Poor Diet, Long Sleep, and Lack of Physical Activity Are Associated with Inflammation among Non-Demented Community-Dwelling Elderly
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
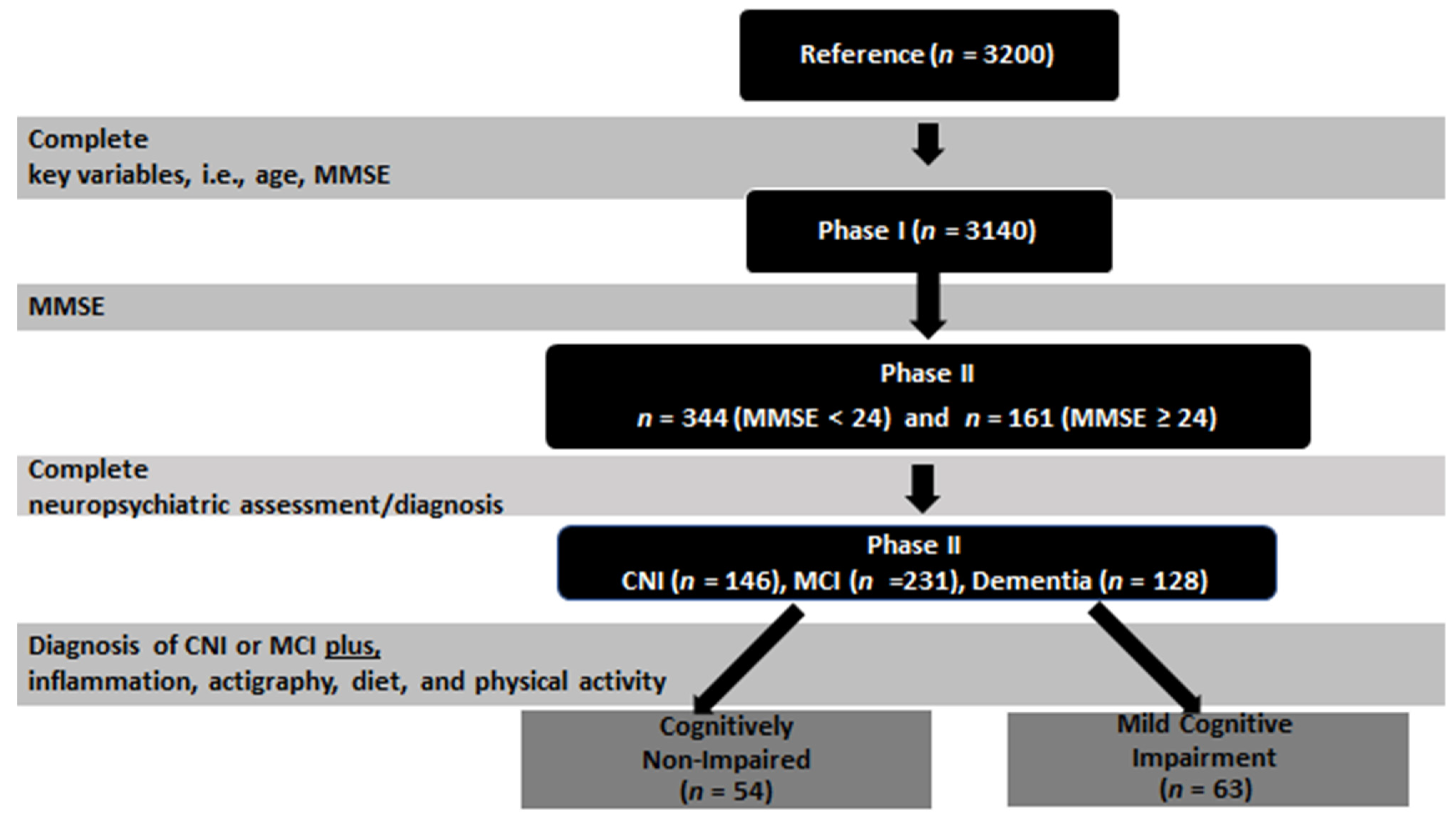
2.2. Participants
2.3. Measurements
2.3.1. Inflammatory Markers
2.3.2. Diet
2.3.3. Sleep Measurements
2.3.4. Physical Activity
2.3.5. Other Variables
2.4. Statistical Analysis
3. Results
3.1. Sample Demographics, Clinical and Preclinical Variables
3.2. Associations of Inflammatory Markers with Sleep, Diet and Physical Activity
4. Discussion
4.1. Strengths and Limitations
4.2. Implications
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Irwin, M.; Opp, M. Sleep Health: Reciprocal Regulation of Sleep and Innate Immunity. Neuropsychopharmacology 2017, 42, 129–155. Available online: https://pubmed.ncbi.nlm.nih.gov/27510422/ (accessed on 6 March 2021). [CrossRef] [Green Version]
- Ferrucci, L.; Fabbri, E. Inflammageing: Chronic Inflammation in Ageing, Cardiovascular Disease, and Frailty. Nat. Rev. Cardiol. 2018, 15, 505–522. Available online: https://www.nature.com/articles/s41569-018-0064-2 (accessed on 7 March 2021). [CrossRef]
- Franceschi, C.; Campisi, J. Chronic Inflammation (Inflammaging) and Its Potential Contribution to Age-Associated Diseases. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69 (Suppl. 1), S4–S9. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24833586 (accessed on 3 August 2019). [CrossRef]
- Kennedy, B.K.; Berger, S.L.; Brunet, A.; Campisi, J.; Cuervo, A.M.; Epel, E.S.; Franceschi, C.; Lithgow, G.J.; Morimoto, R.I.; Pessin, J.E.; et al. Geroscience: Linking aging to chronic disease. Cell 2014, 159, 709–713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngandu, T.; Lehtisalo, J.; Solomon, A.; Levälahti, E.; Ahtiluoto, S.; Antikainen, R.; Bäckman, L.; Hänninen, T.; Jula, A.; Laatikainen, T.; et al. A 2 Year Multidomain Intervention of Diet, Exercise, Cognitive Training, and Vascular Risk Monitoring versus Control to Prevent Cognitive Decline in at-Risk Elderly People (FINGER): A Randomised Controlled Trial. Lancet 2015, 385, 2255–2263. Available online: https://pubmed.ncbi.nlm.nih.gov/25771249/ (accessed on 6 March 2021). [CrossRef]
- Ticinesi, A.; Meschi, T.; Lauretani, F.; Felis, G.; Franchi, F.; Pedrolli, C.; Barichella, M.; Benati, G.; Di Nuzzo, S.; Ceda, G.P.; et al. Nutrition and Inflammation in Older Individuals: Focus on Vitamin, D, n-3 Polyunsaturated Fatty Acids and Whey Proteins. Nutrients 2016, 8, 186. Available online: https://pubmed.ncbi.nlm.nih.gov/27043616/ (accessed on 7 March 2021). [CrossRef] [Green Version]
- Custodero, C.; Mankowski, R.T.; Lee, S.A.; Chen, Z.; Wu, S.; Manini, T.M.; Echeverri, J.; Sabbà, C.; Beavers, D.P.; Cauley, J.A.; et al. Evidence-Based Nutritional and Pharmacological Interventions Targeting Chronic Low-Grade Inflammation in Middle-Age and Older Adults: A Systematic Review and Meta-Analysis. Ageing Res. Rev. 2018, 46, 42–59. Available online: https://pubmed.ncbi.nlm.nih.gov/29803716/ (accessed on 7 March 2021). [CrossRef]
- Milaneschi, Y.; Bandinelli, S.; Penninx, B.W.; Vogelzangs, N.; Corsi, A.M.; Lauretani, F.; Kisialiou, A.; Vazzana, R.; Terracciano, A.; Guralnik, J.M.; et al. Depressive Symptoms and Inflammation Increase in a Prospective Study of Older Adults: A Protective Effect of a Healthy (Mediterranean-Style) Diet. Mol. Psychiatry 2011, 16, 589–590. Available online: https://jhu.pure.elsevier.com/en/publications/depressive-symptoms-and-inflammation-increase-in-a-prospective-st-4 (accessed on 7 March 2021). [CrossRef] [Green Version]
- Luciano, M.; Mõttus, R.; Starr, J.M.; McNeill, G.; Jia, X.; Craig, L.C.A.; Deary, I.L. Depressive Symptoms and Diet: Their Effects on Prospective Inflammation Levels in the Elderly. Brain Behav. Immun. 2012, 26, 717–720. Available online: https://abdn.pure.elsevier.com/en/publications/depressive-symptoms-and-diet-their-effects-on-prospective-inflamm (accessed on 7 March 2021). [CrossRef] [PubMed]
- Ancoli-Israel, S.; Poceta, J.S.; Stepnowsky, C.; Martin, J.; Gehrman, P. Identification and Treatment of Sleep Problems in the Elderly. Sleep Med. Rev. 1997, 1, 3–17. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15310520 (accessed on 3 August 2019). [CrossRef]
- Martin, J.; Shochat, T.; Ancoli-Israel, S. Assessment and Treatment of Sleep Disturbances in Older Adults. Clin. Psychol. Rev. 2000, 20, 783–805. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10983268 (accessed on 3 August 2019). [CrossRef]
- Loiselle, M.M.; Means, M.K.; Edinger, J.D. Sleep Disturbances in Aging. Adv. Cell Aging Gerontol. 2005, 17, 33–59. Available online: https://www.sciencedirect.com/science/article/abs/pii/S1566312404170028 (accessed on 3 August 2019).
- Vgontzas, A.N.; Zoumakis, M.; Bixler, E.O.; Lin, H.-M.; Prolo, P.; Vela-Bueno, A.; Kales, A.; Chrousos, G.P. Impaired Nighttime Sleep in Healthy Old Versus Young Adults Is Associated with Elevated Plasma Interleukin-6 and Cortisol Levels: Physiologic and Therapeutic Implications. J. Clin. Endocrinol. Metab. 2003, 88, 2087–2095. Available online: https://academic.oup.com/jcem/article-lookup/doi/10.1210/jc.2002-021176 (accessed on 12 August 2019). [CrossRef] [PubMed] [Green Version]
- Von Känel, R.; Dimsdale, J.E.; Ancoli-Israel, S.; Mills, P.J.; Patterson, T.L.; McKibbin, C.L.; Ms, C.A.; Grant, I. Poor Sleep Is Associated with Higher Plasma Proinflammatory Cytokine Interleukin-6 and Procoagulant Marker Fibrin D-Dimer in Older Caregivers of People with Alzheimer’s Disease. J. Am. Geriatr. Soc. 2006, 54, 431–437. Available online: http://doi.wiley.com/10.1111/j.1532-5415.2005.00642.x (accessed on 12 August 2019). [CrossRef] [PubMed]
- Vgontzas, A.N.; Zoumakis, M.; Papanicolaou, D.A.; Bixler, E.O.; Prolo, P.; Lin, H.-M.; Vela-Bueno, A.; Kales, A.; Chrousos, G.P.; Vgontzas, A.N.; et al. Chronic Insomnia Is Associated with a Shift of Interleukin-6 and Tumor Necrosis Factor Secretion from Nighttime to Daytime. Metabolism 2002, 51, 887–892. Available online: http://www.ncbi.nlm.nih.gov/pubmed/12077736 (accessed on 26 July 2019). [CrossRef] [PubMed]
- Burgos, I.; Richter, L.; Klein, T.; Fiebich, B.; Feige, B.; Lieb, K.; Voderholzer, U.; Riemann, D. Increased Nocturnal Interleukin-6 Excretion in Patients with Primary Insomnia: A Pilot Study. Brain Behav. Immun. 2006, 20, 246–253. Available online: https://www.sciencedirect.com/science/article/pii/S0889159105001108?via%3Dihub (accessed on 12 August 2019). [CrossRef]
- Taheri, S.; Austin, D.; Lin, L.; Nieto, F.J.; Young, T.; Mignot, E. Correlates of Serum C-Reactive Protein (CRP)—No Association with Sleep Duration or Sleep Disordered Breathing. Sleep 2007, 30, 991–996. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17702268 (accessed on 12 August 2019). [CrossRef] [PubMed] [Green Version]
- Matthews, K.A.; Zheng, H.; Kravitz, H.M.; Sowers, M.; Bromberger, J.T.; Buysse, D.J.; Owens, J.F.; Sanders, M.; Hall, M. Are Inflammatory and Coagulation Biomarkers Related to Sleep Characteristics in Mid-Life Women?: Study of Women’s Health across the Nation Sleep Study. Sleep 2010, 33, 1649–1655. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21120127 (accessed on 12 August 2019). [CrossRef] [PubMed]
- Abramson, J.L.; Vaccarino, V. Relationship between Physical Activity and Inflammation among Apparently Healthy Middle-Aged and Older US Adults. Arch. Intern. Med. 2002, 162, 1286–1292. Available online: https://pubmed.ncbi.nlm.nih.gov/12038947/ (accessed on 7 March 2019). [CrossRef] [Green Version]
- Wannamethee, S.G.; Lowe, G.D.O.; Whincup, P.H.; Rumley, A.; Walker, M.; Lennon, L. Physical Activity and Hemostatic and Inflammatory Variables in Elderly Men. Circulation 2002, 105, 1785–1790. Available online: https://pubmed.ncbi.nlm.nih.gov/11956120/ (accessed on 7 March 2019). [CrossRef] [Green Version]
- Reuben, D.B.; Judd-Hamilton, L.; Harris, T.B.; Seeman, T.E. The Associations between Physical Activity and Inflammatory Markers in High-Functioning Older Persons: MacArthur Studies of Successful Aging. J. Am. Geriatr. Soc. 2003, 51, 1125–1130. Available online: http://doi.wiley.com/10.1046/j.1532-5415.2003.51380.x (accessed on 18 April 2019). [CrossRef] [PubMed]
- Colbert, L.H.; Visser, M.; Simonsick, E.M.; Tracy, R.P.; Newman, A.B.; Kritchevsky, S.B.; Pahor, M.; Taaffe, D.; Brach, J.; Rubin, S.; et al. Physical Activity, Exercise, and Inflammatory Markers in Older Adults: Findings from the Health, Aging and Body Composition Study. J. Am. Geriatr. Soc. 2004, 52, 1098–1104. Available online: https://pubmed.ncbi.nlm.nih.gov/15209647/ (accessed on 7 March 2019). [CrossRef] [PubMed]
- Jankord, R.; Jemiolo, B. Influence of physical activity on serum IL-6 and IL-10 levels in healthy older men. Med. Sci. Sports Exerc. 2004, 36, 960–964. [Google Scholar] [CrossRef]
- Elosua, R.; Bartali, B.; Ordovas, J.M.; Corsi, A.M.; Lauretani, F.; Ferrucci, L. Association Between Physical Activity, Physical Performance, and Inflammatory Biomarkers in an Elderly Population: The InCHIANTI Study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2005, 60, 760–767. Available online: https://academic.oup.com/biomedgerontology/article-abstract/60/6/760/590349 (accessed on 18 April 2019). [CrossRef] [Green Version]
- Nicklas, B.J.; Beavers, D.P.; Mihalko, S.L.; Miller, G.D.; Loeser, R.F.; Messier, S.P. Relationship of Objectively-Measured Habitual Physical Activity to Chronic Inflammation and Fatigue in Middle-Aged and Older Adults. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2016, 71, 1437–1443. Available online: https://pubmed.ncbi.nlm.nih.gov/27382039/ (accessed on 7 March 2019). [CrossRef]
- Draganidis, D.; Jamurtas, A.; Stampoulis, T.; Laschou, V.; Deli, C.; Georgakouli, K.; Papanikolaou, K.; Chatzinikolaou, A.; Michalopoulou, M.; Papadopoulos, C.; et al. Disparate Habitual Physical Activity and Dietary Intake Profiles of Elderly Men with Low and Elevated Systemic Inflammation. Nutrients 2018, 10, 566. Available online: https://www.mdpi.com/2072-6643/10/5/566 (accessed on 7 March 2019). [CrossRef] [PubMed] [Green Version]
- Public Health England, 10 Minutes Brisk Walking Each Day in Mid-Life for Health Benefits and towards Achieving Physical Activity Recom-Mendations. Evidence Summary. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/639030/Health_benefits_of_10_mins_brisk_walking_evidence_summary.pdf (accessed on 7 March 2019).
- Alghadir, A.H.; Gabr, S.A.; Al-Momani, M.; Al-Momani, F. Moderate Aerobic Training Modulates Cytokines and Cortisol Profiles in Older Adults with Cognitive Abilities. Cytokine 2021, 138, 155373. Available online: https://pubmed.ncbi.nlm.nih.gov/33248912/ (accessed on 7 March 2019). [CrossRef]
- Stigger, F.S.; Zago Marcolino, M.A.; Portela, K.M.; Della Méa Plentz, R. Effects of Exercise on Inflammatory, Oxidative, and Neurotrophic Biomarkers on Cognitively Impaired Individuals Diagnosed with Dementia or Mild Cognitive Impairment: A Systematic Review and Meta-Analysis. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2019, 74, 616–624. Available online: https://pubmed.ncbi.nlm.nih.gov/30084942/ (accessed on 7 March 2019). [CrossRef]
- Petersen, R. Mild Cognitive Impairment. N. Engl. J. Med. 2011, 364, 2227–2234. [Google Scholar] [CrossRef] [Green Version]
- Petersen, R. Mild Cognitive Impairment. Continuum Lifelong Learn. Neurol. 2016, 22, 404–418. [Google Scholar] [CrossRef]
- Roberts, R.; Knopman, D. Classification and Epidemiology of MCI. Clin. Geriart. Med. 2013, 29, 753–772. [Google Scholar] [CrossRef] [Green Version]
- Zaganas, I.V.; Simos, P.; Basta, M.; Kapetanaki, S.; Panagiotakis, S.; Koutentaki, I.; Fountoulakis, N.; Bertsias, A.; Duijker, G.; Tziraki, C.; et al. The Cretan Aging Cohort: Cohort Description and Burden of Dementia and Mild Cognitive Impairment. Am. J. Alzheimer’s Dis. Other Dement.® 2019, 34, 23–33. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30259758 (accessed on 11 September 2019). [CrossRef] [PubMed]
- Fountoulakis, K.N.; Tsolaki, M.; Chantzi, H.; Kazis, A. Mini Mental State Examination (MMSE): A Validation Study in Greece. Am. J. Alzheimer’s Dis. Other Dement.® 2000, 15, 342–345. Available online: http://aja.sagepub.com/cgi/doi/10.1177/153331750001500604 (accessed on 11 September 2019). [CrossRef]
- Dardiotis, E.; Kosmidis, M.H.; Yannakoulia, M.; Hadjigeorgiou, G.M.; Scarmeas, N. The Hellenic Longitudinal Investigation of Aging and Diet (HELIAD): Rationale, Study Design, and Cohort Description. Neuroepidemiology 2014, 43, 9–14. Available online: https://www.karger.com/Article/FullText/362723 (accessed on 11 September 2019). [CrossRef]
- Winblad, B.; Palmer, K.; Kivipelto, M.; Jelic, V.; Fratiglioni, L.; Wahlund, L.-O.; Nordberg, A.; Bäckman, L.; Albert, M.; Almkvist, O.; et al. Mild Cognitive Impairment—Beyond Controversies, towards a Consensus: Report of the International Working Group on Mild Cognitive Impairment. J. Intern. Med. 2004, 256, 240–246. Available online: http://doi.wiley.com/10.1111/j.1365-2796.2004.01380.x (accessed on 22 March 2021). [CrossRef]
- Simos, P.; Papastefanakis, E.; Panou, T.; Kasselimis, D. The Greek Memory Scale; Laboratory of Applied Psychology, University of Crete: Rethymno, Greece, 2011. [Google Scholar]
- Bountziouka, V.; Bathrellou, E.; Giotopoulou, A.; Katsagoni, C.; Bonou, M.; Vallianou, N.; Barbetseas, J.; Avgerinos, P.C.; Panagiotakos, D.B. Development, Repeatability and Validity Regarding Energy and Macronutrient Intake of a Semi-Quantitative Food Frequency Questionnaire: Methodological Considerations. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 659–667. Available online: https://pubmed.ncbi.nlm.nih.gov/21269818/ (accessed on 7 March 2021). [CrossRef]
- Panagiotakos, D.B.; Pitsavos, C.; Stefanadis, C. Dietary Patterns: A Mediterranean Diet Score and Its Relation to Clinical and Biological Markers of Cardiovascular Disease Risk. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 559–568. Available online: https://pubmed.ncbi.nlm.nih.gov/17126772/ (accessed on 7 March 2021). [CrossRef]
- Vgontzas, A.N.; Liao, D.; Pejovic, S.; Calhoun, S.; Karataraki, M.; Basta, M.; Fernández-Mendoza, J.; Bixler, E.O. Insomnia with short sleep duration and mortality: The Penn State cohort. Sleep 2010, 33, 1159–1164. [Google Scholar] [CrossRef]
- Mednick, S.C.; Ehrman, M. Take a Nap! Change Your Life, 1st ed.; Workman Publishing: New York, NY, USA, 2006; ISBN 978-0-7611-4290-4. [Google Scholar]
- Basta, M.; Koutentaki, E.; Vgontzas, A.N.; Zaganas, I.; Vogiatzi, E.; Gouna, G.; Bourbouli, M.; Panagiotakis, S.; Kapetanaki, S.; Fernandez-Mendoza, J.; et al. Objective daytime napping is associated with disease severity and inflammation in patients with mild to moderate dementia. J. Alzheimer’s Dis. 2020, 74, 803–815. [Google Scholar] [CrossRef]
- Basta, M.; Simos, P.; Vgontzas, A.; Koutentaki, E.; Tziraki, S.; Zaganas, I.; Panagiotakis, S.; Kapetanaki, S.; Fountoulakis, N.; Lionis, C. Associations between sleep duration and cognitive impairment in mild cognitive impairment. J. Sleep Res. 2019, 28, e12864. [Google Scholar] [CrossRef]
- Department of Health. Start Active Stay Active. 2011. Available online: https://www.gov.uk/government/publications/start-active-stay-active-a-report-on-physical-activity-from-the-four-home-countries-chief-medical-officers (accessed on 7 March 2019).
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.G. Statistical Power Analyses Using G*Power 3.1: Tests for Correlation and Regression Analyses. Behav. Res. Methods 2009, 41, 1149–1160. Available online: https://pubmed.ncbi.nlm.nih.gov/19897823/ (accessed on 9 April 2021). [CrossRef] [Green Version]
- Puzianowska-Kuźnicka, M.; Owczarz, M.; Wieczorowska-Tobis, K.; Nadrowski, P.; Chudek, J.; Slusarczyk, P.; Skalska, A.; Jonas, M.; Franek, E.; Mossakowska, M. Interleukin-6 and C-reactive protein, successful aging, and mortality: The PolSenior study. Immun. Ageing 2016, 13, 21. [Google Scholar] [CrossRef] [Green Version]
- Charlton, R.A.; Lamar, M.; Zhang, A.; Ren, X.; Ajilore, O.; Pandey, G.N.; Kumar, A. Associations between pro-inflammatory cytokines, learning, and memory in late-life depression and healthy aging. Int. J. Geriatr. Psychiatry 2018, 33, 104–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Root, M.M.; Mcginn, M.C.; Nieman, D.C.; Henson, D.A.; Heinz, S.A.; Shanely, R.A.; Knab, A.M.; Jin, F. Combined Fruit and Vegetable Intake Is Correlated with Improved Inflammatory and Oxidant Status from a Cross-Sectional Study in a Community Setting. Nutrients 2012, 4, 29–41. Available online: https://pubmed.ncbi.nlm.nih.gov/22347616/ (accessed on 7 March 2021). [CrossRef]
- Piccand, E.; Vollenweider, P.; Guessous, I.; Marques-Vidal, P. Association between Dietary Intake and Inflammatory Markers: Results from the CoLaus Study. Public Health Nutr. 2019, 22, 498–505. Available online: https://pubmed.ncbi.nlm.nih.gov/30333073/ (accessed on 11 March 2021). [CrossRef] [Green Version]
- Jiang, Y.; Wu, S.H.; Shu, X.O.; Xiang, Y.B.; Ji, B.T.; Milne, G.L.; Cai, Q.; Zhang, X.; Gao, Y.; Zheng, W.; et al. Cruciferous Vegetable Intake Is Inversely Correlated with Circulating Levels of Proinflammatory Markers in Women. J. Acad. Nutr. Diet. 2014, 114, 700–708.e2. Available online: https://pubmed.ncbi.nlm.nih.gov/24630682/ (accessed on 7 March 2021). [CrossRef] [Green Version]
- Hildreth, K.L.; Van Pelt, R.E.; Moreau, K.L.; Grigsby, J.; Hoth, K.F.; Pelak, V.; Anderson, C.A.; Parnes, B.; Kittelson, J.; Wolfe, P.; et al. Effects of Pioglitazone or Exercise in Older Adults with Mild Cognitive Impairment and Insulin Resistance: A Pilot Study. Dement. Geriatr. Cogn. Dis. Extra 2015, 5, 51–63. Available online: https://pubmed.ncbi.nlm.nih.gov/25852732/ (accessed on 7 March 2021). [CrossRef]
- Tabung, F.K.; Smith-Warner, S.A.; Chavarro, J.E.; Wu, K.; Fuchs, C.S.; Hu, F.B.; Chan, A.T.; Willett, W.C.; Giovannucci, E.L. Development and validation of an empirical dietary inflammatory index. J. Nutr. 2016, 146, 1560–1570. [Google Scholar] [CrossRef]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean Diet Pyramid: A Cultural Model for Healthy Eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. Available online: https://pubmed.ncbi.nlm.nih.gov/7754995/ (accessed on 7 March 2021). [CrossRef]
- Bertsias, G.; Linardakis, M.; Mammas, I.; Kafatos, A. Fruit and vegetables consumption in relation to health and diet of medical students in Crete, Greece. Int. J. Vitam. Nutr. Res. 2005, 75, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Rehm, C.D.; Peñalvo, J.L.; Afshin, A.; Mozaffarian, D. Dietary Intake among US Adults, 1999–2012. JAMA 2016, 315, 2542–2553. [Google Scholar] [CrossRef]
- Hoy, M.K.; Goldman, J.D.; Sebastian, R.S. Fruit and vegetable intake of US adults estimated by two methods: What We Eat in America, National Health and Nutrition Examination Survey 2009–2012. Public Health Nutr. 2016, 19, 2508–2512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nicklett, E.J.; Kadell, A.R. Fruit and vegetable intake among older adults: A scoping review. Maturitas 2013, 75, 305–312. [Google Scholar] [CrossRef] [Green Version]
- Anastasiou, C.A.; Yannakoulia, M.; Kosmidis, M.H.; Dardiotis, E.; Hadjigeorgiou, G.M.; Sakka, P.; Arampatzi, X.; Bougea, A.; Labropoulos, I.; Scarmeas, N. Mediterranean diet and cognitive health: Initial results from the Hellenic Longitudinal Investigation of Ageing and Diet. PLoS ONE 2017, 12, e0182048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, L.; Lu, R.; Jiang, L.; Liu, Z.; Peng, Y. Expression and Genetic Analysis of Tumor Necrosis Factor-α (TNF-α) G-308A Polymorphism in Sporadic Alzheimer’s Disease in a Southern China Population. Brain Res. 2009, 1247, 178–181. Available online: https://pubmed.ncbi.nlm.nih.gov/18992723/ (accessed on 7 March 2021). [CrossRef]
- Bagyinszky, E.; Van Giau, V.; Shim, K.; Suk, K.; An, S.S.A.; Kim, S.Y. Role of Inflammatory Molecules in the Alzheimer’s Disease Progression and Diagnosis. J. Neurol. Sci. 2017, 376, 242–254. Available online: https://pubmed.ncbi.nlm.nih.gov/28431620/ (accessed on 7 March 2021). [CrossRef] [PubMed]
- Kaplin, A.; Carroll, K.A.L.; Cheng, J.; Allie, R.; Lyketsos, C.G.; Calabresi, P.; Kaplin, A. IL-6 Release by LPS-Stimulated Peripheral Blood Mononuclear Cells as a Potential Biomarker in Alzheimer’s Disease. Int. Psychogeriatr. 2009, 21, 413–414. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/pmid/19040786/?tool=EBI (accessed on 7 March 2021). [CrossRef] [PubMed] [Green Version]
- Weiner, M.W.; Veitch, D.P.; Aisen, P.S.; Beckett, L.A.; Cairns, N.J.; Green, R.C.; Harvey, D.; Jack, C.R.; Jagust, W.; Liu, E.; et al. The Alzheimer’s Disease Neuroimaging Initiative: A Review of Papers Published since Its Inception. Alzheimer’s Dement. 2013, 9, e111–e194. Available online: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4108198/ (accessed on 7 March 2019). [CrossRef] [Green Version]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Bennett, D.A.; Aggarwal, N.T. MIND Diet Associated with Reduced Incidence of Alzheimer’s Disease. Alzheimer’s Dement. 2015, 11, 1007–1014. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25681666 (accessed on 8 October 2019). [CrossRef] [Green Version]
- Irwin, M.R.; Olmstead, R.; Carroll, J.E. Sleep disturbance, sleep duration, and inflammation: A systematic review and meta-analysis of cohort studies and experimental sleep deprivation. Biol. Psychiatry 2016, 80, 40–52. [Google Scholar] [CrossRef] [Green Version]
- Smagula, S.F.; Harrison, S.; Cauley, J.A.; Ancoli-Israel, S.; Cawthon, P.M.; Cummings, S.; Stone, K.L. Determinants of Change in Objectively Assessed Sleep Duration among Older Men. Am. J. Epidemiol. 2017, 185, 933–940. [Google Scholar] [CrossRef] [Green Version]
- Kripke, D.F.; Langer, R.D.; Elliott, J.A.; Klauber, M.R.; Rex, K.M. Mortality Related to Actigraphic Long and Short Sleep. Sleep Med. 2011, 12, 28–33. Available online: https://pubmed.ncbi.nlm.nih.gov/20870457/ (accessed on 7 March 2021). [CrossRef] [Green Version]
- Irwin, M.R.; Vitiello, M.V. Implications of sleep disturbance and inflammation for Alzheimer’s disease dementia. Lancet Neurol. 2019, 18, 296–306. [Google Scholar] [CrossRef]
- Spira, A.P.; Stone, K.L.; Redline, S.; Ensrud, K.E.; Ancoli-Israel, S.; Cauley, J.A.; Yaffe, K. Actigraphic Sleep Duration and Fragmentation in Older Women: Associations with Performance across Cognitive Domains. Sleep 2017, 40, zsx073. [Google Scholar] [CrossRef] [Green Version]
- Jike, M.; Itani, O.; Watanabe, N.; Buysse, D.J.; Kaneita, Y. Long sleep duration and health outcomes: A systematic review, meta-analysis and meta-regression. Sleep Med. Rev. 2018, 39, 25–36. [Google Scholar] [CrossRef]
- Azevedo Da Silva, M.; Singh-Manoux, A.; Shipley, M.J.; Vahtera, J.; Brunner, E.J.; Ferrie, J.E.; Kivimäki, M.; Nabi, H. Sleep duration and sleep disturbances partly explain the association between depressive symptoms and cardiovascular mortality: The Whitehall II cohort study. J. Sleep Res. 2014, 23, 94–97. [Google Scholar] [CrossRef] [Green Version]
- Patel, S.R.; Zhu, X.; Storfer-Isser, A.; Mehra, R.; Jenny, N.S.; Tracy, R.; Redline, S. Sleep duration and biomarkers of inflammation. Sleep 2009, 32, 200–204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Irwin, M.R. Why Sleep Is Important for Health: A Psychoneuroimmunology Perspective. Annu. Rev. Psychol. 2015, 66, 143–172. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25061767 (accessed on 3 August 2019). [CrossRef] [Green Version]
- Taveras, E.M.; Rifas-Shiman, S.L.; Rich-Edwards, J.W.; Mantzoros, C.S. Maternal Short Sleep Duration Is Associated with Increased Levels of Inflammatory Markers at 3 Years Postpartum. Metabolism 2011, 60, 982–986. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21040938 (accessed on 23 October 2019). [CrossRef] [Green Version]
- Miller, M.A.; Kandala, N.-B.; Kivimaki, M.; Kumari, M.; Brunner, E.J.; Lowe, G.D.O.; Marmot, M.G.; Cappuccio, F.P. Gender Differences in the Cross-Sectional Relationships between Sleep Duration and Markers of Inflammation: Whitehall II Study. Sleep 2009, 32, 857–864. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19639748 (accessed on 7 October 2019).
- Vgontzas, A.N.; Zoumakis, E.; Bixler, E.O.; Lin, H.-M.; Follett, H.; Kales, A.; Chrousos, G.P. Adverse Effects of Modest Sleep Restriction on Sleepiness, Performance, and Inflammatory Cytokines. J. Clin. Endocrinol. Metab. 2004, 89, 2119–2126. Available online: https://academic.oup.com/jcem/article-lookup/doi/10.1210/jc.2003-031562 (accessed on 26 July 2019). [CrossRef] [Green Version]
- Vgontzas, A.N.; Pejovic, S.; Zoumakis, E.; Lin, H.M.; Bixler, E.O.; Basta, M.; Fang, J.; Sarrigiannidis, A.; Chrousos, G.P. Daytime Napping after a Night of Sleep Loss Decreases Sleepiness, Improves Performance, and Causes Beneficial Changes in Cortisol and Interleukin-6 Secretion. Am. J. Physiol. Metab. 2007, 292, E253–E261. Available online: http://www.physiology.org/doi/10.1152/ajpendo.00651.2005 (accessed on 16 July 2019). [CrossRef]
- Pejovic, S.; Basta, M.; Vgontzas, A.N.; Kritikou, I.; Shaffer, M.L.; Tsaoussoglou, M.; Stiffler, D.; Stefanakis, Z.; Bixler, O.E.; Chrousos, G.P. Effects of Recovery Sleep after One Work Week of Mild Sleep Restriction on Interleukin-6 and Cortisol Secretion and Daytime Sleepiness and Performance. Am. J. Physiol. Metab. 2013, 305, E890–E896. Available online: https://www.physiology.org/doi/10.1152/ajpendo.00301.2013 (accessed on 7 March 2021). [CrossRef] [Green Version]
- Opp, M.R.; Kapas, L.; Toth, L.A. Cytokine involvement in the regulation of sleep. Proc. Soc. Exp. Biol. Med. 1992, 201, 16–27. [Google Scholar] [CrossRef]
- Moldofsky, H.; Lue, F.A.; Eisen, J.; Keystone, E.; Gorczynski, R.M. The relationship of interleukin-1 and immune functions to sleep in humans. Psychosom. Med. 1986, 48, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Vgontzas, A.N.; Papanicolaou, D.A.; Bixler, E.O.; Kales, A.; Tyson, K.; Chrousos, G.P. Elevation of plasma cytokines in disorders of excessive daytime sleepiness: Role of sleep disturbance and obesity. J. Clin. Endocrinol. Metab. 1997, 82, 1313–1316. [Google Scholar] [CrossRef]
- Vgontzas, A.N.; Zoumakis, E.; Lin, H.M.; Bixler, E.O.; Trakada, G.; Chrousos, G.P. Marked decrease in sleepiness in patients with sleep apnea by etanercept, a tumor necrosis factor-alpha antagonist. J. Clin. Endocrinol. Metab. 2004, 89, 4409–4413. [Google Scholar] [CrossRef] [Green Version]
- Moy, M.L.; Teylan, M.; Weston, N.A.; Gagnon, D.R.; Danilack, V.A.; Garshick, E. Daily Step Count Is Associated with Plasma c-Reactive Protein and il-6 in a us Cohort with COPD. Chest 2014, 145, 542–550. Available online: https://pubmed.ncbi.nlm.nih.gov/24091482/ (accessed on 7 March 2021). [CrossRef] [PubMed]
- Wåhlin-Larsson, B.; Carnac, G.; Kadi, F. The Influence of Systemic Inflammation on Skeletal Muscle in Physically Active Elderly Women. Age 2014, 36, 9718. Available online: https://pubmed.ncbi.nlm.nih.gov/25311555/ (accessed on 7 March 2021). [CrossRef] [Green Version]
- Fischer, C.P.; Berntsen, A.; Perstrup, L.B.; Eskildsen, P.; Pedersen, B.K. Plasma Levels of Interleukin-6 and C-Reactive Protein Are Associated with Physical Inactivity Independent of Obesity. Scand. J. Med. Sci. Sport 2007, 17, 580–587. Available online: https://pubmed.ncbi.nlm.nih.gov/17076827/ (accessed on 7 March 2021). [CrossRef] [PubMed]
- Bixler, E.O.; Vgontzas, A.N.; Ten Have, T.; Tyson, K.; Kales, A. Effects of Age on Sleep Apnea in Men. Am. J. Respir. Crit. Care Med. 1998, 157, 144–148. Available online: http://www.atsjournals.org/doi/abs/10.1164/ajrccm.157.1.9706079 (accessed on 8 October 2019). [CrossRef] [PubMed]
- Basta, M.; Zaganas, I.; Simos, P.; Koutentaki, E.; Dimovasili, C.; Mathioudakis, L.; Bourbouli, M.; Panagiotakis, S.; Kapetanaki, S.; Vgontzas, A. Apolipoprotein E ɛ4 (APOE ɛ4) Allele is Associated with Long Sleep Duration among Elderly with Cognitive Impairment. J. Alzheimer’s Dis. 2021, 79, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Moga, D.C.; Taipale, H.; Tolppanen, A.M.; Tanskanen, A.; Tiihonen, J.; Hartikainen, S.; Wu, Q.; Jicha, G.A.; Gnjidic, D. A Comparison of Sex Differences in Psychotropic Medication Use in Older People with Alzheimer’s Disease in the US and Finland. Drugs Aging 2017, 34, 55–65. Available online: https://europepmc.org/articles/PMC5253689 (accessed on 7 March 2021). [CrossRef] [PubMed] [Green Version]
| Total Group|(n = 117) | CNI (n = 54) | MCI (n = 63) | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | p | Cohen’s d | |
| Age (years) | 74.0 | 7.4 | 72.3 | 7.6 | 75.6 | 7.0 | 0.005 | 0.45 |
| Education (years) | 5.1 | 3.1 | 5.4 | 2.7 | 4.8 | 3.5 | 0.3 | 0.19 |
| Gender (%) | 0.3 | 0.18 | ||||||
| Men | 36.3 | - | 40.7 | 28.6 | ||||
| Women | 63.7 | - | 59.3 | 71.4 | ||||
| MMSE | 24.9 | 3.6 | 27.0 | 2.8 | 22.9 | 2.8 | <0.001 | 1.19 |
| GDS | 3.5 | 3.4 | 2.7 | 3.4 | 4.5 | 3.3 | 0.008 | 0.50 |
| HADS-Anxiety | 2.8 | 3.3 | 2.6 | 3.4 | 3.4 | 3.3 | 0.3 | 0.25 |
| Depression (%) | 27.4 | - | 11.1 | - | 39.7 | - | 0.001 | 0.65 |
| Psychotropic medication (%) | 31.9 | - | 24.2 | - | 44.2 | - | 0.011 | 0.53 |
| Benzodiazepine use (%) | 8.8 | - | 9.7 | - | 8.2 | - | 0.9 | 0.11 |
| BMI | 30.0 | 4.8 | 30.5 | 4.9 | 29.3 | 4.5 | 0.06 | 0.26 |
| Mean | SD | Mean | SD | ||
|---|---|---|---|---|---|
| IL-6 (pg/mL) | 1.28 | 0.9 | Energy | 2228.1 | 536.20 |
| TNF-α (pg/mL) | 1.12 | 0.6 | MDS | 34.7 | 4.6 |
| Night TST (min) | 411.2 | 70.9 | Servings per day of: | ||
| 24-h TST (min) | 448.7 | 84.2 | Vegetables | 2.54 | 1.28 |
| Night TST > 450 min (%) | 21.0 | - | Red meat | 1.27 | 0.84 |
| Night TST < 360 min (%) | 26.1 | - | Dairy | 1.06 | 0.80 |
| Night TMB (min) | 503.2 | 78.8 | Legumes | 0.65 | 0.45 |
| 24-h TMB (min) | 560.4 | 107.5 | Non-refined cereal | 0.91 | 0.94 |
| Night Sleep Efficiency | 83.0 | 8.8 | Refined cereal | 2.56 | 1.20 |
| Night WASO | 77.0 | 42.3 | Potatoes | 0.38 | 0.24 |
| Night Sleep latency | 13.0 | 12.0 | Fruit | 2.83 | 1.95 |
| Number of Awakenings | 15.9 | 6.1 | Fish | 0.83 | 0.66 |
| Sleep duration (min) | 394.0 | 115.9 | Poultry | 0.52 | 0.37 |
| Non-refreshing sleep (%) | 12.9 | - | Eggs | 0.19 | 0.19 |
| Leg movement (%) | 1.7 | - | Sweets | 0.44 | 0.45 |
| EDS (%) | 2.6 | - | Alcoholic beverages | 0.54 | 0.97 |
| Sleep Apnea symptoms (%) | 17.2 | - | |||
| Insomnia-type symptoms (%) | 32.5 | - | |||
| Physical activity (%) | 64.0 | - |
| CNI (n = 54) | MCI (n = 63) | p | Cohen’s d | CNI (n = 54) | MCI (n = 63) | p | Cohen’s d | ||
|---|---|---|---|---|---|---|---|---|---|
| IL-6 (pg/mL) | 1.36 (1.0) | 1.21 (0.7) | 0.7 | 0.17 | Energy Intake | 2455.8 (472.2) | 2017.9 (508.7) | 0.2 | 0.89 |
| TNF-α (pg/mL) | 1.07 (0.6) | 1.16 (0.6) | 0.1 | 0.16 | MDS | 35.9 (4.3) | 33.6 (4.6) | 0.016 | 0.49 |
| Night TST (min) | 408.9 (77.8) | 414.5 (64.6) | 0.4 | 0.09 | |||||
| Night TST > 450 min (%) | 16.1 | 25.0 | 0.2 | 0.24 | Servings per day of: | ||||
| Night TST < 360 min (%) | 25.8 | 27.3 | 0.9 | 0.02 | Vegetables | 2.72 (1.30) | 2.41 (1.22) | 0.7 | 0.24 |
| 24-h TST (min) | 438.0 (92.9) | 457.8 (75.3) | 0.2 | 0.24 | Red meat | 1.47 (0.95) | 1.12 (0.68) | 0.002 | 0.55 |
| Night TMB (min) | 493.4 (88.1) | 511.6 (68.3) | 0.14 | 0.25 | Dairy products | 1.08 (0.80) | 1.05 (0.82) | 0.5 | 0.04 |
| 24-h TMB (min) | 539.8 (117.9) | 578.0 (95.9) | 0.1 | 0.36 | Legumes | 0.64 (0.40) | 0.66 (0.49) | 0.8 | 0.05 |
| Night Sleep Efficiency | 82.9 (8.0) | 81.2 (9.3) | 0.3 | 0.22 | Non-refined cereals | 1.14 (1.08) | 0.73 (0.78) | 0.3 | 0.44 |
| Night WASO | 71.1 (41.3) | 82.0 (43.2) | 0.3 | 0.25 | Refined cereals | 2.65 (1.16) | 2.48 (1.24) | 0.2 | 0.13 |
| Night Sleep latency | 11.2 (7.0) | 14.5 (14.7) | 0.1 | 0.28 | Potatoes | 0.38 (0.23) | 0.40 (0.25) | 0.3 | 0.08 |
| Number of Awakenings | 15.4 (6.5) | 16.3 (5.8) | 0.9 | 0.15 | Fruit | 3.14 (1.77) | 2.57 (2.07) | 0.2 | 0.34 |
| Sleep duration (min) | 412.3 (123.1) | 378.0 (106.6) | 0.5 | 0.29 | Fish | 0.89 (0.74) | 0.78 (0.58) | 0.5 | 0.17 |
| Non-refreshing sleep (%) | 11.1 | 14.5 | 0.5 | 0.16 | Poultry | 0.51 (0.36) | 0.53 (0.39) | 0.7 | 0.06 |
| Leg movement (%) | 1.9 | 1.6 | 0.9 | 0.02 | Eggs | 0.24 (0.22) | 0.15 (0.14) | 0.017 | 0.47 |
| EDS (%) | 1.6 | 3.7 | 0.6 | 0.16 | Sweets | 0.54 (0.48) | 0.35 (0.41) | 0.006 | 0.43 |
| Sleep Apnea symptoms (%) | 16.7 | 17.7 | 0.9 | 0.04 | Alcoholic beverages | 0.74 (1.21) | 0.38 (0.69) | 0.1 | 0.39 |
| Insomnia-type symptoms (%) | 27.8 | 36.5 | 0.3 | 0.23 | |||||
| Physical activity (%) | 62.0 | 65.6 | 0.9 | 0.03 |
| TNF-α | IL-6 | |
|---|---|---|
| Age | 0.230 †1 | 0.157 |
| Gender (male) | 0.157 | 0.147 |
| Depression Diagnosis | −0.164 | −0.081 |
| Physical activity | −0.070 | −0.303 †5 |
| Night TST > 450 min | 0.202 *2 | 0.079 |
| Insomnia Symptoms | −0.131 | −0.043 |
| Consumption of vegetables | −0.407 †3 | −0.393 †6 |
| Consumption of red meat | −0.190 *4 | −0.140 |
| Consumption of dairy | 0.142 | 0.059 |
| Consumption of legumes | −0.049 | −0.134 |
| B | 95% CI | p Value | |
|---|---|---|---|
| Night TST > 450 min | 0.230 | (0.010 to 0.449) | 0.04 1 |
| Consumption of vegetables | −0.203 | (−0.289 to −0.117) | 0.001 2 |
| Consumption of red meat | −0.102 | (−0.077 to 0.282) | 0.3 |
| Constant | 0.595 | (−0.741 to 1.931) | 0.4 |
| B | 95% CI | p Value | |
|---|---|---|---|
| Physical activity | −0.538 | (−0.847 to −0.230) | 0.001 1 |
| Consumption of vegetables | −0.300 | (−0.428 to −0.172) | 0.001 2 |
| Constant | 1.887 | (−1.169 to 3.943) | 0.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basta, M.; Belogianni, C.; Yannakoulia, M.; Zaganas, I.; Panagiotakis, S.; Simos, P.; Vgontzas, A.N. Poor Diet, Long Sleep, and Lack of Physical Activity Are Associated with Inflammation among Non-Demented Community-Dwelling Elderly. Healthcare 2022, 10, 143. https://doi.org/10.3390/healthcare10010143
Basta M, Belogianni C, Yannakoulia M, Zaganas I, Panagiotakis S, Simos P, Vgontzas AN. Poor Diet, Long Sleep, and Lack of Physical Activity Are Associated with Inflammation among Non-Demented Community-Dwelling Elderly. Healthcare. 2022; 10(1):143. https://doi.org/10.3390/healthcare10010143
Chicago/Turabian StyleBasta, Maria, Christina Belogianni, Mary Yannakoulia, Ioannis Zaganas, Symeon Panagiotakis, Panagiotis Simos, and Alexandros N. Vgontzas. 2022. "Poor Diet, Long Sleep, and Lack of Physical Activity Are Associated with Inflammation among Non-Demented Community-Dwelling Elderly" Healthcare 10, no. 1: 143. https://doi.org/10.3390/healthcare10010143
APA StyleBasta, M., Belogianni, C., Yannakoulia, M., Zaganas, I., Panagiotakis, S., Simos, P., & Vgontzas, A. N. (2022). Poor Diet, Long Sleep, and Lack of Physical Activity Are Associated with Inflammation among Non-Demented Community-Dwelling Elderly. Healthcare, 10(1), 143. https://doi.org/10.3390/healthcare10010143







