Cataract Surgery in Low-Income Countries: A Good Deal!
Abstract
1. Introduction
2. Materials and Methods
2.1. Cost Functions
| Variables | |
| NCa | Net value of cost of cataract of a person with onset age a, a = 40..99 |
| CTa | Net value of cost of treatment of cataract of a person with onset age a, a = 40..99 |
| CPa | Net value of productivity loss of a person with cataract with onset age a, a = 40..99 |
| CCa | Net value of productivity loss of the caregiver for a person with cataract with onset age a, a = 40..99 |
| NSa | Net value of cost of cataract surgery of a person with onset age a, a = 40..99 |
| NQa | Net value of quality of life of a person with cataract with onset age of a, a = 40..99 |
| Constants | |
| CTM | Cost of treatment of cataract per year of mild/moderate impairment |
| CTS | Cost of treatment of cataract per year of severe impairment |
| CTB | Cost of treatment of cataract per year of blindness |
| La | Life expectancy of a person in age a, a = 40..99 |
| tm | Duration of cataract with mild/moderate impairment |
| ts | Duration of cataract with severe impairment |
| tb | Duration of cataract with blindness |
| r | Interest rate |
| CPM | Productivity loss per year for a person with cataract with mild/moderate impairment |
| CPS | Productivity loss per year for a person with cataract with severe impairment |
| CPB | Productivity loss per year for a person with cataract with blindness |
| CCM | Productivity loss of a caregiver per year for a person with cataract with mild/moderate impairment |
| CCS | Productivity loss of a caregiver per year for a person with cataract with severe impairment |
| CCB | Productivity loss of a caregiver per year for a person with cataract with blindness |
| ßt | (dummy variable for pension age) |
| P | Pension age |
| s | Year of surgery |
| CS | Cost of surgery |
| Qm | Quality of life of a person with cataract with mild/moderate impairment |
| Qs | Quality of life of a person with cataract with severe impairment |
| Qb | Quality of life of a person with cataract with blindness |
- -
- Mild or moderate impairment: visual acuity worse than 6/12 to 6/60.
- -
- Severe impairment: visual acuity worse than 6/60 to 3/60
- -
- Blindness: visual acuity worse than 3/60
2.2. Quality of Life
2.3. Data
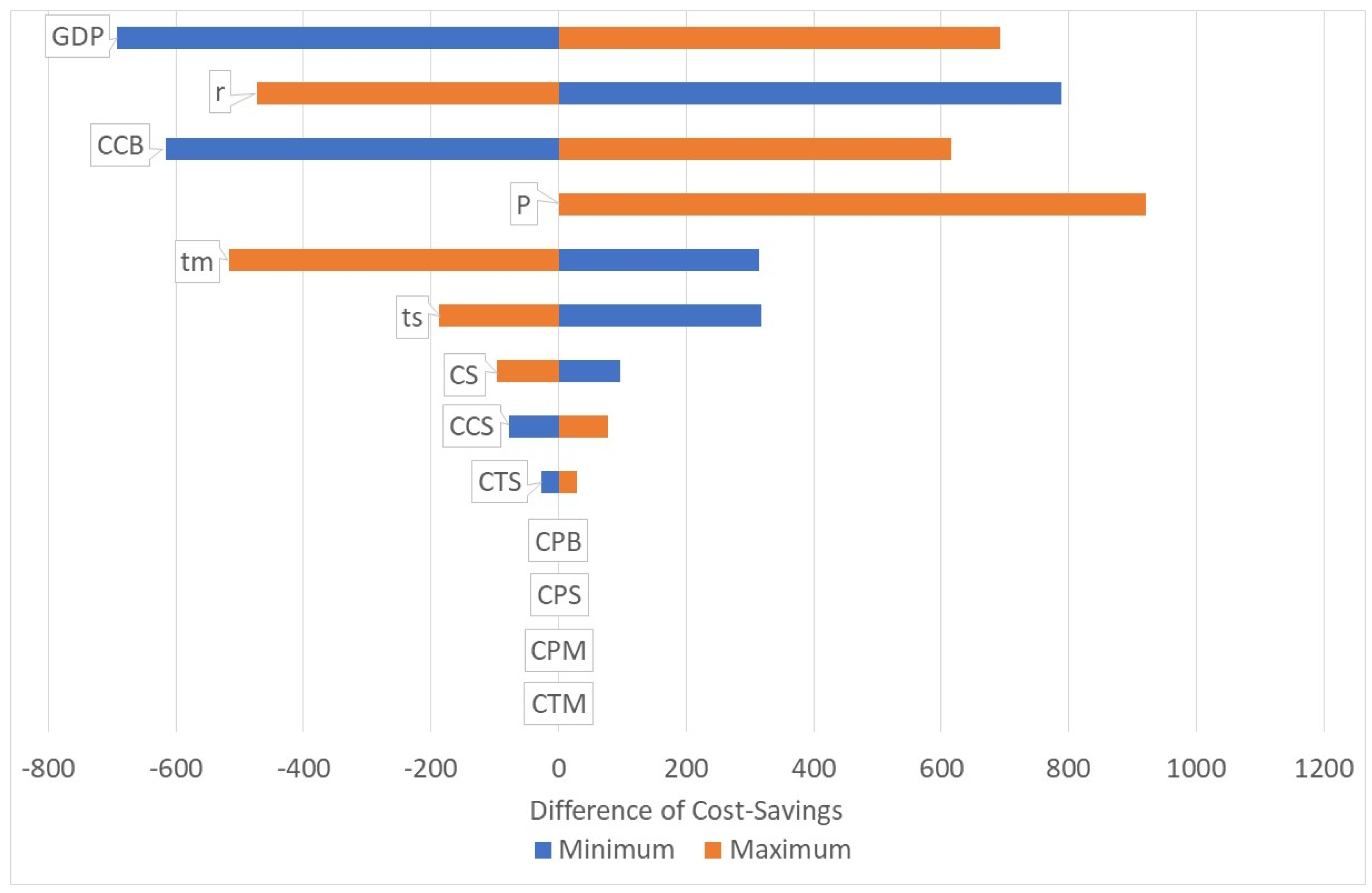
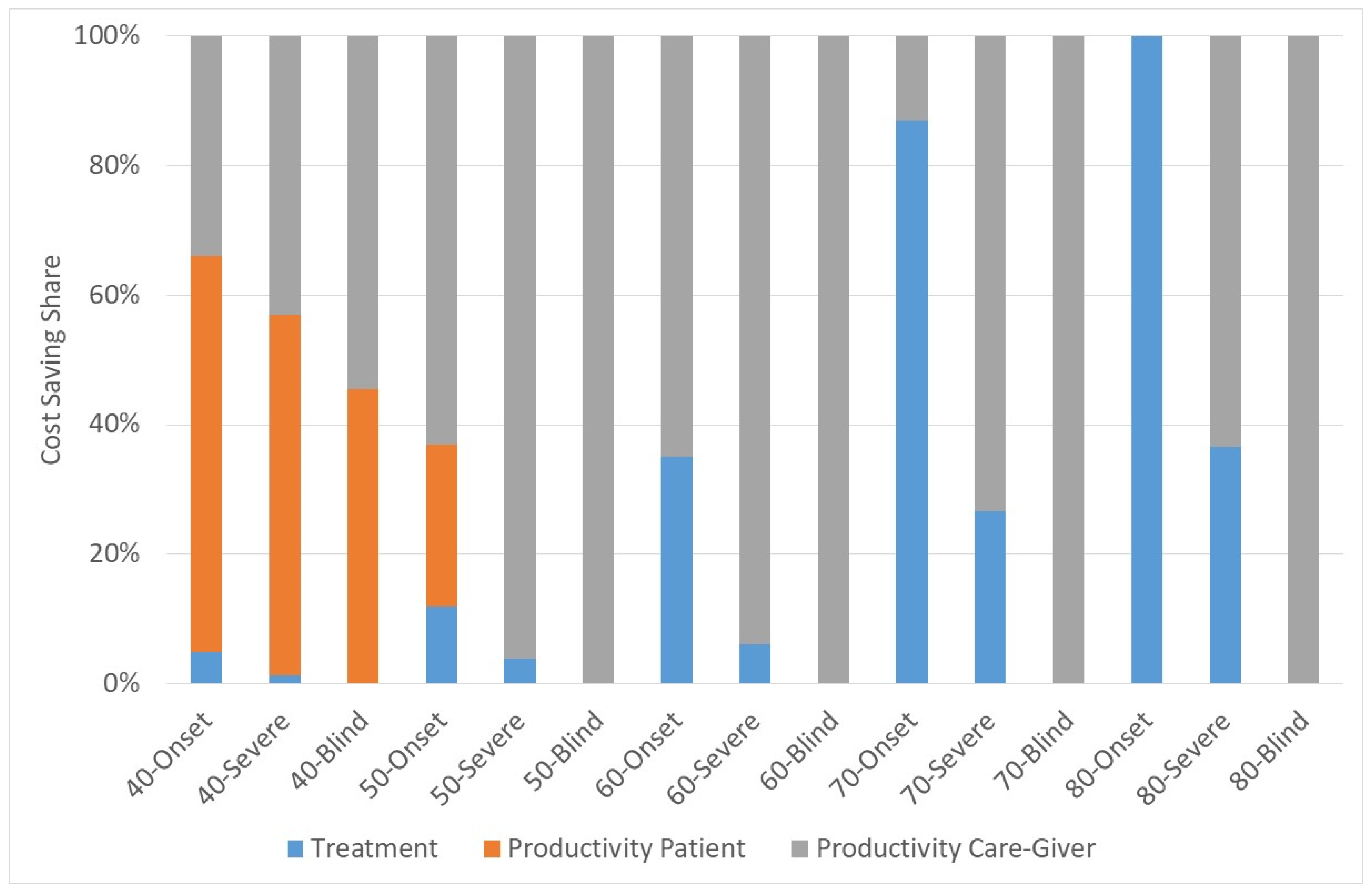
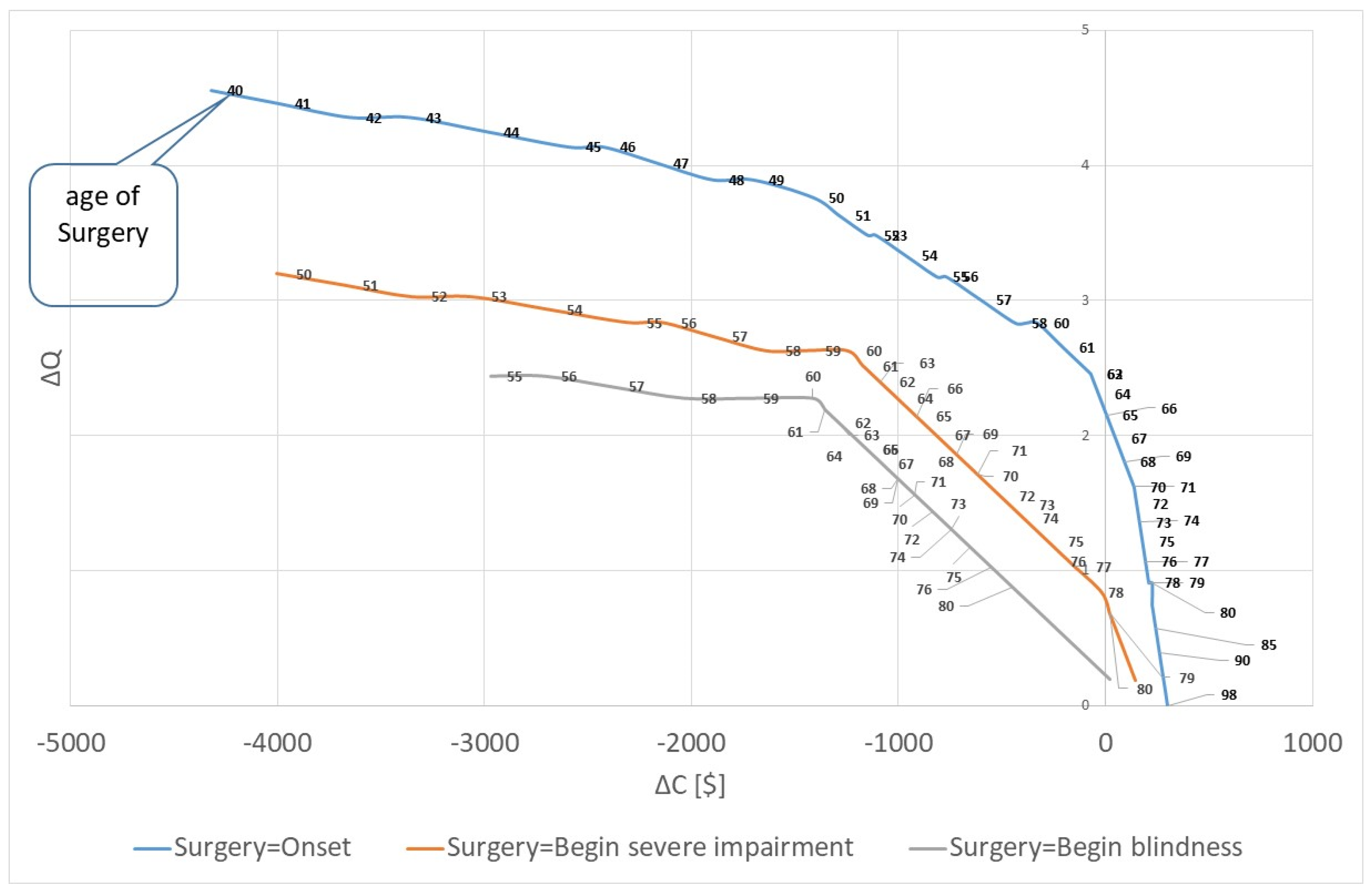
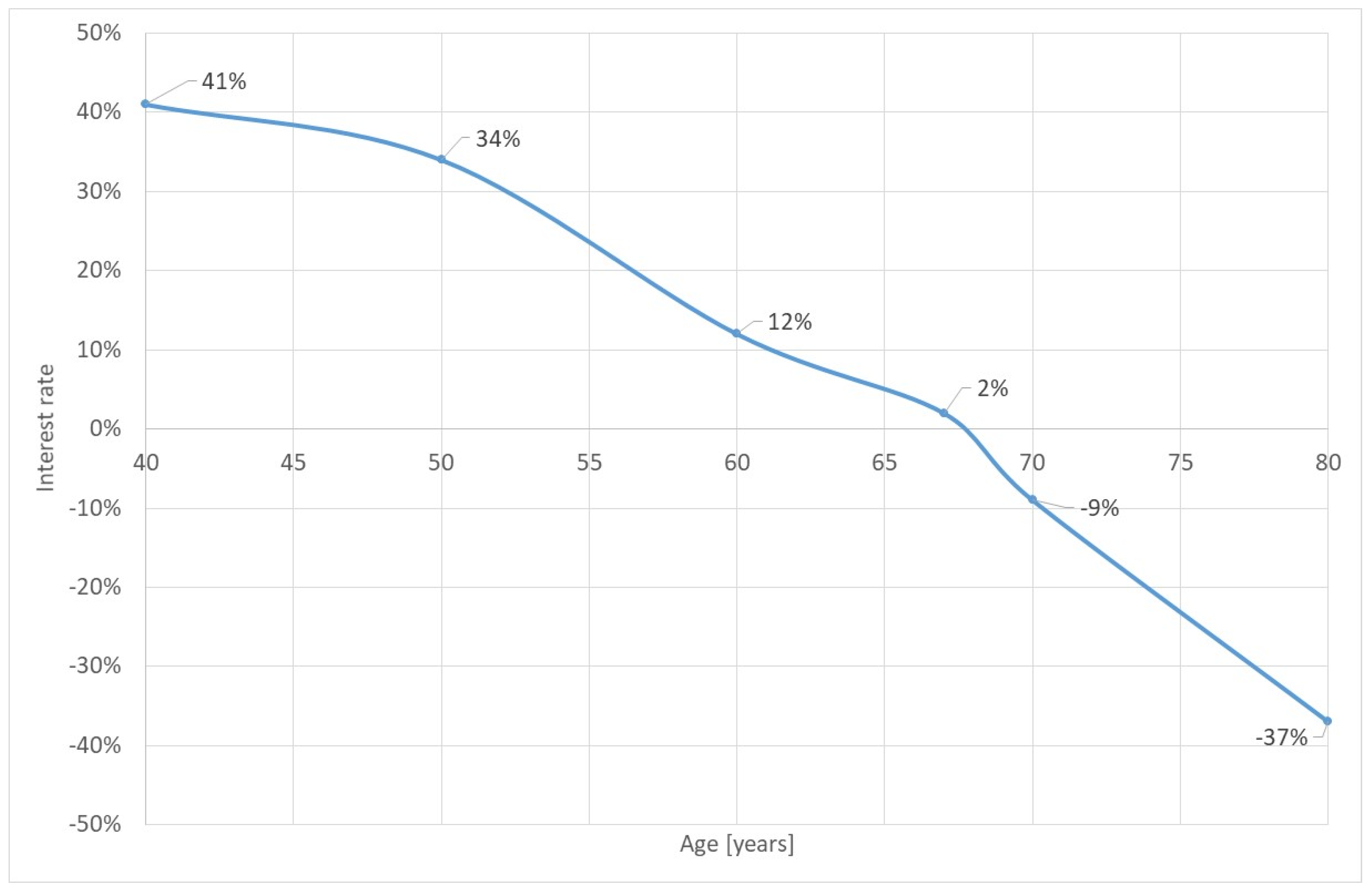
3. Results
- (1)
- Year of Surgery = Year of onset
- (2)
- Year of Surgery = First year of severe visual impairment
- (3)
- Year of Surgery = First year of blindness
4. Discussion
- Limitations of data. There are a number of limitations of this study due to missing or unreliable data while some data might be wrong for specific situations. Although the sensitivity analyses show that the parameters might change strongly without having a major impact on the thresholds of cost-savings and cost-effectiveness, there is still a possibility that the parameters of a real situation might be very different. For instance, our model does not apply to African patients flying for cataract surgery to Arabic countries as the costs of this surgery are much higher than what we assumed here.Furthermore, costs and outcomes depend on the professionalism of services provided. Poor quality of services will result in a completely different result of the health economic analysis of cataract surgery in low-income countries. However, the vast majority of cataract surgeries in these countries are performed professionally and have a tremendous result on the visual strength of the patient and his ability to live an independent and satisfying life—and they are cost-saving or cost-effective in almost all circumstances.
- Limitations of methodology: It would be ideal to use distributions and more advanced modelling techniques to produce more than averages. Stochastic models, such as Markov, discrete event simulation and agent-based simulations have the capacity to produce these distributions and allow a risk assessment. However, with the limited data given in the literature, such models could not be applied. Instead, they would pretend a degree of precision which does not exist. There is a great need to conduct more research in this field in order to produce data which might support more advanced modelling techniques in future.Another consequence of poor data is that a probabilistic sensitivity analysis would not really provide new insights. Usually, one would expect that a distribution of parameters can be taken from the literature, permitting an analysis of the impact of randomly varied parameters on major outcomes. However, in the absence of reliable data, this method cannot be applied as it would pretend a degree of precision which does not exist.
- Simplifying assumptions: We made some assumptions which could be challenged. We assume that the quality of life in the year of surgery is not reduced by the procedure. Thus, we assume that the surgery is successful and has no complications. As there might be severe problems associated with the surgery, we might over-estimate the gain of quality of life due to cataract surgery.
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Burton, M.J.; Ramke, J.; Marques, A.P.; Bourne, R.R.; Congdon, N.; Jones, I.; Tong, B.A.A.; Arunga, S.; Bachani, D.; Bascaran, C. The lancet global health commission on global eye health: Vision beyond 2020. Lancet Glob. Health 2021, 9, e489–e551. [Google Scholar] [CrossRef] [PubMed]
- WHO. Blindness and Vision Impairment. Available online: https://www.who.int/news-room/fact-sheets/detail/blindness-and-visual-impairment (accessed on 3 February 2022).
- IAPD. Vision Atlas. Available online: https://www.iapb.org/learn/vision-atlas/ (accessed on 3 February 2022).
- Psychrembel. Katarakt. Available online: https://www.pschyrembel.de/Katarakt/K0BGL (accessed on 4 February 2022).
- Rao, G.N.; Khanna, R.; Payal, A. The global burden of cataract. Curr. Opin. Ophthalmol. 2011, 22, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Teller, C.; Hailemariam, A. The Demographic Transition and Development in Africa; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Marques, A.P.; Ramke, J.; Cairns, J.; Butt, T.; Zhang, J.H.; Muirhead, D.; Jones, I.; Tong, B.A.A.; Swenor, B.K.; Faal, H. Global economic productivity losses from vision impairment and blindness. EClinicalMedicine 2021, 35, 100852. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. World Report on Vision; World Health Organization: Geneva, Switzerland, 2019.
- Busbee, B.G.; Brown, M.M.; Brown, G.C.; Sharma, S. Incremental cost-effectiveness of initial cataract surgery. Ophthalmology 2002, 109, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Baltussen, R.; Sylla, M.; Mariotti, S.P. Cost-effectiveness analysis of cataract surgery: A global and regional analysis. Bull. World Health Organ. 2004, 82, 338–345. [Google Scholar]
- Kuper, H.; Polack, S.; Mathenge, W.; Eusebio, C.; Wadud, Z.; Rashid, M.; Foster, A. Does cataract surgery alleviate poverty? Evidence from a multi-centre intervention study conducted in Kenya, the Philippines and Bangladesh. PLoS ONE 2010, 5, e15431. [Google Scholar] [CrossRef]
- Polack, S.; Eusebio, C.; Mathenge, W.; Wadud, Z.; Rashid, M.; Foster, A.; Kuper, H. The impact of cataract surgery on activities and time-use: Results from a longitudinal study in Kenya, Bangladesh and the Philippines. PLoS ONE 2010, 5, e10913. [Google Scholar] [CrossRef] [PubMed]
- Lansingh, V.C.; Carter, M.J.; Martens, M. Global cost-effectiveness of cataract surgery. Ophthalmology 2007, 114, 1670–1678. [Google Scholar] [CrossRef]
- WHO. Scaling Up Action against Noncommunicable Diseases: How Much Will It Cost? World Health Organization: Geneva, Switzerland, 2011.
- Hunter, E.; Mac Namee, B.; Kelleher, J.D. A taxonomy for agent-based models in human infectious disease epidemiology. J. Artif. Soc. Soc. Simul. 2017, 20, 2. [Google Scholar] [CrossRef]
- World Health Organization. Who Methods and Data Sources for Global Burden of Disease Estimates 2000–2019; World Health Organization: Geneva, Switzerland, 2020.
- World Bank. World Development Indicators. Available online: https://databank.worldbank.org/source/world-development-indicators# (accessed on 1 July 2021).
- Knoema. Weltdatenatlas. Available online: https://knoema.com/atlas/United-Republic-of-Tanzania/topics/Demographics/Age/Life-expectancy-at-age-60-years (accessed on 4 February 2022).
- Aboobaker, S.; Courtright, P. Barriers to cataract surgery in Africa: A systematic review. Middle East Afr. J. Ophthalmol. 2016, 23, 145–149. [Google Scholar] [CrossRef]
- Jaggernath, J.; Gogate, P.; Moodley, V.; Naidoo, K.S. Comparison of cataract surgery techniques: Safety, efficacy, and cost-effectiveness. Eur. J. Ophthalmol. 2014, 24, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Flessa, S.; Moeller, M.; Ensor, T.; Hornetz, K. Basing care reforms on evidence: The Kenya health sector costing model. BMC Health Serv. Res. 2011, 11, 128. [Google Scholar] [CrossRef] [PubMed]
- Lindfield, R.; Vishwanath, K.; Ngounou, F.; Khanna, R.C. The challenges in improving outcome of cataract surgery in low and middle income countries. Indian J. Ophthalmol. 2012, 60, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Grimes, C.E.; Bowman, K.G.; Dodgion, C.M.; Lavy, C.B. Systematic review of barriers to surgical care in low-income and middle-income countries. World J. Surg. 2011, 35, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Mailu, E.W.; Virendrakumar, B.; Bechange, S.; Jolley, E.; Schmidt, E. Factors associated with the uptake of cataract surgery and interventions to improve uptake in low- and middle-income countries: A systematic review. PLoS ONE 2020, 15, e0235699. [Google Scholar] [CrossRef] [PubMed]
- McCormick, I.; Butcher, R.; Evans, J.R.; Mactaggart, I.Z.; Limburg, H.; Jolley, E.; Sapkota, Y.D.; Oye, J.E.; Mishra, S.K.; Bastawrous, A. Effective cataract surgical coverage in adults aged 50 years and older: Estimates from population-based surveys in 55 countries. Lancet Glob. Health 2022, 10, e1744–e1753. [Google Scholar] [CrossRef]
- Ramke, J.; Evans, J.R.; Habtamu, E.; Mwangi, N.; Silva, J.C.; Swenor, B.K.; Congdon, N.; Faal, H.B.; Foster, A.; Friedman, D.S. Grand challenges in global eye health: A global prioritisation process using delphi method. Lancet Healthy Longev. 2022, 3, e31–e41. [Google Scholar] [CrossRef]
- Singh, A.; Garner, P.; Floyd, K. Cost-effectiveness of public-funded options for cataract surgery in Mysore, India. Lancet 2000, 355, 180–184. [Google Scholar] [CrossRef]
- Marseille, E. Cost-effectiveness of cataract surgery in a public health eye care programme in Nepal. Bull. World Health Organ. 1996, 74, 319–324. [Google Scholar]
- Horton, S.; Gelband, H.; Jamison, D.; Levin, C.; Nugent, R.; Watkins, D. Ranking 93 health interventions for low-and middle-income countries by cost-effectiveness. PLoS ONE 2017, 12, e0182951. [Google Scholar] [CrossRef]
- Grimes, C.E.; Henry, J.A.; Maraka, J.; Mkandawire, N.C.; Cotton, M. Cost-effectiveness of surgery in low-and middle-income countries: A systematic review. World J. Surg. 2014, 38, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Huntington, H.G.; Weyant, J.P.; Scweeney, J.L. Modelling for insights, not numbers: The experience of the energy modelling forum, OMEGA. Int. J. Manag. Sci. 1982, 10, 449. [Google Scholar]

| MSVI or Blindness Causes That Are… | Disease | USD (2018) |
|---|---|---|
| … treatable or addressable | Cataract | 8,768,759,000 |
| Unaddressed Refractive Error (Distance) | 6,988,223,000 | |
| Unaddressed Refractive Error (Near) | 9,035,476,000 | |
| Total | 24,792,458,000 | |
| … preventable | Diabetic Retinopathy | 19,858,251,000 |
| Trachoma | 494,077,000 | |
| Glaucoma | 11,744,642,000 | |
| Total | 32,096,970,000 |
| Variable | Definition | Value | Source |
|---|---|---|---|
| CTM | Cost of treatment of cataract per year of mild/moderate impairment | 20 USD | Expert estimates |
| CTS | Cost of treatment of cataract per year of severe impairment | 20 USD | Expert estimates |
| CTB | Cost of treatment of cataract per year of blindness | 20 USD | Expert estimates |
| La | Life expectancy of a person in age a, a = 40..99 | Linear interpolation between values | [18] |
| tm | Duration of cataract with mild/moderate impairment | 10 years | Expert estimates |
| ts | Duration of cataract with severe impairment | 5 years | Expert estimates |
| tb | Duration of cataract with blindness | Until end of life | Expert estimates |
| GDP | Gross domestic product per capita per annum | 1100 USD | [17] |
| r | Interest rate | 5% | standard |
| CPM | Productivity loss per year for a person with cataract with mild/moderate impairment | 55 USD, i.e., 5% productivity loss | [1,7,8] |
| CPS | Productivity loss per year for a person with cataract with severe impairment | 330 USD, i.e., 30% productivity loss | [1,7,8] |
| CPB | Productivity loss per year for a person with cataract with blindness | 660 USD, i.e., 60% productivity loss | [1,7,8] |
| CCM | Productivity loss of a caregiver per year for a person with cataract with mild/moderate impairment | 0 USD, i.e., 0% productivity loss | [1,7,8] |
| CCS | Productivity loss of a caregiver per year for a person with cataract with severe impairment | 55 USD, i.e., 5% productivity loss | [1,7,8] |
| CCB | Productivity loss of a caregiver per year for a person with cataract with blindness | 275 USD, i.e., 25% productivity loss | [1,7,8] |
| P | Pension age | 60 years | [17] |
| s | Year of surgery | At year of onset, beginning of severe impairment or beginning of blindness | Assumption for scenarios |
| CS | Cost of surgery | 300.00 USD | [19,20,21] |
| Qm | Quality of life of a person with cataract with mild/moderate impairment | 0.7 | [16,22] |
| Qs | Quality of life of a person with cataract with severe impairment | 0.6 | [16,22] |
| Qb | Quality of life of a person with cataract with blindness | 0.5 | [16,22] |
| Qo | Quality of life of a person with cataract after operation | 0.9 | [16,22] |
| Parameter | Without Surgery | With Surgery |
|---|---|---|
| Net-value treatment | 217.97 | 162.16 |
| Net-value productivity loss patient | 445.93 | 445.93 |
| Net-value productivity loss caregiver | 1,384.54 | 0.00 |
| Cost operation | 0.00 | 193.38 |
| Total net-value cost: | 2048.45 | 801.47 |
| Cost saving | 1246.98 | |
| Quality of life [QALY] | 9.59 | 12.22 |
| Benefit [QALY] | 2.63 | |
| Variable | Definition | Original Value | Cost-Saving Threshold | Cost-Effectiveness Threshold | |
|---|---|---|---|---|---|
| ≤1∙GDP | ≤2∙GDP | ||||
| CTM | Cost of treatment of cataract per year of mild/moderate impairment | 20 USD | - | - | - |
| CTS | Cost of treatment of cataract per year of severe impairment | 20 USD | - | - | - |
| CTB | Cost of treatment of cataract per year of blindness | 20 USD | - | - | - |
| La | Life expectancy | 77 yrs. | 73 | 61 | |
| tm | Duration of cataract with mild/moderate impairment | 10 | 15 | - | - |
| ts | Duration of cataract with severe impairment | 5 | - | - | - |
| GDP | Gross domestic product per capita per annum | 1100 USD | 220 USD p.c. | 25 USD p.c. | |
| CPM, CPS, CPB | Productivity loss of patient | 55.00 USD, 330.00 USD, 660.00 USD | - | - | - |
| CCM, CCS, CCB | Productivity loss of a caregiver | 0.00 USD, 55.00 USD, 275.00 USD | - | - | - |
| P | Pension age | 60 | - | - | - |
| CS | Cost of surgery | 300.00 USD | 630.00 USD | 1800.00 USD | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flessa, S. Cataract Surgery in Low-Income Countries: A Good Deal! Healthcare 2022, 10, 2580. https://doi.org/10.3390/healthcare10122580
Flessa S. Cataract Surgery in Low-Income Countries: A Good Deal! Healthcare. 2022; 10(12):2580. https://doi.org/10.3390/healthcare10122580
Chicago/Turabian StyleFlessa, Steffen. 2022. "Cataract Surgery in Low-Income Countries: A Good Deal!" Healthcare 10, no. 12: 2580. https://doi.org/10.3390/healthcare10122580
APA StyleFlessa, S. (2022). Cataract Surgery in Low-Income Countries: A Good Deal! Healthcare, 10(12), 2580. https://doi.org/10.3390/healthcare10122580






