Analyzing Overlaid Foreign Objects in Chest X-rays—Clinical Significance and Artificial Intelligence Tools
Abstract
1. Introduction
2. Inclusion/Exclusion Criteria
- Search keywords: (chest X-ray OR chest radiograph) AND (foreign object detection) OR object detection.
- Search spaces: PubMed and Web of Science.
3. Clinical Significance
4. AI-Guided Tools for NBFO and BFO
5. Data Description
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Raoof, S.; Feigin, D.; Sung, A.; Raoof, S.; Irugulpati, L.; Rosenow III, E.C. Interpretation of plain chest roentgenogram. Chest 2012, 141, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Zohora, F.T.; Santosh, K. Circular foreign object detection in chest x-ray images. In Proceedings of the International Conference on Recent Trends in Image Processing and Pattern Recognition, Bidar, India, 16–17 December 2016; 2016; pp. 391–401. [Google Scholar]
- El-Baz, A.; Beache, G.M.; Gimel’farb, G.; Suzuki, K.; Okada, K.; Elnakib, A.; Soliman, A.; Abdollahi, B. Computer-aided diagnosis systems for lung cancer: Challenges and methodologies. Int. J. Biomed. Imaging 2013, 2013, 942353. [Google Scholar] [CrossRef] [PubMed]
- Tuberculosis (TB). Available online: https://www.who.int/news-room/fact-sheets/detail/tuberculosis (accessed on 25 July 2022).
- Cancer. Available online: https://www.who.int/health-topics/cancer#tab=tab_1 (accessed on 25 July 2022).
- Antani, S. Automatic x-ray screening for tuberculosis. SPIE Newsroom. 2015. Available online: http://www.spie.org/x115742.xml (accessed on 3 January 2016).
- Santosh, K.; Vajda, S.; Antani, S.; Thoma, G.R. Edge map analysis in chest X-rays for automatic pulmonary abnormality screening. Int. J. Comput. Assist. Radiol. Surg. 2016, 11, 1637–1646. [Google Scholar] [CrossRef] [PubMed]
- Baltruschat, I.M.; Nickisch, H.; Grass, M.; Knopp, T.; Saalbach, A. Comparison of deep learning approaches for multi-label chest X-ray classification. Sci. Rep. 2019, 9, 6381. [Google Scholar] [CrossRef] [PubMed]
- Vajda, S.; Karargyris, A.; Jaeger, S.; Santosh, K.; Candemir, S.; Xue, Z.; Antani, S.; Thoma, G. Feature selection for automatic tuberculosis screening in frontal chest radiographs. J. Med. Syst. 2018, 42, 1–11. [Google Scholar] [CrossRef]
- WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/#:~:text=Globally%2C%20as%20of%206%3A45pm,vaccine%20doses%20have%20been%20administered (accessed on 25 July 2022).
- Santosh, K.; Ghosh, S.; GhoshRoy, D. Deep learning for COVID-19 screening using chest x-rays in 2020: A systematic review. Int. J. Pattern Recognit. Artif. Intell. 2022, 36, 2252010. [Google Scholar] [CrossRef]
- Santosh, K.; Ghosh, S. COVID-19 imaging tools: How big data is big? J. Med. Syst. 2021, 45, 1–8. [Google Scholar] [CrossRef]
- Santosh, K. AI-driven tools for coronavirus outbreak: Need of active learning and cross-population train/test models on multitudinal/multimodal data. J. Med. Syst. 2020, 44, 1–5. [Google Scholar] [CrossRef]
- Mahbub, M.K.; Biswas, M.; Gaur, L.; Alenezi, F.; Santosh, K. Deep features to detect pulmonary abnormalities in chest X-rays due to infectious diseaseX: COVID-19, pneumonia, and tuberculosis. Inf. Sci. 2022, 592, 389–401. [Google Scholar] [CrossRef]
- Santosh, K.; Antani, S. Automated chest X-ray screening: Can lung region symmetry help detect pulmonary abnormalities? IEEE Trans. Med. Imaging 2017, 37, 1168–1177. [Google Scholar] [CrossRef]
- Brown, J.C.; Murray, K.F.; Javid, P.J. Hidden attraction: A menacing meal of magnets and batteries. J. Emerg. Med. 2012, 43, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Pugmire, B.S.; Lin, T.K.; Pentiuk, S.; de Alarcon, A.; Hart, C.K.; Trout, A.T. Imaging button battery ingestions and insertions in children: A 15-year single-center review. Pediatr. Radiol. 2017, 47, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, S.; Cano, I.; Benavent, M.I.; Gómez, A. Severe esophageal injuries caused by accidental button battery ingestion in children. J. Emergencies Trauma Shock. 2014, 7, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Gummin, D.D.; Mowry, J.B.; Spyker, D.A.; Brooks, D.E.; Fraser, M.O.; Banner, W. 2016 annual report of the american association of poison control centers’ national poison data system (NPDS): 34th annual report. Clin. Toxicol. 2017, 55, 1072–1254. [Google Scholar] [CrossRef] [PubMed]
- Meyer, T.J.; Grunz, J.-P.; Taeger, J.; Rak, K.; Hagen, R.; Hackenberg, S.; Voelker, J.; Scherzad, A. Systematic analysis of button batteries’, euro coins’, and disk magnets’ radiographic characteristics and the implications for the differential diagnosis of round radiopaque foreign bodies in the esophagus. Int. J. Pediatr. Otorhinolaryngol. 2020, 132, 109917. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.J.; Kubicka, R.A.; Smith, C. Evaluation of cardiopulmonary devices on chest radiographs: Digital vs analog radiographs. Am. J. Roentgenol. 1989, 153, 1165–1168. [Google Scholar] [CrossRef] [PubMed]
- Godoy, M.C.; Leitman, B.S.; De Groot, P.M.; Vlahos, I.; Naidich, D.P. Chest radiography in the ICU: Part 2, Evaluation of cardiovascular lines and other devices. Am. J. Roentgenol. 2012, 198, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Godoy, M.C.; Leitman, B.S.; De Groot, P.M.; Vlahos, I.; Naidich, D.P. Chest radiography in the ICU: Part 1, Evaluation of airway, enteric, and pleural tubes. Am. J. Roentgenol. 2012, 198, 563–571. [Google Scholar] [CrossRef]
- Jennings, P.; Padley, S.P.; Hansell, D.M. Portable chest radiography in intensive care: A comparison of computed and conventional radiography. Br. J. Radiol. 1992, 65, 852–856. [Google Scholar] [CrossRef]
- Grier, D.; Cook, P.; Hartnell, G. Chest radiographs after permanent pacing. Are they really necessary? Clin. Radiol. 1990, 42, 244–249. [Google Scholar] [CrossRef]
- Murthy, P.; Ingle, V.; George, E.; Ramakrishna, S.; Shah, F.A. Sharp foreign bodies in the tracheobronchial tree. Am. J. Otolaryngol. 2001, 22, 154–156. [Google Scholar] [CrossRef] [PubMed]
- Orgill, R.D.; Pasic, T.R.; Peppler, W.W.; Hoffman, M.D. Radiographic evaluation of aspirated metallic foil foreign bodies. Ann. Otol. Rhinol. Laryngol. 2005, 114, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Sink, J.R.; Kitsko, D.J.; Mehta, D.K.; Georg, M.W.; Simons, J.P. Diagnosis of pediatric foreign body ingestion: Clinical presentation, physical examination, and radiologic findings. Ann. Otol. Rhinol. Laryngol. 2016, 125, 342–350. [Google Scholar] [CrossRef]
- Fisher, J.; Mittal, R.; Hill, S.; Wulkan, M.L.; Clifton, M.S. Yield of chest radiography after removal of esophageal foreign bodies. Pediatrics 2013, 131, e1497–e1501. [Google Scholar] [CrossRef] [PubMed]
- Denney, W.; Ahmad, N.; Dillard, B.; Nowicki, M.J. Children will eat the strangest things: A 10-year retrospective analysis of foreign body and caustic ingestions from a single academic center. Pediatr. Emerg. Care 2012, 28, 731–734. [Google Scholar] [CrossRef] [PubMed]
- Huyett, P.; Shaffer, A.; Flom, L.; Simons, J.P.; Jabbour, N. Accuracy of chest X-Ray measurements of pediatric esophageal coins. Int. J. Pediatr. Otorhinolaryngol. 2018, 113, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Raney, L.H.; Losek, J.D. Child with esophageal coin and atypical radiograph. J. Emerg. Med. 2008, 34, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Tander, B.; Yazici, M.; Rizalar, R.; Ariturk, E.; Ayyildiz, S.H.; Bernay, F. Coin ingestion in children: Which size is more risky? J. Laparoendosc. Adv. Surg. Tech. 2009, 19, 241–243. [Google Scholar] [CrossRef] [PubMed]
- Rybojad, B.; Niedzielska, G.; Niedzielski, A.; Rudnicka-Drozak, E.; Rybojad, P. Esophageal foreign bodies in pediatric patients: A thirteen-year retrospective study. Sci. World J. 2012, 2012, 102642. [Google Scholar] [CrossRef]
- Schlesinger, A.E.; Crowe, J.E. Sagittal orientation of ingested coins in the esophagus in children. Am. J. Roentgenol. 2011, 196, 670–672. [Google Scholar] [CrossRef]
- Ullal, A.; Mundra, R.; Gupta, Y.; Mishra, S. Virtual bronchoscopy: Highly sensitive time and life saving investigation in the diagnosis of foreign body aspiration—Our experience. Indian J. Otolaryngol. Head Neck Surg. 2019, 71, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Morrier, J.; Chrétien, M.; Martin, A.-G.; Vigneault, É.; Beaulieu, L. A more efficient, radiation-free alternative to systematic chest x-ray for the detection of embolized seeds to the lung. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 1052–1056. [Google Scholar] [CrossRef] [PubMed]
- Kero, P.; Puhakka, H.; Erkinjuntti, M.; Iisalo, E.; Vilkki, P. Foreign body in the airways of children. Int. J. Pediatr. Otorhinolaryngol. 1984, 6, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Candemir, S.; Antani, S.; Long, L.R.; Jaeger, S.; Demner-Fushman, D.; Thoma, G.R. Foreign object detection in chest X-rays. In Proceedings of the 2015 IEEE international conference on bioinformatics and biomedicine (BIBM), Washington, DC, USA, 9–12 November 2015; pp. 956–961. [Google Scholar]
- Zohora, F.T.; Santosh, K. Foreign circular element detection in chest x-rays for effective automated pulmonary abnormality screening. Int. J. Comput. Vis. Image Process. 2017, 7, 36–49. [Google Scholar] [CrossRef]
- Hogeweg, L.; Sánchez, C.I.; Melendez, J.; Maduskar, P.; Story, A.; Hayward, A.; van Ginneken, B. Foreign object detection and removal to improve automated analysis of chest radiographs. Med. Phys. 2013, 40, 071901. [Google Scholar] [CrossRef]
- Schultheiss, M.; Schober, S.A.; Lodde, M.; Bodden, J.; Aichele, J.; Müller-Leisse, C.; Renger, B.; Pfeiffer, F.; Pfeiffer, D. A robust convolutional neural network for lung nodule detection in the presence of foreign bodies. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Deshpande, H.; Harder, T.; Saalbach, A.; Sawarkar, A.; Buelow, T. Detection of foreign objects in chest radiographs using deep learning. In Proceedings of the 2020 IEEE 17th International Symposium on Biomedical Imaging Workshops (ISBI Workshops), Iowa City, IA, USA, 4 April 2020; pp. 1–4. [Google Scholar]
- Santosh, K.; Dhar, M.K.; Rajbhandari, R.; Neupane, A. Deep neural network for foreign object detection in chest X-rays. In Proceedings of the 2020 IEEE 33rd International symposium on computer-based medical systems (CBMS), Rochester, MN, USA, 28–30 July 2020; 2020; pp. 538–541. [Google Scholar]
- Santosh, K.; Roy, S.; Allu, S. Generic Foreign Object Detection in Chest X-rays. In Proceedings of the Recent Trends in Image Processing and Pattern Recognition RTIP2R 2021 Communications in Computer and Information Science, TX, USA, 1–2 December 2022. [Google Scholar] [CrossRef]
- Demner-Fushman, D.; Kohli, M.D.; Rosenman, M.B.; Shooshan, S.E.; Rodriguez, L.; Antani, S.; Thoma, G.R.; McDonald, C.J. Preparing a collection of radiology examinations for distribution and retrieval. J. Am. Med. Inform. Assoc. 2016, 23, 304–310. [Google Scholar] [CrossRef]
- Demner-Fushman, D.; Antani, S.; Simpson, M.; Thoma, G.R. Design and development of a multimodal biomedical information retrieval system. J. Comput. Sci. Eng. 2012, 6, 168–177. [Google Scholar] [CrossRef]
- Story, A.; Murad, S.; Roberts, W.; Verheyen, M.; Hayward, A.C. Tuberculosis in London: The importance of homelessness, problem drug use and prison. Thorax 2007, 62, 667–671. [Google Scholar] [CrossRef]
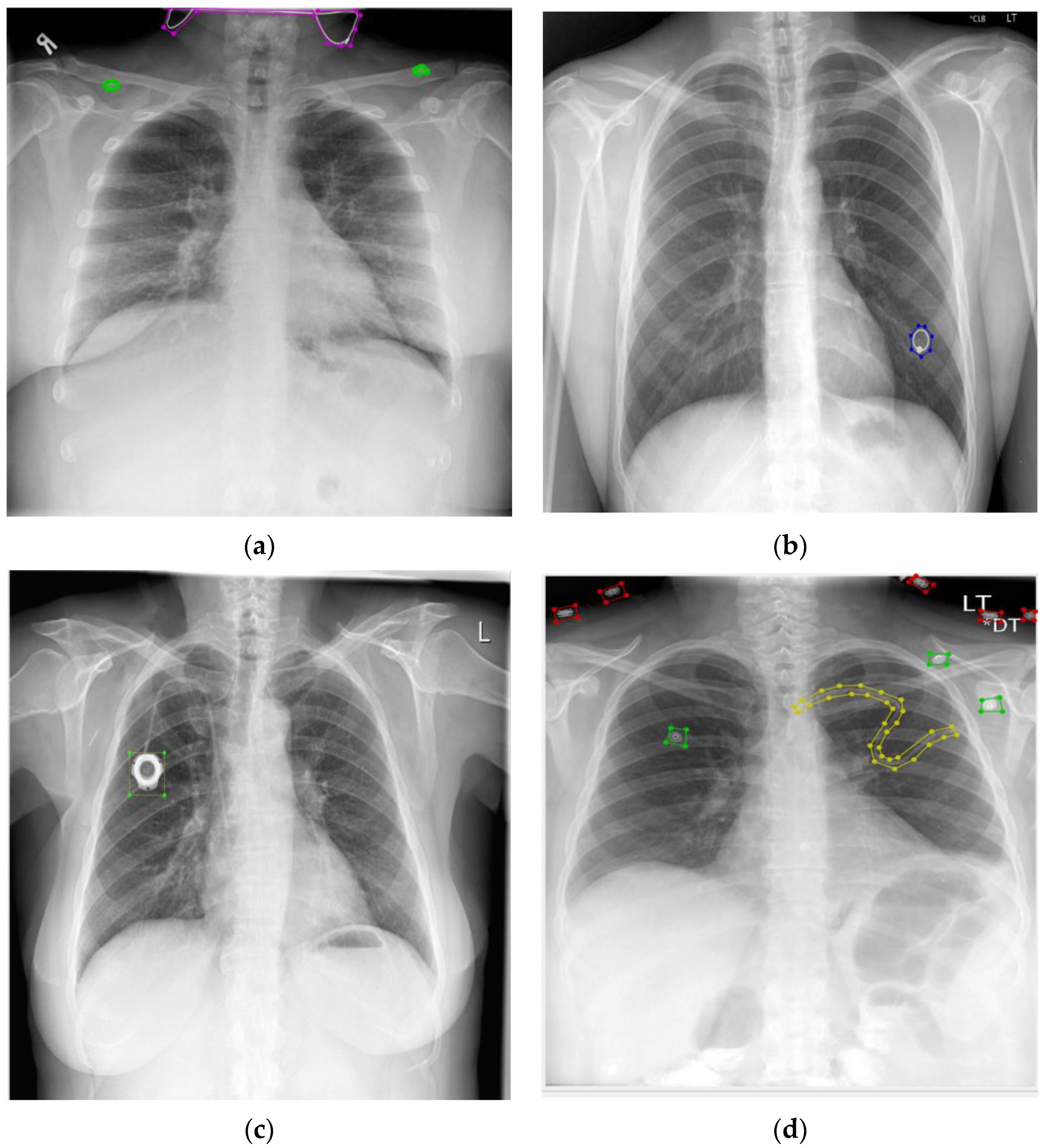
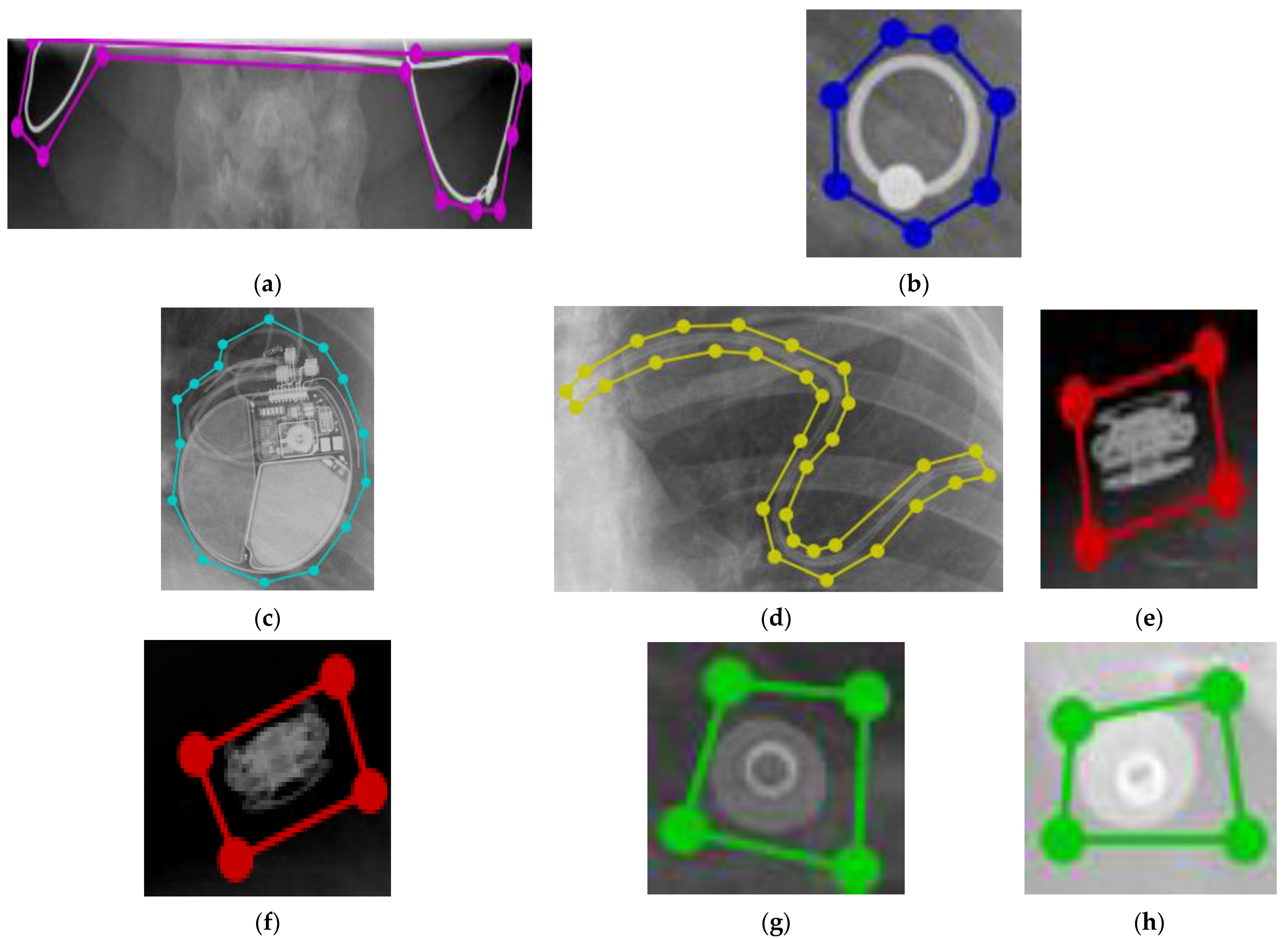
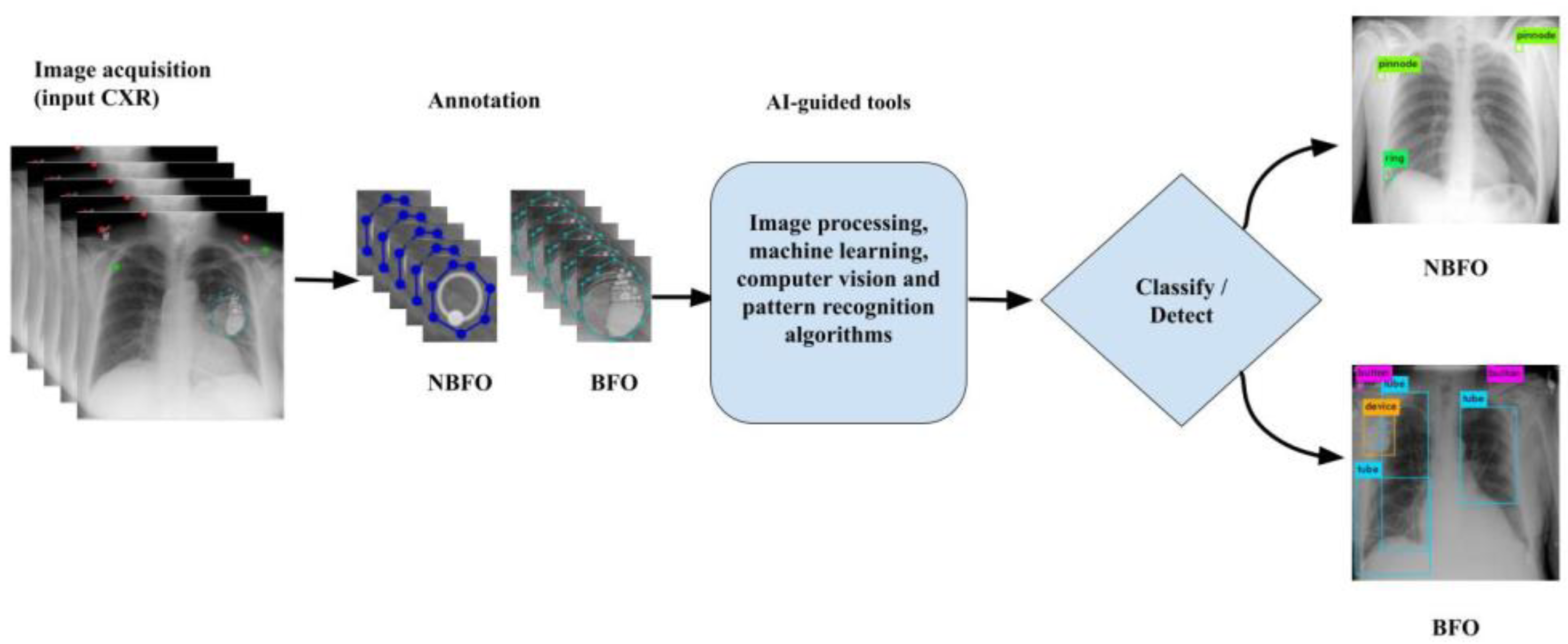
| Authors | Foreign Objects | Method | Dataset (Case) (Subjects) |
|---|---|---|---|
| Brown et al. (2012) [16] | Magnets and batteries | Case Study | 1 Subject |
| Pugmire et al. (2016) [17] | Button Battery | Review | 276 cases |
| Fuentes et al. (2014) [18] | Button Battery | Review and Case Study | 3 Cases and 29 Review |
| Meyer et al. (2020)) [20] | Button Battery, Coins, Disk magnets | Case Study | 20 subjects |
| Thompson et al. (1989) [21] | Cardiopulmonary devices | Digital and Analog | 40 subjects |
| Godoy et al. (2012) [22] | Catheters, Pacemakers, Automatic implantable cardioverter defibrillators, intra-aortic counter pulsation balloon pump, ventricular assist devices | Case Study | - |
| Godoy et al. (2012) [23] | Endotracheal and tracheostomy tubes, Chest tubes, and Nasogastric and Nasoenteric tubes | Case Study | - |
| Jennings et al. (1992) [24] | Cardiopulmonary devices | Phosphor plate system | 50 subjects |
| Grier et al. (1990) [25] | Pacemaker | Case Study | 600 subjects |
| Murthy et al. (2001) [26] | Scarf pin | Case Study | 6 subjects |
| Orgill et al. (2018) [27] | Multi-layer metallic candy wrappers | Duel-energy radio-graph | 1 subject |
| Huyett et al. (2018)) [31] | Coins | Case Study | 4 subjects |
| Raney, Losek (2007)) [32] | Coins | Case Study | 1 subject |
| Schlesinger, Crowe (2011) [35] | Coins | Case Study | 8 subjects |
| Tander et al. (2009) [33] | Coins | Case Study | 62 subjects |
| Ullal et al. (2018) [36] Morrier et al. (2010) [37] | Foreign body | Case Study | 150 subjects |
| Seed-migration | A gamma scintillation | 737 Subjects | |
| Kero et al. (1983) [38] Rybojad et al. (2012) [34] | Foreign bodies | Case Study | 57 subjects |
| Esophageal foreign bodies | Chi-square test | 192 subjects |
| Authors | NBFO, BFO | Methods | Datasets (Size of Images) | Performance (in %) | ||
|---|---|---|---|---|---|---|
| Precision | Recall | F1-Score | ||||
| Xue et al. (2015) [39] | Buttons | Hand-crafted features | 505 | 0.84 | 0.88 | - |
| Zohora, Santosh (2017) [2] | Buttons, medical devices | Hand-crafted features | 50 | 100 | 100 | - |
| Zohora, Santosh (2017) [40] | Circular (buttons, medical devices) | Hand-crafted features | 400 | 0.96 | 0.90 | 0.92 |
| Hogeweg et al. (2013) [41] | Buttons, brassier clips, jewelry, or pacemakers and wires | kNN classifier | 257 | 0.949 (pixel level value) | - | - |
| Schulthesiss et al. [42] | Nodule detection | CNN (RatinaNet) | 411 | - | 0.87 (ROC) | - |
| Santosh et al. (2020) [44] | Circle like (e.g., coins/buttons) | R-CNN | 400 | 0.97 | 0.90 | 0.93 |
| Santosh et al. (2022) [45] | Buttons, coins, ring, pinnode, medical devices, tube | YOLOv4 | 400 | 0.85 | 0.93 | 0.89 |
| Dataset | Size | Authors |
|---|---|---|
| US NLM, National Institute of Health (Indiana Dataset) | 278 | Xue et al. (2015) [39] |
| US NLM, National Institute of Health | 50 | Zohora, Santosh (2017) [2] |
| US NLM, National Institute of Health | 400 | Zohora, Santosh (2017) [40] Santosh et al. (2020,2022) [44,45] |
| Digital Diagnost Trixel, Philips Healthcare, the Netherlands | 257 | Hogeweg et al. (2013) [41] |
| Japanese Society of Radiological Technology (JSRT) | 411 | Schulthesiss et al. [42] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roy, S.; Santosh, K. Analyzing Overlaid Foreign Objects in Chest X-rays—Clinical Significance and Artificial Intelligence Tools. Healthcare 2023, 11, 308. https://doi.org/10.3390/healthcare11030308
Roy S, Santosh K. Analyzing Overlaid Foreign Objects in Chest X-rays—Clinical Significance and Artificial Intelligence Tools. Healthcare. 2023; 11(3):308. https://doi.org/10.3390/healthcare11030308
Chicago/Turabian StyleRoy, Shotabdi, and KC Santosh. 2023. "Analyzing Overlaid Foreign Objects in Chest X-rays—Clinical Significance and Artificial Intelligence Tools" Healthcare 11, no. 3: 308. https://doi.org/10.3390/healthcare11030308
APA StyleRoy, S., & Santosh, K. (2023). Analyzing Overlaid Foreign Objects in Chest X-rays—Clinical Significance and Artificial Intelligence Tools. Healthcare, 11(3), 308. https://doi.org/10.3390/healthcare11030308







