Two-Day Cardiopulmonary Exercise Testing in Females with a Severe Grade of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Comparison with Patients with Mild and Moderate Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Cardiopulmonary Exercise Testing (CPET)
2.2. Disability Metric
2.3. Statistical Analysis
3. Results
3.1. Baseline Characteristics
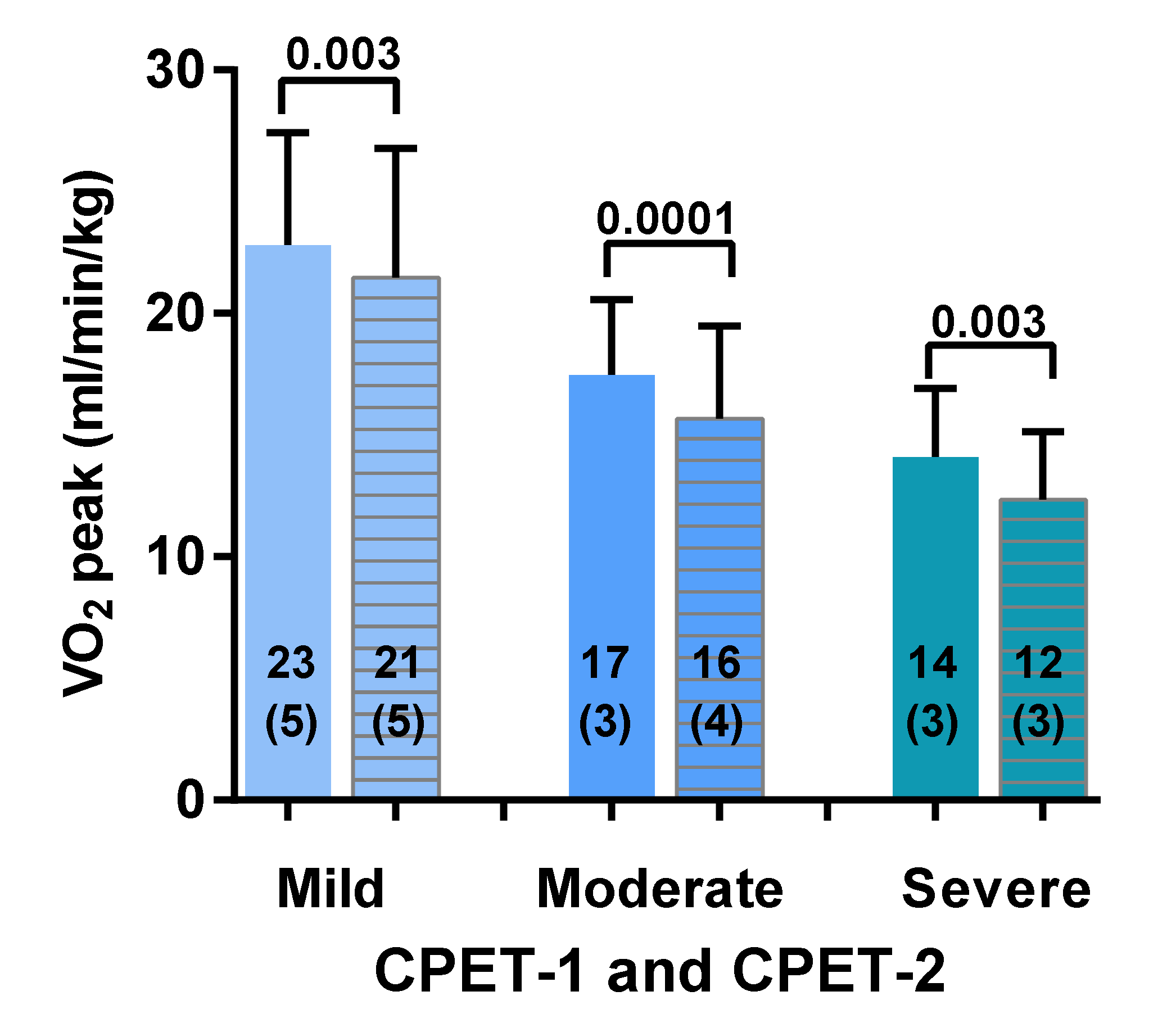
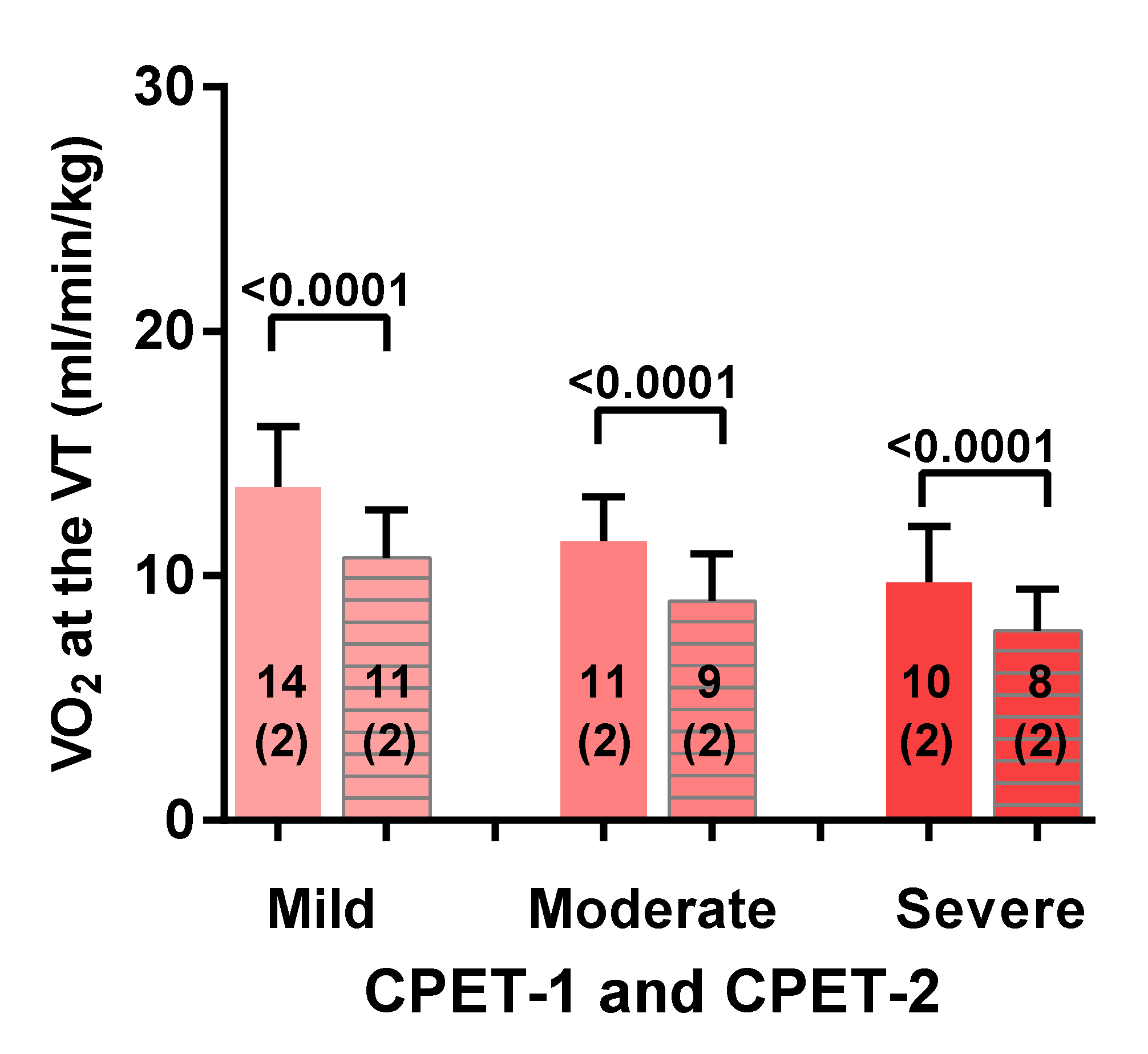
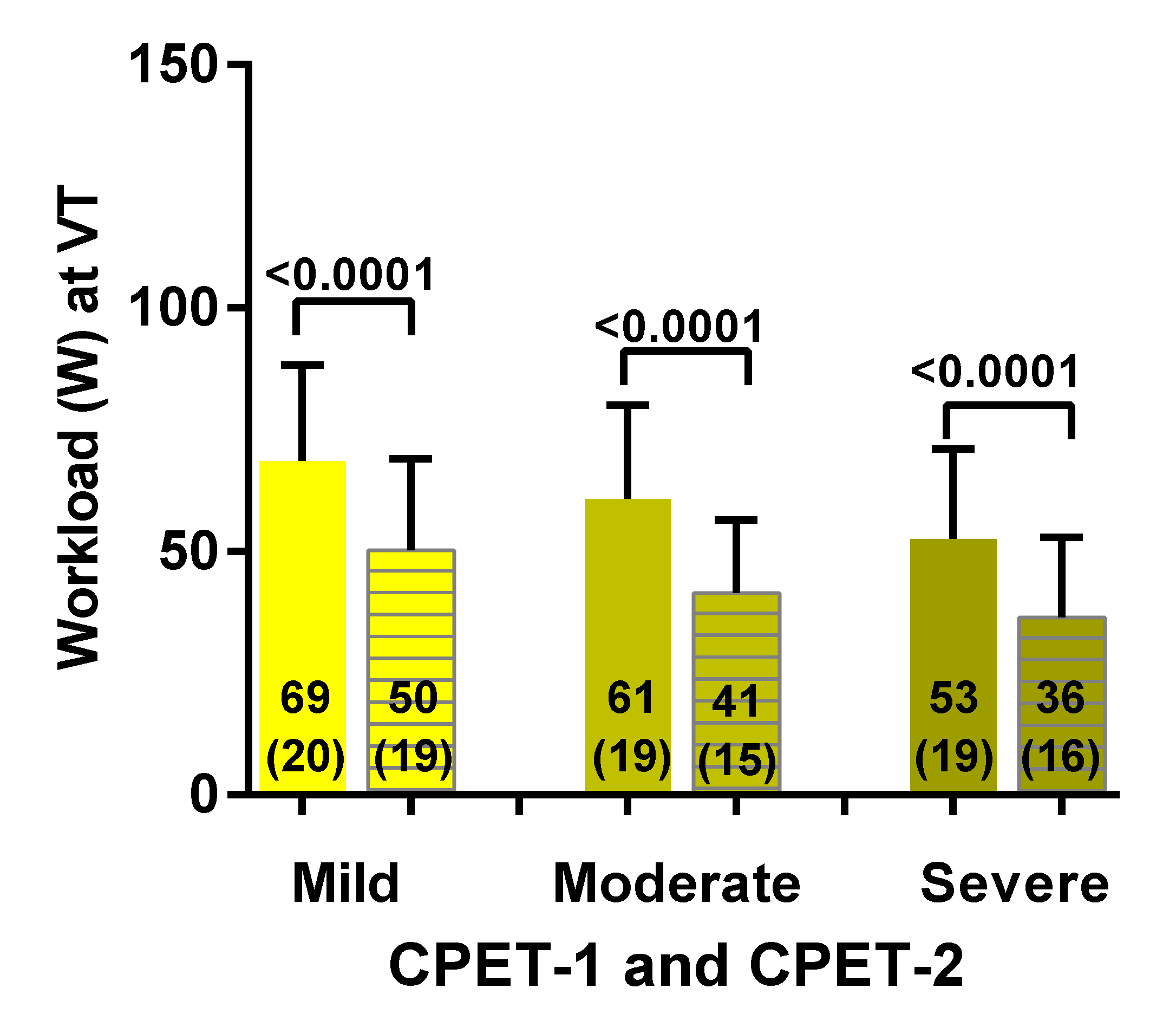
3.2. Two-Day CPET Data for ME/CFS Female Patients with Severe, Moderate and Mild Disease
3.3. Comparison of ME/CFS Patients with Severe, Moderate and Mild Disease for CPET Day-1 and Day-2 Variables
4. Discussion
4.1. Two-Day Cardiopulmonary Exercise Test Studies Reported in Literature
4.2. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fukuda, K. The Chronic Fatigue Syndrome: A Comprehensive Approach to Its Definition and Study. Ann. Intern. Med. 1994, 121, 953–959. [Google Scholar] [CrossRef] [PubMed]
- Carruthers, B.M.; van de Sande, M.I.; de Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.P.; Speight, N.; Vallings, R.; et al. Myalgic encephalomyelitis: International consensus criteria. J. Intern. Med. 2011, 270, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Clayton, E.W. Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. JAMA 2015, 313, 1101. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (IOM). Beyond Mayalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness; The National Academies Press: Washington, DC, USA, 2015. [Google Scholar]
- Paul, L.; Wood, L.; Behan, W.M.; MacLaren, W.M. Demonstration of delayed recovery from fatiguing exercise in chronic fatigue syndrome. Eur. J. Neurol. 1999, 6, 63–69. [Google Scholar] [CrossRef]
- Jones, D.E.J.; Hollingsworth, K.G.; Taylor, R.; Blamire, A.; Newton, J.L. Abnormalities in PH handling by peripheral muscle and potential regulation by the autonomic nervous system in chronic fatigue syndrome. J. Intern. Med. 2010, 267, 394–401. [Google Scholar] [CrossRef]
- Mitchell, J.H.; Sproule, B.J.; Chapman, C.B. The physiological meaning of the maximal oxygen intake test. J. Clin. Investig. 1958, 37, 538–547. [Google Scholar] [CrossRef]
- Saltin, B.; Astrand, P.O. Maximal oxygen uptake in athletes. J. Appl. Physiol. 1967, 23, 353–358. [Google Scholar] [CrossRef]
- Mezzani, A.; Agostoni, P.; Cohen-Solal, A.; Corrà, U.; Jegier, A.; Kouidi, E.; Mazic, S.; Meurin, P.; Piepoli, M.; Simon, A.; et al. Standards for the use of cardiopulmonary exercise testing for the functional evaluation of cardiac patients: A report from the Exercise Physiology Section of the European Association for Cardiovascular Prevention and Rehabilitation. Eur. J. Cardiovasc. Prev. Rehab. 2009, 16, 249–267. [Google Scholar] [CrossRef]
- Guazzi, M.; Adams, V.; Conraads, V.; Halle, M.; Mezzani, A.; Vanhees, L.; Arena, R.; Fletcher, G.F.; Forman, D.E.; Kitzman, D.W.; et al. EACPR/AHA Scientific Statement. Clinical recommendations for cardiopulmonary exercise testing data assessment in specific patient populations. Circulation 2012, 126, 2261–2274. [Google Scholar] [CrossRef]
- De Becker, P.; Roeykens, J.; Reynders, M.; McGregor, N.; De Meirleir, K. Exercise capacity in chronic fatigue syndrome. Arch. Intern. Med. 2000, 160, 3270–3277. [Google Scholar] [CrossRef]
- Fulcher, K.Y.; White, P.D. Strength and physiological response to exercise in patients with chronic fatigue syndrome. J. Neurol. Neurosurg. Psychiatry 2000, 69, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Hodges, L.; Nielsen, T.; Baken, D. Physiological measures in participants with chronic fatigue syndrome, multiple sclerosis and healthy controls following repeated exercise: A pilot study. Clin. Physiol. Funct. Imaging 2017, 38, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Jammes, Y.; Steinberg, J.G.; Mambrini, O.; Bregeon, F.; Delliaux, S. Chronic fatigue syndrome: Assessment of increased oxidative stress and altered muscle excitability in response to incremental exercise. J. Intern. Med. 2005, 257, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Keller, B.A.; Pryor, J.L.; Giloteaux, L. Inability of myalgic encephalomyelitis/chronic fatigue syndrome patients to reproduce VO2peak indicates functional impairment. J. Transl. Med. 2014, 12, 104. [Google Scholar] [CrossRef] [PubMed]
- Sargent, C.; Scroop, G.C.; Nemeth, P.M.; Burnet, R.B.; Buckley, J.D. Maximal oxygen uptake and lactate metabolism are normal in chronic fatigue syndrome. Med. Sci. Sports Exerc. 2002, 34, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Sisto, S.A.; LaManca, J.; Cordero, D.L.; Bergen, M.T.; Ellis, S.P.; Drastal, S.; Boda, W.L.; Tapp, W.N.; Natelson, B.H. Metabolic and cardiovascular effects of a progressive exercise test in patients with chronic fatigue syndrome. Am. J. Med. 1996, 100, 634–640. [Google Scholar] [CrossRef]
- Snell, C.R.; Stevens, S.R.; Davenport, T.E.; Van Ness, J.M. Discriminative validity of metabolic and workload measurements for identifying people with chronic fatigue syndrome. Phys. Ther. 2013, 93, 1484–1492. [Google Scholar] [CrossRef]
- Vanness, J.M.; Snell, C.R.; Stevens, S.R. Diminished cardiopulmonary capacity during post-exertional malaise. J. Chronic Fatigue Syndr. 2007, 14, 77–85. [Google Scholar] [CrossRef]
- Vermeulen, R.C.W.; Kurk, R.M.; Visser, F.C.; Sluiter, W.; Scholte, H.R. Patients with chronic fatigue syndrome performed worse than controls in a controlled repeated exercise study despite a normal oxidative phosphorylation capacity. J. Transl. Med. 2010, 8, 93. [Google Scholar] [CrossRef]
- Vermeulen, R.C.W.; van Eck, I.W.V. Decreased oxygen extraction during cardiopulmonary exercise test in patients with chronic fatigue syndrome. J. Transl. Med. 2014, 12, 20. [Google Scholar] [CrossRef]
- Wallman, K.; Morton, A.R.; Goodman, C.; Grove, J.R. Physiological responses during a submaximal cycle test in chronic fatigue syndrome. Med. Sci. Sports Exerc. 2004, 36, 1682–1688. [Google Scholar] [CrossRef] [PubMed]
- Lien, K.; Johansen, B.; Veierød, M.B.; Haslestad, A.S.; Bøhn, S.K.; Melsom, M.N.; Kardel, K.R.; Iversen, P.O. Abnormal blood lactate accumulation during repeated exercise testing in myalgic encephalomyelitis/chronic fatigue syndrome. Physiol. Rep. 2019, 7, e14138. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.J.; Buckley, J.D.; Thomson, R.L.; Clark, D.; Kwiatek, R.; Davison, K. Diagnostic sensitivity of 2-day cardiopulmonary exercise testing in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. J. Transl. Med. 2019, 17, 80. [Google Scholar] [CrossRef] [PubMed]
- Van Campen, C.L.M.; Visser, F.C. Validity of 2-day cardiopulmonary exercise testing in female patients with myalgic encephalomyelitis/chronic fatigue syndrome. Int. J. Curr. Res. 2020, 12, 10436–10442. [Google Scholar]
- Van Campen, C.L.M.; Rowe, P.C.; Visser, F.C. Validity of 2-day cardiopulmonary exercise testing in male patients with myalgic encephalomyelitis/Chronic fatigue syndrome. Adv. Phys. Educ. 2020, 10, 68–80. [Google Scholar] [CrossRef][Green Version]
- Glaser, S.; Koch, B.; Ittermann, T.; Schaper, C.; Dorr, M.; Felix, S.B.; Henry, V.; Ewert, R.; Hansen, J.E. Influence of age, sex, body size, smoking, and beta blockade on key gas exchange exercise parameters in an adult population. Eur. J. Cardiovasc. Prev. Rehabil. 2010, 17, 469–476. [Google Scholar]
- Cureton, K.; Bishop, P.; Hutchinson, P.; Newland, H.; Vickery, S.; Zwiren, L. Sex difference in maximal oxygen uptake. Effect of equating haemoglobin concentration. Eur. J. Appl. Physiol. Occup. Physiol. 1986, 54, 656–660. [Google Scholar] [CrossRef]
- Fletcher, G.F.; Balady, G.J.; Amsterdam, E.A.; Chaitman, B.; Eckel, R.; Fleg, J.; Froelicher, V.F.; Leon, A.S.; Piña, I.L.; Rodney, R.; et al. Exercise standards for testing and training: A statement for healthcare professionals from the American Heart Association. Circulation 2001, 104, 1694–1740. [Google Scholar] [CrossRef]
- Weber, K.T.; Janicki, J.S. Cardiopulmonary exercise testing for evaluation of chronic cardiac failure. Am. J. Cardiol. 1985, 55, A22–A31. [Google Scholar] [CrossRef]
- Martina, J.R.; Westerhof, B.E.; Van Goudoever, J.; De Beaumont, E.M.F.H.; Truijen, J.; Kim, Y.-S.; Immink, R.V.; Jöbsis, D.A.; Hollmann, M.W.; Lahpor, J.R.; et al. Noninvasive continuous arterial blood pressure monitoring with Nexfin®. Anesthesiology 2012, 116, 1092–1103. [Google Scholar] [CrossRef]
- Beaver, W.L.; Wasserman, K.; Whipp, B.J. A new method for detecting anaerobic threshold by gas exchange. J. Appl. Physiol. 1986, 60, 2020–2027. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.G.; Wyndham, C.H.; Kok, R.; Rahden, M.J.E. Effect of training on maximum oxygen intake and on anaerobic metabolism in man. Graefe’s Arch. Clin. Exp. Ophthalmol. 1967, 24, 18–23. [Google Scholar] [CrossRef]
- Wasserman, K.; McIlroy, M.B. Detecting the threshold of anaerobic metabolism in cardiac patients during exercise. Am. J. Cardiol. 1964, 14, 844–852. [Google Scholar] [CrossRef]
- Coen, B.; Urhausen, A.; Kindermann, W. Individual anaerobic threshold: Methodological aspects of its assessment in running. Int. J. Sports Med. 2001, 22, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.D.; Berkowitz, R.; LeJemtel, T.H. Anaerobic threshold detection in patients with congestive heart failure. Am. J. Cardiol. 1992, 69, 1565–1569. [Google Scholar] [CrossRef]
- Yeh, M.P.; Gardner, R.M.; Adams, T.D.; Yanowitz, F.G.; Crapo, R.O. “Anaerobic threshold”: Problems of determination and validation. J. Appl. Physiol. 1983, 55, 1178–1186. [Google Scholar] [CrossRef]
- Myers, J.; Ashley, E. Dangerous curves. A perspective on exercise, lactate, and the anaerobic threshold. Chest 1997, 111, 787–795. [Google Scholar] [CrossRef]
- Solberg, G.; Robstad, B.; Skjønsberg, O.H.; Borchsenius, F. Respiratory gas exchange indices for estimating the anaerobic threshold. J. Sports Sci. Med. 2005, 4, 29–36. [Google Scholar]
- Mezzani, A. Cardiopulmonary exercise testing: Basics of methodology and measurements. Ann. Am. Thorac. Soc. 2017, 14, S3–S11. [Google Scholar] [CrossRef]
- Brown, A.; Jones, D.E.; Walker, M.; Newton, J.L. Abnormalities of AMPK activation and glucose uptake in cultured skeletal muscle cells from individuals with chronic fatigue syndrome. PLoS ONE 2015, 10, e0122982. [Google Scholar] [CrossRef]
- Jones, D.E.J.; Hollingsworth, K.G.; Jakovljevic, D.G.; Fattakhova, G.; Pairman, J.; Blamire, A.; Trenell, M.; Newton, J.L. Loss of capacity to recover from acidosis on repeat exercise in chronic fatigue syndrome: A case-control study. Eur. J. Clin. Investig. 2011, 42, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Van Campen, C.M.C.; Rowe, P.C.; Verheugt, F.W.A.; Visser, F.C. Physical activity measures in patients with myalgic encephalomyalitis/chronic fatigue syndrome: Correlations between peak oxygen consumption, the physical functioning scale of the SF-36 scale, and the number of steps from an activity meter. J. Transl. Med. 2020, 18, 228–238. [Google Scholar] [CrossRef] [PubMed]

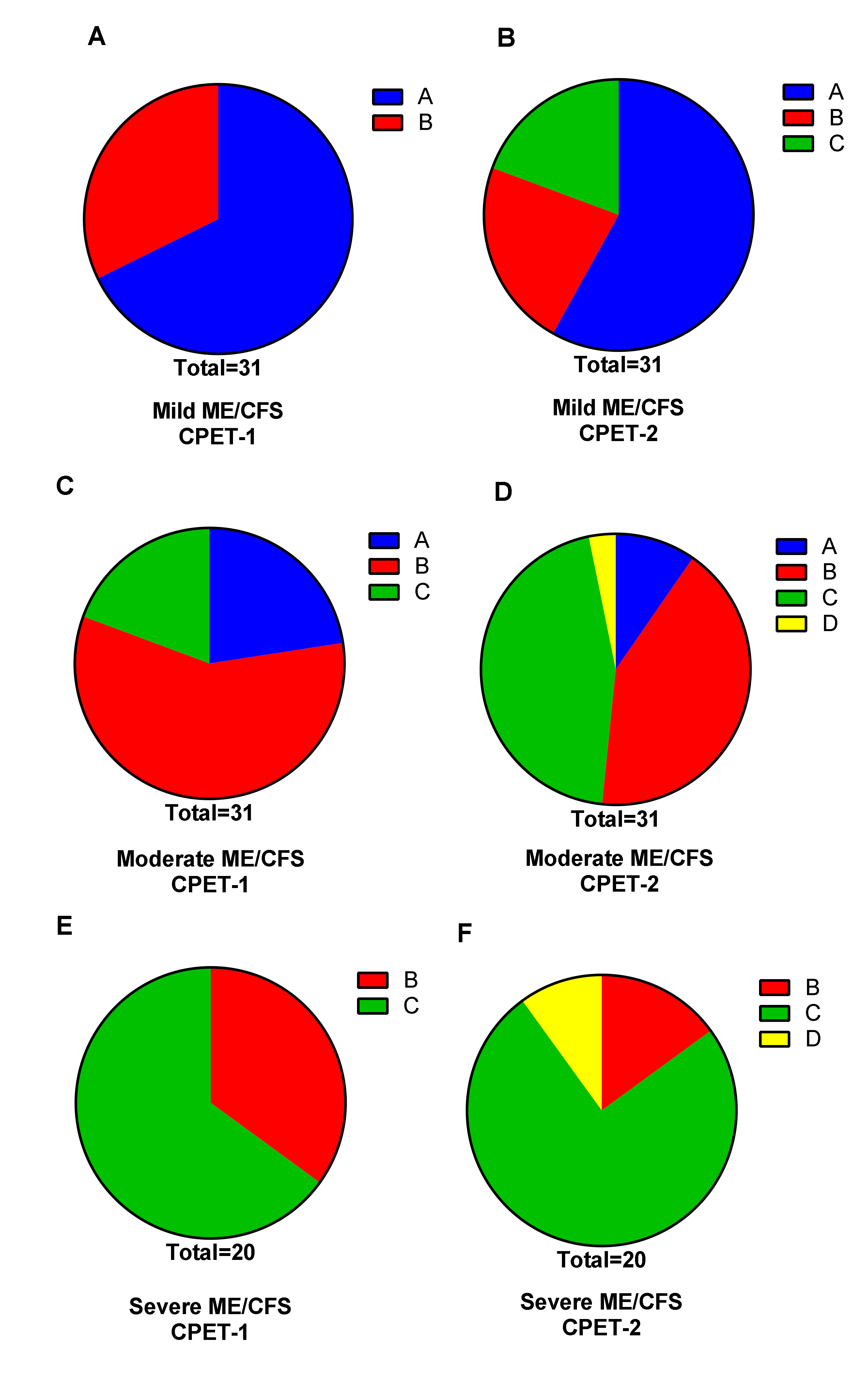
| Weber Class | Oxygen Consumption | Aerobic Capacity |
|---|---|---|
| Class A | >20 mL/min/kg | No impairment |
| Class B | 16–20 mL/min/kg | Mild to moderate impairment |
| Class C | 10–15 mL/min/kg | Moderate to severe impairment |
| Class D | <10 mL/min/kg | Severe impairment |
| Group 1 Severe (n = 20) | Group 2 Moderate (n = 31) | Group 3 Mild (n = 31) | ANOVA/Kruskal-Wallis Test | |
|---|---|---|---|---|
| Age (years) | 39 (10) | 41 (10) | 42 (9) | F (2, 79) = 0.46; p = 0.63 |
| Height (cm) | 171 (7) | 171 (7) | 169 (6) | F (2, 79) = 1.56; p = 0.22 |
| Weight (kg) | 70 (61–77) | 69 (63–80) | 65 (60–72) | X2(2) = 1.144; p = 0.56 |
| BSA (m2) | 1.4 (1.2–1.8) | 1.4 (1.3–1.5) | 1.3 (1.2–1.4) | X2(2) = 1.690; p = 0.43 |
| BMI (kg/m2) | 22.7 (21.8–27.6) | 23.9 (20.7–27.6) | 23.4 (21.4–26.7) | X2(2) = 0.032; p = 0.98 |
| Disease duration (years) | 15.9 (9.3) | 13.3 (8.9) | 13.5 (9.3) | F (2, 79) = 0.56; p = 0.57 |
| Percent Difference CPET-2 Minus CPET-1 | Group 1 Severe (n = 20) | Group 2 Moderate (n = 31) | Group 3 Mild (n = 31) | ANOVA and Post-Hoc Tukey’s Test |
|---|---|---|---|---|
| VO2 peak (mL/min/kg) | −12 (14) | −11 (14) | −6 (11) | F (2, 79) = 1.28; p = 0.28 |
| HR peak (bpm) | −7 (6) | −6 (7) | −3 (6) | F (2, 79) = 2.08; p = 0.13 |
| Workload peak (Watts) | −19 (11) | −16 (15) | −10 (8) | F (2, 79) = 4.37; p = 0.016. Post-hoc tests: 1 vs. 2 p = 0.083; 1 vs. 3 p = 0.019 and 2 vs. 3 p = 0.68 |
| VO2 VT (mL/min/kg) | −19 (11) | −21 (12) | −21 (11) | F (2, 79) = 0.26; p = 0.77 |
| HR VT (bpm) | −7 (5) | −9 (6) | −8 (7) | F (2, 79) = 0.78; p = 0.46 |
| Workload VT (Watts) | −33 (20) | −31 (18) | −26 (18) | F (2, 79) = 0.54; p = 0.58 |
| RER | −5 (7) | −2 (9) | −3 (7) | F (2, 79) = 0.83; p = 0.44 |
| Group 1 Severe (n = 20) | Group 2 Moderate (n = 31) | Group 3 Mild (n = 31) | ANOVA |
|---|---|---|---|
| CPET day-1 | |||
| 1.08 (0.09) | 1.09 (0.09) | 1.13 (0.11) | F(2.79) = 2.02; p = 0.14 |
| CPET day-2 | |||
| 1.02 (0.11) | 1.07 (0.11) | 1.10 (0.11 | F(2.79) = 2.77; p = 0.07 |
| Literature Day-1 | Literature Day-2 | Present Study Day-1 | Present Study Day-2 | |
|---|---|---|---|---|
| Peak VO2 (mL/min/kg) | 19–26 | 17–21 | 14–23 | 12–21 |
| VT VO2 (mL/min/kg) | 12–15 | 9–12 | 10–14 | 8–11 |
| Peak Workload (Watt) | 110–132 | 102–125 | 90–144 | 73–130 |
| VT Workload (Watt) | 50–62 | 22–54 | 53–69 | 36–50 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Campen, C.M.C.; Rowe, P.C.; Visser, F.C. Two-Day Cardiopulmonary Exercise Testing in Females with a Severe Grade of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Comparison with Patients with Mild and Moderate Disease. Healthcare 2020, 8, 192. https://doi.org/10.3390/healthcare8030192
van Campen CMC, Rowe PC, Visser FC. Two-Day Cardiopulmonary Exercise Testing in Females with a Severe Grade of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Comparison with Patients with Mild and Moderate Disease. Healthcare. 2020; 8(3):192. https://doi.org/10.3390/healthcare8030192
Chicago/Turabian Stylevan Campen, C (Linda) M. C., Peter C. Rowe, and Frans C. Visser. 2020. "Two-Day Cardiopulmonary Exercise Testing in Females with a Severe Grade of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Comparison with Patients with Mild and Moderate Disease" Healthcare 8, no. 3: 192. https://doi.org/10.3390/healthcare8030192
APA Stylevan Campen, C. M. C., Rowe, P. C., & Visser, F. C. (2020). Two-Day Cardiopulmonary Exercise Testing in Females with a Severe Grade of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Comparison with Patients with Mild and Moderate Disease. Healthcare, 8(3), 192. https://doi.org/10.3390/healthcare8030192





