Cost-Effectiveness of Glucosamine in Osteoarthritis Treatment: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Selection Criteria
- The interventions discussed used glucosamine as the non-combined formulation;
- The paper was written in English and either evaluated the cost-effectiveness or contained any other type of economic evaluation;
- The main topic of the paper was osteoarthritis therapy with a viable duration;
- The paper contained specific information reporting the ICER value;
- The research presented a clear conclusion as to whether glucosamine was cost-effective or not.
- The study combined glucosamine with other compounds;
- The study was not available to read in the English language;
- The study did not discuss osteoarthritis treatment, or did not focus on glucosamine;
- The study did not relate to OA treatments;
- The study contained an unclear statement or lacked information about the ICER.
2.3. Data Extraction
2.4. Quality Assessment of Selected Articles
3. Results
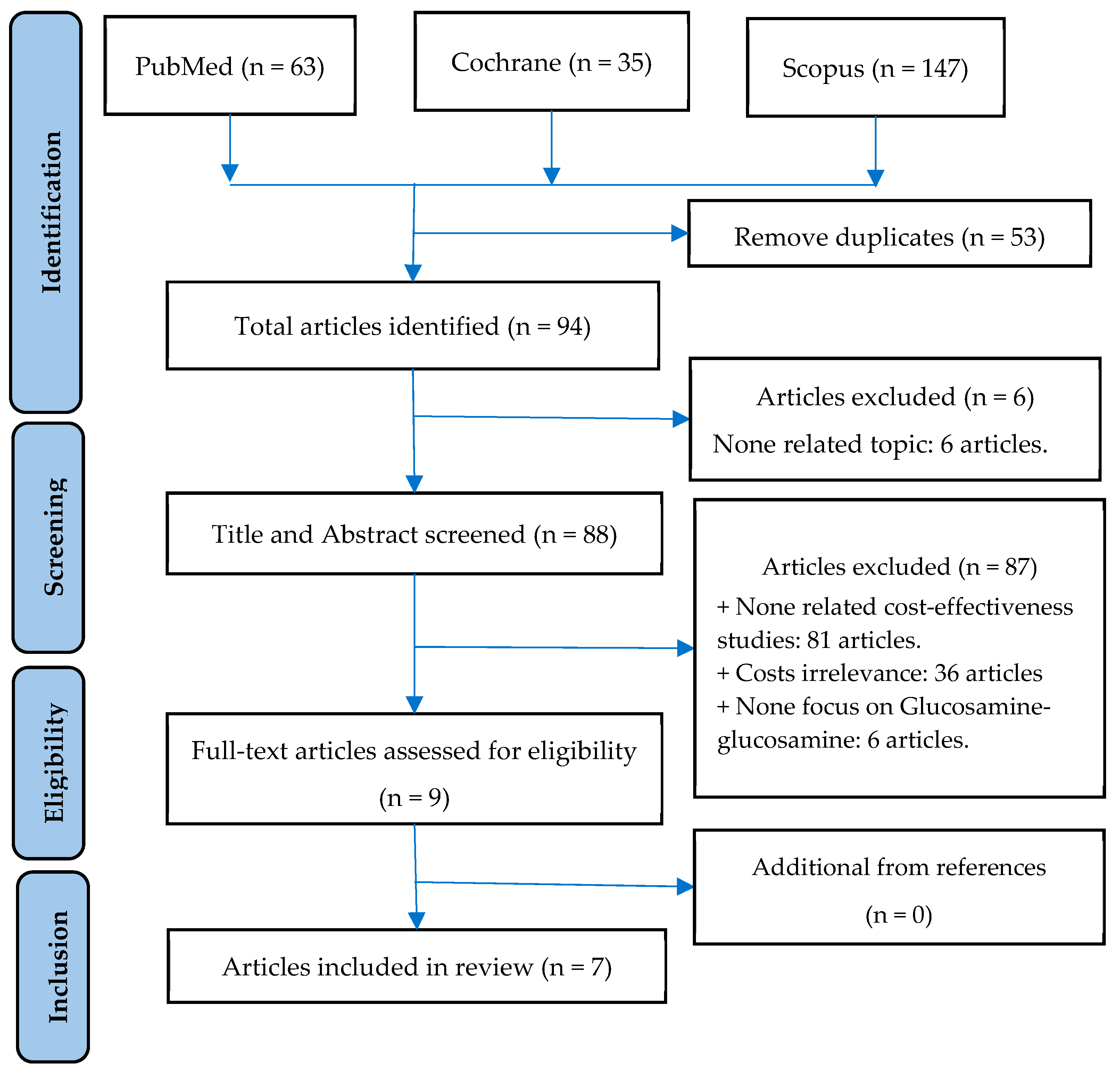
3.1. Study Selection Process
3.2. Characteristics of Included Studies
3.3. Quality Assessment by QHES Instrument
3.4. Keypoint Data Related to Cost-Effectiveness
4. Discussion
5. Conclusions
6. Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Litwic, A.; Edwards, M.H.; Dennison, E.M.; Cooper, C. Epidemiology and burden of osteoarthritis. Br. Med. Bull. 2013, 105, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Hutton, C.W. Osteoarthritis: The cause not result of joint failure? Ann. Rheum. Dis. 1989, 48, 958–961. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393, 1745–1759. [Google Scholar] [CrossRef] [PubMed]
- Kolasinski, S.L.; Neogi, T.; Hochberg, M.C.; Oatis, C.; Guyatt, G.; Block, J.; Callahan, L.; Copenhaver, C.; Dodge, C.; Felson, D.; et al. 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee. Arthritis Rheumatol. 2020, 72, 220–233. [Google Scholar] [CrossRef] [PubMed]
- Institute_for_Health_Metrics_and_Evaluation. Global Burden of Disease Study 2019 (GBD 2019) Data Resources. Available online: https://ghdx.healthdata.org/gbd-2019 (accessed on 17 May 2023).
- Institute_for_Health_Metrics_and_Evaluation. Global Burden of Disease Study 2019 (GBD 2019) Results. Osteoarthritis-Level 3 Cause. Available online: https://www.healthdata.org/results/gbd_summaries/2019/osteoarthritis-level-3-cause#:~:text=Summary%20Osteoarthritis%20(OA)%20resulted%20in,%25%20of%20OA%20YLDs%2C%20respectively (accessed on 17 May 2023).
- Neogi, T. The epidemiology and impact of pain in osteoarthritis. Osteoarthr. Cartil. 2013, 21, 1145–1153. [Google Scholar] [CrossRef] [PubMed]
- Fransen, M.; Bridgett, L.; March, L.; Hoy, D.; Penserga, E.; Brooks, P. The epidemiology of osteoarthritis in Asia. Int. J. Rheum. Dis. 2011, 14, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.A.; Goodman, R.A.; Holtzman, D.; Posner, S.F.; Northridge, M.E. Aging in the United States: Opportunities and challenges for public health. Am. J. Public Health 2012, 102, 393–395. [Google Scholar] [CrossRef] [PubMed]
- Christensen, K.; Doblhammer, G.; Rau, R.; Vaupel, J.W. Ageing populations: The challenges ahead. Lancet 2009, 374, 1196–1208. [Google Scholar] [CrossRef]
- Wortley, D.; An, J.Y.; Heshmati, A. Tackling the Challenge of the Aging Society: Detecting and Preventing Cognitive and Physical Decline through Games and Consumer Technologies. Healthc. Inform. Res. 2017, 23, 87–93. [Google Scholar] [CrossRef][Green Version]
- Safiri, S.; Kolahi, A.A.; Smith, E.; Hill, C.; Bettampadi, D.; Mansournia, M.A.; Hoy, D.; Ashrafi-Asgarabad, A.; Sepidarkish, M.; Almasi-Hashiani, A.; et al. Global, regional and national burden of osteoarthritis 1990-2017: A systematic analysis of the Global Burden of Disease Study 2017. Ann. Rheum. Dis. 2020, 79, 819–828. [Google Scholar] [CrossRef]
- Diseases, G.B.D.; Injuries, C. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Zhang, Y.; Jordan, J.M. Epidemiology of osteoarthritis. Clin. Geriatr. Med. 2010, 26, 355–369. [Google Scholar] [CrossRef] [PubMed]
- Luksameesate, P.; Tanavalee, A.; Taychakhoonavudh, S. An economic evaluation of knee osteoarthritis treatments in Thailand. Front. Pharmacol. 2022, 13, 926431. [Google Scholar] [CrossRef] [PubMed]
- Minh Hoa, T.T.; Darmawan, J.; Chen, S.L.; Van Hung, N.; Thi Nhi, C.; Ngoc An, T. Prevalence of the rheumatic diseases in urban Vietnam: A WHO-ILAR COPCORD study. J. Rheumatol. 2003, 30, 2252–2256. [Google Scholar] [PubMed]
- Garstang, S.V.; Stitik, T.P. Osteoarthritis: Epidemiology, risk factors, and pathophysiology. Am. J. Phys. Med. Rehabil. 2006, 85 (Suppl. 11), S2–S11; quiz S12–S14. [Google Scholar] [CrossRef] [PubMed]
- Allen, K.D.; Golightly, Y.M. State of the evidence. Curr. Opin. Rheumatol. 2015, 27, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Reyes, C.; Leyland, K.M.; Peat, G.; Cooper, C.; Arden, N.K.; Prieto-Alhambra, D. Association Between Overweight and Obesity and Risk of Clinically Diagnosed Knee, Hip, and Hand Osteoarthritis: A Population-Based Cohort Study. Arthritis Rheumatol. 2016, 68, 1869–1875. [Google Scholar] [CrossRef]
- Zamri, N.A.A.; Harith, S.; Yusoff, N.A.M.; Hassan, N.M.; Ong, Y.Q. Prevalence, Risk Factors and Primary Prevention of Osteoarthritis in Asia: A Scoping Review. Elder. Health J. 2019, 5, 19–31. [Google Scholar]
- Bijlsma, J.W.; Knahr, K. Strategies for the prevention and management of osteoarthritis of the hip and knee. Best Pract. Res. Clin. Rheumatol. 2007, 21, 59–76. [Google Scholar] [CrossRef]
- Andrianakos, A.A.; Kontelis, L.K.; Karamitsos, D.G.; Aslanidis, S.I.; Georgountzos, A.I.; Kaziolas, G.O.; Pantelidou, K.V.; Vafiadou, E.V.; Dantis, P.C.; Group, E.S. Prevalence of symptomatic knee, hand, and hip osteoarthritis in Greece. The ESORDIG study. J. Rheumatol. 2006, 33, 2507–2513. [Google Scholar]
- Australian Bureau of Statistics. National Health Survey: Summary of Results; Australian Bureau of Statistics: Canberra, Australia, 1995.
- Segal, L.; Day, S.E.; Chapman, A.B.; Osborne, R.H. Can we reduce disease burden from osteoarthritis? Med. J. Aust. 2004, 180 (Suppl. 5), S11–S17. [Google Scholar] [CrossRef] [PubMed]
- Cui, A.; Li, H.; Wang, D.; Zhong, J.; Chen, Y.; Lu, H. Global, regional prevalence, incidence and risk factors of knee osteoarthritis in population-based studies. EClinicalMedicine 2020, 29–30, 100587. [Google Scholar] [CrossRef] [PubMed]
- March, L.M.; Bachmeier, C.J. Economics of osteoarthritis: A global perspective. Baillieres Clin. Rheumatol. 1997, 11, 817–834. [Google Scholar] [CrossRef] [PubMed]
- Leifer, V.P.; Katz, J.N.; Losina, E. The burden of OA-health services and economics. Osteoarthr. Cartil. 2022, 30, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Salmon, J.H.; Rat, A.C.; Sellam, J.; Michel, M.; Eschard, J.P.; Guillemin, F.; Jolly, D.; Fautrel, B. Economic impact of lower-limb osteoarthritis worldwide: A systematic review of cost-of-illness studies. Osteoarthr. Cartil. 2016, 24, 1500–1508. [Google Scholar] [CrossRef] [PubMed]
- Le Pen, C.; Reygrobellet, C.; Gerentes, I. Financial cost of osteoarthritis in France. The “COART” France study. Jt. Bone Spine 2005, 72, 567–570. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Prevalence of self-reported arthritis or chronic joint symptoms among adults--United States, 2001. MMWR Morb. Mortal. Wkly. Rep. 2002, 51, 948–950. [Google Scholar]
- Lawrence, R.C.; Helmick, C.G.; Arnett, F.C.; Deyo, R.A.; Felson, D.T.; Giannini, E.H.; Heyse, S.P.; Hirsch, R.; Hochberg, M.C.; Hunder, G.G.; et al. Estimates of the prevalence of arthritis and selected musculoskeletal disorders in the United States. Arthritis Rheumatol. 1998, 41, 778–799. [Google Scholar] [CrossRef]
- Yelin, E.; Weinstein, S.; King, T. The burden of musculoskeletal diseases in the United States. Semin. Arthritis Rheumatol. 2016, 46, 259–260. [Google Scholar] [CrossRef]
- Torio, C.M.; Moore, B.J. National Inpatient Hospital Costs: The Most Expensive Conditions by Payer, 2013. In Healthcare Cost and Utilization Project (HCUP) Statistical Briefs; HCUP: Rockville, MD, USA, 2006. [Google Scholar]
- Papanicolas, I.; Woskie, L.R.; Jha, A.K. Health Care Spending in the United States and Other High-Income Countries. JAMA 2018, 319, 1024–1039. [Google Scholar] [CrossRef]
- Chen, A.; Gupte, C.; Akhtar, K.; Smith, P.; Cobb, J. The Global Economic Cost of Osteoarthritis: How the UK Compares. Arthritis 2012, 2012, 698709. [Google Scholar] [CrossRef] [PubMed]
- Kotlarz, H.; Gunnarsson, C.L.; Fang, H.; Rizzo, J.A. Osteoarthritis and absenteeism costs: Evidence from US National Survey Data. J. Occup. Environ. Med. 2010, 52, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, M.C.; Altman, R.D.; April, K.T.; Benkhalti, M.; Guyatt, G.; McGowan, J.; Towheed, T.; Welch, V.; Wells, G.; Tugwell, P.; et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 2012, 64, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Nuki, G.; Moskowitz, R.W.; Abramson, S.; Altman, R.D.; Arden, N.K.; Bierma-Zeinstra, S.; Brandt, K.D.; Croft, P.; Doherty, M.; et al. OARSI recommendations for the management of hip and knee osteoarthritis: Part III: Changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthr. Cartil. 2010, 18, 476–499. [Google Scholar] [CrossRef] [PubMed]
- Bruyère, O.; Cooper, C.; Al-Daghri, N.M.; Dennison, E.M.; Rizzoli, R.; Reginster, J.Y. Inappropriate claims from non-equivalent medications in osteoarthritis: A position paper endorsed by the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO). Aging Clin. Exp. Res. 2018, 30, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Vetter, G. Lokale Therapie der Arthrosen mit Glukosaminen (Dona 200) [Topical therapy of arthroses with glucosamines (Dona 200)]. Munch Med. Wochenschr. 1969, 111, 1499–1502. [Google Scholar] [PubMed]
- Scholtissen, S.; Bruyère, O.; Neuprez, A.; Severens, J.L.; Herrero-Beaumont, G.; Rovati, L.; Hiligsmann, M.; Reginster, J.Y. Glucosamine sulphate in the treatment of knee osteoarthritis: Cost-effectiveness comparison with paracetamol. Int. J. Clin. Pract. 2010, 64, 756–762. [Google Scholar] [CrossRef]
- Terwee, C.B.; Jansma, E.P.; Riphagen, I.I.; Vet, H.C.W.d. Development of a methodological PubMed search filter for finding studies on measurement properties of measurement instruments. Qual. Life Res. 2009, 18, 1115–1123. [Google Scholar] [CrossRef]
- Shi, J.; Fan, K.; Yan, L.; Fan, Z.; Li, F.; Wang, G.; Liu, H.; Liu, P.; Yu, H.; Li, J.J.; et al. Cost Effectiveness of Pharmacological Management for Osteoarthritis: A Systematic Review. Appl. Health Econ. Health Policy 2022, 20, 351–370. [Google Scholar] [CrossRef]
- Prieto, L.; Sacristan, J.A. Problems and solutions in calculating quality-adjusted life years (QALYs). Health Qual. Life Outcomes 2003, 1, 80. [Google Scholar] [CrossRef]
- Ofman, J.J.; Sullivan, S.D.; Neumann, P.J.; Chiou, C.F.; Henning, J.M.; Wade, S.W.; Hay, J.W. Examining the value and quality of health economic analyses: Implications of utilizing the QHES. J. Manag. Care Pharm. 2003, 9, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Bruyere, O.; Detilleux, J.; Reginster, J.Y. Health Technology Assessment of Different Glucosamine Formulations and Preparations Currently Marketed in Thailand. Medicines 2023, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- Bruyère, O.; Detilleux, J.; Reginster, J.Y. Cost-Effectiveness Assessment of Different Glucosamines in Patients with Knee Osteoarthritis: A Simulation Model Adapted to Germany. Curr. Aging Sci. 2021, 14, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Bruyère, O.; Reginster, J.Y.; Honvo, G.; Detilleux, J. Cost-effectiveness evaluation of glucosamine for osteoarthritis based on simulation of individual patient data obtained from aggregated data in published studies. Aging Clin. Exp. Res. 2019, 31, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Black, C.; Clar, C.; Henderson, R.; MacEachern, C.; McNamee, P.; Quayyum, Z.; Royle, P.; Thomas, S. The clinical effectiveness of glucosamine and chondroitin supplements in slowing or arresting progression of osteoarthritis of the knee: A systematic review and economic evaluation. Health Technol. Assess. 2009, 13, 1–148. [Google Scholar] [CrossRef] [PubMed]
- Segal, L.D.S.; Chapman, A.; Osborne, R.H. Priority Setting in Osteoarthritis; ResearchGate: Berlin, Germany, 2004. [Google Scholar]
- Weinstein, M.C.; Stason, W.B. Foundations of cost-effectiveness analysis for health and medical practices. N. Engl. J. Med. 1977, 296, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Blumenschein, K.; Johannesson, M.; Yokoyama, K.K.; Freeman, P.R. Hypothetical versus real willingness to pay in the health care sector: Results from a field experiment. J. Health Econ. 2001, 20, 441–457. [Google Scholar] [CrossRef]
- Cohen, M.; Wolfe, R.; Mai, T.; Lewis, D. A randomized, double blind, placebo controlled trial of a topical cream containing glucosamine sulfate, chondroitin sulfate, and camphor for osteoarthritis of the knee. J. Rheumatol. 2003, 30, 523–528. [Google Scholar]
- Pavelká, K.; Gatterová, J.; Olejarová, M.; Machacek, S.; Giacovelli, G.; Rovati, L.C. Glucosamine sulfate use and delay of progression of knee osteoarthritis: A 3-year, randomized, placebo-controlled, double-blind study. Arch. Intern. Med. 2002, 162, 2113–2123. [Google Scholar] [CrossRef]
- Kim, D.D.; Wilkinson, C.L.; Pope, E.F.; Chambers, J.D.; Cohen, J.T.; Neumann, P.J. The influence of time horizon on results of cost-effectiveness analyses. Expert Rev. Pharmacoecon. Outcomes Res. 2017, 17, 615–623. [Google Scholar] [CrossRef]
- Iino, H.; Hashiguchi, M.; Hori, S. Estimating the range of incremental cost-effectiveness thresholds for healthcare based on willingness to pay and GDP per capita: A systematic review. PLoS ONE 2022, 17, e0266934. [Google Scholar] [CrossRef]
- Bertram, M.Y.; Lauer, J.A.; De Joncheere, K.; Edejer, T.; Hutubessy, R.; Kieny, M.P.; Hill, S.R. Cost-effectiveness thresholds: Pros and cons. Bull. World Health Organ. 2016, 94, 925–930. [Google Scholar] [CrossRef]
- Ii, M.I.A.; Nakamura, R. Considering the Costs and Benefits of Medical Care; National Institute of Public Health: Saitama, Japan, 2019. (In Japanese)
- Zhang, B.D.; Liang, Z.J.; Zhang, H.T.; He, M.T.; Li, D.S. Cost-effectiveness analysis on the treatment of knee osteoarthritis by glucosamine hydrochloride and glucosamine sulfate. Chin. J. Tissue Eng. Res. 2012, 16, 9867–9872. [Google Scholar]
| Number of Articles | ||
|---|---|---|
| Drugs used | Glucosamine and OTC drugs (NSAIDs, paracetamol) | 3 |
| Glucosamine | 4 | |
| Type of glucosamine | pCGS | 3 |
| pCGS and OFG | 4 | |
| OA site | Knee | 5 |
| All | 2 |
| (a) | |||||||
| No. | Study, Year, and Country | Subjects | Intervention | Perspective | Method | Time Horizon | Costs of Glucosamine |
| 1 | Bruyère et al. [46], 2023, Thailand | OA patients | pCGS vs. OFG vs. placebo | Healthcare | CEA | - |
USD 27.78/powder pCGS, USD 27.22/tablet pCGS. USD 14.61/powder OFG, USD 10.80/tablet OFG. |
| 2 | Luksameesate et al. [15], 2022, Thailand |
Patients ≥ 45 years old with mild-to-moderate pain and no comorbidities |
pCGS combined with etoricoxib vs. glucosamine monotherapy | Societal | CEA | Lifetime | - |
| 3 | Bruyère et al. [47], 2021, Germany |
OA patients >40 years old | pCGS vs. OFG | Healthcare | CEA | - | - |
| 4 | Bruyère et al. [48], 2019, |
OA patients >40 years old | pCGS vs. OFG | Healthcare | CEA | - |
0.9 EUR/day for pCGS, 0.55 EUR/day for OFG |
| 5 | Scholtissen et al. [41], 2010 Spain, Portugal |
Knee OA patients with average age of 63 years old |
GS vs paracetamol vs. placebo | Healthcare | CEA | 6 months | - |
| 6 | Black et al. [49], 2009, UK |
Knee OA patients | GS/GH vs. chondroitin sulfate vs. GS and chondroitin | National healthcare system | CEA | Lifetime | £221 (1-year) |
| 7 | Segal et al. [50], 2004, Australia | OA patients | Interventions for arthritis, including glucosamine | National healthcare system | CUA | - | USD 180 (1-year) |
| (b) | |||||||
| No. | Study, Year, and Country | Subjects | Intervention | Model Type | Duration | Sensitivity Analysis | Discount Rate |
| 1 | Bruyère et al. [46], 2023, Thailand | OA patients | pCGS vs. OFG vs. placebo |
Grootendorst model | 6 months | - | - |
| 2 | Luksameesate et al. [15], 2022, Thailand |
Patients ≥ 45 years old with mild-to-moderate pain and no comorbidities |
pCGS combined with etoricoxib vs. glucosamine monotherapy | Markov model | 6 months | One-way; PSA | 3% |
| 3 | Bruyère et al. [47], 2021, Germany |
OA patients >40 years old | pCGS vs. OFG |
Grootendorst model | 3 years | - | - |
| 4 | Bruyère et al. [48], 2019, |
OA patients >40 years old | pCGS vs. OFG |
Grootendorst model | 3 years | One-way | - |
| 5 | Scholtissen et al. [41], 2010 Spain, Portugal | Knee OA patients with average age of 63 years old | GS vs. paracetamol vs. placebo | Mathematical—decision model | 6 months | PSA | - |
| 6 | Black et al. [49], 2009, UK | OA patients |
Interventions for OA including glucosamine | Mathematical—decision model | 1 year | - | 5% |
| 7 | Segal et al. [50], 2004, Australia | Knee OA patients | GS Sulfate/hydrochloride vs. chondroitin sulfate vs. GS and chondroitin | Cohort model | 1 year | One-way | 3.5% |
| No. | Study, Year, and Country | Comparator | Cost | QALY Gain | ICER | Conclusion |
|---|---|---|---|---|---|---|
| 1 | Bruyère et al. [46], 2023, Thailand | pCGS vs. OFG | At 3 months pCGS: USD 53.805 OFG: USD 100.44 At 6 months pCGS: USD 126.1359 | At 3 months pCGS: 0.017 OFG: 0.0031 At 6 months pCGS: 0.0411 OFG: 0.0048 | At 3 months pCGS/PBO: 3165 USD/QALY OFG/PBO: 32,400 USD/QALY At 6 months pCGS/PBO: 3069 USD/QALY OFG/PBO: placebo better |
pCGS is cost-effective at threshold of 3260 USD/QALY pCGS is more cost-effective than OFG |
| 2 | Luksameesate et al. [15], 2022, Thailand | pCGS + standard care vs. standard care | - | 0.87 | Dominant | Early addition of pCGS into standard care treatment early is cost-saving and more effective compared with standard care alone |
| 3 | Bruyère et al. [47], 2021, Germany | pCGS vs. OFG | At 3 months pCGS: EUR 77.0964 OFG: EUR 208.854 At 6 months pCGS: EUR 183.0003 At 36 months pCGS: EUR 2785.2712 | At 3 months pCGS: 0.0164 OFG: 0.0036 At 6 months pCGS: 0.0413 OFG: 0.0044 At 36 months pCGS: 0.2701 | At 3 months pCGS/PBO: 4701 EUR/QALY OFG/PBO: 58,015 EUR/QALY At 6 months pCGS/PBO: 4431 EUR/QALY OFG/PBO: Placebo better At 36 months pCGS/PBO: 10,312 EUR/QALY | pCGS is more cost-effective than OFG |
| 4 | Bruyère et al. [48], 2019 | pCGS vs. OFG | At 3 months pCGS: EUR 90.234 OFG: EUR 151.009 At 6 months pCGS: EUR 209.413 At 36 months pCGS: EUR 3162.910 | At 3 months pCGS: 0.0169 OFG: 0.00303 At 6 months pCGS: 0.0435 OFG: 0.00424 At 36 months pCGS: 0.2742 | At 3 months pCGS/PBO: 5347.2 EUR/QALY OFG/PBO: 49,737.4 EUR/QALY At 6 months pCGS/PBO: 4807.2 EUR/QALY OFG/PBO: Placebo better At 36 months pCGS/PBO: 11,535.5 EUR/QALY | pCGS is more cost-effective than OFG |
| 5 |
Scholtissen et al. [41], 2010 Spain, Portugal |
GS vs. paracetamol, GS vs. placebo | - | - |
GS/paracetamol: −1376 EUR/QALY GS/placebo: 3617.47 EUR/QALY | GS is highly cost-effective vs. paracetamol |
| 6 | Black et al. [49], 2009, UK | GS adding conventional vs. conventional care | GBP 2346.85 | 0.11 | 21,335 GBP/QALY | Addition of GS therapy to current care is cost-effective at threshold of 22,000 GBP/QALY |
| 7 | Segal et al. [50], 2004, Australia | GS vs. NSAIDs | USD 180.024 | 0.052 | 3462 USD/QALY | Glucosamine is cost-effective |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vo, N.X.; Le, N.N.H.; Chu, T.D.P.; Pham, H.L.; Dinh, K.X.A.; Che, U.T.T.; Ngo, T.T.T.; Bui, T.T. Cost-Effectiveness of Glucosamine in Osteoarthritis Treatment: A Systematic Review. Healthcare 2023, 11, 2340. https://doi.org/10.3390/healthcare11162340
Vo NX, Le NNH, Chu TDP, Pham HL, Dinh KXA, Che UTT, Ngo TTT, Bui TT. Cost-Effectiveness of Glucosamine in Osteoarthritis Treatment: A Systematic Review. Healthcare. 2023; 11(16):2340. https://doi.org/10.3390/healthcare11162340
Chicago/Turabian StyleVo, Nam Xuan, Ngan Nguyen Hoang Le, Trinh Dang Phuong Chu, Huong Lai Pham, Khang Xuan An Dinh, Uyen Thi Thuc Che, Thanh Thi Thanh Ngo, and Tien Thuy Bui. 2023. "Cost-Effectiveness of Glucosamine in Osteoarthritis Treatment: A Systematic Review" Healthcare 11, no. 16: 2340. https://doi.org/10.3390/healthcare11162340
APA StyleVo, N. X., Le, N. N. H., Chu, T. D. P., Pham, H. L., Dinh, K. X. A., Che, U. T. T., Ngo, T. T. T., & Bui, T. T. (2023). Cost-Effectiveness of Glucosamine in Osteoarthritis Treatment: A Systematic Review. Healthcare, 11(16), 2340. https://doi.org/10.3390/healthcare11162340







