Far-Reaching Dispersal of Borrelia burgdorferi Sensu Lato-Infected Blacklegged Ticks by Migratory Songbirds in Canada
Abstract
1. Introduction
2. Materials and Methods
2.1. Tick Collection
2.2. Molecular Tick Identification
2.3. Spirochete Detection
3. Results
3.1. Tick Collection
3.2. Molecular Tick Identification
3.3. Spirochete Detection
3.4. Spirochete Amplicon Sequences
4. Discussion
4.1. Photoperiod to Molt Blacklegged Ticks
4.2. Obstacles Facing Migratory Songbirds
4.3. Bird Parasitism during Spring Migration
4.4. Spirochete-Positive Blacklegged Ticks in Central Manitoba
4.5. Bird-Feeding Ticks Positive for Lyme Disease Spirochete in Québec
4.6. Songbird-Transported Ticks Infected with Lyme Disease Bacterium in Alberta
4.7. Ticks Positive for Lyme Disease Spirochete in Cape Breton, Nova Scotia
4.8. Ticks Infected with Lyme Disease Spirochete in Newfoundland and Labrador
4.9. Songbirds Establish Tick Populations
4.10. Viability of B. burgdorferi Sensu Lato in Bird-Feeding Ticks
4.11. Implications of Human Lyme Disease
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Burgdorfer, W.; Barbour, A.G.; Hayes, S.F.; Benach, J.L.; Grunwaldt, E.; Davis, J.P. Lyme disease—A tick-borne spirochetosis? Science 1982, 216, 1317–1319. [Google Scholar] [CrossRef] [PubMed]
- Keirans, J.E.; Hutcheson, H.J.; Durden, L.A.; Klompen, J.S.H. Ixodes (Ixodes) scapularis (Acari: Ixodidae): Redescription of all active stages, distribution, hosts, geographical variation, and medical and veterinary importance. J. Med. Entomol. 1996, 33, 297–318. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Foley, J.E.; Young, M.R.; Durden, L.A. First report of a blacklegged tick, Ixodes scapularis Say (Acari: Ixodidae), parasitizing a raptor in Canada. Syst. Appl. Acarol. 2017, 22, 208–216. [Google Scholar] [CrossRef]
- Scott, J.D.; Fernando, K.; Banerjee, S.N.; Durden, L.A.; Byrne, S.K.; Banerjee, M.; Mann, R.B.; Morshed, M.G. Birds disperse ixodid (Acari: Ixodidae) and Borrelia burgdorferi-infected ticks in Canada. J. Med. Entomol. 2001, 38, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Anderson, J.F.; Durden, L.A. Widespread dispersal of Borrelia burgdorferi-infected ticks collected from songbirds across Canada. J. Parasitol. 2012, 98, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Durden, L.A. New records of the Lyme disease bacterium in ticks collected from songbirds in central and eastern Canada. Int. J. Acarol. 2015, 41, 241–249. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Lin, T.; Gao, L.; Grubhoffer, L.; Oliver, J.H., Jr. Delineation of a new species of the Borrelia burgdorferi sensu lato complex, Borrelia americana sp. nov. J. Clin. Microbiol. 2009, 47, 3875–3880. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Foley, J.E. Detection of Borrelia americana in avian coastal tick, Ixodes auritulus (Acari: Ixodidae), collected from a bird captured in Canada. Open J. Anim. Sci. 2016, 6, 207–216. [Google Scholar] [CrossRef]
- Baranton, G.; Postic, D.; Saint Girons, I.; Boerlin, P.; Piffaretti, J.-C.; Assous, M.; Girmont, P.A.D. Delineation of Borrelia burgdorferi sensu stricto, Borrelia garinii sp. nov. and group VS461 associated with Lyme borreliosis. Int. J. Syst. Bacteriol. 1992, 42, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.N.; Christensen, C.I.; Scott, J.D. Isolation of Borrelia burgdorferi on mainland Ontario. Can. Commun. Dis. Rep. 1995, 21, 85–86. [Google Scholar] [PubMed]
- Margos, G.; Hojgaard, A.; Lane, R.S.; Cornet, M.; Fingerle, V.; Rudenko, N.; Ogden, N.; Aanensen, D.M.; Fish, D.; Piesman, J. Multilocus sequence analysis of Borrelia bissettii strains from North America reveals a new Borrelia species, Borrelia kurtenbachii. Ticks Tick-Borne Dis. 2010, 1, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Margos, G.; Fedorova, N.; Kleinjan, J.E.; Hartberger, C.; Schwan, T.G.; Sing, A.; Fingerle, V. Borrelia lanei sp. nov. extends the diversity of Borrelia species in California. Int. J. Syst. Evol. Microbiol. 2017, 67, 3872–3876. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Clark, K.L.; Foley, J.E.; Anderson, J.F.; Durden, L.A.; Manord, J.M.; Smith, M.L. Detection of Borrelia genomospecies 2 in Ixodes spinipalpis ticks collected from a rabbit in Canada. J. Parasitol. 2017, 103, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.P., Jr.; Muzaffar, S.B.; Lavers, J.; Lacombe, E.H.; Cahill, B.K.; Lubelczyk, C.B.; Kinsler, A.; Mathers, A.J.; Rand, P.W. Borrelia garinii in seabird ticks (Ixodes uriae), Atlantic coast, North America. Emerg. Infect. Dis. 2006, 12, 1909–1912. [Google Scholar] [CrossRef] [PubMed]
- Postic, D.; Ras, N.M.; Lane, R.S.; Hendson, M.; Baranton, G. Expanded diversity among Californian Borrelia isolates and description of Borrelia bissettii sp. nov. (formerly Borrelia group DN127). J. Clin. Microbiol. 1998, 36, 3497–3504. [Google Scholar] [PubMed]
- Banerjee, S.N.; Banerjee, M.; Smith, J.A.; Fernando, K. Lyme Disease in British Columbia—An update. In Proceedings of the VII Annual Lyme Disease Foundation International Conference, Stamford, CT, USA, 22–23 April 1994; Lyme Disease Foundation: Hartford, CT, USA, 1994; pp. 1–88. [Google Scholar]
- Margos, G.; Lane, R.S.; Fedorova, N.; Koloczek, J.; Piesman, J.; Hojgaard, A.; Sing, A.; Fingerle, V. Borrelia bissettiae sp. nov. and Borrelia californiensis sp. nov. prevail in diverse enzootic transmission cycles. Int. J. Syst. Evol. Microbiol. 2016, 66, 1447–1452. [Google Scholar] [CrossRef] [PubMed]
- DeLuca, W.V.; Woodworth, B.K.; Rimmer, C.C.; Marra, P.P.; Taylor, P.D.; McFarland, K.P.; Mackenzie, S.A.; Norris, D.R. Transoceanic migration by a 12 g songbird. Biol. Lett. 2015, 11, 20141045. [Google Scholar] [CrossRef] [PubMed]
- Morshed, M.G.; Scott, J.D.; Fernando, K.; Beati, L.; Mazerolle, D.F.; Geddes, G.; Durden, L.A. Migratory songbirds disperse ticks across Canada, and first isolation of the Lyme disease spirochete, Borrelia burgdorferi, from the avian tick, Ixodes auritulus. J. Parasitol. 2005, 91, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Ogden, N.H.; Lindsay, L.R.; Hanincová, K.; Barker, I.K.; Bigras-Poulin, M.; Charron, D.F.; Heagy, A.; Francis, C.M.; O’Callaghan, C.J.; Schwartz, I.; et al. Role of migratory birds in introduction and range expansion of I. scapularis ticks and of Borrelia burgdorferi and Anaplasma phagocytophilum in Canada. Appl. Environ. Microbiol. 2008, 74, 1780–1790, Erratum: Appl. Environ. Microbiol. 2008, 74, 3919. doi:10.1128/AEM.00857-0819. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Lee, M.-K.; Fernando, K.; Durden, L.A.; Jorgensen, D.R.; Mak, S.; Morshed, M.G. Detection of Lyme disease spirochete, Borrelia burgdorferi sensu lato, including three novel genotypes in ticks (Acari: Ixodidae) collected from songbirds (Passeriformes) across Canada. J. Vect. Ecol. 2010, 35, 124–139. [Google Scholar] [CrossRef]
- Clifford, C.M.; Anastos, G.; Elbl, A. The larval ixodid ticks of the eastern United States. Misc. Publ. Entomol. Soc. Am. 1961, 2, 213–237. [Google Scholar]
- Keirans, J.E.; Clifford, C.M. The genus Ixodes in the United States: A scanning electron microscope study and key to the adults. J. Med. Entomol. 1978, 15 (Suppl. 2), 1–38. [Google Scholar] [CrossRef]
- Durden, L.A.; Keirans, J.E. Nymphs of the Genus Ixodes (Acari: Ixodidae) of the United States: Taxonomy, Identification Key, Distribution, Hosts, and Medical/Veterinary Importance. Monographs; Thomas Say Publications in Entomology, Entomological Society of America: Lanham, MD, USA, 1996; p. 95. ISBN 0-938522-57-4. [Google Scholar]
- Barbour, A.G.; Bunikis, J.; Travinsky, B.; Hoen, A.G.; Diuk-Wasser, M.A.; Fish, D.; Tsao, J.I. Niche partitioning of Borrelia burgdorferi and Borrelia miyamotoi in the same tick vector and mammalian reservoir species. Am. J. Trop. Med. Hyg. 2009, 81, 1120–1131. [Google Scholar] [CrossRef] [PubMed]
- Clark, K.; Hendricks, A.; Burge, D. Molecular identification and analysis of Borrelia burgdorferi sensu lato in lizards in the southeastern United States. Appl. Environ. Microbiol. 2005, 71, 2616–2625. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.D.; Collins, R.A.; Boyer, S.; Lefort, M.C.; Malumbres-Olarte, J.; Vink, C.J.; Cruickshank, R.H. Spider: An R package for the analysis of species identity and evolution, with particular reference to DNA barcoding. Mol. Ecol. Resour. 2012, 12, 562–565. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Scott, C.M. Lyme disease propelled by Borrelia burgdorferi-infected blacklegged ticks, wild birds and public awareness—Not climate change. J. Veter. Sci. Med. 2018, 6, 8. [Google Scholar] [CrossRef]
- Sogge, M.K.; Ahlers, D.; Sferra, S.J. A Natural History Summary and Survey Protocol for the Southwestern Willow Flycatcher; U.S. Geological Survey Techniques and Methods, 2A-10; U.S. Geological Survey: Reston, VA, USA, 2010.
- Blancher, P. Estimated number of birds killed by house cats (Felis catus) in Canada. Avian Conserv. Ecol. 2013, 8, 3. [Google Scholar] [CrossRef]
- Sillett, T.S.; Holmes, R.T. Variation in survivorship of a migratory songbird throughout its annual cycle. J. Anim. Ecol. 2002, 71, 296–308. [Google Scholar] [CrossRef]
- Mannelli, A.; Kitron, U.; Jones, C.J.; Slajchert, T.L. Influence of season and habitat on Ixodes scapularis infestation on white-footed mice in northwestern Illinois. J. Parasitol. 1994, 80, 1038–1043. [Google Scholar] [CrossRef] [PubMed]
- Rollend, L.; Fish, D.; Childs, J.E. Transovarial transmission of Borrelia spirochetes by Ixodes scapularis: A summary of the literature and recent observations. Ticks Tick-Borne Dis. 2013, 4, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.N.; Banerjee, M.; Fernando, K.; Dong, M.Y.; Smith, J.A.; Cook, D. Isolation of Borrelia burgdorferi, the Lyme disease spirochete from rabbit ticks, Haemaphysalis leporispalustris from Alberta. J. Spir. Tick-Borne Dis. 1995, 2, 23–24. [Google Scholar]
- Eisen, L.; Lane, R.S. Vectors of Borrelia burgdorferi sensu lato. In Lyme Borreliosis: Biology, Epidemiology and Control; Gray, J., Kahl, O., Lane, R.S., Stanek, G., Eds.; CAB International: Wallingford, UK, 2002; pp. 91–115. ISBN 978-0851996325. [Google Scholar]
- Anderson, J.F.; Magnarelli, L.A. Avian and mammalian hosts for spirochete-infected ticks and insects in a Lyme disease focus in Connecticut. Yale J. Biol. Med. 1984, 57, 627–641. [Google Scholar] [PubMed]
- Anderson, J.F.; Magnarelli, L.A.; Stafford, K.C., III. Bird-feeding ticks transstadially transmit Borrelia burgdorferi that infect Syrian hamsters. J. Wildl. Dis. 1990, 26, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D.; Scott, C.M.; Anderson, J.F. The establishment of a blacklegged tick population by migratory songbirds in Ontario, Canada. J. Veter. Sci. Med. 2014, 2, 5. [Google Scholar] [CrossRef]
- Scott, J.D.; Durden, L.A. Amblyomma dissimile Koch (Acari: Ixodidae) parasitizes bird captured in Canada. Syst. Appl. Acarol. 2015, 20, 854–860. [Google Scholar] [CrossRef]
- Scott, J.D.; Durden, L.A. First record of Amblyomma rotundatum tick (Acari: Ixodidae) parasitizing a bird collected in Canada. Syst. Appl. Acarol. 2015, 20, 155–161. [Google Scholar] [CrossRef]
- Hersh, M.H.; Osfeld, R.S.; McHenry, D.J.; Tibbetts, M.; Brunner, J.L.; Killilea, M.E.; LoGiudice, K.; Schmidt, K.A.; Keesing, F. Co-infestation of blacklegged ticks with Babesia microti and Borrelia burgdorferi is higher than expected and acquired from small mammal hosts. PLoS ONE 2014, 9, e99348. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.D. First record of locally acquired human babesiosis in Canada caused by Babesia duncani: A case report. Sage Open Med. Case Rep. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, R.W. Integument and ecdysis. In Biology of Ticks, 2nd ed.; Sonenshine, D.E., Roe, R.M., Eds.; Oxford University Press: Oxford, UK, 2013; Volume 1, pp. 99–121. ISBN 9780199744053. [Google Scholar]
- Straubinger, R.K.; Straubinger, A.F.; Summers, B.A.; Jacobson, R.H. Status of Borrelia burgdorferi infection after antibiotic treatment and the effects of corticosteroids: An experimental study. J. Inf. Dis. 2000, 181, 1069–1081. [Google Scholar] [CrossRef] [PubMed]
- Ladislav, S.; Sonenshine, D.E.; Park, Y.; Žitňan, D. Nervous and sensory systems: Structure, function, genomics, and proteomics. In Biology of Ticks, 2nd ed.; Sonenshine, D.E., Roe, R.M., Eds.; Oxford University Press: Oxford, England, 2014; Volume 1, pp. 309–367. ISBN 978-0195084313. [Google Scholar]
- Stafford, K.C., III. Tick-associated diseases. In Tick Management Handbook: An Integrated Guide for Homeowners, Pest Control Operators, and Public Health Officials for the Prevention of Tick-Associated Disease; The Connecticut Agricultural Experiment Station; EPS Printing II, LLC: South Windsor, CT, USA, 2007; pp. 20–34. [Google Scholar]
- Cook, M.J. Lyme borreliosis: A review of data on transmission time after tick attachment. Int. J. Gen. Med. 2015, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Des Vignes, F.; Piesman, J.; Heffernan, R.; Schulze, T.L.; Stafford, K.C., III; Fish, D. Effect of tick removal on transmission of Borrelia burgdorferi and Ehrlichia phagocytophila by Ixodes scapularis nymphs. J. Infect. Dis. 2001, 183, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Ebel, G.D.; Kramer, L.D. Short report: Duration of tick attachment required for transmission of Powassan virus by deer ticks. Am. J. Trop. Med. Hyg. 2004, 71, 268–271. [Google Scholar] [PubMed]
- Mehlhorn, H.; Schein, E. The piroplasms: Life cycle and sexual stages. Adv. Parasitol. 1985, 23, 37–103. [Google Scholar] [CrossRef]
- Johnson, L.; Mankoff, J.; Stricker, R.B. Severity of chronic Lyme disease compared to other chronic conditions: A quality of life survey. PeerJ 2014, 2, e322. [Google Scholar] [CrossRef] [PubMed]
- Stonehouse, A.; Studdiford, J.S.; Henry, C.A. An update on the diagnosis and treatment of early Lyme disease: “focusing on the bull’s eye, you may miss the mark”. J. Emerg. Med. 2010, 39, e147–e151. [Google Scholar] [CrossRef] [PubMed]
- Schutzer, S.E.; Berger, B.W.; Krueger, J.G.; Eshoo, M.W. Atypcial erythema migrans in patients with PCR-positive Lyme disease. Emerg. Inf. Dis. 2013, 19, 815–817. [Google Scholar] [CrossRef] [PubMed]
- Schuttelaar, M.L.; Laeijendecker, R.; Heinhuis, R.J.; Van Joost, T. Erythema multiforme and persistent erythema as early cutaneous manifestations of Lyme disease. J. Am. Acad. Dermatol. 1997, 37, 873–875. [Google Scholar] [CrossRef]
- Berger, B.W. Dermatologic manifestations of Lyme disease. Rev. Infect. Dis. 1989, 11 (Suppl. 6), S1475–S1481. [Google Scholar] [CrossRef]
- Cameron, D.J.; Johnson, L.B.; Maloney, E.L. Evidence assessments and guideline recommendations in Lyme disease: The clinical management of known tick bites, erythema migrans rashes and persistent disease. Expert Rev. Anti-Infect. Ther. 2014, 12, 1103–1135. [Google Scholar] [CrossRef] [PubMed]
- Miklossy, J. Bacterial amyloid and DNA are important constituents of senile plaques: Further evidence of the spirochetal and biofilm nature of senile plaques. J. Alzheimers Dis. 2016, 53, 1459–1473. [Google Scholar] [CrossRef] [PubMed]
- Sapi, E.; Bastain, S.L.; Mpoy, C.M.; Scott, S.; Rattelle, A.; Pabbati, N.; Poruri, A.; Burugu, D.; Theophilus, P.A.S.; Pham, T.V.; et al. Characterization of biofilm formation by Borrelia burgdorferi in vitro. PLoS ONE 2012, 7, e48277. [Google Scholar] [CrossRef] [PubMed]
- Dorward, D.W.; Fischer, E.R.; Brooks, D.M. Invasion and cytopathic killing of human lymphocytes by spirochetes causing Lyme disease. Clin. Infect. Dis. 1997, 25 (Suppl. 1), S2–S8. [Google Scholar] [CrossRef]
- Zijkowska, J.M.; Hermanowska-Szpakowicz, T. New aspects of the pathogenesis of Lyme disease. Prz. Epidemiol. 2002, 56 (Suppl. 1), S57–S67. [Google Scholar]
- Horowitz, R.I. Lyme and inflammation. In Why Can’t I Get Better? Solving the Mystery of Lyme & Chronic Disease; St. Martin’s Press: New York, NY, USA, 2013; pp. 185–201. ISBN 978-1-250-01940-0. [Google Scholar]
- Asbrink, E.; Hovmark, A.; Weber, K. Acrodermatitis chronica atrophicans. In Aspects of Lyme Borreliosis; Weber, K., Burgdorfer, W., Eds.; Springer-Verlag: Berlin/Heidelberg, Germany, 1993; pp. 193–204. ISBN 3-540-55628-1. [Google Scholar]
- Chagnon, S.L.; Naik, M.; Abdel-Hamid, H. Child neurology: Tick paralysis: A diagnosis not to miss. Neurology 2014, 82, e91–e93. [Google Scholar]
- Scott, J.D.; Scott, C.M.; Anderson, J.F. Tick paralysis in a snowshoe hare by Ixodes pacificus ticks in British Columbia, Canada. J. Veter. Sci. Med. 2014, 2, 5. [Google Scholar]
- Peacock, B.N.; Gherezghiher, T.B.; Hilario, J. New insights into Lyme disease. Redox Biol. 2015, 5, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Butler, T. The Jarisch-Herxheimer reaction after antibiotic treatment of spirochetal infections: A review of recent cases and our understanding of pathogenesis. Am. J. Trop. Med. Hyg. 2017, 96, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Eppes, S.C.; Childs, J.A. Comparative study of cefuroxime axetil versus amoxicillin in children with early Lyme disease. Pediatrics 2002, 109, 1173–1177. [Google Scholar] [CrossRef] [PubMed]
- Middelveen, M.J.; Sapi, E.; Filush, K.R.; Franco, A.; Fesler, M.C.; Stricker, R.B. Persistent Borrelia infection in patients with ongoing symptoms of Lyme disease. Healthcare 2018, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Oksi, J.; Kalimo, H.; Marttila, R.J.; Marjamäki, M.; Sonninen, P.; Nikoskelainen, J.; Viljanen, M.K. Inflammatory brain changes in Lyme borreliosis: A report on three patients and review of literature. Brain 1996, 119, 2143–2154. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, A.B. Alzheimer’s neuroborreliosis with trans-synaptic spread of infection and neurofibrillary tangles derived from intraneuronal spirochetes. Med. Hypotheses 2007, 68, 822–825. [Google Scholar] [CrossRef] [PubMed]
- Miklossy, J. Alzheimer’s disease—A neurospirochetosis. Analysis of the evidence following Koch’s and Hill’s criteria. J. Neuroinflamm. 2011, 8, 90. [Google Scholar] [CrossRef] [PubMed]
- Häupl, T.; Hahn, G.; Rittig, M.; Krause, A.; Schoerner, C.; Schönherr, U.; Kalden, J.R.; Burmester, G.R. Persistence of Borrelia burgdorferi in ligamentous tissue from a patient with chronic Lyme borreliosis. Arthritis Rheum. 1993, 36, 1621–1626. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.E. Damage of collagen and elastic fibres by Borrelia burgdorferi—Known and new clinical histopathogical aspects. Open Neurol. J. 2012, 6 (Suppl. 1), S179–S186. [Google Scholar] [CrossRef] [PubMed]
- Oksi, J.; Mertsola, J.; Reunanen, M.; Marjamäki, M.; Viljanen, M.K. Subacute multiple-site osteomyelitis caused by Borrelia burgdorferi. Clin. Infect. Dis. 1994, 19, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Preac-Mursic, V.; Pfister, H.W.; Spiegel, H.; Burk, R.; Wilske, B.; Reinhardt, S.; Böhmer, R. First isolation of Borrelia burgdorferi from an iris biopsy. J. Clin. Neuroophthalmol. 1993, 13, 155–161. [Google Scholar] [PubMed]
- Frey, M.; Jaulhac, B.; Piemont, Y.; Marcellin, L.; Boohs, P.-M.; Vautravers, P.; Jesel, M.; Kuntz, J.-L.; Monteil, H.; Sibilia, J. Detection of Borrelia burgdorferi DNA in muscle of patients with chronic myalgia related to Lyme disease. Am. J. Med. 1998, 104, 591–594. [Google Scholar] [CrossRef]
- Ramesh, G.; Borda, J.T.; Dufour, J.; Kaushal, D.; Ramamoorthy, R.; Lackner, A.A.; Philipp, M.T. Interaction of the Lyme disease spirochete Borrelia burgdorferi with brain parenchyma elicits inflammatory mediators from glial cells as well as glial and neuronal apoptosis. Am. J. Pathol. 2008, 173, 1415–1427. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, G.; Santana-Gould, L.; Inglis, F.M.; England, J.D.; Philipp, M.T. The Lyme disease spirochete Borrelia burgdorferi induces inflammation and apoptosis in cells from dorsal root ganglia. J. Neuroinflamm. 2013, 10, 88. [Google Scholar] [CrossRef] [PubMed]
- Girschick, H.J.; Huppertz, H.I.; Rüssmann, H.; Krenn, V.; Karch, H. Intracellular persistence of Borrelia burgdorferi in human synovial cells. Rheumatol. Int. 1996, 16, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Klempner, M.S.; Noring, R.; Rogers, R.A. Invasion of human skin fibroblasts by the Lyme disease spirochete, Borrelia burgdorferi. J. Infect. Dis. 1993, 167, 1074–1081. [Google Scholar] [CrossRef] [PubMed]
- Benach, J.L.; Bosler, E.M.; Hanrahan, J.P.; Coleman, J.L.; Habicht, G.S.; Bast, T.F.; Cameron, D.J.; Ziegler, J.L.; Barbour, A.G.; Burgdorfer, W. Spirochetes isolated from the blood of two patients with Lyme disease. N. Engl. J. Med. 1983, 308, 740–742. [Google Scholar] [CrossRef] [PubMed]
- Sapi, E.; Pabbati, N.; Datar, A.; Davies, E.M.; Rattelle, A.; Kuo, B.A. Improved culture conditions for the growth and detection of Borrelia from human serum. Int. J. Med. Sci. 2013, 10, 362–376. [Google Scholar] [CrossRef] [PubMed]
- Meriläinen, L.; Herranen, A.; Schwarzbach, A.; Gilbert, L. Morphological and biochemical features of Borrelia burgdorferi pleomorphic forms. Microbiology 2015, 161, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Embers, M.E.; Hasenkampf, N.R.; Jacobs, M.B.; Tardo, A.C.; Doyle-Meyers, A.; Philipp, M.T.; Hodzic, E. Variable manifestations, diverse seroreactivity and post-treatment persistence in non-human primates exposed to Borrelia burgdorferi by tick feeding. PLoS ONE 2017, 12, e0189071. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Brown, A.V.; Matluck, M.E.; Hu, L.T.; Lewis, K. Borrelia burgdorferi, the causative agent of Lyme disease, forms drug-tolerant persister cells. Antimicrob. Agents Chemother. 2015, 59, 4616–4624. [Google Scholar] [CrossRef] [PubMed]
- Cabello, F.C.; Godfrey, H.P.; Bugrysheva, J.V.; Newman, S.A. Sleeper cells: The stringent response and persistence in the Borreliella (Borrelia) burgdorferi enzootic cycle. Environ. Microbiol. 2017, 19, 3846–3862. [Google Scholar] [CrossRef] [PubMed]
- Middelveen, M.J.; Burke, J.; Sapi, E.; Bandoski, C.; Filush, K.R.; Wang, Y.; Franco, A.; Timmaraju, A.; Schlinger, H.A.; Mayne, P.J.; et al. Culture and identification of Borrelia spirochetes in human vaginal and seminal secretions. F1000Research 2015, 3, 309. [Google Scholar] [CrossRef] [PubMed]
- Stricker, R.B.; Middelveen, M.J. Sexual transmission of Lyme disease: Challenging the tickborne disease paradigm. Expert Rev. Anti-Infect. Ther. 2015, 13, 1303–1306. [Google Scholar] [CrossRef] [PubMed]
- Embers, M.E.; Barthold, S.W.; Borda, J.T.; Bowers, L.; Doyle, L.; Hodzic, E.; Jacobs, M.B.; Hasenkampf, N.R.; Martin, D.S.; Narasimhan, D.S.; et al. Persistence of Borrelia burgdorferi in rhesus macaques following antibiotic treatment of disseminated infection. PLoS ONE 2012, 7, e29914. [Google Scholar] [CrossRef]
- Hodzic, E.; Imai, D.; Feng, S.; Barthold, S.W. Resurgence of persisting non-cultivable Borrelia burgdorferi following antibiotic treatment in mice. PLoS ONE 2014, 9, e86907. [Google Scholar] [CrossRef] [PubMed]
- Liegner, K.B.; Duray, P.; Agricola, M.; Rosenkilde, C.; Yannuzzi, L.A.; Ziska, M.; Tilton, R.C.; Hulinska, D.; Hubbard, J.; Fallon, B.A. Lyme disease and the clinical spectrum of antibiotic responsive chronic meningoencephalomyelitides. J. Spir. Tick-Borne Dis. 1997, 4, 61–73. [Google Scholar]
- Bransfield, R.C. Suicide and Lyme and associated diseases. Neuropsychiatr. Dis. Treat. 2017, 13, 1575–1587. [Google Scholar] [CrossRef] [PubMed]
- Bransfield, R.C. Aggressiveness, violence, homocidality, homicide, and Lyme disease. Neuropsychiatr. Dis. Treat. 2018, 14, 693–713. [Google Scholar] [CrossRef] [PubMed]


| Bird Species | No. of Birds | Hlp | Iaf | Imu | Isc | No. Ticks | No. Ticks | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| L | N | N | L | F | L | N | Collected | Pos/Tested (%) | |||||
| Eastern white-crowned Sparrow, Zonotrichia leucophrys (Forster) | 1 | - | - | 0/1 | - | - | - | - | 1 | 0/1 (0) | |||
| Song Sparrow, Melospiza melodia (Wilson) | 2 | - | - | - | - | - | 0/1 | 1/1 | 2 | 1/2 (50) | |||
| Brown Thrasher, Toxostoma rufum (L.) | 1 | - | - | - | - | - | - | 1/1 | 1 | 1/1 (100) | |||
| Northern House Wren, Troglodytes aedon (Vieillot) | 4 | - | - | 0/1 | 0/3 | - | 3/5 | 9 | 3/9 (33) | ||||
| Hermit Thrush, Catharus guttatus (Pallas) | 1 | - | - | - | - | - | - | 0/1 | 1 | 0/1 (0) | |||
| Swamp Sparrow, Melospiza georgiana (Latham) | 1 | - | - | - | - | - | - | 0/1 | 1 | 0/1 (0) | |||
| Ovenbird, Seiurus aurocapillus (L.) | 2 | - | - | - | - | - | - | 0/2 | 2 | 0/2 (0) | |||
| White-throated Sparrow, Zonotrichia albicollis (Gmelin) | 3 | - | - | - | - | 0/1 | - | 1/4 | 5 | 1/5 (20) | |||
| Northern Waterthrush, Parkesia noveboracensis (Gmelin) | 2 | - | - | - | - | - | - | 1/3 | 3 | 1/3 (33) | |||
| Common Yellowthroat, Geothlypis trichas (L.) | 10 | - | - | 0/2 | - | - | - | 8/17 | 19 | 8/19 (42) | |||
| Magnolia Warbler, Setophaga magnolia (Wilson) | 1 | - | - | - | - | - | - | 0/2 | 2 | 0/2 (0) | |||
| Veery, Catharus fuscescens (Stephens) | 2 | - | - | 0/1 | - | - | 0/1 | 0/3 | 5 | 0/5 (0) | |||
| Gray-cheeked Thrush, Catharus minimus (Lafresnaye) | 2 | - | - | - | - | - | - | 1/2 | 2 | 1/2 (50) | |||
| Swainson’s Thrush, Catharus ustulatus (Nuttall) | 5 | 5 † | 1 † | - | - | - | 0/2 | 6/9 | 17 | 6/11 (55) | |||
| Indigo Bunting, Passerina cyanea (L.) | 1 | - | - | - | - | - | - | 1/4 | 4 | 1/4 (25) | |||
| Mourning Warbler, Geothlypis philadelphia (Wilson) | 1 | - | - | - | - | - | - | 0/2 | 2 | 0/2 (0) | |||
| Dark-eyed Junco, Junco hyemalis (L.) | 1 | - | - | - | - | - | - | 1/2 | 2 | 1/2 (50) | |||
| Lincoln’s Sparrow, Melospiza lincolnii (Audubon) | 2 | - | - | - | - | - | - | 0/3 | 3 | 0/3 (0) | |||
| Chipping Sparrow, Spizella passerina (Bechstein) | 1 | 0/4 | 0/2 | - | - | - | - | - | 6 | 0/6 (0) | |||
| Gray Catbird, Dumetella carolinensis (L.) | 1 | - | - | - | 2 ‡ | - | - | - | 2 | 0/1 (0) | |||
| Total: 20 species | 44 | 14 | 3 | 5 | 3 | 4 | 4 | 24/62 (39) | 89 | 24/87 (28) | |||
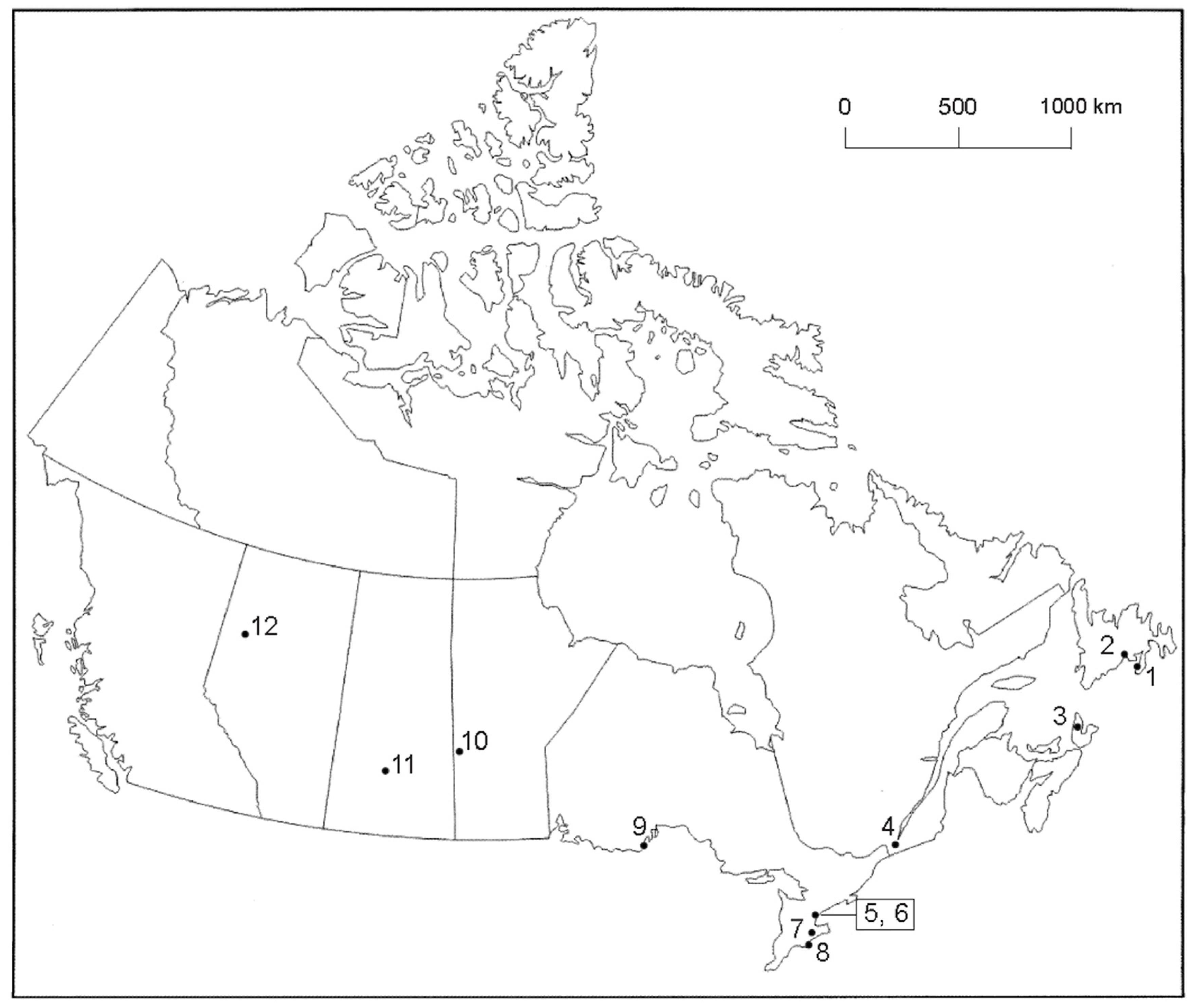
| Tick No. | Geographic Location | Prov. | Site | Host | GenBank Accession No. | Lab |
|---|---|---|---|---|---|---|
| 17-5A4 | Ruthven Park | ON | 7 | Song Sparrow | MG952944 | JEF |
| 17-5A6 | Ruthven Park | ON | 7 | Brown Thrasher | MG958137 | JEF |
| 17-5A7B | Ste-Anne-de-Belleville | QC | 4 | Northern House Wren | MG958138 | JEF |
| CN17-5A32 | Toronto | ON | 6 | Northern House Wren | MH290726 | KLC |
| CN17-5A35 | Toronto | ON | 6 | Ovenbird | MH290727 | KLC |
| CN17-5A38 | Ste-Anne-de-Bellevue | QC | 4 | Northern Waterthrush | MH290728 | KLC |
| CN17-5A39-2 | Ste-Anne-de-Bellevue | QC | 4 | Common Yellowthroat † | MH290729 | KLC |
| CN17-5A42-1 | Ste-Anne-de-Bellevue | QC | 4 | Common Yellowthroat † | MH290730 | KLC |
| CN17-5A51A, C | Swan River | MB | 10 | Swainson’s Thrush | MH290731–MH290732 | KLC |
| CN17-5A62-2 | Long Point | ON | 8 | Indigo Bunting | MH290733 | KLC |
| CN17-5A74-2 | McKellar Island | ON | 9 | Dark-eyed Junco | MH290734 | KLC |
| CN17-5A77B-2, 3 | Toronto | ON | 5 | Swainson’s Thrush | MH290735–MH290736 | KLC |
| 17-5A80A, B | Peace River | AB | 12 | Swainson’s Thrush | MG967647–MG967648 | JEF |
| 17-5A81A, B | Middle River | NS | 3 | Common Yellowthroat | MG967649–MG967650 | JEF |
| 17-5A82B, C | Grand Bank | NL | 1 | Common Yellowthroat | MG967651–MG967652 | JEF |
| CN17-5A83B | St. Albans | NL | 2 | Gray-cheeked Thrush | MH 290737 | KLC |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scott, J.D.; Clark, K.L.; Foley, J.E.; Bierman, B.C.; Durden, L.A. Far-Reaching Dispersal of Borrelia burgdorferi Sensu Lato-Infected Blacklegged Ticks by Migratory Songbirds in Canada. Healthcare 2018, 6, 89. https://doi.org/10.3390/healthcare6030089
Scott JD, Clark KL, Foley JE, Bierman BC, Durden LA. Far-Reaching Dispersal of Borrelia burgdorferi Sensu Lato-Infected Blacklegged Ticks by Migratory Songbirds in Canada. Healthcare. 2018; 6(3):89. https://doi.org/10.3390/healthcare6030089
Chicago/Turabian StyleScott, John D., Kerry L. Clark, Janet E. Foley, Bradley C. Bierman, and Lance A. Durden. 2018. "Far-Reaching Dispersal of Borrelia burgdorferi Sensu Lato-Infected Blacklegged Ticks by Migratory Songbirds in Canada" Healthcare 6, no. 3: 89. https://doi.org/10.3390/healthcare6030089
APA StyleScott, J. D., Clark, K. L., Foley, J. E., Bierman, B. C., & Durden, L. A. (2018). Far-Reaching Dispersal of Borrelia burgdorferi Sensu Lato-Infected Blacklegged Ticks by Migratory Songbirds in Canada. Healthcare, 6(3), 89. https://doi.org/10.3390/healthcare6030089




