 0.01 |
| Dependent Variable | Independent Variables | Milk Product Intake | Green Leafy Vegetables Intake | Fruit Intake | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| R2 | β | P | R2 | β | p | R2 | β | p | ||
| Birth weight of newborn (g) | Sex, parity, gestational age at birth | 19.8 (0.87) | 16.9 | <0.001 | 23.1 (1.63) | 6.7 | <0.05 | 22.1 (0.98) | 5.8 | <0.01 |
| Sex, parity, gestational age at birth and baseline weight of mother | 29.3 (1.8) | 17.5 | <0.001 | 23.4 (0.47) | 5.1 | <0.05 | 31.6 (0.83) | 9.1 | <0.05 | |
| Dependent Variable | Independent Variable | Physical Activity | Maternal Well-being |
|---|---|---|---|
| Birth Weight | P1 (Sex, parity, and baseline weight of mother) | <0.05 | <0.05 |
| P1 and intake of milk products, GLV, and fruits | <0.001 | <0.05 | |
| Crown–Heel Length | P1 (Sex, parity, and baseline weight of mother) | <0.01 | <0.05 |
| P1 and intake of milk products, GLV, and fruits | <0.05 | <0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paulsamy, P.; Easwaran, V.; Ashraf, R.; Alshahrani, S.H.; Venkatesan, K.; Qureshi, A.A.; Arrab, M.M.; Prabahar, K.; Periannan, K.; Vasudevan, R.; et al. Association of Maternal Observation and Motivation (MOM) Program with m-Health Support on Maternal and Newborn Health. Healthcare 2021, 9, 1629. https://doi.org/10.3390/healthcare9121629
Paulsamy P, Easwaran V, Ashraf R, Alshahrani SH, Venkatesan K, Qureshi AA, Arrab MM, Prabahar K, Periannan K, Vasudevan R, et al. Association of Maternal Observation and Motivation (MOM) Program with m-Health Support on Maternal and Newborn Health. Healthcare. 2021; 9(12):1629. https://doi.org/10.3390/healthcare9121629
Chicago/Turabian StylePaulsamy, Premalatha, Vigneshwaran Easwaran, Rizwan Ashraf, Shadia Hamoud Alshahrani, Krishnaraju Venkatesan, Absar Ahmed Qureshi, Mervat Moustafa Arrab, Kousalya Prabahar, Kalaiselvi Periannan, Rajalakshimi Vasudevan, and et al. 2021. "Association of Maternal Observation and Motivation (MOM) Program with m-Health Support on Maternal and Newborn Health" Healthcare 9, no. 12: 1629. https://doi.org/10.3390/healthcare9121629
APA StylePaulsamy, P., Easwaran, V., Ashraf, R., Alshahrani, S. H., Venkatesan, K., Qureshi, A. A., Arrab, M. M., Prabahar, K., Periannan, K., Vasudevan, R., Kandasamy, G., Chidambaram, K., Pappiya, E. M., Venkatesan, K., & Manoharan, V. (2021). Association of Maternal Observation and Motivation (MOM) Program with m-Health Support on Maternal and Newborn Health. Healthcare, 9(12), 1629. https://doi.org/10.3390/healthcare9121629








