Effectiveness of Physiotherapy in Patients with Ankylosing Spondylitis: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source and Search Strategy
2.2. Inclusion Criteria and Study Selection
2.3. Data Extraction and Management
2.4. Assessment of Risk of Bias in Included Studies
2.5. Data Synthesis
2.6. Summary of Findings
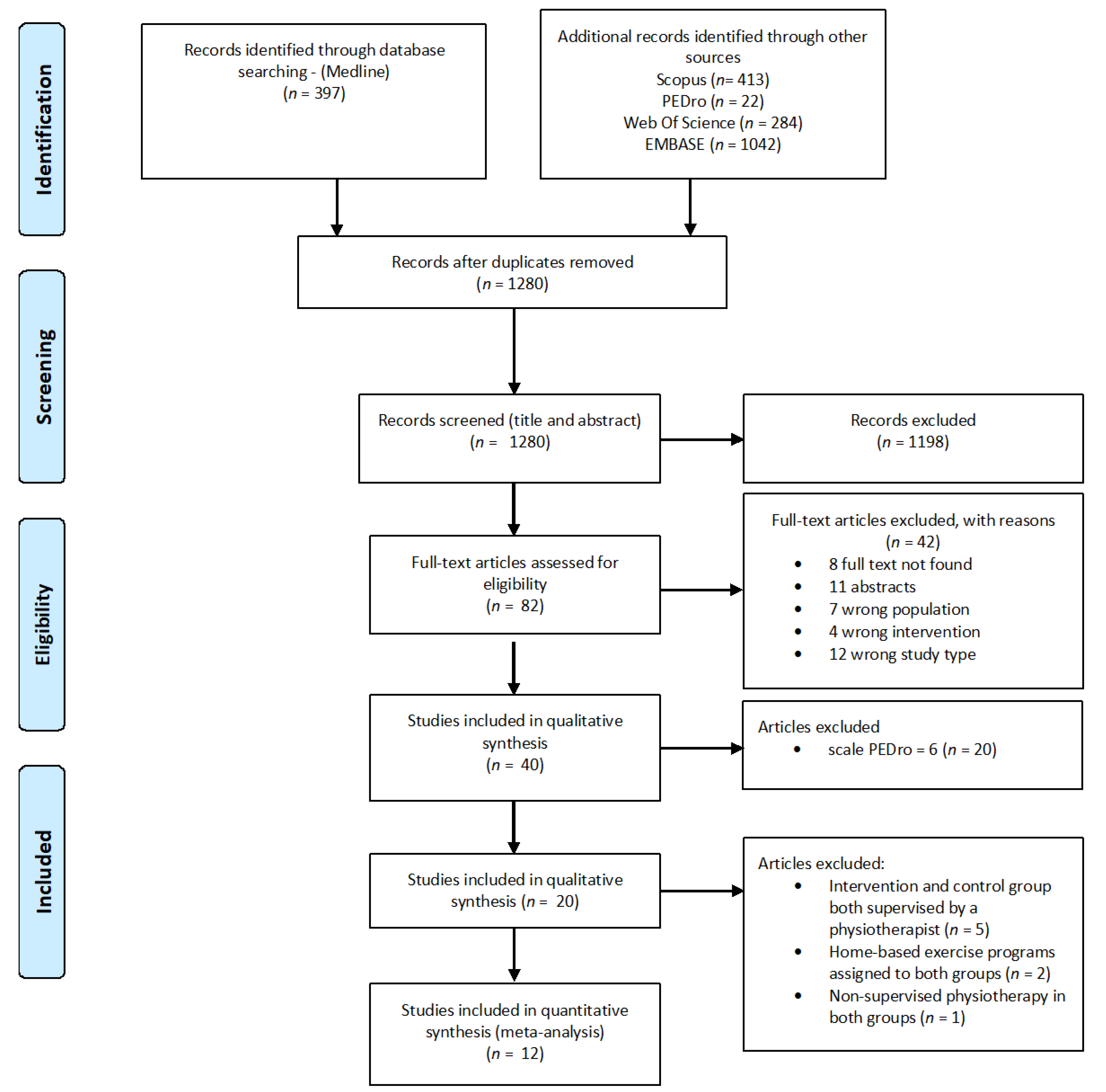
3. Results
3.1. Study Characteristics
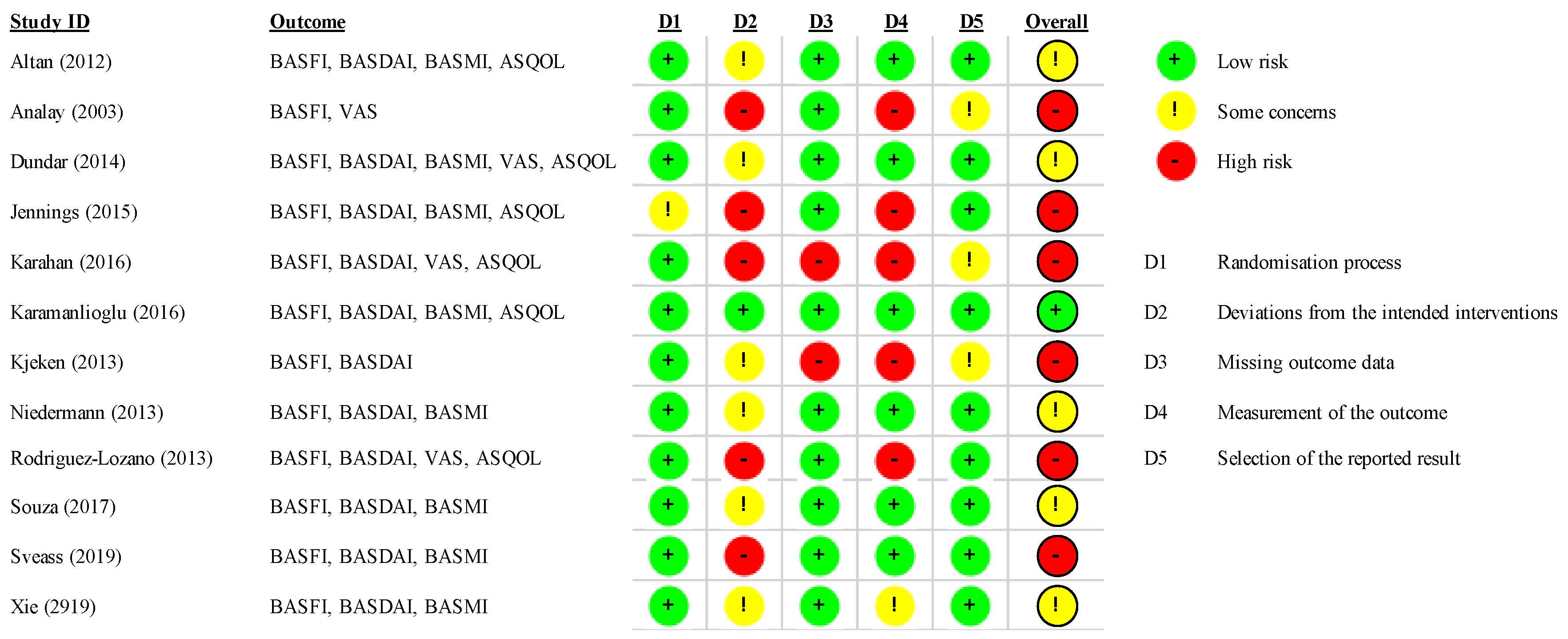
3.2. Risk of Bias
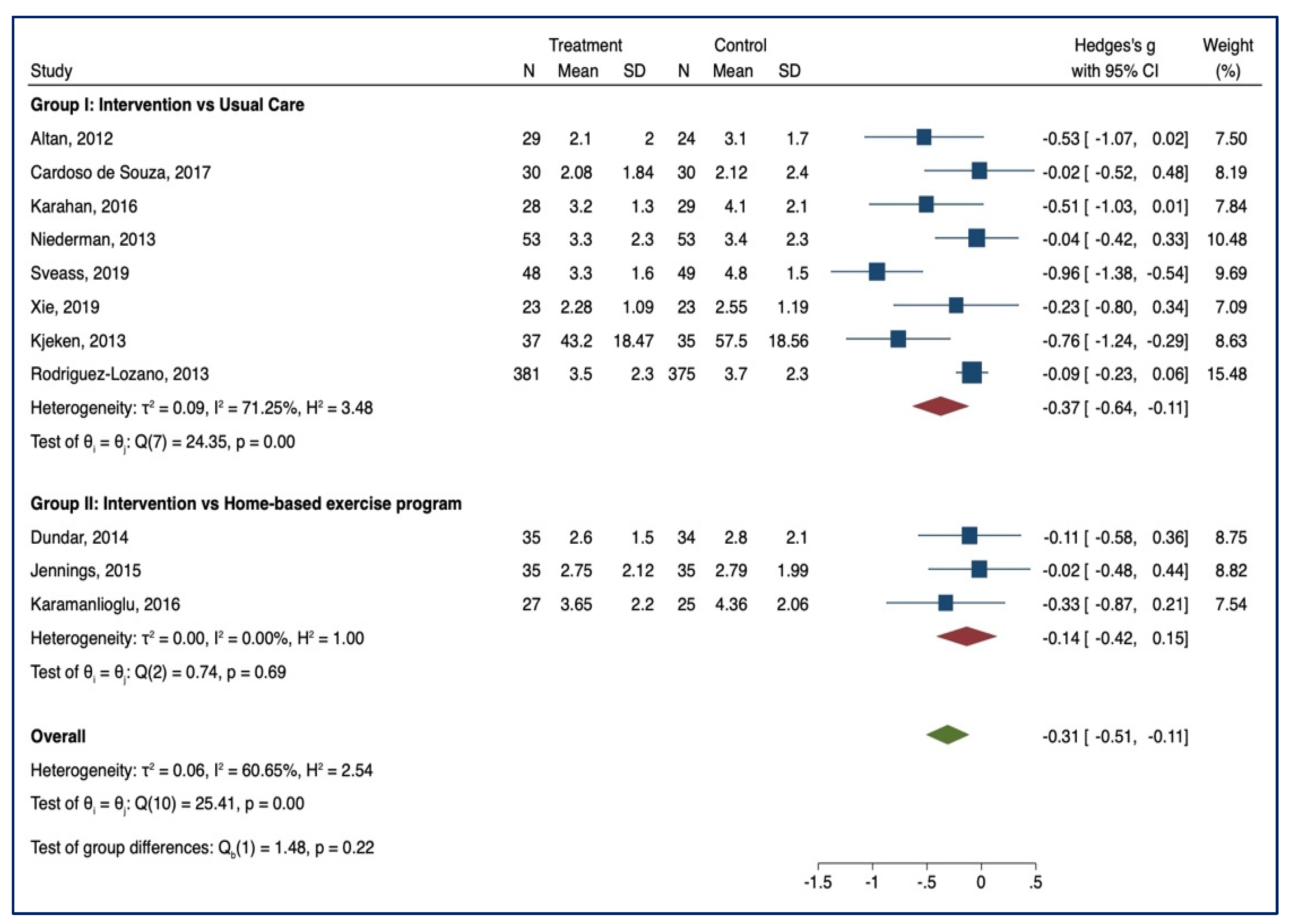
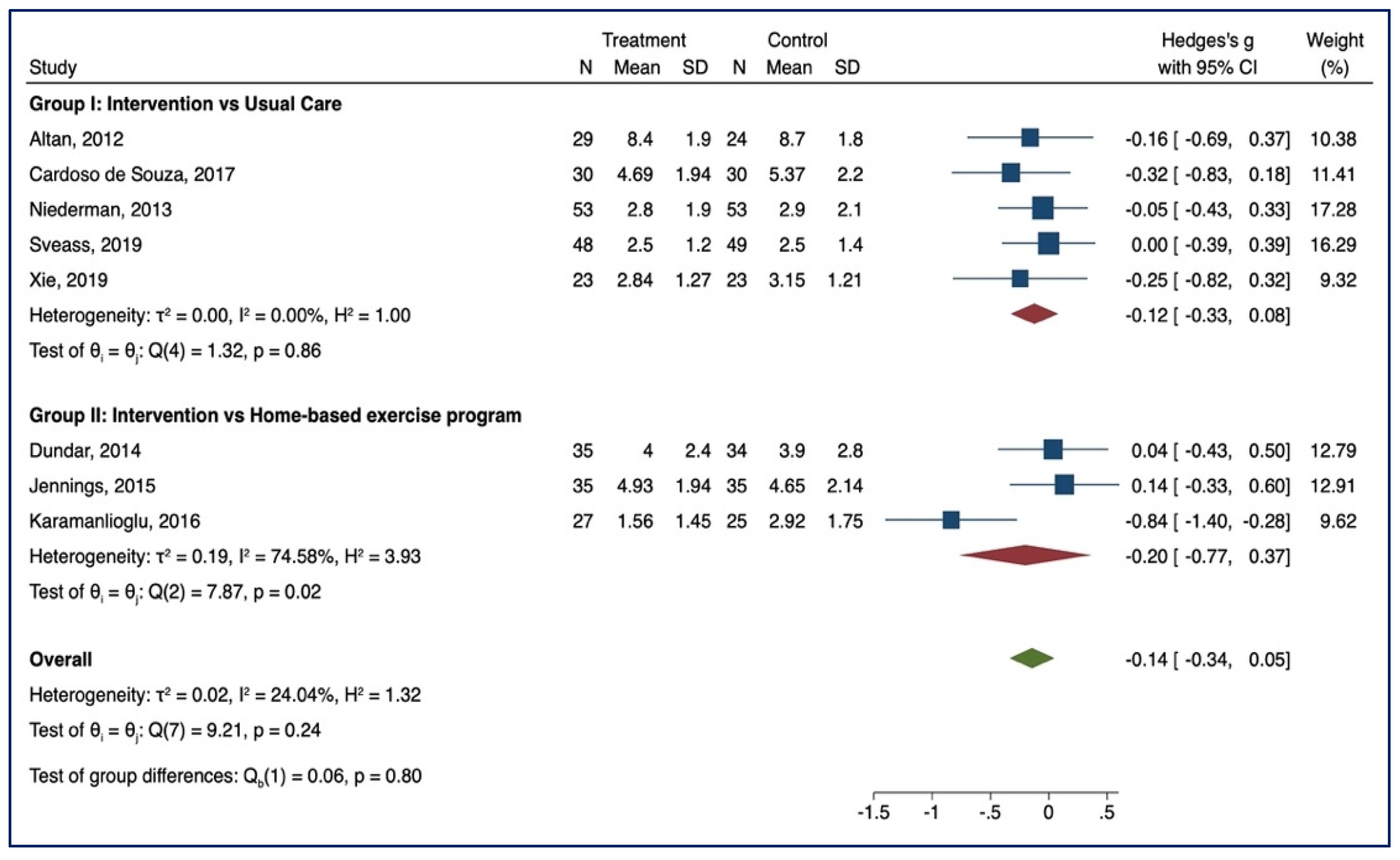
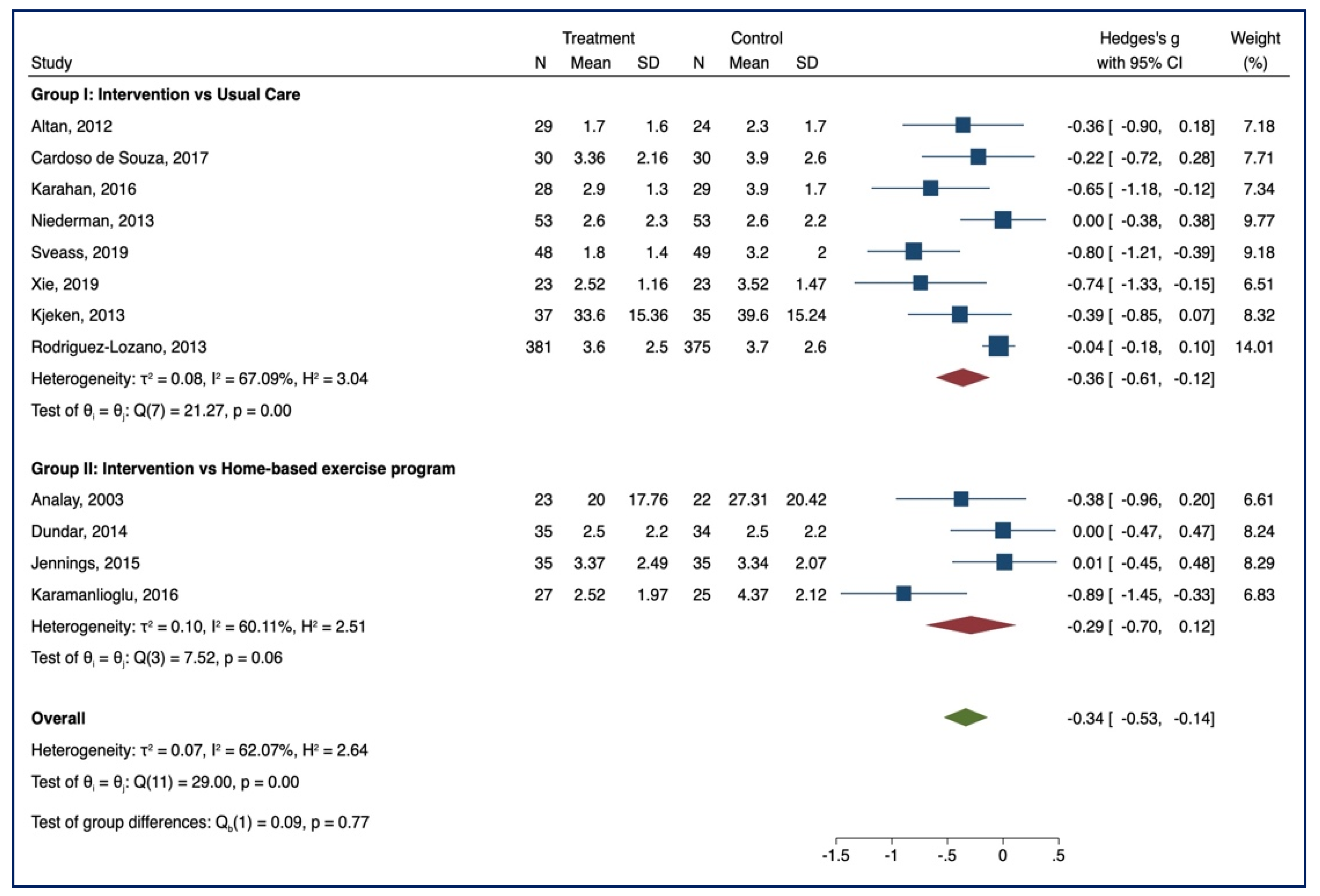
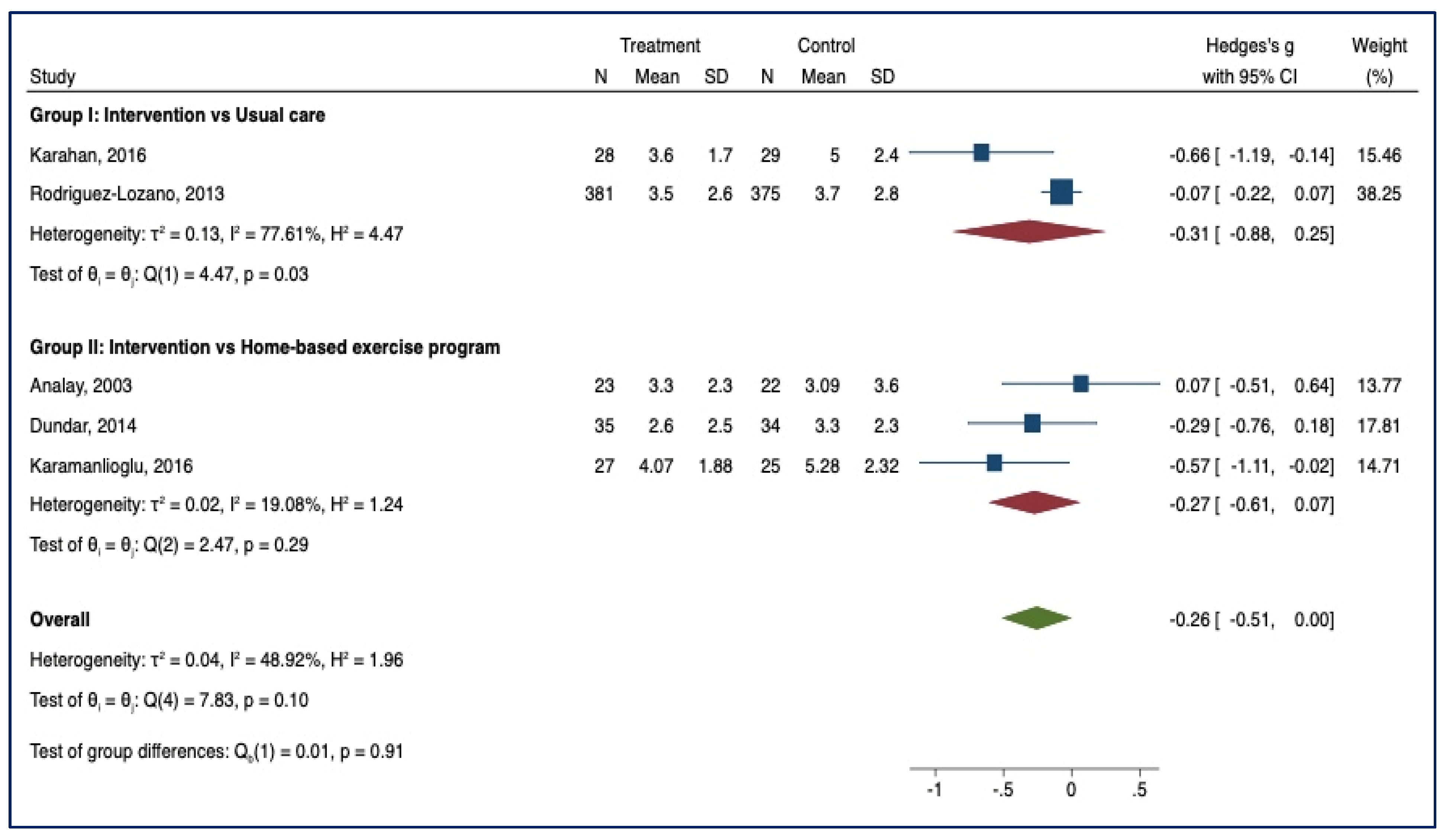
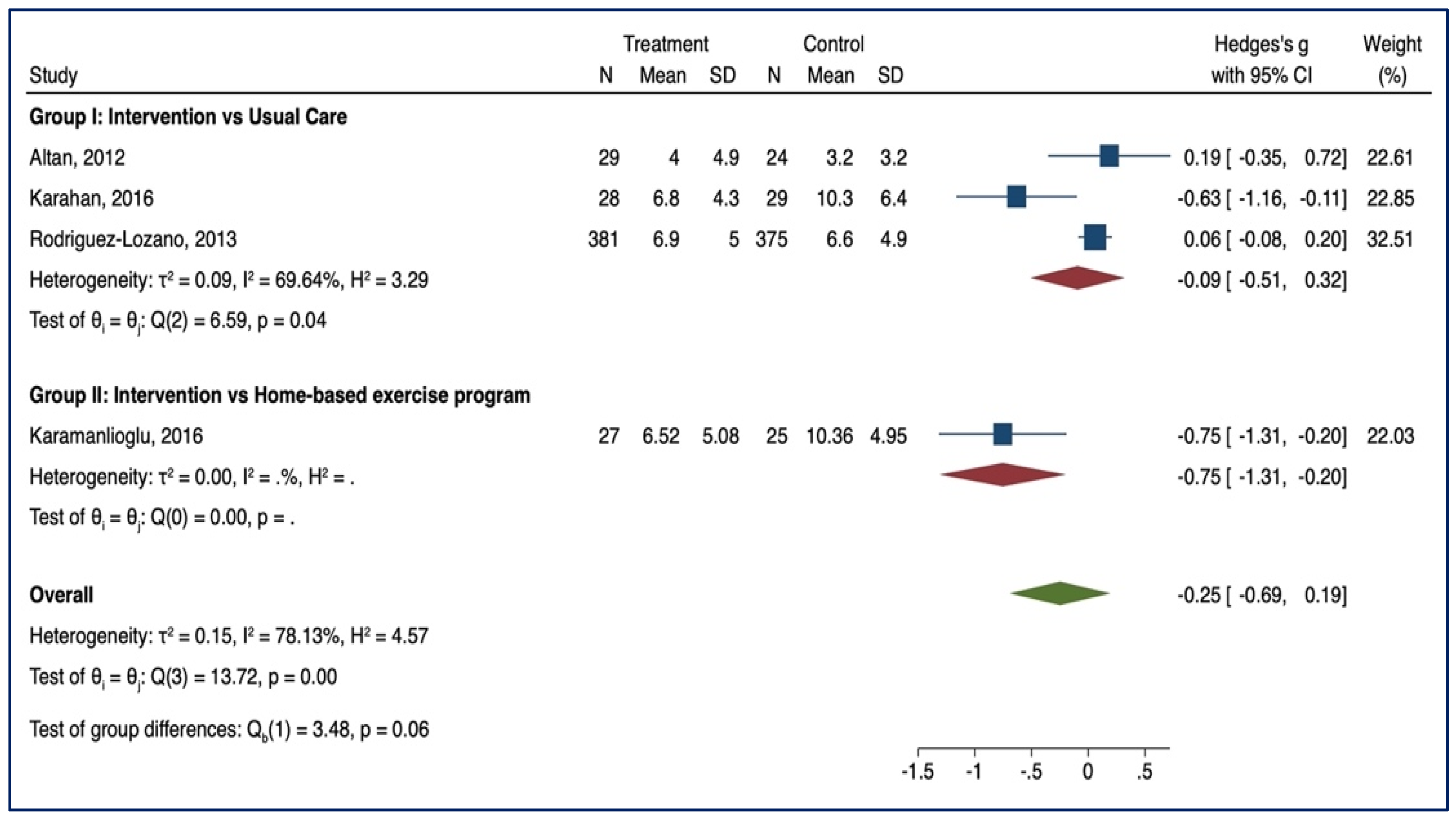
3.3. Meta-Analysis of Outcome
3.3.1. BASDAI
3.3.2. BASMI
3.3.3. BASFIs
3.3.4. VAS
3.3.5. ASQoL
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, W.; He, X.; Cheng, K.; Zhang, L.; Chen, D.; Wang, X.; Qiu, G.; Cao, X.; Weng, X. Ankylosing spondylitis: Etiology, pathogenesis, and treatments. Bone Res. 2019, 7, 22. [Google Scholar] [CrossRef]
- Shahid, A.; Mushtaq, H.S.; Azfar, S.M. Spontaneous bilateral quadriceps rupture in a patient with ankylosing spondylitis: A case report. J. Pak. Med. Assoc. 2017, 67, 150–152. [Google Scholar] [PubMed]
- Exarchou, S.; Lindstrom, U.; Askling, J.; Eriksson, J.K.; Forsblad-d’Elia, H.; Neovius, M.; Turesson, C.; Kristensen, L.E.; Jacobsson, L.T. The prevalence of clinically diagnosed ankylosing spondylitis and its clinical manifestations: A nationwide register study. Arthritis Res. Ther. 2015, 17, 118. [Google Scholar] [CrossRef]
- Stolwijk, C.; Boonen, A.; van Tubergen, A.; Reveille, J.D. Epidemiology of spondyloarthritis. Rheum. Dis. Clin. N. Am. 2012, 38, 441–476. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.H.; Chen, Y.M.; Lai, K.L.; Hsieh, T.Y.; Hung, W.T.; Lin, C.T.; Tseng, C.W.; Tang, K.T.; Chou, Y.Y.; Wu, Y.D.; et al. Gender difference in ASAS HI among patients with ankylosing spondylitis. PLoS ONE 2020, 15, e0235678. [Google Scholar] [CrossRef] [PubMed]
- Akkoc, N.; Direskeneli, H.; Erdem, H.; Gul, A.; Kabasakal, Y.; Kiraz, S.; Tezer, D.B.; Hacibedel, B.; Hamuryudan, V. Direct and indirect costs associated with ankylosing spondylitis and related disease activity scores in Turkey. Rheumatol. Int. 2015, 35, 1473–1478. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Azevedo, V.F.; Rossetto, C.N.; Lorencetti, P.G.; Tramontin, M.Y.; Fornazari, B.; Araujo, D.V. Indirect and direct costs of treating patients with ankylosing spondylitis in the Brazilian public health system. Rev. Bras. Reumatol. Engl. Ed. 2016, 56, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Li, J.; He, C.; Li, D.; Tong, W.; Zou, Y.; Xu, W. Role of HLA-B27 in the pathogenesis of ankylosing spondylitis (Review). Mol. Med. Rep. 2017, 15, 1943–1951. [Google Scholar] [CrossRef] [PubMed]
- Reveille, J.D.; Weisman, M.H. The epidemiology of back pain, axial spondyloarthritis and HLA-B27 in the United States. Am. J. Med. Sci. 2013, 345, 431–436. [Google Scholar] [CrossRef] [PubMed]
- International Genetics of Ankylosing Spondylitis Consortium; Cortes, A.; Hadler, J.; Pointon, J.P.; Robinson, P.C.; Karaderi, T.; Leo, P.; Cremin, K.; Pryce, K.; Harris, J.; et al. Identification of multiple risk variants for ankylosing spondylitis through high-density genotyping of immune-related loci. Nat. Genet. 2013, 45, 730–738. [Google Scholar] [CrossRef]
- Wang, R.; Ward, M.M. Epidemiology of axial spondyloarthritis: An update. Curr. Opin. Rheumatol. 2018, 30, 137–143. [Google Scholar] [CrossRef]
- Collantes-Estevez, E.; del Mazo, A.C.; Munoz-Gomariz, E. Assessment of 2 systems of spondyloarthropathy diagnostic and classification criteria (Amor and ESSG) by a Spanish multicenter study. European Spondyloarthropathy Study Group. J. Rheumatol. 1995, 22, 246–251. [Google Scholar]
- Moll, J.M. Ankylosing spondylitis. Nurs. Times 1973, 69, 985–988. [Google Scholar]
- Van der Linden, S.; Valkenburg, H.A.; Cats, A. Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum. 1984, 27, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.; Sieper, J. Ankylosing spondylitis. Lancet 2007, 369, 1379–1390. [Google Scholar] [CrossRef]
- Smolen, J.S.; Braun, J.; Dougados, M.; Emery, P.; Fitzgerald, O.; Helliwell, P.; Kavanaugh, A.; Kvien, T.K.; Landewe, R.; Luger, T.; et al. Treating spondyloarthritis, including ankylosing spondylitis and psoriatic arthritis, to target: Recommendations of an international task force. Ann. Rheum. Dis. 2014, 73, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.; van den Berg, R.; Baraliakos, X.; Boehm, H.; Burgos-Vargas, R.; Collantes-Estevez, E.; Dagfinrud, H.; Dijkmans, B.; Dougados, M.; Emery, P.; et al. 2010 update of the ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann. Rheum. Dis. 2011, 70, 896–904. [Google Scholar] [CrossRef]
- Daikh, D.I.; Chen, P.P. Advances in managing ankylosing spondylitis. F1000Prime Rep. 2014, 6, 78. [Google Scholar] [CrossRef]
- Tahir, H. Therapies in ankylosing spondylitis-from clinical trials to clinical practice. Rheumatology 2018, 57, vi23–vi28. [Google Scholar] [CrossRef]
- Arem, H.; Moore, S.C.; Patel, A.; Hartge, P.; de Gonzalez, A.B.; Visvanathan, K.; Campbell, P.T.; Freedman, M.; Weiderpass, E.; Adami, H.O.; et al. Leisure time physical activity and mortality: A detailed pooled analysis of the dose-response relationship. JAMA Intern. Med. 2015, 175, 959–967. [Google Scholar] [CrossRef]
- Millner, J.R.; Barron, J.S.; Beinke, K.M.; Butterworth, R.H.; Chasle, B.E.; Dutton, L.J.; Lewington, M.A.; Lim, E.G.; Morley, T.B.; O’Reilly, J.E.; et al. Exercise for ankylosing spondylitis: An evidence-based consensus statement. Semin. Arthritis Rheum. 2016, 45, 411–427. [Google Scholar] [CrossRef] [PubMed]
- Vidoni, E.D.; Johnson, D.K.; Morris, J.K.; Van Sciver, A.; Greer, C.S.; Billinger, S.A.; Donnelly, J.E.; Burns, J.M. Dose-Response of Aerobic Exercise on Cognition: A Community-Based, Pilot Randomized Controlled Trial. PLoS ONE 2015, 10, e0131647. [Google Scholar] [CrossRef] [PubMed]
- Elyan, M.; Khan, M.A. The role of nonsteroidal anti-inflammatory medications and exercise in the treatment of ankylosing spondylitis. Curr. Rheumatol. Rep. 2006, 8, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Dagfinrud, H.; Kvien, T.K.; Hagen, K.B. Physiotherapy interventions for ankylosing spondylitis. Cochrane Database Syst. Rev. 2008, 1, CD002822. [Google Scholar] [CrossRef]
- Wang, C.Y.; Chiang, P.Y.; Lee, H.S.; Wei, J.C. The effectiveness of exercise therapy for ankylosing spondylitis: A review. Int. J. Rheum. Dis. 2009, 12, 207–210. [Google Scholar] [CrossRef]
- Van den Berg, R.; Baraliakos, X.; Braun, J.; van der Heijde, D. First update of the current evidence for the management of ankylosing spondylitis with non-pharmacological treatment and non-biologic drugs: A systematic literature review for the ASAS/EULAR management recommendations in ankylosing spondylitis. Rheumatology 2012, 51, 1388–1396. [Google Scholar] [CrossRef]
- Regnaux, J.P.; Davergne, T.; Palazzo, C.; Roren, A.; Rannou, F.; Boutron, I.; Lefevre-Colau, M.M. Exercise programmes for ankylosing spondylitis. Cochrane Database Syst. Rev. 2019, 10, CD011321. [Google Scholar] [CrossRef]
- Korpi, H.; Peltokallio, L.; Piirainen, A. The story model of physiotherapy students’ professional development. Narrative research. Eur. J. Physiother. 2014, 16, 219–229. [Google Scholar] [CrossRef]
- Edwards, I.; Jones, M.; Carr, J.; Braunack-Mayer, A.; Jensen, G.M. Clinical reasoning strategies in physical therapy. Phys. Ther. 2004, 84, 312–330; discussion 331–335. [Google Scholar] [CrossRef]
- Friedrich, M.; Gittler, G.; Halberstadt, Y.; Cermak, T.; Heiller, I. Combined exercise and motivation program: Effect on the compliance and level of disability of patients with chronic low back pain: A randomized controlled trial. Arch. Phys. Med. Rehabil. 1998, 79, 475–487. [Google Scholar] [CrossRef]
- Schünemann, H.J.; Higgins, J.P.T.; Vist, G.E.; Glasziou, P.; Guyatt, G.H. Presenting results and ‘Summary of findings’ tables. In Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (Updated March 2011); Higgins, J.P.T., Green, S., Eds.; The Cochrane Collaboration: London, UK, 2011. [Google Scholar]
- Schünemann, H.J.; Vist, G.E.; Higgins, J.P.T.; Deeks, J.J.; Glasziou, P. Chapter 12: Interpreting results and drawing conclusions. In Cochrane Handbook for Systematic Review of Interventions Version 5.1.0 (Updated March 2011); Higgins, J.P.T., Green, S., Eds.; The Cochrane Collaboration: London, UK, 2011. [Google Scholar]
- Altan, L.; Korkmaz, N.; Dizdar, M.; Yurtkuran, M. Effect of Pilates training on people with ankylosing spondylitis. Rheumatol. Int. 2012, 32, 2093–2099. [Google Scholar] [CrossRef] [PubMed]
- Analay, Y.; Ozcan, E.; Karan, A.; Diracoglu, D.; Aydin, R. The effectiveness of intensive group exercise on patients with ankylosing spondylitis. Clin. Rehabil. 2003, 17, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Dundar, U.; Solak, O.; Toktas, H.; Demirdal, U.S.; Subasi, V.; Kavuncu, V.; Evcik, D. Effect of aquatic exercise on ankylosing spondylitis: A randomized controlled trial. Rheumatol. Int. 2014, 34, 1505–1511. [Google Scholar] [CrossRef]
- Jennings, F.; Oliveira, H.A.; de Souza, M.C.; Vda, G.C.; Natour, J. Effects of Aerobic Training in Patients with Ankylosing Spondylitis. J. Rheumatol. 2015, 42, 2347–2353. [Google Scholar] [CrossRef] [PubMed]
- Karahan, A.Y.; Tok, F.; Yildirim, P.; Ordahan, B.; Turkoglu, G.; Sahin, N. The Effectiveness of Exergames in Patients with Ankylosing Spondylitis: A Randomized Controlled Trial. Adv. Clin. Exp. Med. 2016, 25, 931–936. [Google Scholar] [CrossRef]
- Kjeken, I.; Bo, I.; Ronningen, A.; Spada, C.; Mowinckel, P.; Hagen, K.B.; Dagfinrud, H. A three-week multidisciplinary in-patient rehabilitation programme had positive long-term effects in patients with ankylosing spondylitis: Randomized controlled trial. J. Rehabil. Med. 2013, 45, 260–267. [Google Scholar] [CrossRef]
- Niedermann, K.; Sidelnikov, E.; Muggli, C.; Dagfinrud, H.; Hermann, M.; Tamborrini, G.; Ciurea, A.; Bischoff-Ferrari, H. Effect of cardiovascular training on fitness and perceived disease activity in people with ankylosing spondylitis. Arthritis Care Res. 2013, 65, 1844–1852. [Google Scholar] [CrossRef]
- Rodriguez-Lozano, C.; Juanola, X.; Cruz-Martinez, J.; Pena-Arrebola, A.; Mulero, J.; Gratacos, J.; Collantes, E.; Spondyloarthropathies Study Group of the Spanish Society of Rheumatology. Outcome of an education and home-based exercise programme for patients with ankylosing spondylitis: A nationwide randomized study. Clin. Exp. Rheumatol. 2013, 31, 739–748. [Google Scholar]
- Karamanlioglu, D.S.; Aktas, I.; Ozkan, F.U.; Kaysin, M.; Girgin, N. Effectiveness of ultrasound treatment applied with exercise therapy on patients with ankylosing spondylitis: A double-blind, randomized, placebo-controlled trial. Rheumatol. Int. 2016, 36, 653–661. [Google Scholar] [CrossRef]
- Souza, M.C.; Jennings, F.; Morimoto, H.; Natour, J. Swiss ball exercises improve muscle strength and walking performance in ankylosing spondylitis: A randomized controlled trial. Rev. Bras. Reumatol. Engl. Ed. 2017, 57, 45–55. [Google Scholar] [CrossRef]
- Sveaas, S.H.; Bilberg, A.; Berg, I.J.; Provan, S.A.; Rollefstad, S.; Semb, A.G.; Hagen, K.B.; Johansen, M.W.; Pedersen, E.; Dagfinrud, H. High intensity exercise for 3 months reduces disease activity in axial spondyloarthritis (axSpA): A multicentre randomised trial of 100 patients. Br. J. Sports Med. 2020, 54, 292–297. [Google Scholar] [CrossRef]
- Xie, Y.; Guo, F.; Lu, Y.; Guo, Y.; Wei, G.; Lu, L.; Ji, W.; Qian, X. A 12-week Baduanjin Qigong exercise improves symptoms of ankylosing spondylitis: A randomized controlled trial. Complement. Ther. Clin. Pract. 2019, 36, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Higgins, J.P.T.; Sterne, J.A.C. Chapter 13: Assessing risk of bias due to missing results in a synthesis. In Cochrane Handbook for Systematic Reviews of Interventions Version 6.2; Cochrane, Ed.; Cochrane: London, UK, 2021. [Google Scholar]
- Sharan, D.; Rajkumar, J.S. Physiotherapy for Ankylosing Spondylitis: Systematic Review and a Proposed Rehabilitation Protocol. Curr. Rheumatol. Rev. 2017, 13, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Passalent, L.A.; Soever, L.J.; O’Shea, F.D.; Inman, R.D. Exercise in ankylosing spondylitis: Discrepancies between recommendations and reality. J. Rheumatol. 2010, 37, 835–841. [Google Scholar] [CrossRef] [PubMed]
- Giannotti, E.; Trainito, S.; Arioli, G.; Rucco, V.; Masiero, S. Effects of physical therapy for the management of patients with ankylosing spondylitis in the biological era. Clin. Rheumatol. 2014, 33, 1217–1230. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.C.; van der Heijde, D.; Dougados, M.; Woolley, J.M. Reductions in health-related quality of life in patients with ankylosing spondylitis and improvements with etanercept therapy. Arthritis Rheum. 2005, 53, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Salaffi, F.; Carotti, M.; Gasparini, S.; Intorcia, M.; Grassi, W. The health-related quality of life in rheumatoid arthritis, ankylosing spondylitis, and psoriatic arthritis: A comparison with a selected sample of healthy people. Health Qual. Life Outcomes 2009, 7, 25. [Google Scholar] [CrossRef]
- Ward, M.M. Health-related quality of life in ankylosing spondylitis: A survey of 175 patients. Arthritis Care Res. 1999, 12, 247–255. [Google Scholar] [CrossRef]
- Barlow, J.H.; Macey, S.J.; Struthers, G.R. Gender, depression, and ankylosing spondylitis. Arthritis Care Res. 1993, 6, 45–51. [Google Scholar] [CrossRef]
- Pecourneau, V.; Degboe, Y.; Barnetche, T.; Cantagrel, A.; Constantin, A.; Ruyssen-Witrand, A. Effectiveness of Exercise Programs in Ankylosing Spondylitis: A Meta-Analysis of Randomized Controlled Trials. Arch. Phys. Med. Rehabil. 2018, 99, 383–389.e1. [Google Scholar] [CrossRef]
| First Author | Country | Year | Sample IG | Sample CG | Mean Age * | Exercise Description | Training Frequency | Mode of Combination | Training Mode | No. of Sessions | CG Activity | BASFI | BASDAI | BASMI | VAS | ASQoL | Main Results |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Altan | Turkey | 2012 | 29 | 24 | 45.23 | Pilates | 3 times a week for 12 weeks | Alternative | group | 36 | Usual care | yes | yes | yes | no | yes | Significant difference for BASDAI at 12 weeks (p < 0.01) and BASMI and BASFI at 24 weeks (p < 0.05) in the intervention group |
| Analay | Turkey | 2003 | 23 | 22 | 36 | Intensive exercise programme | 3 times a week for 6 weeks | Simultaneous | group | 18 | Home-based exercise | yes | no | no | yes | no | Statistically significant difference in all parameters except pain (p < 0.05) |
| Dundar | Turkey | 2014 | 35 | 34 | 42.7 | Aquatic exercise | 5 times a week for 4 weeks | Alternative | group | 20 | Home-based exercise | yes | yes | yes | yes | yes | Significant improvements for all parameters in both groups after treatment at week 4 and week 12 (p < 0.05) |
| Jennings | Brazil | 2015 | 35 | 35 | 41.6 | Aerobic and stretching exercise | 3 times a week for 12 weeks | Alternative | individual | 36 | Stretching exercise | yes | yes | yes | no | yes | Significant improvement pre-post (p < 0.05), but no difference between groups |
| Karahan | Turkey | 2016 | 28 | 29 | 36.4 | Exercise with videogame | 5 days a week for 8 weeks | Alternative | individual | 40 | Usual care | yes | yes | no | yes | yes | Significant differences between the two groups in VAS, BASFI, BASDAI and ASQoL; considerable improvement in the intervention group (p < 0.05) |
| Karamanlioglu | Turkey | 2016 | 27 | 25 | 39.65 | Ultrasound therapy and education programme | US: 10 sessions—Exercise: 5 times a week for 2 weeks | Alternative | individual | 10 | Instruction on exercise therapy | yes | yes | yes | no | yes | Significant results in intervention group for BASMI (p < 0.05) after 2 weeks and daily pain (p < 0.01), BASDAI (p < 0.05), and ASQoL (p < 0.05) after 6 weeks. |
| Kjeken | Norway | 2013 | 37 | 35 | 49.2 | Rehabilitation program | Pool: 3–5 sessions/week—Gym: 2–3 sessions/week—Outdoors: 3 sessions/week | Alternative | individual | NA | Usual care | yes | yes | no | no | no | Significant improvement in the intervention group for BASDAI (p < 0.05) |
| Niederman ** | Switzerland | 2013 | 53 | 53 | 48.9 | Cardiovascular training | 2 times a week for 12 weeks | Alternative | individual | 24 | Usual care | yes | yes | yes | no | no | After 3 months, significant improvement in the intervention group (p < 0.001) |
| Rodriguez-Lozano | Spain | 2013 | 381 | 375 | 45.5 | Education programme + exercise | 2 h informative session | Alternative | group | NA | Usual care | yes | yes | no | yes | yes | After 6 months, significant difference in intervention group for BASDAI (p < 0.01), BASFI (p < 0.01), VAS (p = 0.02), and ASQoL (p < 0.01) |
| Souza | Brazil | 2017 | 30 | 30 | 44.4 | Exercise with Swiss ball | 2 times a week for 16 weeks | Alternative | group | 32 | Usual care | yes | yes | yes | no | no | No significant differences between groups for BASFI and BASMI. |
| Sveass | Norway | 2019 | 48 | 49 | 45.7 | Cardiorespiratory and muscular strength exercise | 2 times per week for 12 weeks | Alternative | individual | 24 | Usual care | yes | yes | yes | no | no | Significant improvement in the intervention group for BASDAI (p < 0.001) and BASFI (p < 0.001) |
| Xie | China | 2019 | 23 | 23 | 18–60 * | Baduanjin Qigong exercise | First phase: twice per week for 4 weeks—Second phase: 3 times per week for 8 weeks | Alternative | NA | 32 | Usual care | yes | yes | yes | no | no | No difference between groups |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gravaldi, L.P.; Bonetti, F.; Lezzerini, S.; De Maio, F. Effectiveness of Physiotherapy in Patients with Ankylosing Spondylitis: A Systematic Review and Meta-Analysis. Healthcare 2022, 10, 132. https://doi.org/10.3390/healthcare10010132
Gravaldi LP, Bonetti F, Lezzerini S, De Maio F. Effectiveness of Physiotherapy in Patients with Ankylosing Spondylitis: A Systematic Review and Meta-Analysis. Healthcare. 2022; 10(1):132. https://doi.org/10.3390/healthcare10010132
Chicago/Turabian StyleGravaldi, Luca Pontone, Francesca Bonetti, Simona Lezzerini, and Fernando De Maio. 2022. "Effectiveness of Physiotherapy in Patients with Ankylosing Spondylitis: A Systematic Review and Meta-Analysis" Healthcare 10, no. 1: 132. https://doi.org/10.3390/healthcare10010132
APA StyleGravaldi, L. P., Bonetti, F., Lezzerini, S., & De Maio, F. (2022). Effectiveness of Physiotherapy in Patients with Ankylosing Spondylitis: A Systematic Review and Meta-Analysis. Healthcare, 10(1), 132. https://doi.org/10.3390/healthcare10010132






