Lyme Disease: Diversity of Borrelia Species in California and Mexico Detected Using a Novel Immunoblot Assay
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Data Collection
2.2. Antigen Preparation
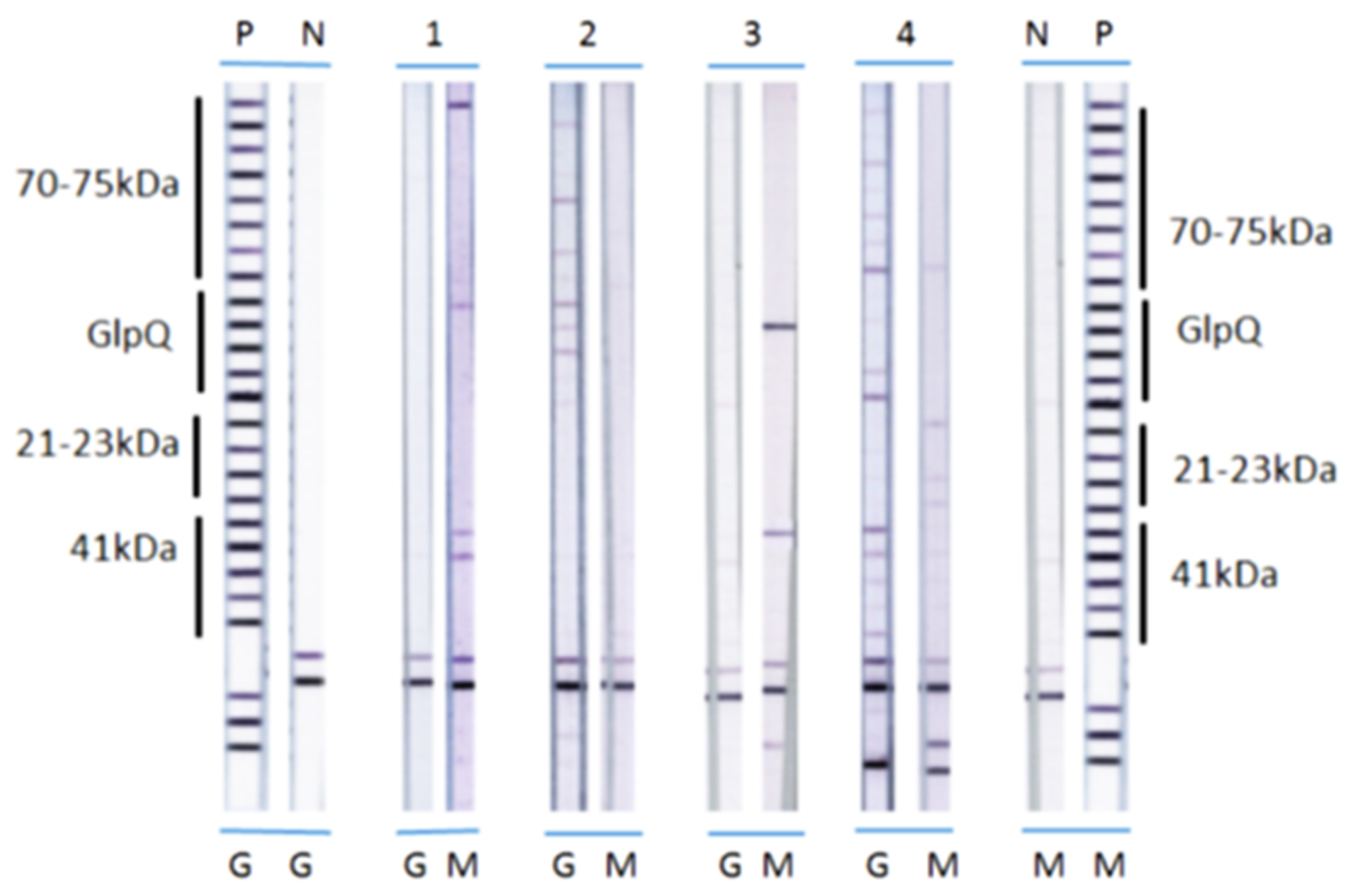
2.3. Preparation of Antigen Strips
2.4. Detection of Antibody Reactivity
2.5. Scoring of Immunoblots
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Appendix A

| # | State/Country | Bb IgM | Bb IgG | Bb IgM and IgG | RFB IgM | RFB IgG | RFB IgM and IgG | Species | Bbss IgM or IgG |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Mex | CDC+ | + | + | − | − | − | B. afzelii/B. garinii | |
| 2 | CA | + | + | + | − | − | − | Bbss | + |
| 3 | CA | − | − | − | + | − | − | B. hermsii | |
| 4 | CA | − | − | − | − | + | − | RFB sp. | |
| 5 | CA | CDC+ | − | − | − | − | − | B. mayonii B. spielmanii | |
| 6 | CA | CDC+ | − | − | − | − | − | Bbss | + |
| 7 | CA | − | − | − | + | − | − | RFB sp. | |
| 8 | CA | CDC+ | − | − | − | − | − | Bbsl, Bbss | + |
| 9 | CA | − | − | − | − | + | − | B. miyamotoi | |
| 10 | CA | CDC+ | + | + | − | − | − | B. californiensis | |
| 11 | Mex | CDC+ | − | − | − | − | − | Bb sp. | |
| 12 | CA | + | − | − | − | − | − | Bbsl, Bbss | + |
| 13 | CA | − | − | − | + | − | − | RFB sp. | |
| 14 | CA | + | − | − | + | − | − | Bb sp. B. hermsii | |
| 15 | CA | − | − | − | + | + | + | B. miyamotoi RFB sp. | |
| 16 | CA | CDC+ | − | − | + | − | − | B. spielmanii RFB sp. | |
| 17 | CA | − | − | − | − | + | − | B. miyamotoi | |
| 18 | CA | − | − | − | + | − | − | RFB sp. | |
| 19 | CA | − | − | − | + | − | − | RFB sp. | |
| 20 | CA | − | − | − | − | + | − | RFB sp. | |
| 21 | CA | − | − | − | − | + | − | B. turicatae | |
| 22 | CA | CDC+ | CDC+ | + | − | − | − | B. spielmanii (IgM) B. mayonii (IgG), Bbss | + |
| 23 | CA | − | − | − | + | + | + | RFB sp. | |
| 24 | CA | − | − | − | − | + | − | RFB sp. | |
| 25 | Mex | − | − | − | − | + | − | B. turicatae | |
| 26 | CA | − | − | − | + | − | − | RFB Sp. | |
| 27 | CA | − | − | − | − | + | − | B. miyamotoi | |
| 28 | CA | − | − | − | − | + | − | B. turicatae | |
| 29 | CA | CDC+ | + | + | − | − | − | B. spielmanii | |
| 30 | Mex | − | − | − | + | − | − | RFB sp. | |
| 31 | CA | CDC+ | CDC+ | + | + | − | − | B. californiensis RFB sp. Bbss | + |
| 32 | CA | CDC+ | − | − | − | − | − | B. afzelii/B. garinii | |
| 33 | CA | − | − | − | + | − | − | RFB sp. | |
| 34 | CA | − | − | − | + | − | − | RFB sp. | |
| 35 | Mex | CDC+ | + | + | − | − | − | B. californiensis | |
| 36 | CA | − | − | − | − | + | − | RFB sp. | |
| 37 | CA | CDC+ | − | − | + | − | − | B. afzelii/B. garinii B. turicatae | |
| 38 | Mex | − | − | − | + | − | − | RFB sp. | |
| 39 | CA | − | − | − | − | + | − | RFB sp. | |
| 40 | CA | − | − | − | + | − | − | B. turicatae | |
| 41 | Mex | − | − | − | − | + | − | B. turicatae | |
| 42 | CA | − | − | − | + | − | − | RFB sp. | |
| 43 | CA | CDC+ | + | + | − | − | − | B. californiensis, B. mayonii, B. afzelii/B. garinii | |
| 44 | CA | CDC+ | − | − | − | − | − | B. spielmanii | |
| 45 | CA | CDC+ | − | − | − | + | − | B. afzelii/B. garinii RFB sp. | |
| 46 | CA | − | − | − | + | − | − | B. turicatae | |
| 47 | CA | CDC+ | CDC+ | + | − | − | − | Bbss | + |
| 48 | CA | + | − | − | − | + | − | B. californiensis RFB sp. | |
| 49 | CA | − | − | − | + | − | − | RFB sp. | |
| 50 | Mex | − | − | − | + | − | − | B. hermsii B. turcica bipA + | |
| 51 | CA | CDC+ | − | − | − | − | − | Bb sp. Bbss (Strong+ to all OspCs) | + |
| 52 | CA | CDC+ | − | − | − | − | − | B. spielmanii Bbss | + |
| 53 | CA | − | − | − | + | − | − | B. turcica | |
| 54 | Mex | CDC+ | − | − | − | − | − | B. afzelii/B. garinii | |
| 55 | CA | − | − | − | + | − | − | RFB sp. | |
| 56 | CA | + | − | − | − | − | − | B. spielmanii | |
| 57 | CA | − | − | − | + | − | − | RFB sp. B. miyamotoi | |
| 58 | CA | − | − | − | + | − | − | RFB sp. | |
| 59 | CA | − | − | − | + | − | RFB sp. | ||
| 60 | CA | CDC+ | − | − | − | − | − | Bb sp | |
| 61 | CA | − | − | − | + | + | + | B. hermsii B. miyamotoi | |
| 62 | CA | − | − | − | − | + | − | B. miyamotoi | |
| 63 | CA | CDC+ | − | − | IND | − | − | B. californiensis Bbss | + |
| 64 | CA | + | + | + | − | − | − | B. afzelii/B. garinii Bbss | + |
| 65 | CA | CDC+ | − | − | + | − | − | Bbss RFB sp. | + |
| 66 | Mex | − | − | − | − | + | − | B. hermsii | |
| 67 | CA | CDC+ | − | − | IND | IND | − | B. spielmanii B. afzelii/B. garinii | |
| 68 | CA | − | − | − | + | − | − | RFB sp. | |
| 69 | CA | − | + | − | − | − | − | B. californiensis | |
| 70 | CA | − | − | − | − | + | − | B. turicatae | |
| 71 | CA | − | − | − | − | + | − | B. turicatae | |
| 72 | CA | − | + | − | − | − | − | B. spielmanii | |
| 73 | CA | − | − | − | − | + | − | B. hermsii | |
| 74 | CA | − | − | − | + | − | − | B. hermsii | |
| 75 | CA | − | + | − | − | − | − | Bb sp. | |
| 76 | Mex | − | − | − | + | − | − | RFB sp. | |
| 77 | CA | − | − | − | − | + | − | B. hermsii | |
| 78 | CA | − | + | − | − | − | − | B. californiensis | |
| 79 | CA | − | − | − | − | + | − | B. hermsii | |
| 80 | CA | − | − | − | − | + | − | RFB sp. | |
| 81 | Mex | − | + | − | − | − | − | B. afzelii/B. garinii | |
| 82 | Mex | − | + | − | − | − | − | B. spielmanii | |
| 83 | CA | − | + | − | − | − | − | B. afzelii/B. garinii | |
| 84 | CA | − | − | − | + | − | − | RFB sp. | |
| 85 | CA | − | − | − | + | − | − | RFB sp. | |
| 86 | CA | − | + | − | − | − | − | Bbsl, Bbss | + |
| 87 | CA | − | − | − | − | + | − | RFB sp. | |
| 88 | CA | − | + | − | − | − | − | Bbsl, Bbss | + |
| 89 | CA | − | + | − | − | − | − | Bb sp. | |
| 90 | CA | − | + | − | − | + | − | Bb sp. RFB sp. | |
| Total | IgM only 20 | IgG only 11 | IgM and IgG 10 | IgM only 29 | IgG only 25 | IgM and IgG 3 | Bbss IgM or IgG 14 |
| Immunoblot | Total |
|---|---|
| Group 1 Bbsl positive | 42 |
| Group 2 RFB positive | 56 |
| Dual Group 1 and 2 positive | 8 |
| Group 1 Bbsl positive samples | 34 (38%) |
| Bbsl alone | 8 |
| Bbss alone | 4 |
| B. afzelii/garinii alone | 6 |
| B. californiensis alone | 6 |
| B. spielmanii alone | 6 |
| B. mayonii + B. speilmanii | 2 |
| B. spielmanii + B. afzelii/garinii | 1 |
| B. afzelii/garinii, B. californiensis, B. mayonii | 1 |
| Group 2. RFB Positive Samples | 48 (53%) |
| RFB alone | 25 |
| B. hermsii alone | 7 |
| B. miyamotoi alone | 4 |
| B. turicatae alone | 8 |
| B. turcica alone | 2 |
| B. hermsii + B. turcica | 1 |
| B. hermsii + B. miyamotoi | 1 |
| Dual Group 1 and 2 | 8 (9%) |
| Bb + RFB | 2 |
| Bb + B. hermsii | 1 |
| B. californiensis + RFB | 2 |
| B. spielmanii + RFB | 1 |
| B. afzelii/garinii + RFB | 1 |
| B. afzelii/garinii + B. turicatae | 1 |
| Total cases | 90 |
| Source | Characteristic | Total No. of Sera | Bbsl Immunoblots (+) | RFB Immunoblots (+) |
|---|---|---|---|---|
| CDC Reference Set (n = 25) | Fibromyalgia | 2 | 0 | 0 |
| Healthy endemic | 7 | 1 | 0 | |
| Healthy non-endemic | 6 | 0 | 0 | |
| Mononucleosis | 2 | 0 | 0 | |
| Multiple sclerosis | 2 | 0 | 1 | |
| Rheumatoid arthritis | 2 | 0 | 0 | |
| Severe Periodontitis | 2 | 0 | 0 | |
| Syphilis | 2 | 0 | 1 | |
| CAP and NYSHD (n = 42) Autoimmunity and Allergy | Autoimmune | |||
| ANA (+) | 3 | 0 | 0 | |
| ANA (−) | 2 | 0 | 0 | |
| DNA (+) | 1 | 0 | 0 | |
| Rheumatoid factor (+) | 9 | 0 | 0 | |
| Rheumatoid factor (−) | 8 | 0 | 0 | |
| Allergy (n = 19) | ||||
| IgG (+) | 13 | 0 | 1 | |
| Spec. IgE (+) | 4 | 0 | 0 | |
| Spec. IgE (−) | 2 | 0 | 0 | |
| NYB (n = 21) Viruses and RPR (+) | Epstein–Barr virus (EBV) | 4 | 1 | 0 |
| Human immunodeficiency virus (HIV) | 4 | 0 | 0 | |
| Cytomegalovirus (CMV) | 5 | 0 | 1 | |
| Hepatitis C virus (HCV) | 0 | 0 | 0 | |
| RPR (+) | 8 | 2 | 1 | |
| IGeneX (n = 87) | Bartonella henselae infection | 7 | 0 | 0 |
| Human granulocytic anaplasmosis | 16 | 0 | 0 | |
| Babesia microti infection | 14 | 0 | 0 | |
| Babesia duncani Infection | 41 | 0 | 0 | |
| Human monocytic ehrlichiosis | 5 | 0 | 0 | |
| Negative controls | 4 | 0 | 0 | |
| False Positives | 175 | 4 | 5 | |
| Specificity | 97.7% | 97.1% | ||
References
- Liu, S.; Cruz, I.D.; Ramos, C.C.; Taleon, P.; Ramasamy, R.; Shah, J. Pilot study of immunoblots with recombinant Borrelia burgdorferi antigens for laboratory diagnosis of Lyme disease. Healthcare 2018, 14, 99. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.S.; Liu, S.; Du Cruz, I.; Poruri, A.; Maynard, R.; Shkilna, M. Line immunoblot assay for tick-borne relapsing fever and findings in patient sera from Australia, Ukraine and the USA. Healthcare 2019, 7, 121. [Google Scholar] [CrossRef]
- Todar, K. Todar’s Online Textbook of Bacteriology; University of Wisconsin-Madison, Department of Bacteriology: Madison, WI, USA, 2006. [Google Scholar]
- Cutler, S.J. Relapsing fever Borreliae: A global review. Clin. Lab Med. 2015, 35, 847–865. [Google Scholar] [CrossRef]
- Cutler, S.J.; Ruzic-Sabljic, E.; Potkonjak, A. Emerging borreliae—Expanding beyond Lyme borreliosis. Mol. Cell Probes 2017, 31, 22–27. [Google Scholar] [CrossRef]
- Rose, I.; Yoshimizu, M.H.; Bonilla, D.L.; Fedorova, N.; Lane, R.S.; Padgett, K.A. Phylogeography of Borrelia spirochetes in Ixodes pacificus and Ixodes spinipalpis ticks highlights differential acarological risk of tick-borne disease transmission in northern versus southern California. PLoS ONE 2019, 14, e0214726. [Google Scholar] [CrossRef] [PubMed]
- Panetta, J.L.; Sima, R.; Calvani, N.E.D.; Hajdušek, O. Reptile-associated Borrelia species in the goanna tick (Bothriocroton undatum) from Sydney, Australia. Parasit Vectors 2017, 20, 616. [Google Scholar] [CrossRef] [PubMed]
- WHO. Lyme Borreliosis (Lyme Disease). Available online: https://www.who.int/ith/diseases/lyme/en/ (accessed on 24 October 2019).
- Rochlin, I.; Ninivaggi, D.V.; Benach, J.L. Malaria and Lyme disease - the largest vector-borne US epidemics in the last 100 years: Success and failure of public health. BMC Public Health 2019, 19, 804. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, A.M.; Hinckley, A.F.; Mead, P.S.; Hook, S.A.; Kugeler, K.J. Surveillance for Lyme Disease—United States, 2008–2015. MMWR Surveill. Summ. 2017, 66, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Maraspin, V.; Ruzic-Sabljic, E.; Strle, F. Lyme Borreliosis and Borrelia spielmanii. Emerg. Infect. Dis. 2006, 12, 1177. [Google Scholar] [CrossRef]
- Dworkin, M.S.; Schwan, T.G.; Anderson, D.E., Jr.; Borchardt, S.M. Tick-borne relapsing fever. Infect. Dis. Clin. N. Am. 2008, 22, 449–468. [Google Scholar] [CrossRef]
- Lopez, J.E.; Krishnavahjala, A.; Garcia, M.N.; Bermudez, S. Tick-borne relapsing fever spirochetes in the Americas. Vet. Sci. 2016, 3, 16. [Google Scholar] [CrossRef] [PubMed]
- Telford, S.R., III; Goethert, H.K.; Molloy, P.J.; Berardi, V.P.; Chowdri, H.R.; Gugliotta, J.L. Borrelia miyamotoi disease: Neither Lyme disease nor relapsing fever. Clin. Lab. Med. 2015, 35, 867–882. [Google Scholar] [CrossRef] [PubMed]
- Middelveen, M.J.; Shah, J.S.; Fesler, M.C.; Stricker, R.B. Relapsing fever Borrelia in California: A pilot serological study. Int. J. Gen. Med. 2018, 11, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Barbour, A.G. Relapsing Fever and Other Borrelia Diseases. In Tropical Infectious Diseases: Principles, Pathogens and Practice, 3rd ed.; Guerrant, R.L., Walker, D.H., Weller, P.F., Eds.; W.B. Saunders: Edinburgh, UK, 2011; pp. 295–302. [Google Scholar]
- CDC. Tick-Borne Relapsing Fever (TBRF). Information for Clinicians; 2016. Available online: https://www.cdc.gov/relapsing-fever/clinicians/index.html (accessed on 24 October 2019).
- Koton, Y.; Bisharat, N. Tick-borne relapsing fever with severe Jarisch-Herxheimer reaction. ISR Med. Assoc. J. 2018, 20, 62–63. [Google Scholar]
- Eickhoff, C.; Blaylock, J. Tickborne diseases other than Lyme in the United States. Cleve Clin. J. Med. 2017, 84, 555–567. [Google Scholar] [CrossRef]
- Stricker, R.B.; Johnson, L. Lyme wars: Let’s tackle the testing. BMJ 2007, 335, 1008. [Google Scholar] [CrossRef]
- Stricker, R.B.; Johnson, L. Serologic tests for Lyme disease: More smoke and mirrors. Clin. Infect. Dis. 2008, 47, 1111–1112. [Google Scholar] [CrossRef][Green Version]
- Cook, M.J.; Puri, B.K. Commercial test kits for detection of Lyme borreliosis: A meta-analysis of test accuracy. Int. J. Gen. Med. 2016, 9, 427–440. [Google Scholar] [CrossRef]
- Cameron, D.J.; Johnson, L.B.; Maloney, E.L. Evidence assessments and guideline recommendations in Lyme disease: The clinical management of known tick bites, erythema migrans rashes and persistent disease. Expert Rev. Anti Infect. Ther. 2014, 12, 1103–1135. [Google Scholar] [CrossRef]
- Stricker, R.B.; Fesler, M.C. Chronic Lyme Disease: A Working Case Definition. Am. J. Infect. Dis. 2018, 14, 1–44. [Google Scholar] [CrossRef]
- Golovchenko, M.; Vancová, M.; Clark, K.; Oliver, J.H., Jr.; Grubhoffer, L.; Rudenko, N. A divergent spirochete strain isolated from a resident of the southeastern United States was identified by multilocus sequence typing as Borrelia bissettii. Parasit Vectors 2016, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Hinckley, A.F.; Connally, N.P.; Meek, J.I.; Johnson, B.J.; Kemperman, M.M.; Feldman, K.A.; White, J.L.; Mead, P.S. Lyme disease testing by large commercial laboratories in the United States. Clin. Infect. Dis. 2014, 59, 676–681. [Google Scholar] [CrossRef] [PubMed]
- Davidsson, M. The financial implications of a well-hidden and ignored chronic Lyme disease pandemic. Healthcare 2018, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, N.; Golovchenko, M.; Grubhoffer, L.; Oliver, J.H., Jr. Updates on Borrelia burgdorferi sensu lato complex with respect to public health. Ticks Tick Borne Dis. 2011, 2, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Parola, P.; Diatta, G.; Socolovschi, C.; Mediannikov, O.; Tall, A.; Bassene, H.; Trape, J.F.; Raoult, D. Tick-Borne Relapsing Fever Borreliosis, Rural Senegal. Emerg. Infect. Dis. 2011, 17, 883–885. [Google Scholar] [CrossRef] [PubMed]
- Sarih, M.; Garnier, M.; Boudebouch, N.; Bouattour, A.; Rihani, A.; Hassar, M.; Gern, L.; Postic, D.; Cornet, M. Borrelia hispanica relapsing fever, Morocco. Emerg. Infect. Dis. 2009, 15, 1626–1629. [Google Scholar] [CrossRef]
- Karan, L.; Makenov, M.; Kolyasnikova, N.; Stukolova, O.; Toporkova, M.; Olenkova, O. Dynamics of spirochetemia and early PCR detection of Borrelia miyamotoi. Emerg. Infect. Dis. 2018, 24, 860–867. [Google Scholar] [CrossRef]
- Krause, P.J.; Carroll, M.; Fedorova, N. Human Borrelia miyamotoi infection in California: Serodiagnosis is complicated by multiple endemic Borrelia species. PLoS ONE 2018, 13, e0191725. [Google Scholar] [CrossRef]
- Forrester, J.D.; Kjemtrup, A.M.; Fritz, C.L. Tickborne Relapsing Fever—United States, 1990–2011. MMWR 2015, 64, 58–60. [Google Scholar]
- Burkot, T.R.; Mullen, G.R.; Anderson, R.; Schneider, B.S.; Happ, C.M.; Zeidner, N.S. Borrelia lonestari DNA in adult Amblyomma americanum ticks, Alabama. Emerg. Infect. Dis. 2001, 7, 471–473. [Google Scholar] [CrossRef]
- Bunikis, J.; Tsao, J.; Garpmo, U.; Berglund, J.; Fish, D.; Barbour, A.G. Typing of Borrelia relapsing fever group strains. Emerg. Infect. Dis. 2004, 10, 1661–1664. [Google Scholar] [CrossRef] [PubMed]
- Schwan, T.G.; Raffel, S.J.; Schrumpf, M.E.; Schrumpf, M.E.; Webster, L.S.; Marques, A.R.; Spano, R.; Rood, M.; Burns, J.; Hu, R. Tick-borne relapsing fever and Borrelia hermsii, Los Angeles County, California, USA. Emerg. Infect. Dis. 2009, 15, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Vuyyuru, R.; Liu, H.; Manser, T.; Alugupalli, K.R. Characteristics of Borrelia hermsii infection in human hematopoietic stem cell-engrafted mice mirror those of human relapsing fever. Proc. Natl. Acad. Sci. USA 2011, 108, 20707–20712. [Google Scholar] [CrossRef] [PubMed]
- Wagemakers, A.; Staarink, P.J.; Sprong, H.; Hovius, J.W.R. Borrelia miyamotoi: A widespread tick-borne relapsing fever spirochete. Trends Parasitol. 2015, 31, 260–269. [Google Scholar] [CrossRef]
- Nieto, N.C.; Teglas, M.B. Relapsing fever group Borrelia in Southern California rodents. J. Med. Entomol. 2014, 51, 1029–1034. [Google Scholar] [CrossRef]
- Padgett, K.; Bonilla, D.; Kjemtrup, A. Large scale spatial risk and comparative prevalence of Borrelia miyamotoi and Borrelia burgdorferi sensu lato in Ixodes pacificus. PLoS ONE 2014, 9, e110853. [Google Scholar] [CrossRef]
- Cook, V.J.; Fedorova, N.; Macdonald, W.P. Unique strain of Borrelia miyamotoi in Ixodes pacificus ticks, California, USA. Emerg. Infect. Dis. 2016, 22, 2205–2207. [Google Scholar] [CrossRef]
- Nguyen, N.T.T.; Röttgerding, F.; Devraj, G.; Lin, Y.-P.; Koenigs, A.; Kraiczy, P. The complement binding and inhibitory protein CbiA of Borrelia miyamotoi degrades extracellular matrix components by interacting with plasmin(ogen). Front. Cell Infect. Microbiol. 2018, 8, 23. [Google Scholar] [CrossRef]
- Middelveen, M.J.; Cruz, I.D.; Fesler, M.C.; Stricker, R.B.; Shah, J.S. Detection of tick-borne infection in Morgellons disease patients by serological and molecular techniques. Clin. Cosmet. Investig. Dermatol. 2018, 11, 561–569. [Google Scholar] [CrossRef]
- Krause, P.J.; Barbour, A.G. Borrelia miyamotoi: The newest infection brought to us by deer ticks. Ann. Intern Med. 2015, 163, 141–142. [Google Scholar] [CrossRef]
- Mun, J.; Eisen, R.J.; Eisen, L.; Lane, R.S. Detection of a Borrelia miyamotoi sensu lato relapsing-fever group spirochete from Ixodes pacificus in California. J. Med. Entomol. 2006, 43, 120–123. [Google Scholar] [CrossRef]
- Barbour, A.G. Phylogeny of a relapsing fever Borrelia species transmitted by the hard tick Ixodes scapularis. Infect. Genet. Evol. 2014, 27, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Takano, A.; Toyomane, K.; Konnai, S.; Ohashi, K.; Nakao, M.; Ito, T. Tick surveillance for relapsing fever spirochete Borrelia miyamotoi in Hokkaido, Japan. PLoS ONE 2014, 9, e104532. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.J.; Fish, D.; Narasimhan, S.; Barbour, A.G. Borrelia miyamotoi infection in nature and in humans. Clin. Microbiol. Infect. 2015, 21, 631–639. [Google Scholar] [CrossRef]
- Mukhacheva, T.A.; Salikhova, I.I.; Kovalev, S.Y. Multilocus spacer analysis revealed highly homogeneous genetic background of Asian type of Borrelia miyamotoi. Infect. Genet. Evol. 2015, 31, 257–262. [Google Scholar] [CrossRef]
- Wroblewski, D.; Gebhardt, L.; Prusinski, M.A.; Meehan, L.J.; Halse, T.A.; Miusser, K.A. Detection of Borrelia miyamotoi and other tick-borne pathogens in human clinical specimens and Ixodes scapularis ticks in New York State, 2012–2015. Ticks Tick Borne Dis. 2017, 8, 407–411. [Google Scholar] [CrossRef]
- Gofton, A.W.; Margos, G.; Fingerle, V.; Hepner, S.; Loh, S.M.; Ryan, U.; Irwin, P.; Oskam, C.L. Genome-wide analysis of Borrelia turcica and ‘Candidatus Borrelia tachyglossi’ shows relapsing fever-like genomes with unique genomic links to Lyme disease Borrelia. Infect. Genet. Evol. 2018, 66, 72–81. [Google Scholar] [CrossRef]
- Bergström, S.; Normark, J. Microbiological features distinguishing Lyme disease and relapsing fever spirochetes. Wien Klin. Wochenschr. 2018, 130, 484–490. [Google Scholar] [CrossRef]
- Margos, G.; Marosevic, D.; Cutler, S.; Derdakova, M.; Diuk Wasser, M.; Emler, S.; Kahl, O. There is inadequate evidence to support the division of the genus Borrelia. Int. J. Syst. Evol. Microbiol. 2017, 67, 1081–1084. [Google Scholar] [CrossRef]
- Hynote, E.D.; Mervine, P.C.; Stricker, R.B. Clinical evidence for rapid transmission of Lyme disease following a tickbite. Diagn Microbiol. Infect. Dis. 2012, 72, 188–192. [Google Scholar] [CrossRef]
- Cook, M.J. Lyme borreliosis: A review of data on transmission time after tick attachment. Int. J. Gen. Med. 2014, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hofhuis, A.; van de Kassteele, J.; Sprong, H.; van den Wijngaard, C.C.; Harms, M.G.; Fonville, M. Predicting the risk of Lyme borreliosis after a tick bite, using a structural equation model. PLoS ONE 2017, 12, e0181807. [Google Scholar] [CrossRef] [PubMed]
- Sertour, N.; Cotté, V.; Garnier, M.; Malandrin, L.; Ferquel, E.; Choumet, V. Infection kinetics and tropism of Borrelia burgdorferi sensu lato in mouse after natural (via ticks) or artificial (needle) infection depends on the bacterial strain. Front. Microbiol. 2018, 9, 1722. [Google Scholar] [CrossRef] [PubMed]
- Pospisilova, T.; Urbanova, V.; Hes, O.; Kopacek, P.; Hajdusek, O.; Sima, R. Tracking of Borrelia afzelii transmission from infected Ixodes ricinus nymphs to mice. Infect. Immun. 2019, 87, e00896-18. [Google Scholar] [CrossRef]
- Schwan, T.G.; Piesman, J. Vector interactions and molecular adaptations of Lyme disease and relapsing fever spirochetes associated with transmission by ticks. Emerg. Infect. Dis. 2002, 8, 115–121. [Google Scholar] [CrossRef]
- Boyle, W.K.; Wilder, H.K.; Lawrence, A.M.; Lopez, J.E. Transmission dynamics of Borrelia turicatae from the arthropod vector. PLoS Negl. Trop. Dis. 2014, 8, e2767. [Google Scholar] [CrossRef]
- Breuner, N.E.; Dolan, M.C.; Replogle, A.J. Transmission of Borrelia miyamotoi sensu lato relapsing fever group spirochetes in relation to duration of attachment by Ixodes scapularis nymphs. Ticks Tick Borne Dis. 2017, 8, 677–681. [Google Scholar] [CrossRef]
- Shah, J.S.; Cruz, I.D.; Narciso, W.; Lo, W.; Harris, N.S. Improved sensitivity of Lyme disease Western blots prepared with a mixture of Borrelia burgdorferi strains 297 and B31. Chronic Dis. Int. 2014, 1, 7. [Google Scholar]
- CDC. Lyme Disease (Borrelia burgdorferi): 2017 Case Definition. 2017. Available online: https://wwwn.cdc.gov/nndss/conditions/lyme-disease/case-definition/2017/ (accessed on 24 October 2019).
- Greene, R.T.; Walker, R.L.; Greene, C.E. Pseudospirochetes in animal blood being cultured for Borrelia burgdorferi. J. Vet. Diagn. Investig. 1991, 3, 350–352. [Google Scholar] [CrossRef]
- Craft, J.E.; Fischer, D.K.; Shimamoto, G.T.; Steere, A.C. Antigens of Borrelia burgdorferi recognized during Lyme disease. Appearance of a new immunoglobulin M response and expansion of the immunoglobulin G response late in the illness. J. Clin. Investig. 1986, 78, 934–939. [Google Scholar] [CrossRef]
- Kalish, R.A.; Leong, J.M.; Steere, A.C. Early and late antibody responses to full-length and truncated constructs of outer surface protein A of Borrelia burgdorferi in Lyme disease. Infect. Immun. 1995, 63, 2228–2235. [Google Scholar] [CrossRef]
- Lomholt, H.; Lebech, A.M.; Hansen, K.; Brandrup, F.; Halkier-Sørenson, L. Long-term serological follow-up of patients treated for chronic cutaneous borreliosis or culture-positive erythema migrans. Acta Derm. Venereol. 2000, 80, 362–366. [Google Scholar]
- Middelveen, M.J.; Sapi, E.; Burke, J.; Filush, K.R.; Franco, A.; Fesler, M.C.; Stricker, R.B. Persistent Borrelia infection in patients with ongoing symptoms of Lyme disease. Healthcare 2018, 6, 33. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fesler, M.C.; Shah, J.S.; Middelveen, M.J.; Du Cruz, I.; Burrascano, J.J.; Stricker, R.B. Lyme Disease: Diversity of Borrelia Species in California and Mexico Detected Using a Novel Immunoblot Assay. Healthcare 2020, 8, 97. https://doi.org/10.3390/healthcare8020097
Fesler MC, Shah JS, Middelveen MJ, Du Cruz I, Burrascano JJ, Stricker RB. Lyme Disease: Diversity of Borrelia Species in California and Mexico Detected Using a Novel Immunoblot Assay. Healthcare. 2020; 8(2):97. https://doi.org/10.3390/healthcare8020097
Chicago/Turabian StyleFesler, Melissa C., Jyotsna S. Shah, Marianne J. Middelveen, Iris Du Cruz, Joseph J. Burrascano, and Raphael B. Stricker. 2020. "Lyme Disease: Diversity of Borrelia Species in California and Mexico Detected Using a Novel Immunoblot Assay" Healthcare 8, no. 2: 97. https://doi.org/10.3390/healthcare8020097
APA StyleFesler, M. C., Shah, J. S., Middelveen, M. J., Du Cruz, I., Burrascano, J. J., & Stricker, R. B. (2020). Lyme Disease: Diversity of Borrelia Species in California and Mexico Detected Using a Novel Immunoblot Assay. Healthcare, 8(2), 97. https://doi.org/10.3390/healthcare8020097






