The Activities and Impact of State Programs to Address Hereditary Breast and Ovarian Cancer, 2011–2014
Abstract
:1. Introduction
2. Experimental Section and Methods
3. Results and Discussion
3.1. Broad Overview of State Activities
Impact of Selected, Key State Activities
| State | Activity | Brief Description of Activity | Type of Activity: Education, Surveillance, or Policy |
|---|---|---|---|
| Michigan Department of Health and Human Services (Michigan) | Identification of educational needs followed by development and dissemination of a free online educational module with CMEs | Michigan identified health care provider knowledge gaps by using surveys and other data sources. A free, online, educational module was then developed with attached Continuing Medical Education credits (CMEs) called, Hereditary Breast and Ovarian Cancer: Is Your Patient at High Risk? | Education |
| Honoring health insurance plans for having evidence-based genomic services policies | Michigan reviewed health insurance company policies on BRCA counseling, testing, and related clinical services and gave nonmonetary awards to health plans for having written policies consistent with evidence-based recommendations. They also held focus groups with eight health plan administrators to understand and address barriers and facilitators to the uptake of evidence-based policy by health plans. | Policy | |
| Georgia Department of Public Health (Georgia) | Incorporating a hereditary cancer risk assessment tool into clinical practice | Georgia incorporated a risk assessment tool, the Breast Cancer Genetics Referral Screening Tool (B-RST), into clinical practice in 9 of 18 public health districts across the state. The screening tool quickly identified women seen at these public health clinics who were appropriate for referral to genetic counseling. Before the incorporation of the tool in the nine health centers, an educational program was provided for all clinical and clerical staff who provided services to women. | Surveillance and Education |
| Oregon Health Authority (Oregon) | Tracking and promotion of genomics services in the Oregon Medicaid program. | Oregon worked closely with its state Medicaid program to track and promote use of evidence-based genomic tests. | Surveillance and Policy |
| Bidirectional reporting between the cancer registry and cancer survivors and physicians. | Oregon implemented bidirectional reporting (i.e., cancer survivors who were likely to be appropriate for BRCA counseling were identified, and they and their doctors were notified and received educational materials through the Oregon State Cancer Registry). | Education and Surveillance |
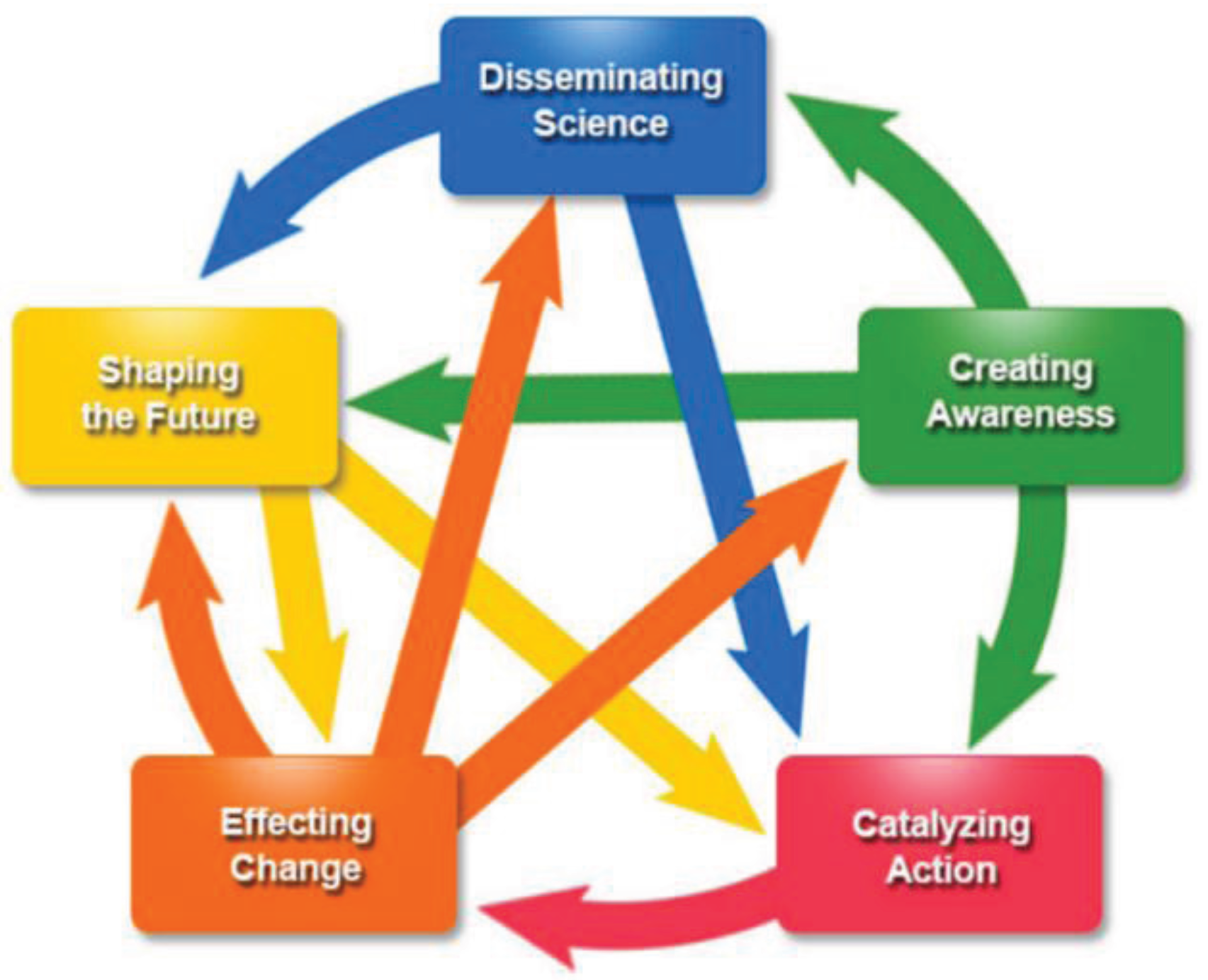
| Domain of Influence | Potential Measureable Indicators |
|---|---|
| Disseminating Science: Generating and communicating knowledge by the producer |
|
| Creating Awareness: The uptake of knowledge and further dissemination and dialogue by the user, and acceptance of a concept or findings by others |
|
| Catalyzing Action: Adoption of knowledge resulting in specific actions |
|
| Effecting Change: Changes in current or existing situations, directions, strategies, policies, or practice |
|
| Shaping the Future: Implementing new or furthering improvements and changes |
|
3.2. Michigan Department of Health and Human Services (Michigan) Activities
3.2.1. Education: Identification of Educational Need Followed by Development and Dissemination of a Free Online Educational Module with CMEs
Disseminating Science
Creating Awareness
Catalyzing Action
Shaping the Future
3.2.2. Policy: Honoring Health Insurance Plans for Having Evidence-Based Genomic Services Policies
Catalyzing Action
Disseminating Science
Effecting Change
3.3. Georgia Department of Public Health (Georgia) Activities
3.3.1. Surveillance: Incorporating a Hereditary Cancer Risk Assessment Tool into Clinical Practice
Catalyzing Action
Effecting Change
Disseminating Science
3.4. Oregon Health Authority (Oregon) Activities
3.4.1. Surveillance/Policy: Tracking and Promotion of Genomics Services in the Oregon Medicaid Program
- Creating Awareness. Starting in 2011, a key collaborator of the Oregon Health Authority chaired and facilitated the Genetic Advisory Council (GAC) to the Health Evidence Review Commission (HERC). The HERC provides coverage guidance and sets priorities for health spending in the Oregon Health Plan and promotes evidence-based medical practice statewide. Oregon and the GAC worked closely to create guidelines and a decision-making algorithm for nonprenatal genetic testing, thus creating awareness among key stakeholders and reflecting acceptance of the concept that genetic services are important. Oregon also worked closely with GAC and the HERC Value-based Benefits Subcommittee to review updated molecular pathology CPT codes and recommended coverage of appropriate tests to ensure that coverage priority was set by using the most current evidence-based genetic information.
- Disseminating Science. Oregon conducted and disseminated results of analyses of Medicaid claims data annually to the Oregon Medicaid program (i.e., to the HERC, the medical director, and managed care directors). These results showed increases in the number of BRCA tests ordered, from 6.7 per 100,000 persons in 2008, to 33.2 per 100,000 persons in 2012, but usage was still lower than expected on the basis of the population covered by Medicaid, indicating underutilization. However, they were unable to determine appropriateness of testing given the incomplete personal and family history data available. These data are paramount as Oregon is one of the few states in which Medicaid covers genetic testing, and these data were used to track progress towards improving coverage for these services.
- Creating Awareness. Because of the dissemination of the claims data, ongoing discussions between Oregon and the Oregon Health Plan ensued. On the basis of feedback from the Medicaid program and their review of the results, they concluded that the low usage in the program was likely caused by a lack of knowledge about coverage for these services.
- Catalyzing Action. Because of the lack of knowledge about coverage for these services, Oregon developed a form for clinicians to use when ordering genetic tests for Medicaid clients to aid in coverage decisions by managed care directors. In addition, Oregon added information about Medicaid coverage for genetics in their educational materials in 2012. Medicaid’s health plan policy, which includes coverage of genetic counseling and testing for BRCA based on USPSTF and NCCN Guidelines®, Washington, PA, USA, was implemented in 2011 and has been updated regularly. Collaborations between the two programs continue.
3.4.2. Education: Bidirectional Reporting between the Cancer Registry and Cancer Survivors and Physicians
Catalyzing Action
Disseminating Science
Effecting Change
3.4.3. Other Noteworthy Impacts of the FOA not Directly Linked to above
Effecting Change
Shaping the Future
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anglian Breast Cancer Study Group. Prevalence and penetrance of BRCA1 and BRCA2 mutations in a population-based series of breast cancer cases. Br. J. Cancer 2000, 83, 1301–1308. [Google Scholar]
- Antoniou, A.C.; Gayther, S.A.; Stratton, J.F.; Ponder, B.A.; Easton, D.F. Risk models for familial ovarian and breast cancer. Genet. Epidemiol. 2000, 18, 173–190. [Google Scholar] [CrossRef]
- Antoniou, A.C.; Pharoah, P.D.; McMullan, G.; Day, N.E.; Stratton, M.R.; Peto, J.; Ponder, B.J.; Easton, D.F. A comprehensive model for familial breast cancer incorporating BRCA1, BRCA2 and other genes. Br. J. Cancer 2002, 86, 76–83. [Google Scholar]
- Peto, J.; Collins, N.; Barfoot, R.; Seal, S.; Warren, W.; Rahman, N.; Easton, D.F.; Evans, C.; Deacon, J.; Stratton, M.R. Prevalence of BRCA1 and BRCA2 gene mutations in patients with early-onset breast cancer. J. Natl. Cancer Inst. 1999, 91, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Antoniou, A.; Pharoah, P.D.; Narod, S.; Risch, H.A.; Eyfjord, J.E.; Hopper, J.L.; Loman, N.; Olsson, H.; Johannsson, O.; Borg, A.; et al. Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case Series unselected for family history: A combined analysis of 22 studies. Am. J. Hum. Genet. 2003, 72, 1117–1130. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Parmigiani, G. Meta-analysis of BRCA1 and BRCA2 penetrance. J. Clin. Oncol. 2007, 25, 1329–1333. [Google Scholar] [CrossRef] [PubMed]
- Domchek, S.M.; Friebel, T.M.; Singer, C.F.; Evans, D.G.; Lynch, H.T.; Isaacs, C.; Garber, J.E.; Neuhausen, S.L.; Matloff, E.; Eeles, R.; et al. Association of risk-reducing surgery in BRCA1 or BRCA2 mutation carriers with cancer risk and mortality. JAMA 2010, 304, 967–975. [Google Scholar] [CrossRef] [PubMed]
- Skytte, A.B.; Cruger, D.; Gerster, M.; Laenkholm, A.V.; Lang, C.; Brondum-Nielsen, K.; Andersen, M.K.; Sunde, L.; Kolvraa, S.; Gerdes, A.M. Breast cancer after bilateral risk-reducing mastectomy. Clin. Genet. 2011, 79, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.G.; Gaarenstroom, K.N.; Stirling, D.; Shenton, A.; Maehle, L.; Dorum, A.; Steel, M.; Lalloo, F.; Apold, J.; Porteous, M.E.; et al. Screening for familial ovarian cancer: Poor survival of BRCA1/2 related cancers. J. Med. Genet. 2009, 46, 593–597. [Google Scholar] [CrossRef] [PubMed]
- Nelson, H.D.; Smith, M.E.; Griffin, J.C.; Fu, R. Use of medications to reduce risk for primary breast cancer: A systematic review for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2013, 158, 604–614. [Google Scholar] [CrossRef] [PubMed]
- Moyer, V.A. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer in women: U.S. Preventive Services Task Force recommendation statement. Ann. Intern. Med. 2014, 160, 271–281. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. Available online: www.nccn.org (accessed on 31 August 2015).
- U.S. Preventive Services Task Force. Genetic risk assessment and BRCA mutation testing for breast and ovarian cancer susceptibility: Recommendation statement. Ann. Intern. Med. 2005, 143, 355–361. [Google Scholar]
- Frieden, T.R. Six components necessary for effective public health program implementation. Am. J. Public Health 2014, 104, 17–22. [Google Scholar] [CrossRef] [PubMed]
- St Pierre, J.; Bach, J.; Duquette, D.; Oehlke, K.; Nystrom, R.; Silvey, K.; Zlot, A.; Giles, R.; Johnson, J.; Anders, H.M.; et al. Strategies, actions, and outcomes of pilot state programs in public health genomics, 2003–2008. Prev. Chronic Dis. 2014, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- 2020 Topics and Objectives-Objectives A–Z. Available online: http://www.healthypeople.gov/2020/topicsobjectives2020/default (accessed on 31 August 2015).
- Patient Protection and Affordable Care Act. Available online: http://legcounsel.house.gov/Comps/Patient%20Protection%20And%20Affordable%20Care%20Act.pdf (accessed on 31 August 2015).
- Science Impact. Available online: http://www.cdc.gov/od/science/impact/ (accessed on 31 August 2015).
- The Institute of Medicine: What Makes It Great? President’s Address, Institute of Medicine Annual Meeting. Available online: http://iom.nationalacademies.org/~/media/Files/About%20the%20IOM/PresidentsAddress2013.pdf (accessed on 31 August 2015).
- Advisory Committee on Breast Cancer in Young Women (ACBCYW). Enhancing Breast Cancer Genomics Best Practices and Policies in the State of Michigan. Available online: http://www.michigan.gov/documents/mdch/ACBCYW2014_SM_jvb_477688_7.pdf (accessed on 31 August 2015).
- Hereditary Breast and Ovarian Cancer: Is Your Patient at Risk? Available online: http://www.nchpeg.org/hboc/ (accessed on 31 August 2015).
- Inside Knowledge: Get the Facts About Gynecologic Cancer. Available online: http://www.cdc.gov/cancer/knowledge/ (accessed on 31 August 2015).
- Breast Cancer in Young Women. Available online: http://www.cdc.gov/cancer/breast/young_women/index.htm (accessed on 31 August 2015).
- Bellcross, C. Further development and evaluation of a breast/ovarian cancer genetics referral screening tool. Genet. Med. 2010. [Google Scholar] [CrossRef] [PubMed]
- Bellcross, C.A.; Lemke, A.A.; Pape, L.S.; Tess, A.L.; Meisner, L.T. Evaluation of a breast/ovarian cancer genetics referral screening tool in a mammography population. Genet. Med. 2009, 11, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Breast Cancer Genetics Referral Screening Tool (B-RSTTM). Available online: https://www.breastcancergenescreen.org/ (accessed on 31 August 2015).
- Brannon Traxler, L.; Martin, M.L.; Kerber, A.S.; Bellcross, C.A.; Crane, B.E.; Green, V.; Matthews, R.; Paris, N.M.; Gabram, S.G. Implementing a screening tool for identifying patients at risk for hereditary breast and ovarian cancer: A statewide initiative. Ann. Surg. Oncol. 2014, 21, 3342–3347. [Google Scholar] [CrossRef] [PubMed]
- Cox, S.; George, R.; Soule, J.; Parkman, A.; Kovak, K.; Patil, M.; Shipley, D. Increasing awareness of hereditary breast and ovarian cancer syndrome (HBOC) among survivors and their providers through the Oregon State Cancer Registry (OSCaR). In Proceedings of the 7th Biennial Survivorship Conference, Atlanta, GA, USA, 7 July 2014.
- Shipley, D.; Patil, M.; Soule, J.; Evangelista, S.; Cox, S.; George, R.; Parkman, A.; Kovak, K. Oregon’s approach to increasing awareness of hereditary breast and ovarian cancer syndrome (HBOC) among clinicians and patients. In Proceedings of the NAACCR Annual Conference, Ottawa, ON, Canada, 24 June 2014.
- H.R.83—Consolidated and Further Continuing Appropriations Act. Available online: https://www.congress.gov/bill/113th-congress/house-bill/83 (accessed on 31 August 2015).
- Genomics Application Toolkit. Available online: http://www.cdc.gov/genomics/implementation/toolkit/tier1.htm (accessed on 31 August 2015).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trivers, K.F.; Rodriguez, J.L.; Cox, S.L.; Crane, B.E.; Duquette, D. The Activities and Impact of State Programs to Address Hereditary Breast and Ovarian Cancer, 2011–2014. Healthcare 2015, 3, 948-963. https://doi.org/10.3390/healthcare3040948
Trivers KF, Rodriguez JL, Cox SL, Crane BE, Duquette D. The Activities and Impact of State Programs to Address Hereditary Breast and Ovarian Cancer, 2011–2014. Healthcare. 2015; 3(4):948-963. https://doi.org/10.3390/healthcare3040948
Chicago/Turabian StyleTrivers, Katrina F., Juan L. Rodriguez, Summer L. Cox, Barbara E. Crane, and Debra Duquette. 2015. "The Activities and Impact of State Programs to Address Hereditary Breast and Ovarian Cancer, 2011–2014" Healthcare 3, no. 4: 948-963. https://doi.org/10.3390/healthcare3040948
APA StyleTrivers, K. F., Rodriguez, J. L., Cox, S. L., Crane, B. E., & Duquette, D. (2015). The Activities and Impact of State Programs to Address Hereditary Breast and Ovarian Cancer, 2011–2014. Healthcare, 3(4), 948-963. https://doi.org/10.3390/healthcare3040948





