Pancreatic Cancer Cachexia: The Role of Nutritional Interventions
Abstract
1. Introduction
2. Nutritional Interventions to Support Pancreatic Cancer Cachexia Management
2.1. Calorie Intake
2.2. Oral Nutritional Supplementation and Parenteral Nutrition
2.3. Ketogenic Diet
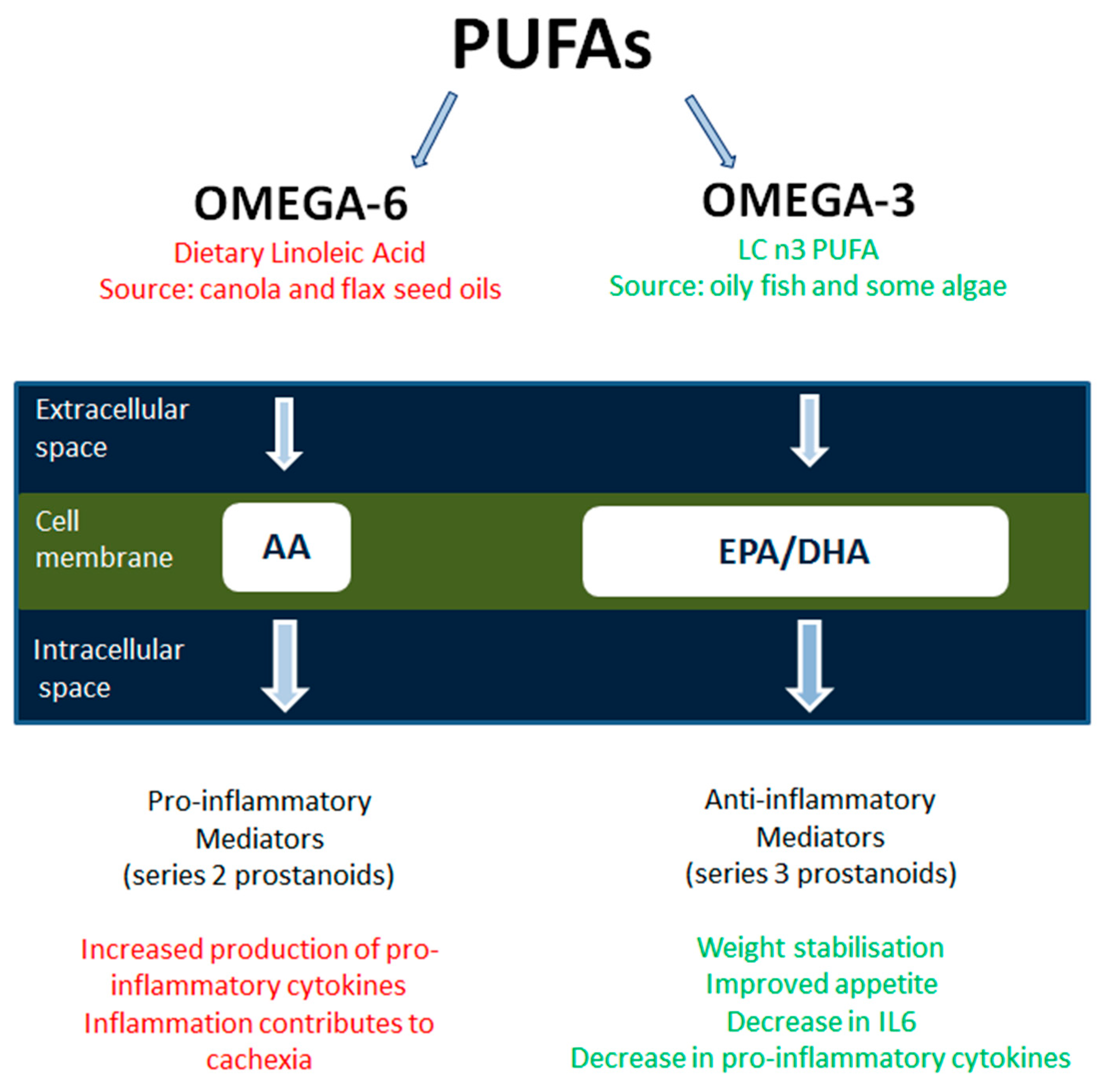
2.4. Fish Oil
2.5. Amino Acids
2.6. Branch Chain Amino Acids
2.7. L-Carnitine
2.8. Pancreatic Enzyme Replacement
3. Limitations
- be sufficiently powered to accommodate a large dropout rate;
- multi-centred to ensure sufficient enrolment over a relatively short time and to ensure results are generalisable across populations;
- provide plenty of support for study participants to minimise drop out;
- include single and complex interventions;
- and finally, utilise several different tools to strengthen the data on dietary intake and adherence to the intervention.
4. Conclusions
Funding
Conflicts of Interest
References
- Fearon, K.C.; Voss, A.C.; Hustead, D.S. Definition of cancer cachexia: Effect of weight loss, reduced food intake, and systemic inflammation on functional status and prognosis. Am. J. Clin. Nutr. 2006, 83, 1345–1350. [Google Scholar] [CrossRef] [PubMed]
- Fearon, K.C.; Glass, D.J.; Guttridge, D.C. Cancer Cachexia: Mediators, Signaling, and Metabolic Pathways. Cell Metab. 2012, 16, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Bachmann, J.; Ketterer, K.; Marsch, C.; Fechtner, K.; Krakowski-Roosen, H.; Buchler, M.W.; Friess, H.; Martignoni, M.E. Pancreatic cancer related cachexia: Influence on metabolism and correlation to weight loss and pulmonary function. BMC Cancer 2009, 9, 255. [Google Scholar] [CrossRef] [PubMed]
- The American Cancer Society. Pancreatic Cancer Risk Factors. 2016. Available online: https://www.cancer.org/cancer/pancreatic-cancer/causes-risks-prevention/risk-factors.html (accessed on 3 December 2018).
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics, 2016. CA Cancer J. Clin. 2016, 66, 271–289. [Google Scholar] [CrossRef] [PubMed]
- Cancer Research UK. Pancreatic Cancer Survival Statistics. Available online: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/pancreatic-cancer/survival (accessed on 3 December 2018).
- Vanhoutte, G.; van de Wiel, M.; Wouters, K.; Sels, M.; Bartolomeeussen, L.; De Keersmaecker, S.; Verschueren, C.; De Vroey, V.; De Wilde, A.; Smits, E.; et al. Cachexia in cancer: What is in the definition? BMJ Open Gastroenterol. 2016, 3, e000097. [Google Scholar] [CrossRef] [PubMed]
- Fearon, K.; Strasser, F.; Anker, S.D.; Bosaeus, I.; Bruera, E.; Fainsinger, R.L.; Jatoi, A.; Loprinzi, C.; MacDonald, N.; Mantovani, G.; et al. Definition and classification of cancer cachexia: An international consensus. Lancet Oncol. 2011, 12, 489–495. [Google Scholar] [CrossRef]
- Evans, W.J.; Morley, J.E.; Argilés, J.; Bales, C.; Baracos, V.; Guttridge, D.; Jatoi, A.; Kalantar-Zadeh, K.; Lochs, H.; Mantovani, G.; et al. Cachexia: A new definition. Clin. Nutr. 2008, 27, 793–799. [Google Scholar]
- Mueller, T.C.; Burmeister, M.A.; Bachmann, J.; Martignoni, M.E. Cachexia and pancreatic cancer: Are there treatment options? World J. Gastroenterol. 2014, 20, 9361–9373. [Google Scholar] [PubMed]
- Patel, S.; Alvarez-Guaita, A.; Melvin, A.; Rimmington, D.; Dattilo, A.; Miedzybrodzka, E.L.; Cimino, I.; Maurin, A.C.; Roberts, G.P.; Meek, C.L.; et al. GDF15 Provides an Endocrine Signal of Nutritional Stress in Mice and Humans. Cell Metab. 2019, 29, 707–718. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, B.; Tucker, S.L.; Li, D.; Abbruzzese, J.L.; Kurzrock, R. Cytokines in pancreatic carcinoma. Cancer 2004, 101, 2727–2736. [Google Scholar] [CrossRef] [PubMed]
- Guttridge, D.C. A TGF-beta pathway associated with cancer cachexia. Nat. Med. 2015, 21, 1248–1249. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Penafuerte, C.A.; Gagnon, B.; Sirois, J.; Murphy, J.; MacDonald, N.; Tremblay, M.L. Identification of neutrophil-derived proteases and angiotensin II as biomarkers of cancer cachexia. Br. J. Cancer 2016, 114, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Lerner, L.; Hayes, T.G.; Tao, N.; Krieger, B.; Feng, B.; Wu, Z.; Nicoletti, R.; Chiu, M.I.; Gyuris, J.; Garcia, J.M. Plasma growth differentiation factor 15 is associated with weight loss and mortality in cancer patients. J. Cachexia Sarcopenia Muscle 2015, 6, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Dev, R.; Wong, A.; Hui, D.; Bruera, E. The Evolving Approach to Management of Cancer Cachexia. Oncology 2017, 31, 23–32. [Google Scholar] [PubMed]
- Tuca, A.; Jimenez-Fonseca, P.; Gascon, P. Clinical evaluation and optimal management of cancer cachexia. Crit. Rev. Oncol. Hematol. 2013, 88, 625–636. [Google Scholar] [CrossRef]
- Jackman, R.W.; Floro, J.; Yoshimine, R.; Zitin, B.; Eiampikul, M.; El-Jack, K.; Seto, D.N.; Kandarian, S.C. Continuous Release of Tumor-Derived Factors Improves the Modeling of Cachexia in Muscle Cell Culture. Front. Physiol. 2017, 8, 738. [Google Scholar] [CrossRef]
- Russell, S.T.; Tisdale, M.J. Effect of a tumour-derived lipid-mobilising factor on glucose and lipid metabolism in vivo. Br. J. Cancer 2002, 87, 580–584. [Google Scholar] [CrossRef]
- Todorov, P.T.; Deacon, M.; Tisdale, M.J. Structural analysis of a tumor-produced sulfated glycoprotein capable of initiating muscle protein degradation. J. Biol. Chem. 1997, 272, 12279–12288. [Google Scholar] [CrossRef]
- Cariuk, P.; Lorite, M.J.; Todorov, P.T.; Field, W.N.; Wigmore, S.J.; Tisdale, M.J. Induction of cachexia in mice by a product isolated from the urine of cachectic cancer patients. Br. J. Cancer 1997, 76, 606–613. [Google Scholar] [CrossRef]
- Lorite, M.J.; Cariuk, P.; Tisdale, M.J. Induction of muscle protein degradation by a tumour factor. Br. J. Cancer 1997, 76, 1035–1040. [Google Scholar] [CrossRef]
- Deutsch, J.; Kolhouse, J.F. Assessment of gastrointestinal function and response to megesterol acetate in subjects with gastrointestinal cancers and weight loss. Support. Care Cancer 2004, 12, 503–510. [Google Scholar] [CrossRef]
- Aoyagi, T.; Terracina, K.P.; Raza, A.; Matsubara, H.; Takabe, K. Cancer cachexia, mechanism and treatment. World J. Gastrointest Oncol. 2015, 7, 17–29. [Google Scholar] [CrossRef]
- Warnold, I.; Lundholm, K.; Schersten, T. Energy balance and body composition in cancer patients. Cancer Res. 1978, 38, 1801–1807. [Google Scholar] [PubMed]
- Dempsey, D.T.; Mullen, J.L. Macronutrient requirements in the malnourished cancer patient. How much of what and why? Cancer 1985, 55, 290–294. [Google Scholar] [CrossRef]
- Baracos, V.E.; Martin, L.; Korc, M.; Guttridge, D.C.; Fearon, K.C.H. Cancer-associated cachexia. Nat. Rev. Dis. Primers 2018, 4, 17105. [Google Scholar] [CrossRef] [PubMed]
- DeWys, W.D. Pathophysiology of cancer cachexia: Current understanding and areas for future research. Cancer Res. 1982, 42, 721s–726s. [Google Scholar] [PubMed]
- Bye, A.; Jordhoy, M.S.; Skjegstad, G.; Ledsaak, O.; Iversen, P.O.; Hjermstad, M.J. Symptoms in advanced pancreatic cancer are of importance for energy intake. Support. Care Cancer 2013, 21, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Okusaka, T.; Okada, S.; Ishii, H.; Ikeda, M.; Kosakamoto, H.; Yoshimori, M. Prognosis of advanced pancreatic cancer patients with reference to calorie intake. Nutr. Cancer 1998, 32, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Samra, R. Chapter 15: Fats and Satiety. In Fat Detection: Taste, Texture, and Post Ingestive Effects; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2010. Available online: https://www.ncbi.nlm.nih.gov/books/NBK53550/ (accessed on 11 January 2019).
- Nasrah, R.; Kanbalian, M.; Van Der Borch, C.; Swinton, N.; Wing, S.; Jagoe, R.T. Defining the role of dietary intake in determining weight change in patients with cancer cachexia. Clin. Nutr. 2018, 37, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Davidson, W.; Ash, S.; Capra, S.; Bauer, J.; Cancer Cachexia Study Group. Weight stabilisation is associated with improved survival duration and quality of life in unresectable pancreatic cancer. Clin. Nutr. 2004, 23, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Solheim, T.S.; Laird, B.J.A.; Balstad, T.R.; Stene, G.B.; Bye, A.; Johns, N.; Pettersen, C.H.; Fallon, M.; Fayers, P.; Fearon, K.; et al. A randomized phase II feasibility trial of a multimodal intervention for the management of cachexia in lung and pancreatic cancer. J. Cachexia Sarcopenia Muscle 2017, 8, 778–788. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Capra, S.; Battistutta, D.; Davidson, W.; Ash, S.; Cancer Cachexia Study Group. Compliance with nutrition prescription improves outcomes in patients with unresectable pancreatic cancer. Clin. Nutr. 2005, 24, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Simpson, F.; Doig, G.S. Parenteral vs. enteral nutrition in the critically ill patient: A meta-analysis of trials using the intention to treat principle. Intensive Care Med. 2005, 31, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Bozzetti, F.; Forbes, A. The ESPEN clinical practice Guidelines on Parenteral Nutrition: Present status and perspectives for future research. Clin. Nutr. 2009, 28, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Pelzer, U.; Arnold, D.; Govercin, M.; Stieler, J.; Doerken, B.; Riess, H.; Oettle, H. Parenteral nutrition support for patients with pancreatic cancer. Results of a phase II study. BMC Cancer 2010, 10, 86. [Google Scholar] [CrossRef] [PubMed]
- Cotogni, P.; De Carli, L.; Passera, R.; Amerio, M.L.; Agnello, E.; Fadda, M.; Ossola, M.; Monge, T.; De Francesco, A.; Bozzetti, F. Longitudinal study of quality of life in advanced cancer patients on home parenteral nutrition. Cancer Med. 2017, 6, 1799–1806. [Google Scholar] [CrossRef] [PubMed]
- Kruger, J.; Meffert, P.J.; Vogt, L.J.; Gartner, S.; Steveling, A.; Kraft, M.; Mayerle, J.; Lerch, M.M.; Aghdassi, A.A. Early Parenteral Nutrition in Patients with Biliopancreatic Mass Lesions, a Prospective, Randomized Intervention Trial. PLoS ONE 2016, 11, e0166513. [Google Scholar] [CrossRef]
- Wigmore, S.J.; Ross, J.A.; Falconer, J.S.; Plester, C.E.; Tisdale, M.J.; Carter, D.C.; Fearon, K.C. The effect of polyunsaturated fatty acids on the progress of cachexia in patients with pancreatic cancer. Nutrition 1996, 12, S27–S30. [Google Scholar] [CrossRef]
- Barber, M.D.; Ross, J.A.; Voss, A.C.; Tisdale, M.J.; Fearon, K.C. The effect of an oral nutritional supplement enriched with fish oil on weight-loss in patients with pancreatic cancer. Br. J. Cancer 1999, 81, 80–86. [Google Scholar] [CrossRef]
- Wigmore, S.J.; Barber, M.D.; Ross, J.A.; Tisdale, M.J.; Fearon, K.C. Effect of oral eicosapentaenoic acid on weight loss in patients with pancreatic cancer. Nutr. Cancer 2000, 36, 177–184. [Google Scholar] [CrossRef]
- Barber, M.D.; Fearon, K.C.; Tisdale, M.J.; McMillan, D.C.; Ross, J.A. Effect of a fish oil-enriched nutritional supplement on metabolic mediators in patients with pancreatic cancer cachexia. Nutr. Cancer 2001, 40, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Bruera, E.; Strasser, F.; Palmer, J.L.; Willey, J.; Calder, K.; Amyotte, G.; Baracos, V. Effect of fish oil on appetite and other symptoms in patients with advanced cancer and anorexia/cachexia: A double-blind, placebo-controlled study. J. Clin. Oncol. 2003, 21, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Fearon, K.C.; Von Meyenfeldt, M.F.; Moses, A.G.; Van Geenen, R.; Roy, A.; Gouma, D.J.; Giacosa, A.; Van Gossum, A.; Bauer, J.; Barber, M.D.; et al. Effect of a protein and energy dense N-3 fatty acid enriched oral supplement on loss of weight and lean tissue in cancer cachexia: A randomised double blind trial. Gut 2003, 52, 1479–1486. [Google Scholar] [CrossRef] [PubMed]
- Werner, K.; Kullenberg de Gaudry, D.; Taylor, L.A.; Keck, T.; Unger, C.; Hopt, U.T.; Massing, U. Dietary supplementation with n-3-fatty acids in patients with pancreatic cancer and cachexia: Marine phospholipids versus fish oil—A randomized controlled double-blind trial. Lipids Health Dis. 2017, 16, 104. [Google Scholar] [CrossRef] [PubMed]
- Abe, K.; Uwagawa, T.; Haruki, K.; Takano, Y.; Onda, S.; Sakamoto, T.; Gocho, T.; Yanaga, K. Effects of omega-3 Fatty Acid Supplementation in Patients with Bile Duct or Pancreatic Cancer Undergoing Chemotherapy. Anticancer Res. 2018, 38, 2369–2375. [Google Scholar]
- Nakamura, K.; Tonouchi, H.; Sasayama, A.; Ashida, K. A Ketogenic Formula Prevents Tumor Progression and Cancer Cachexia by Attenuating Systemic Inflammation in Colon 26 Tumor-Bearing Mice. Nutrients 2018, 10, 206. [Google Scholar] [CrossRef]
- Shukla, S.K.; Gebregiworgis, T.; Purohit, V.; Chaika, N.V.; Gunda, V.; Radhakrishnan, P.; Mehla, K.; Pipinos, I.I.; Powers, R.; Yu, F.; et al. Metabolic reprogramming induced by ketone bodies diminishes pancreatic cancer cachexia. Cancer Metab. 2014, 2, 18. [Google Scholar] [CrossRef]
- Chung, H.Y.; Park, Y.K. Rationale, Feasibility and Acceptability of Ketogenic Diet for Cancer Treatment. J. Cancer Prev. 2017, 22, 127–134. [Google Scholar] [CrossRef]
- Klement, R.J.; Kammerer, U. Is there a role for carbohydrate restriction in the treatment and prevention of cancer? Nutr. Metab. 2011, 8, 75. [Google Scholar] [CrossRef]
- Schmidt, M.; Pfetzer, N.; Schwab, M.; Strauss, I.; Kammerer, U. Effects of a ketogenic diet on the quality of life in 16 patients with advanced cancer: A pilot trial. Nutr. Metab. 2011, 8, 54. [Google Scholar] [CrossRef]
- Poff, A.M.; Ari, C.; Seyfried, T.N.; D’Agostino, D.P. The ketogenic diet and hyperbaric oxygen therapy prolong survival in mice with systemic metastatic cancer. PLoS ONE 2013, 8, e65522. [Google Scholar] [CrossRef] [PubMed]
- Poff, A.M.; Ward, N.; Seyfried, T.N.; Arnold, P.; D’Agostino, D.P. Non-Toxic Metabolic Management of Metastatic Cancer in VM Mice: Novel Combination of Ketogenic Diet, Ketone Supplementation, and Hyperbaric Oxygen Therapy. PLoS ONE 2015, 10, e0127407. [Google Scholar] [CrossRef] [PubMed]
- Otto, C.; Kaemmerer, U.; Illert, B.; Muehling, B.; Pfetzer, N.; Wittig, R.; Voelker, H.U.; Thiede, A.; Coy, J.F. Growth of human gastric cancer cells in nude mice is delayed by a ketogenic diet supplemented with omega-3 fatty acids and medium-chain triglycerides. BMC Cancer 2008, 8, 122. [Google Scholar] [CrossRef] [PubMed]
- Fine, E.J.; Segal-Isaacson, C.J.; Feinman, R.D.; Herszkopf, S.; Romano, M.C.; Tomuta, N.; Bontempo, A.F.; Negassa, A.; Sparano, J.A. Targeting insulin inhibition as a metabolic therapy in advanced cancer: A pilot safety and feasibility dietary trial in 10 patients. Nutrition 2012, 28, 1028–1035. [Google Scholar] [CrossRef] [PubMed]
- Champ, C.E.; Palmer, J.D.; Volek, J.S.; Werner-Wasik, M.; Andrews, D.W.; Evans, J.J.; Glass, J.; Kim, L.; Shi, W. Targeting metabolism with a ketogenic diet during the treatment of glioblastoma multiforme. J. Neurooncol. 2014, 117, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Klement, R.J.; Sweeney, R.A. Impact of a ketogenic diet intervention during radiotherapy on body composition: I. Initial clinical experience with six prospectively studied patients. BMC Res. Notes 2016, 9, 143. [Google Scholar] [CrossRef] [PubMed]
- Rieger, J.; Bahr, O.; Maurer, G.D.; Hattingen, E.; Franz, K.; Brucker, D.; Walenta, S.; Kammerer, U.; Coy, J.F.; Weller, M.; et al. ERGO: A pilot study of ketogenic diet in recurrent glioblastoma. Int. J. Oncol. 2014, 44, 1843–1852. [Google Scholar] [CrossRef]
- Breitkreutz, R.; Tesdal, K.; Jentschura, D.; Haas, O.; Leweling, H.; Holm, E. Effects of a high-fat diet on body composition in cancer patients receiving chemotherapy: A randomized controlled study. Wien. Klin. Wochenschr. 2005, 117, 685–692. [Google Scholar] [CrossRef]
- Bosaeus, I. Nutritional support in multimodal therapy for cancer cachexia. Support. Care Cancer 2008, 16, 447–451. [Google Scholar] [CrossRef]
- Barber, M.D.; Fearon, K.C.; Ross, J.A. Relationship of serum levels of interleukin-6, soluble interleukin-6 receptor and tumour necrosis factor receptors to the acute-phase protein response in advanced pancreatic cancer. Clin. Sci. 1999, 96, 83–87. [Google Scholar] [CrossRef]
- Hooper, L.; Thompson, R.L.; Harrison, R.A.; Summerbell, C.D.; Ness, A.R.; Moore, H.J.; Worthington, H.V.; Durrington, P.N.; Higgins, J.P.; Capps, N.E.; et al. Risks and benefits of omega 3 fats for mortality, cardiovascular disease, and cancer: Systematic review. BMJ 2006, 332, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Beck, S.A.; Smith, K.L.; Tisdale, M.J. Anticachectic and antitumor effect of eicosapentaenoic acid and its effect on protein turnover. Cancer Res. 1991, 51, 6089–6093. [Google Scholar] [PubMed]
- Calder, P.C.; Davis, J.; Yaqoob, P.; Pala, H.; Thies, F.; Newsholme, E.A. Dietary fish oil suppresses human colon tumour growth in athymic mice. Clin. Sci. 1998, 94, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Endres, S.; Ghorbani, R.; Kelley, V.E.; Georgilis, K.; Lonnemann, G.; van der Meer, J.W.; Cannon, J.G.; Rogers, T.S.; Klempner, M.S.; Weber, P.C.; et al. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N. Engl. J. Med. 1989, 320, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Colomer, R.; Moreno-Nogueira, J.M.; Garcia-Luna, P.P.; Garcia-Peris, P.; Garcia-de-Lorenzo, A.; Zarazaga, A.; Quecedo, L.; del Llano, J.; Usan, L.; Casimiro, C. N-3 fatty acids, cancer and cachexia: A systematic review of the literature. Br. J. Nutr. 2007, 97, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Candela, C.; Villarino Sanz, M.; Horrisberger, A.; Loria Kohen, V.; Bermejo, L.M.; Zamora Aunon, P. Efficacy evaluation of an oral powder supplement enriched with eicosapentaenoic acid in cancer patients. Nutr. Hosp. 2011, 26, 1385–1393. [Google Scholar] [PubMed]
- van der Meij, B.S.; Langius, J.A.; Smit, E.F.; Spreeuwenberg, M.D.; von Blomberg, B.M.; Heijboer, A.C.; Paul, M.A.; van Leeuwen, P.A. Oral nutritional supplements containing (n-3) polyunsaturated fatty acids affect the nutritional status of patients with stage III non-small cell lung cancer during multimodality treatment. J. Nutr. 2010, 140, 1774–1780. [Google Scholar] [CrossRef] [PubMed]
- Burns, C.P.; Halabi, S.; Clamon, G.; Kaplan, E.; Hohl, R.J.; Atkins, J.N.; Schwartz, M.A.; Wagner, B.A.; Paskett, E. Phase II study of high-dose fish oil capsules for patients with cancer-related cachexia. Cancer 2004, 101, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Burns, C.P.; Halabi, S.; Clamon, G.H.; Hars, V.; Wagner, B.A.; Hohl, R.J.; Lester, E.; Kirshner, J.J.; Vinciguerra, V.; Paskett, E. Phase I clinical study of fish oil fatty acid capsules for patients with cancer cachexia: Cancer and leukemia group B study 9473. Clin. Cancer Res. 1999, 5, 3942–3947. [Google Scholar] [PubMed]
- Burri, L.; Hoem, N.; Banni, S.; Berge, K. Marine omega-3 phospholipids: Metabolism and biological activities. Int. J. Mol. Sci. 2012, 13, 15401–15419. [Google Scholar] [CrossRef]
- Ramprasath, V.R.; Eyal, I.; Zchut, S.; Jones, P.J. Enhanced increase of omega-3 index in healthy individuals with response to 4-week n-3 fatty acid supplementation from krill oil versus fish oil. Lipids Health Dis. 2013, 12, 178. [Google Scholar] [CrossRef] [PubMed]
- Ulven, S.M.; Kirkhus, B.; Lamglait, A.; Basu, S.; Elind, E.; Haider, T.; Berge, K.; Vik, H.; Pedersen, J.I. Metabolic effects of krill oil are essentially similar to those of fish oil but at lower dose of EPA and DHA, in healthy volunteers. Lipids 2011, 46, 37–46. [Google Scholar] [CrossRef]
- Taylor, L.A.; Pletschen, L.; Arends, J.; Unger, C.; Massing, U. Marine phospholipids—A promising new dietary approach to tumor-associated weight loss. Support. Care Cancer 2010, 18, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Lenihan-Geels, G.; Bishop, K.S.; Ferguson, L.R. Alternative sources of omega-3 fats: Can we find a sustainable substitute for fish? Nutrients 2013, 5, 1301–1315. [Google Scholar] [CrossRef] [PubMed]
- Eley, H.L.; Russell, S.T.; Baxter, J.H.; Mukerji, P.; Tisdale, M.J. Signaling pathways initiated by beta-hydroxy-beta-methylbutyrate to attenuate the depression of protein synthesis in skeletal muscle in response to cachectic stimuli. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E923–E931. [Google Scholar] [CrossRef]
- Eley, H.L.; Russell, S.T.; Tisdale, M.J. Attenuation of depression of muscle protein synthesis induced by lipopolysaccharide, tumor necrosis factor, and angiotensin II by beta-hydroxy-beta-methylbutyrate. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1409–E1416. [Google Scholar] [CrossRef] [PubMed]
- Eley, H.L.; Russell, S.T.; Tisdale, M.J. Mechanism of attenuation of muscle protein degradation induced by tumor necrosis factor-alpha and angiotensin II by beta-hydroxy-beta-methylbutyrate. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E1417–E1426. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.J.; Wyke, S.M.; Tisdale, M.J. Mechanism of the attenuation of proteolysis-inducing factor stimulated protein degradation in muscle by beta-hydroxy-beta-methylbutyrate. Cancer Res. 2004, 64, 8731–8735. [Google Scholar] [CrossRef] [PubMed]
- Aversa, Z.; Bonetto, A.; Costelli, P.; Minero, V.G.; Penna, F.; Baccino, F.M.; Lucia, S.; Rossi Fanelli, F.; Muscaritoli, M. beta-hydroxy-beta-methylbutyrate (HMB) attenuates muscle and body weight loss in experimental cancer cachexia. Int. J. Oncol. 2011, 38, 713–720. [Google Scholar]
- Smith, H.J.; Mukerji, P.; Tisdale, M.J. Attenuation of proteasome-induced proteolysis in skeletal muscle by {beta}-hydroxy-{beta}-methylbutyrate in cancer-induced muscle loss. Cancer Res. 2005, 65, 277–283. [Google Scholar] [PubMed]
- Nunes, E.A.; Kuczera, D.; Brito, G.A.; Bonatto, S.J.; Yamazaki, R.K.; Tanhoffer, R.A.; Mund, R.C.; Kryczyk, M.; Fernandes, L.C. Beta-hydroxy-beta-methylbutyrate supplementation reduces tumor growth and tumor cell proliferation ex vivo and prevents cachexia in Walker 256 tumor-bearing rats by modifying nuclear factor-kappaB expression. Nutr. Res. 2008, 28, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Caperuto, E.C.; Tomatieli, R.V.; Colquhoun, A.; Seelaender, M.C.; Costa Rosa, L.F. Beta-hydoxy-beta-methylbutyrate supplementation affects Walker 256 tumor-bearing rats in a time-dependent manner. Clin. Nutr. 2007, 26, 117–122. [Google Scholar] [CrossRef] [PubMed]
- May, P.E.; Barber, A.; D’Olimpio, J.T.; Hourihane, A.; Abumrad, N.N. Reversal of cancer-related wasting using oral supplementation with a combination of beta-hydroxy-beta-methylbutyrate, arginine, and glutamine. Am. J. Surg. 2002, 183, 471–479. [Google Scholar] [CrossRef]
- Rathmacher, J.A.; Nissen, S.; Panton, L.; Clark, R.H.; Eubanks May, P.; Barber, A.E.; D’Olimpio, J.; Abumrad, N.N. Supplementation with a combination of beta-hydroxy-beta-methylbutyrate (HMB), arginine, and glutamine is safe and could improve hematological parameters. J. Parenter. Enter. Nutr. 2004, 28, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Berk, L.; James, J.; Schwartz, A.; Hug, E.; Mahadevan, A.; Samuels, M.; Kachnic, L. A randomized, double-blind, placebo-controlled trial of a beta-hydroxyl beta-methyl butyrate, glutamine, and arginine mixture for the treatment of cancer cachexia (RTOG 0122). Support. Care Cancer 2008, 16, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Eley, H.L.; Russell, S.T.; Tisdale, M.J. Effect of branched-chain amino acids on muscle atrophy in cancer cachexia. Biochem. J. 2007, 407, 113–120. [Google Scholar] [CrossRef]
- Peters, S.J.; van Helvoort, A.; Kegler, D.; Argiles, J.M.; Luiking, Y.C.; Laviano, A.; van Bergenhenegouwen, J.; Deutz, N.E.; Haagsman, H.P.; Gorselink, M.; et al. Dose-dependent effects of leucine supplementation on preservation of muscle mass in cancer cachectic mice. Oncol. Rep. 2011, 26, 247–254. [Google Scholar]
- Liu, K.A.; Lashinger, L.M.; Rasmussen, A.J.; Hursting, S.D. Leucine supplementation differentially enhances pancreatic cancer growth in lean and overweight mice. Cancer Metab. 2014, 2, 6. [Google Scholar] [CrossRef]
- Hunter, D.C.; Weintraub, M.; Blackburn, G.L.; Bistrian, B.R. Branched chain amino acids as the protein component of parenteral nutrition in cancer cachexia. Br. J. Surg. 1989, 76, 149–153. [Google Scholar] [CrossRef]
- Cruciani, R.A.; Dvorkin, E.; Homel, P.; Malamud, S.; Culliney, B.; Lapin, J.; Portenoy, R.K.; Esteban-Cruciani, N. Safety, tolerability and symptom outcomes associated with L-carnitine supplementation in patients with cancer, fatigue, and carnitine deficiency: A phase I/II study. J. Pain Symptom Manag. 2006, 32, 551–559. [Google Scholar] [CrossRef]
- Szefel, J.; Kruszewski, W.J.; Ciesielski, M.; Szajewski, M.; Kawecki, K.; Aleksandrowicz-Wrona, E.; Jankun, J.; Lysiak-Szydlowska, W. L-carnitine and cancer cachexia. I. L-carnitine distribution and metabolic disorders in cancer cachexia. Oncol. Rep. 2012, 28, 319–323. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Silverio, R.; Laviano, A.; Rossi Fanelli, F.; Seelaender, M. l-carnitine and cancer cachexia: Clinical and experimental aspects. J. Cachexia Sarcopenia Muscle 2011, 2, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Cruciani, R.A.; Dvorkin, E.; Homel, P.; Culliney, B.; Malamud, S.; Lapin, J.; Portenoy, R.K.; Esteban-Cruciani, N. L-carnitine supplementation in patients with advanced cancer and carnitine deficiency: A double-blind, placebo-controlled study. J. Pain Symptom Manag. 2009, 37, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Cruciani, R.A.; Dvorkin, E.; Homel, P.; Culliney, B.; Malamud, S.; Shaiova, L.; Fleishman, S.; Lapin, J.; Klein, E.; Lesage, P.; et al. L-carnitine supplementation for the treatment of fatigue and depressed mood in cancer patients with carnitine deficiency: A preliminary analysis. Ann. N. Y. Acad. Sci. 2004, 1033, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Gramignano, G.; Lusso, M.R.; Madeddu, C.; Massa, E.; Serpe, R.; Deiana, L.; Lamonica, G.; Dessi, M.; Spiga, C.; Astara, G.; et al. Efficacy of l-carnitine administration on fatigue, nutritional status, oxidative stress, and related quality of life in 12 advanced cancer patients undergoing anticancer therapy. Nutrition 2006, 22, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Kraft, M.; Kraft, K.; Gartner, S.; Mayerle, J.; Simon, P.; Weber, E.; Schutte, K.; Stieler, J.; Koula-Jenik, H.; Holzhauer, P.; et al. L-Carnitine-supplementation in advanced pancreatic cancer (CARPAN)—A randomized multicentre trial. Nutr. J. 2012, 11, 52. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, G.; Maccio, A.; Madeddu, C.; Serpe, R.; Massa, E.; Dessi, M.; Panzone, F.; Contu, P. Randomized phase III clinical trial of five different arms of treatment in 332 patients with cancer cachexia. Oncologist 2010, 15, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Cruciani, R.A.; Zhang, J.J.; Manola, J.; Cella, D.; Ansari, B.; Fisch, M.J. L-carnitine supplementation for the management of fatigue in patients with cancer: An eastern cooperative oncology group phase III, randomized, double-blind, placebo-controlled trial. J. Clin. Oncol. 2012, 30, 3864–3869. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Munoz, J.E. Pancreatic exocrine insufficiency: Diagnosis and treatment. J. Gastroenterol. Hepatol. 2011, 26 (Suppl. 2), 12–16. [Google Scholar] [CrossRef]
- Saito, T.; Hirano, K.; Isayama, H.; Nakai, Y.; Saito, K.; Umefune, G.; Akiyama, D.; Watanabe, T.; Takagi, K.; Hamada, T.; et al. The Role of Pancreatic Enzyme Replacement Therapy in Unresectable Pancreatic Cancer: A Prospective Cohort Study. Pancreas 2017, 46, 341–346. [Google Scholar]
| Criteria | Time | Citation |
|---|---|---|
| Involuntary weight loss >5% | 6 months | [8] |
| BMI < 20 kg/m2 and any degree of involuntary weight loss > 2% | Not stated | [8] |
| Sarcopenia with weight loss > 2% (An appendicular skeletal muscle mass index > 7.26 kg/m2 and > 5.45 kg/m2 in males and females respectively, as defined by DEXA) | Not stated | [8] |
| Weight loss ≥ 5% in the presence of underlying illness, and three of the following criteria: decreased muscle strength, fatigue, anorexia, low fat-free mass index or abnormal biochemistry (CRP, IL6, anaemia, serum albumin) | ≤12 months | [9] reported in [7]. |
| Refs | Study Design | Cancer Type | Nutritional Status | Comparison Details | Duration | Summary of Results | Adverse Effects |
|---|---|---|---|---|---|---|---|
| [41] | Phase II study | 18 Pancreatic cancer patients | Progressive weight loss | 1 g soft gel capsule; Initial dose 2 g/day, increasing to 16 g/day FO | Patients tolerated 12 g/day FO, equivalent 2 g EPA. Weight +0.3 kg (0 to 0.5) a/month compared to −2.9 kg (2 to 4.6) a/month pre-study (p < 0.002) | Offensive tasting reuctations, transient diarrhoea | |
| [42] | Intention to treat | 20 Pancreatic cancer patients | Unresectable adenocarcinoma of the pancreas | ONS (2 cans) providing 610 kcal, 32.2 g protein, 2.2 g EPA, 0.96 g DHA | 7 weeks (n = 13) | Consumed 1.9 cans/day (1.2 to 2);Weight +2.0 kg (−0.4 to 4.6) a (p = 0.033); LBM + 1.9 kg (1.0 to 3.0) a (p = 0.0047); KPS increased 10 (0 to 10) a (p = 0.046) | Steatorrhoea (3)—treated with pancreatic enzyme supplementation |
| [43] | Prospective interventional study | 23 unresectable pancreatic cancer; 3 unresectable ampullary cancers | WHO PS-2, 13% weight loss in 4 months, BMI 23.2 (21.1–27.4)% a | EPA capsule 500 mg Week 1:1 g; Week 2:2 g Week 3:4 g; Weeks 4–12:6 g | 12 weeks | Weight: Pre-study −2 kg (n = 26), week 12 +0.3 kg (n = 14 (p < 0.005 vs. week 0) | Nausea Steatorrhea |
| [44] | 20 Pancreatic cancer patients | Unresectable adenocarcinoma of the pancreas with evidence of cachexia | ONS (2 cans) providing 610 kcal, 32.2 g protein, 2.2 g EPA, 0.96 g DHA | 3 weeks | Consumed 1.9 cans/day (1.25 to 2); Weight +1.0 kg (−0.1 to 2.0) a (p = 0.024); Significant fall in IL6 (p = 0.015); Detected PIF excretion fell from 88% to 40% of patients (p = 0.008) | ||
| [45] | Double blind placebo-controlled study | 60 advanced cancer patients | Advanced cancer, anorexia; weight loss >5% pre-illness body weight | A daily dose of 18 gelatine capsules containing: 1000 mg fish oil (180 mg EPA, 120 mg DHA, and 1 mg vitamin E) or 1,000 mg olive oil. | 2 weeks | Ability to tolerate 18 capsules was limited (9.8 ± 4 b FO, 9.2 ± 3 b PC) A strong trend towards appetite improvement in both groups | Vomiting, belching, nausea, diarrhoea, hematemesis, abdominal pain |
| [46] | Double-blind randomised multicentre trial | 200 unresectable pancreatic cancer patients T n = 95; PC n = 105 | >5% of their pre-illness weight over the previous six months, KPS ˃ 60, life expectancy > 2 months. | ONS two cans/day of 480 mL, 620 kcal, 32 g protein and 2.2 g EPA; Control-identical supplement without ω-3 FA and antioxidants | 8 weeks | Intake averaged 1.4 cans/day in both groups; No net gain of LBM. Significant intervention weight loss −0.25 kg/mth (n = 88), control weight loss −0.37 kg/mth (n = 97) | |
| [47] | Randomised controlled double-blind trial | 60 pancreatic cancer patients N = 31 (FO) N = 29 (MPL) | Weight loss ˃ 5% KPS ˃60% | Capsule either FO or MPL 1 capsule 3 times per day with meals, Equating to ω-3 300 mg/day via MPL or FO (FO capsule included 200 mg Medium chain triglycerides to balance ω-3 FA versus MPL) | 6 weeks | Appetite stabilised–↑ meal portions (FO p = 0.02, MPL p = 0.05); Weight stabilisation vs. pre-study (FO p = 0.001, MPL = 0.003); 50% FO group and 47% of MPL group gained weight; DHA and EPA increased significantly in the blood | FO group-pyrosis, “fishy” regurgitation, loss of appetite and diarrhoea; MPL group-diarrhoea |
| [48] | Pancreatic (n = 19) and bile duct (n = 8) cancer patients | Patients who underwent chemotherapy Nov 2014 to Nov 2016 | Enteral 2–4 packs (200 kcal and 300 mg ω-3 per pack) + pancreatic enzymes weeks 4 to 8 | Baseline, 4–8 weeks | Average 2.42 ± 0.4 b packs consumed per day; ↑ trend for weight (NS), ↑ skeletal muscle mass at week 8; EPA/AA ratio improved week 8; (pancreatic enzymes aided absorption of ω-3FA). |
| Refs | Study Design | Characteristics | Health Status | Details | Duration | Summary of Results | Adverse Effects |
|---|---|---|---|---|---|---|---|
| [97] | Up–down dose finding design | 15 cancer patients 2 dropped out | Moderate to severe fatigue Carnitine deficiency KPS ± 50 | Oral Beginning dose of 250 mg/day, increased in 500 mg increments to 3000 mg | 1 week | LC deficiency and self-reported fatigue 83%; LC supplementation safe up to 1750 mg/day; LC increased from 30.0 ± 6.9 a to 41.0 ± 12.1 a (p = 0.01); BFI improved 73 (46, 82) b vs. 50 (3, 82) b (p = 0.009); CES-D improved 31.3 (16, 48) b vs. 22.0 (6, 40) b (p = 0.028); ESS improved 17.5 (0, 24) b vs. 8 (0, 15) b (p = 0.015) | None reported by participants |
| [98] | Single arm | 12 cancer patients | Locally advanced/metastatic disease with high levels of fatigue/ROS | Daily 6 g LC, in 3 doses of 2 g each Concomitant chemotherapy | 4 weeks (t2) | Fatigue decreased: MFSI-SF: 12.05 ± 12.56 a (p < 0.001); QoL improved: Part A QoL-OS:36.80 ± 15.7 a (p < 0.05); EQ-5DVAS: 73.33 ± 12.4 a (p < 0.001); LBM increased t0:38.0 ± 7.36 a t2:40.39 ± 8.55 a kg (p < 0.05); Appetite increased: t0: 4.75 ± 2.59 a t2: 6.83 ± 1.9 a p = 0.001; | None reported that could be related to L-carnitine |
| [93] | Phase I/II open label clinical trial to test safety, tolerance and establish safe dose range | 27 cancer patients | Moderate to severe fatigue (KPS ≥ 50) and carnitine deficiency | 250 mg/day increased in 500 mg increments to 3000 mg | 1 week | Total Carnitine rose from 32.8 ± 10 a to 54.3 ± 23 a (p < 0.001); Haemoglobin declined from 12.2 ± 2.1 a g/dL to 12.0 ± 1.8 a g/dL (p = 0.03); BFI improved 66.1 ± 12 a vs. 39.7 ± 26 a (p < 0.001); ESS improved 12.9 ± 7 a vs. 9 ± 6 a (p = 0.001); CES-D improved 29.2 ± 12 a vs. 19 ± 12 a (p < 0.001); Supplementation well tolerated upto 3000 mg; | Mild nausea (2) |
| [96] | Double-blind placebo-controlled, then open-label phase | 29 advanced cancer patients treatment 17; placebo 12 | Moderate to severe fatigue (KPS ≥ 50) and carnitine deficiency | 0.5 g/day (2 days), 1 g/day (2 days), 2 g/day (10 days) | 2 weeks placebo-controlled 2 weeks open label | Total Carnitine increased from 32.9 ± 3.8 a to 56.6 ± 20.5 a (p = 0.004); Analysis from the 14-day blinded phase failed to show any significant improvement in fatigue or function compared to the placebo. | Constipation and diarrhoea (1 of each) |
| [100] | Randomised phase III clinical trial (5 arm) | Patients with cancer cachexia n = 332; LC arm n = 88 | Advanced stage; loss of >5% of ideal or pre-illness weight | Randomised to one of 5 arms LC arm received 4 g/day | 16 weeks | LC did not increase LBM, decrease REE or fatigue GPS improved 1.2 ± 0.76 a to 0.9 ± 0.86 a (p = 0.03); ECOG PS improved 1.88 ± 0.88 a to 1.5 ± 0.9 a (p = 0.0001) | Grade 3 or 4 diarrhoea (2) |
| [99] | Prospective multi-centre, placebo-controlled, randomised and double-blinded trial | 72 pancreatic cancer patients; Evaluable n = 26 LC n = 14 PC n = 12 | Advanced pancreatic cancer 10% weight loss in previous 6 months KPS > 60 | Oral LC 4 g/day or placebo | 12 weeks | BMI increase LC: 3.4% ± 1.35 c vs. PC: −1.5% ± 1.4 c p < 0.018; After 12 weeks difference was 4.9% ± 1.9 between groups p < 0.05; BIA showed improved body composition (p < 0.013) and BF (p < 0.041); EORTC-QLQ-C30/PAN26; Improved cognitive function LC: 0.30 versus PC-0.13 (p < 0.034), global health status LC 0.76 versus PC-0.32 (p < 0.041), and gastrointestinal symptoms improved-0.35 while participants on the placebo deteriorated 0.78 (p < 0.033). | |
| [101] | Phase III, randomised, double-blind, placebo-controlled trial followed by an open-label trial | N = 376 LC n = 189 PC n = 187 | Invasive malignant cancer and moderate to severe fatigue | Week 0–4: 2 g oral L-carnitine or placebo; week 5–8: 2 g oral L-carnitine | 4 weeks randomised, double-blind placebo, 4 weeks open label | 33% of participants had LC deficiency; total mean carnitine increased 46.3 (95% CI, 44.1 to 48.4 µM/L) to 66.2 µM/L (95% CI, 62.4 to 69.9 µM/L); PC mean total carnitine increased nominally from 43.6 (95% CI, 41.4 to 45.7 µM/L) to 43.7 (95% CI, 40.7 to 46.7 µM/L; p = 0.004); No measures showed significant improvement when treatment and placebo arm compared, but significant improvement versus baseline suggesting a placebo effect. | Haemoglobin (LC-10, PC-9); Platelets (PC-1); Fatigue (LC-2); Itching /rash (LC-2); Death /disease progression (PC-2); Constipation (LC-1); Diarrhoea (LC-1, PC-4); Nausea /vomiting (LC-5, PC-6); Urinary tract infection (LC-1); Abdominal pain (LC-1, PC-2); Atrial fibrulation (PC-1); Dyspnea (2); Patient odour (PC-1); Extrapyramidal movement (PC-1); Headache (PC-1) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitchell, T.; Clarke, L.; Goldberg, A.; Bishop, K.S. Pancreatic Cancer Cachexia: The Role of Nutritional Interventions. Healthcare 2019, 7, 89. https://doi.org/10.3390/healthcare7030089
Mitchell T, Clarke L, Goldberg A, Bishop KS. Pancreatic Cancer Cachexia: The Role of Nutritional Interventions. Healthcare. 2019; 7(3):89. https://doi.org/10.3390/healthcare7030089
Chicago/Turabian StyleMitchell, Toni, Lewis Clarke, Alexandra Goldberg, and Karen S. Bishop. 2019. "Pancreatic Cancer Cachexia: The Role of Nutritional Interventions" Healthcare 7, no. 3: 89. https://doi.org/10.3390/healthcare7030089
APA StyleMitchell, T., Clarke, L., Goldberg, A., & Bishop, K. S. (2019). Pancreatic Cancer Cachexia: The Role of Nutritional Interventions. Healthcare, 7(3), 89. https://doi.org/10.3390/healthcare7030089






