A Mathematical Model of the Transition from Normal Hematopoiesis to the Chronic and Accelerated-Acute Stages in Myeloid Leukemia
Abstract
1. Introduction
Medical Background
2. The Mathematical Model
2.1. The Normal-Leukemic Dynamic System
- If , then the steady state is asymptotically stable, and the steady state is unstable.
- If , then the steady state is positive and asymptotically stable, and the steady states and are unstable.
- If , then the steady state is asymptotically stable, and the steady state is unstable.
2.2. The Mathematical–Biological Interpretation
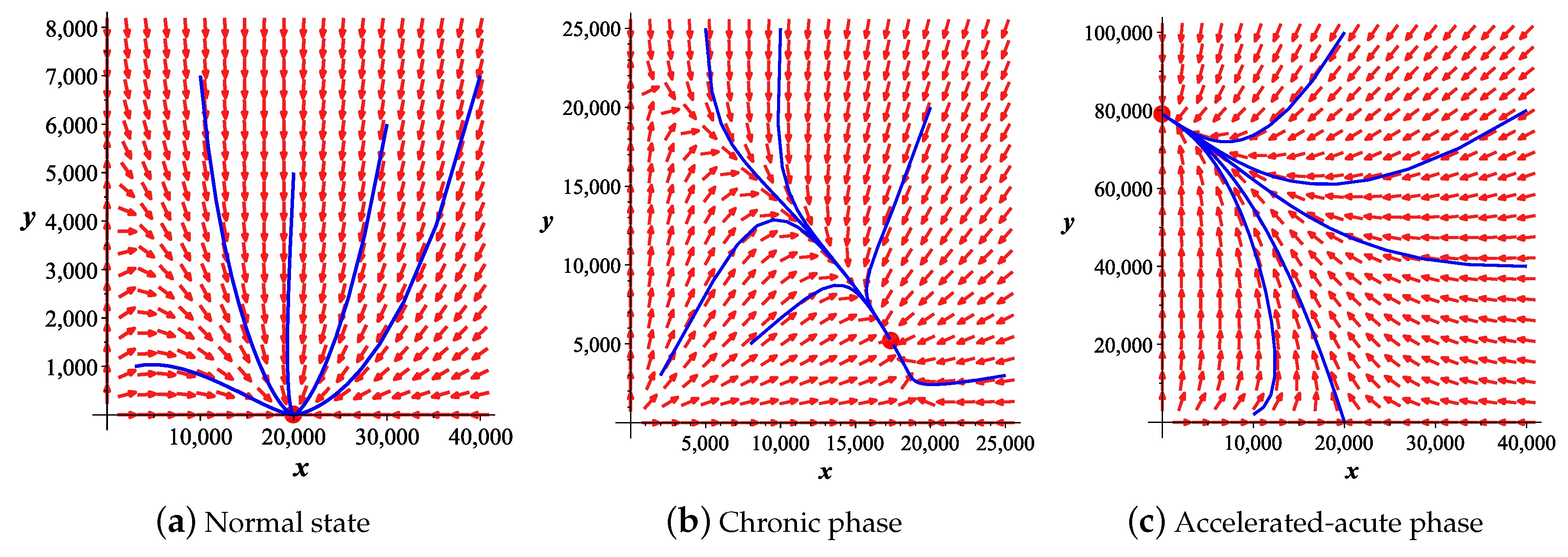
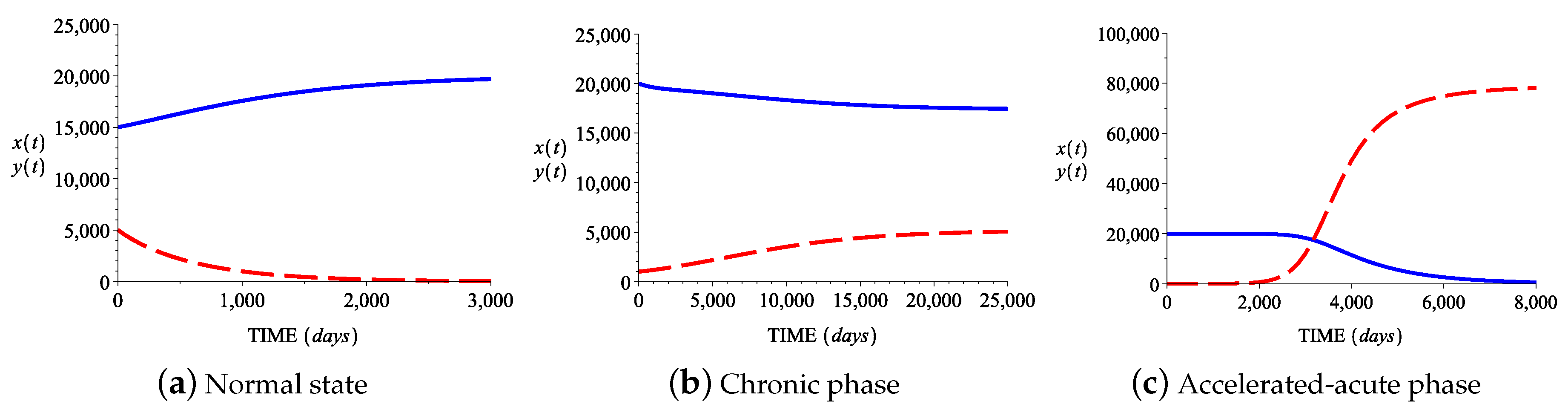
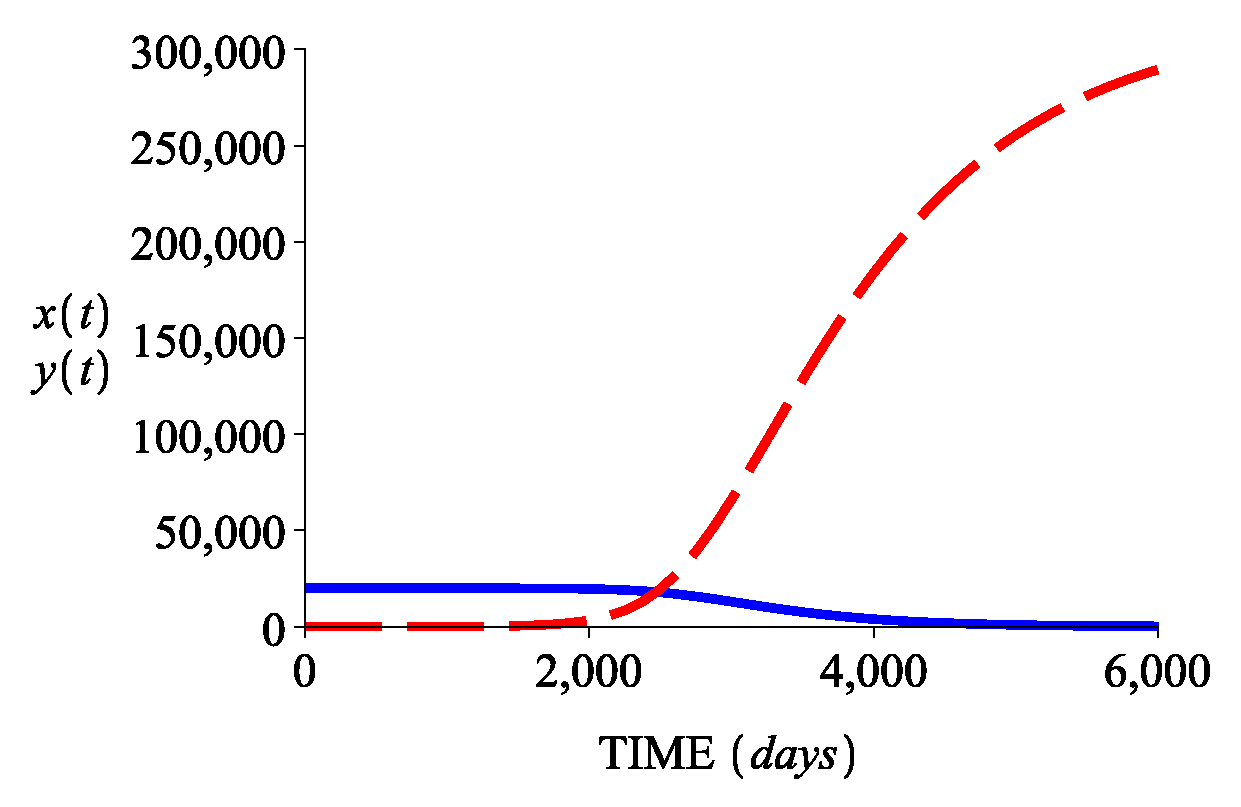
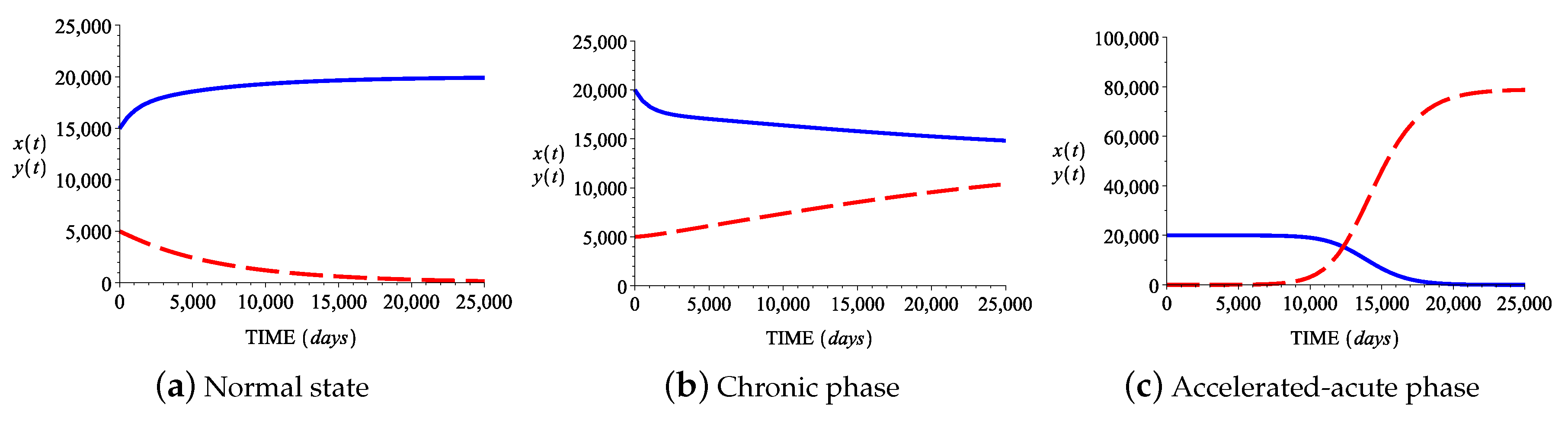
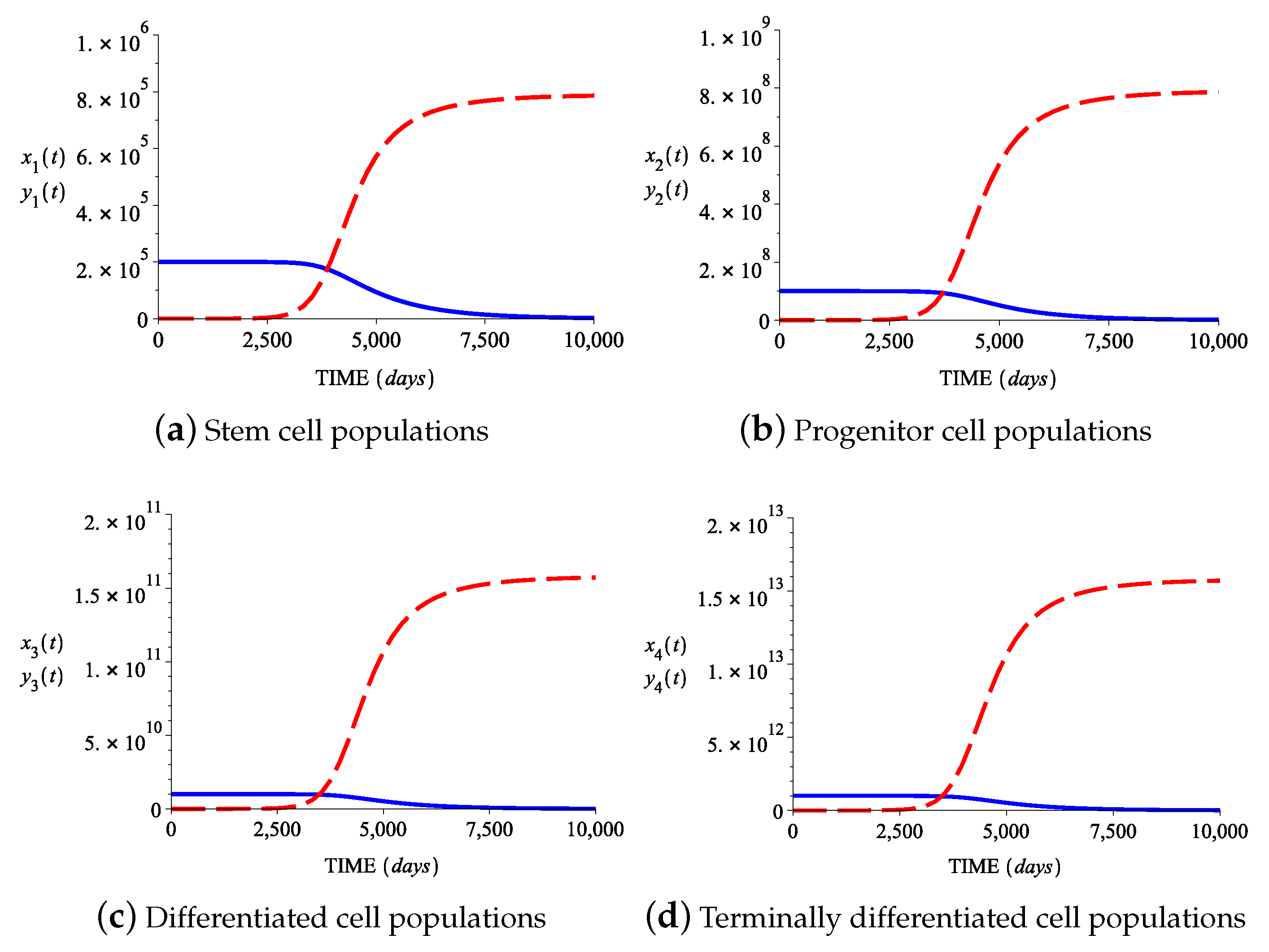
3. Numerical Simulation of the Model
3.1. Parameter Estimations
3.2. Numerical Simulations
4. The Model Extended to Terminally Differentiated Cells
5. Discussion and Conclusions
- The mHSCs proliferation rate is a predictive factor for the development of the accelerated-acute state: an increased rate of proliferation of these cells in comparison to normal stem cells determines the accelerated-acute phase to occur earlier;
- The death rate of leukemic stem cells is predictive for the global evolution of the disease, influencing the shifts between the different phases of the chronic myeloid leukemia.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rubinow, S.I.; Lebowitz, J.L. A mathematical model of neutrophil production and control in normal man. J. Math. Biol. 1975, 1, 187–225. [Google Scholar] [CrossRef]
- Rubinow, S.I.; Lebowitz, J.L. A mathematical model of the acute myeloblastic leukemic state in man. Biophys. J. 1976, 16, 897–910. [Google Scholar] [CrossRef]
- Mackey, M.C.; Glass, L. Oscillation and chaos in physiological control systems. Science 1977, 197, 287–289. [Google Scholar] [CrossRef]
- Mackey, M.C. Unified hypothesis of the origin of aplastic anemia and periodic hematopoiesis. Blood 1978, 51, 941–956. [Google Scholar] [CrossRef]
- Djulbegovic, B.; Svetina, S. Mathematical model of acute myeloblastic leukaemia: An investigation of the relevant kinetic parameters. Cell Prolif. 1985, 18, 307–319. [Google Scholar] [CrossRef]
- Fokas, A.S.; Keller, J.B.; Clarkson, B.D. Mathematical model of granulocytopoiesis and chronic myelogenous leukemia. Cancer Res. 1991, 51, 2084–2091. [Google Scholar]
- Neiman, B. A Mathematical Model of Chronic Myelogenous Leukemia; Oxford University: Oxford, UK, 2000; Available online: https://core.ac.uk/download/files/69/96488.pdf (accessed on 10 May 2019).
- Andersen, L.K.; Mackey, M.C. Resonance in periodic chemotherapy: A case study of acute myelogenous leukemia. J. Theor. Biol. 2001, 209, 113–130. [Google Scholar] [CrossRef]
- Colijn, C.; Mackey, M.C. A mathematical model of hematopoiesis-I. Periodic chronic myelogenous leukemia. J. Theor. Biol. 2005, 237, 117–132. [Google Scholar] [CrossRef]
- Adimy, M.; Crauste, F.; Ruan, S. A mathematical study of the hematopoiesis process with applications to chronic myelogenous leikemia. SIAM J. Appl. Math. 2005, 65, 1328–1352. [Google Scholar] [CrossRef]
- Dingli, D.; Michor, F. Successful therapy must eradicate cancer stem cells. Stem Cells 2006, 24, 2603–2610. [Google Scholar] [CrossRef]
- Kim, P.S.; Lee, P.P.; Levy, D. Dynamics and potential impact of the immune response to chronic myelogenous leukemia. PLoS Comput. Biol. 2008, 4, e1000095. [Google Scholar] [CrossRef] [PubMed]
- Cucuianu, A.; Precup, R. A hypothetical-mathematical model of acute myeloid leukemia pathogenesis. Comput. Math. Methods Med. 2010, 11, 49–65. [Google Scholar] [CrossRef]
- Doumic-Jauffret, M.; Kim, P.S.; Perthame, B. Stability analysis of a simplified yet complete model for chronic myelogenous leukemia. Bull. Math. Biol. 2010, 72, 1732–1759. [Google Scholar] [CrossRef] [PubMed]
- Komarova, N.L. Mathematical modeling of cyclic treatments of chronic myeloid leukemia. Math. Biosci. Eng. 2011, 8, 289–306. [Google Scholar]
- Stiehl, T.; Marciniak-Czochra, A. Mathematical modelling of leukemogenesis and cancer stem cell dynamics. Math. Mod. Nat. Phenom. 2012, 7, 166–202. [Google Scholar] [CrossRef]
- MacLean, A.L.; Lo Celso, C.; Stumpf, M.P.H. Population dynamics of normal and leukaemia stem cells in the haematopoietic stem cell niche show distinct regimes where leukaemia will be controlled. J. R. Soc. Interfaces 2013, 10, 20120968. [Google Scholar] [CrossRef]
- MacLean, A.L.; Filippi, S.; Stumpf, M.P.H. The ecology in the hematopoietic stem cell niche determines the clinical outcome in chronic myeloid leukemia. Proc. Natl. Acad. Sci. USA 2014, 111, 3882–3888. [Google Scholar] [CrossRef]
- Radulescu, I.R.; Candea, D.; Halanay, A. A study on stability and medical implications for a complex delay model for CML with cell competition and treatment. J. Theor. Biol. 2014, 363, 30–40. [Google Scholar] [CrossRef]
- Bianca, C.; Pennisi, M.; Motta, S.; Ragusa, M.A. Immune system network and cancer vaccine. AIP Conf. Proc. 2011, 1389, 945–948. [Google Scholar]
- Bianca, C.; Pappalardo, F.; Pennisi, M.; Ragusa, M.A. Persistence analysis in a Kolmogorov-type model for cancer-immune system competition. AIP Conf. Proc. 2013, 1558, 1797–1800. [Google Scholar]
- Ragusa, M.A.; Russo, G. ODEs approaches in modeling fibrosis: Comment on “Towards a unified approach in the modeling of fibrosis: A review with research perspectives” by Martine Ben Amar and Carlo Bianca. Phys. Life Rev. 2016, 17, 112–113. [Google Scholar] [CrossRef] [PubMed]
- Vincent, P.C.; Rutzen-Loesevitz, L.; Tibken, B.; Heinze, B.; Hofer, E.P.; Fliedner, T.M. Relapse in chronic myeloid leukemia after bone marrow transplantation: Biomathematical modeling as a new approach to understanding pathogenesis. Stem Cells 1999, 17, 9–17. [Google Scholar] [CrossRef] [PubMed]
- De Conde, R.; Kim, P.S.; Levy, D.; Lee, P.P. Post-transplantation dynamics of the immune response to chronic myelogenous leukemia. J. Theor. Biol. 2005, 236, 39–59. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.S.; Lee, P.P.; Levy, D. Mini-Transplants for Chronic Myelogenous Leukemia: A Modeling Perspective, Biology and Control Theory: Current Challenges; Lecture Notes in Control and Information Sciences; Springer: Berlin/Heidelberg, Germany, 2007; Volume 357, pp. 3–20. [Google Scholar]
- Marciniak-Czochra, A.; Stiehl, T. Mathematical models of hematopoietic reconstitution after stem cell transplantation. In Model Based Parameter Estimation: Theory and Applications; Bock, H.G., Carraro, T., Jaeger, W., Koerkel, S., Rannacher, R., Schloeder, J.P., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 191–206. [Google Scholar]
- Precup, R.; Arghirescu, S.; Cucuianu, A.; Serban, M. Mathematical Modeling of cell dynamics after allogeneic bone marrow transplantation. Int. J. Biomath. 2012, 5, 1–18. [Google Scholar] [CrossRef]
- Precup, R.; Serban, M.A.; Trif, D. Asymptotic stability for a model of cellular dynamics after allogeneic bone marrow transplantation. Nonlinear Dyn. Syst. Theory 2013, 13, 79–92. [Google Scholar]
- Precup, R. Mathematical understanding of the autologous stem cell transplantation. Ann. Tiberiu Popoviciu Semin. Funct. Equ. Approx. Convexity 2012, 10, 155–167. [Google Scholar]
- Stiehl, T.; Ho, A.; Marciniak-Czochra, A. The impact of CD34+ cell dose on engraftment after SCTs: Personalized estimates based on mathematical modeling. Bone Marrow Transpl. 2014, 49, 30–37. [Google Scholar] [CrossRef]
- Afenya, E. Mathematical models for cancer and their relevant insights. In Handbook of Cancer Models with Applications; Tan, W.Y., Hanin, L., Eds.; World Scientific: Hackensack, NJ, USA, 2008. [Google Scholar]
- Michor, F. Mathematical models of cancer stem cells. J. Clin. Oncol. 2008, 26, 2854–2861. [Google Scholar] [CrossRef][Green Version]
- Foley, C.; Mackey, M.C. Dynamic hematological disease: A review. J. Math. Biol. 2009, 58, 285–322. [Google Scholar] [CrossRef]
- Clapp, G.; Levy, D. A review of mathematical models for leukemia and lymphoma. Drug Discov. Today Dis. Models 2015, 16, 1–6. [Google Scholar] [CrossRef]
- Jilkine, A.; Gutenkunst, R. Effect of dedifferentiation on time to mutation acquisition in stem cell-driven cancers. PLoS Comput. Biol. 2014, 10, e1003481. [Google Scholar] [CrossRef] [PubMed]
- Driessens, G.; Beck, B.; Caauwe, A.; Simons, B.; Blanpain, C. Defining the mode of tumour growth by clonal analysis. Nature 2012, 488, 527–530. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.; Simons, B. Universal patterns of stem cell fate in cycling adult tissues. Development 2011, 138, 3103–3111. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Garcia, C.; Klein, A.; Simons, B.; Winton, D. Intestinal stem cell replacement follows a pattern of neutral drift. Science 2010, 330, 822–825. [Google Scholar] [CrossRef] [PubMed]
- Snippert, H.; van der Flier, L.; Sato, T.; van Es, J.; van den Born, M.; Kroon-Veenboer, C.; Barker, N.; Klein, A.; van Rheenen, J.; Simons, B.; et al. Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell 2010, 143, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Gou, X.C.; Kong, D.; Tang, X. Contradictory relationships between cancer and normal cells and implications for anti-cancer therapy. Asian Pac. J. Cancer Prev. 2015, 16, 5143–5147. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Precup, R.; Serban, M.A.; Trif, D.; Cucuianu, A. A planning algorithm for correction therapies after allogeneic stem cell transplantation. J. Math. Model. Algorithm 2012, 11, 309–323. [Google Scholar] [CrossRef]
- Michor, F.; Hughes, T.P.; Iwasa, Y.; Branford, S.; Shah, N.P.; Sawyers, C.L.; Nowak, M.A. Dynamics of chronic myeloid leukaemia. Nature 2005, 435, 1267–1270. [Google Scholar] [CrossRef]
- Howard, M.; Hamilton, P.; Britton, R. Haematology; Churchill Livingstone: London, UK, 2013; pp. 10–120. [Google Scholar]
- Young, N. Clinical Hematology; Mosby Elsevier: Philadelphia, PA, USA, 2006; pp. 1035–1456. [Google Scholar]
- Kaushansky, K.; Lichtman, M.A.; Beutler, E.; Kipps, T.J.; Seligsohn, U.; Prchal, J.T. Williams Hematology; McGraw-Hill Medical: New York, NY, USA, 2010; pp. 1150–2439. [Google Scholar]
- Kawamoto, H.; Wada, H.; Katsura, Y. A revised scheme for developmental pathways of hematopoietic cells: The myeloid-based model. Int. Immunol. 2010, 22, 65–70. [Google Scholar] [CrossRef][Green Version]
- Abkowitz, J.L. Evidence that the number of hematopoietic stem cells per animal is conserved in mammals. Blood 2002, 100, 2665–2667. [Google Scholar] [CrossRef]
- Ramalingam, P.; Poulos, M.; Butler, J. Regulation of the hematopoietic stem cell lifecycle by the endothelial niche. Comput. Curr. Opin. Hematol. 2017, 24, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Zon, L. Intrinsic and extrinsic control of haematopoietic stem-cell self-renewal. Nature 2008, 453, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Arber, D.; Orazi, A.; Hasserjian, R.; Thiele, J.; Borowitz, M.; Le Beau, M.; Bloomfield, C.; Cazzola, M.; Vardiman, J. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 2016, 127, 2391–2405. [Google Scholar] [CrossRef] [PubMed]
- Roeder, I.; d’Inverno, M. New experimental and theoretical investigations of hematopoietic stem cells and chronic myeloid leukemia. Blood Cells Mol. Dis. 2009, 43, 88–97. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hemminki, K.; Jiang, Y. Familial myeloid leukemias from the Swedish Family-Cancer Database. Leuk. Res. 2002, 26, 611–613. [Google Scholar] [CrossRef]
- Kantarjian, H.; O’Brien, S.; Jabbour, E.; Garcia-Manero, G.; Quintas-Cardama, A.; Shan, J.; Rios, M.; Ravandi, F.; Faderl, S.; Kadia, T.; et al. Improved survival in chronic myeloid leukemia since the introduction of imatinib therapy: A single-institution historical experience. Blood 2012, 119, 1981–1987. [Google Scholar] [CrossRef]
- Thomas, E. Marrow transplantation for the treatment of chronic myelogenous leukemia. Ann. Intern. Med. 1986, 104, 155–163. [Google Scholar] [CrossRef]
- Alenzi, F.Q.; Alenazi, B.Q.; Ahmad, S.Y.; Salem, M.L.; Al-Jabri, A.A.; Wyse, R.K.H. The haemopoietic stem cell: Between apoptosis and self renewal. Yale J. Biol. Med. 2009, 82, 7–18. [Google Scholar]
- Cisneros, T.; Dillard, D.; Castro, M.; Arredondo-Guerrero, J.; Krams, S.; Esquivel, C.; Martinez, O. The role of natural killer cells in recognition and killing of stem cells and stem cell-derived hepatoblasts. Am. J. Transplant 2017, 17 (Suppl. 3), 115. [Google Scholar]
- Domen, J. The role of apoptosis in regulating hematopoietic stem cell numbers. Apoptosis 2001, 6, 239–252. [Google Scholar] [CrossRef]
- Riether, C.; Schürch, C.M.; Ochsenbein, A.F. Regulation of hematopoietic and leukemic stem cells by the immune system. Cell Death Differ. 2015, 22, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Vivier, E.; Tomasello, E.; Baratin, M.; Walzer, T.; Ugolini, S. Functions of natural killer cells. Nat. Immunol. 2008, 9, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, D.; Glass, L. Understanding Nonlinear Dynamics; Springer: New York, NY, USA, 1995. [Google Scholar]
- Coddington, E.A.; Levinson, N. Theory of Ordinary Differential Equations; Tata McGraw-Hill: New Delhi, India, 1972. [Google Scholar]
- Jones, D.S.; Plank, M.J.; Sleeman, B.D. Differential Equations and Mathematical Biology; CRC Press: London, UK, 2010. [Google Scholar]
- Stine, R.; Matunis, E. Stem cell competition: Finding balance in the niche. Trends Cell Biol. 2013, 23, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Catlin, S.; Busque, L.; Gale, R.; Guttorp, P.; Abkowitz, J. The replication rate of human hematopoietic stem cells in vivo. Blood 2011, 117, 4460–4466. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.; Laurenti, E.; Oser, G.; van der Wath, R.; Blanco-Bose, W.; Jaworski, M.; Offner, S.; Dunant, C.; Eshkind, L.; Bockamp, E.; et al. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell 2008, 135, 1118–1129. [Google Scholar] [CrossRef] [PubMed]
- Sieburg, H.; Rezner, B.; Muller-Sieburg, C. Predicting clonal self-renewal and extinction of hematopoietic stem cells. Proc. Natl. Acad. Sci. USA 2011, 108, 4370–4375. [Google Scholar] [CrossRef] [PubMed]
- Sieburg, H.; Cattarossi, G.; Muller-Sieburg, C. Lifespan differences in hematopoietic stem cells are due to imperfect repair and unstable mean-reversion. PLoS Comput. Biol. 2013, 9, e1003006. [Google Scholar] [CrossRef]
- Enriquez-Navas, P.; Kam, Y.; Das, T.; Hassan, S.; Silva, A.; Foroutan, P.; Ruiz, E.; Martinez, G.; Minton, S.; Gillies, R.; et al. Exploiting evolutionary principles to prolong tumor control in preclinical models of breast cancer. Sci. Transl. Med. 2016, 8, 327ra24. [Google Scholar] [CrossRef]
- Gerlinger, M.; Rowan, A.; Horswell, S.; Larkin, J.; Endesfelder, D.; Gronroos, E.; Martinez, P.; Matthews, N.; Stewart, A.; Tarpey, P.; et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 2012, 366, 883–892. [Google Scholar] [CrossRef]


| Case I | Case II | Case III | Case IV |
|---|---|---|---|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parajdi, L.G.; Precup, R.; Bonci, E.A.; Tomuleasa, C. A Mathematical Model of the Transition from Normal Hematopoiesis to the Chronic and Accelerated-Acute Stages in Myeloid Leukemia. Mathematics 2020, 8, 376. https://doi.org/10.3390/math8030376
Parajdi LG, Precup R, Bonci EA, Tomuleasa C. A Mathematical Model of the Transition from Normal Hematopoiesis to the Chronic and Accelerated-Acute Stages in Myeloid Leukemia. Mathematics. 2020; 8(3):376. https://doi.org/10.3390/math8030376
Chicago/Turabian StyleParajdi, Lorand Gabriel, Radu Precup, Eduard Alexandru Bonci, and Ciprian Tomuleasa. 2020. "A Mathematical Model of the Transition from Normal Hematopoiesis to the Chronic and Accelerated-Acute Stages in Myeloid Leukemia" Mathematics 8, no. 3: 376. https://doi.org/10.3390/math8030376
APA StyleParajdi, L. G., Precup, R., Bonci, E. A., & Tomuleasa, C. (2020). A Mathematical Model of the Transition from Normal Hematopoiesis to the Chronic and Accelerated-Acute Stages in Myeloid Leukemia. Mathematics, 8(3), 376. https://doi.org/10.3390/math8030376





