High-Dimensional Modeling of Huanglongbing Dynamics with Time-Varying Impulsive Control
Abstract
1. Introduction
2. Model Formulation
3. Basic Reproduction Number for a General Impulsive Epidemic System
- (i)
- if and only if ;
- (ii)
- if and only if ;
- (iii)
- if and only if .
4. Main Results for System (2)
4.1. Basic Reproduction Number
4.2. Global Asymptotic Stability of the Disease-Free Periodic Solution
4.3. Uniform Persistence
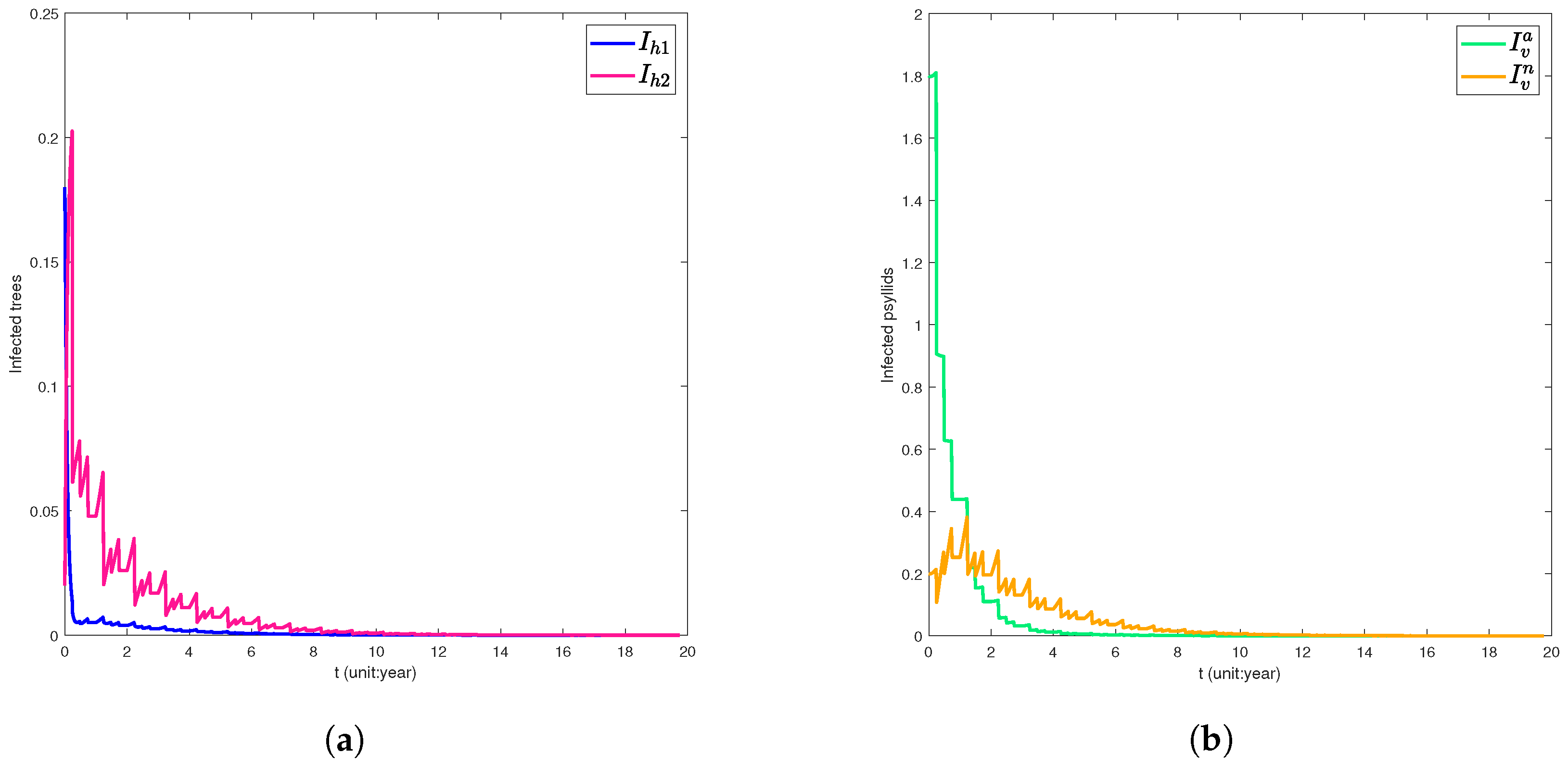
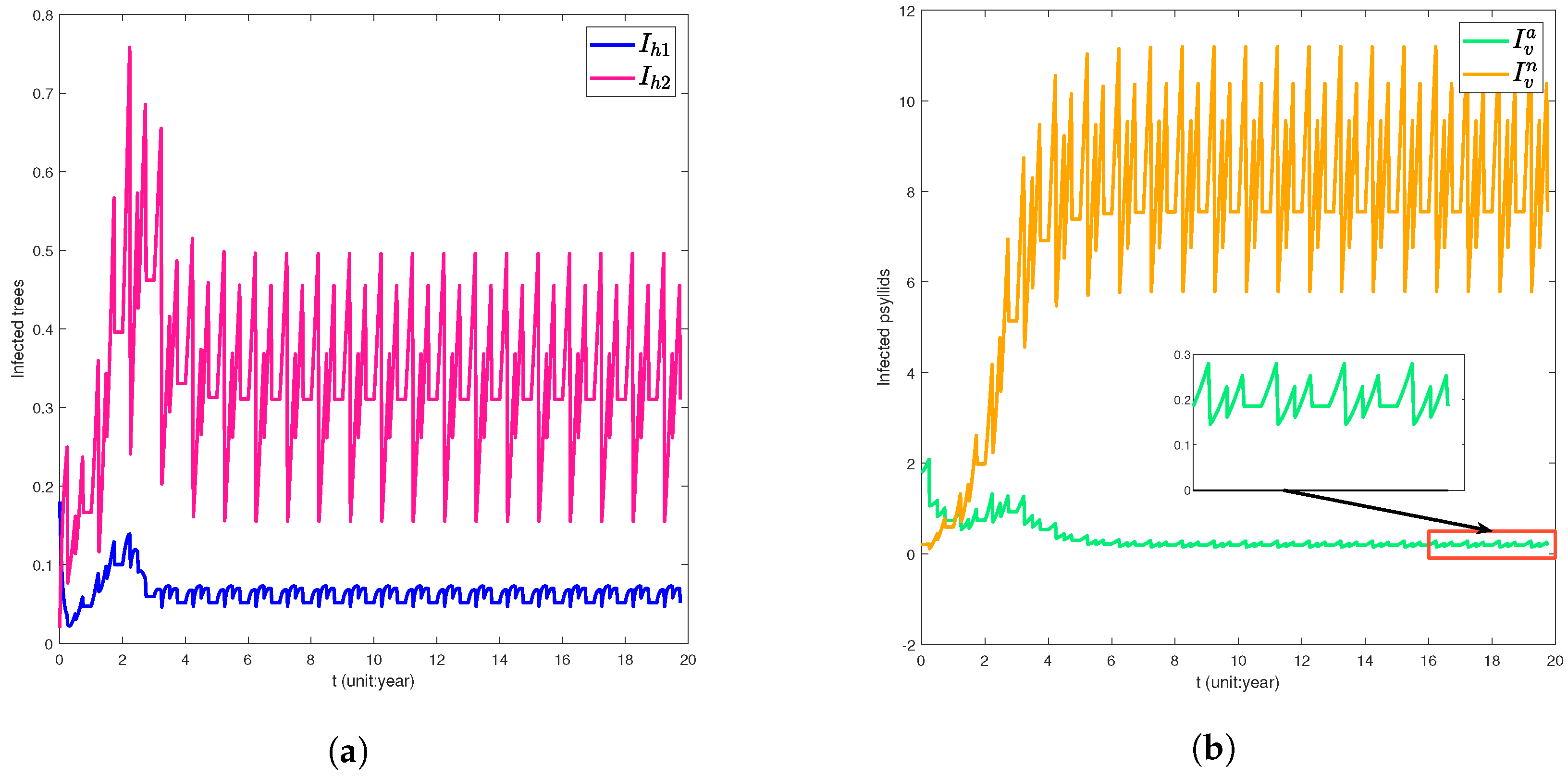
5. Numerical Simulation
6. Conclusions and Discussion
- (i)
- Multi-pronged psyllid population control
- Chemical control: rational use of high-efficiency, low-toxicity pesticides to target adult psyllids, with careful attention to resistance management.
- Cultural management: practices such as pruning water sprouts and synchronizing flush cycles can reduce psyllid oviposition sites and disrupt their reproductive cycle.
- Biological and physical control: combining yellow sticky traps (exploiting adult phototaxis) with natural enemies (e.g., Tamarixia radiata) for eco-friendly pest suppression.
- (ii)
- Application of botanical repellents
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bové, J.M. Huanglongbing: A destructive newly-emerging, century-old disease of citrus. J. Plant Pathol. 2006, 88, 7–37. [Google Scholar]
- Gao, S.; Yu, D.; Meng, X.; Zhang, F. Global dynamics of a stage-structured Huanglongbing model with time delay. Chaos Solitons Fractals 2018, 117, 60–67. [Google Scholar] [CrossRef]
- Jacobsen, K.; Stupiansky, J.; Pilyugin, S.S. Mathematical modeling of citrus groves infected by huanglongbing. Math. Biosci. Eng. 2013, 10, 705–728. [Google Scholar] [PubMed]
- Zhou, C. The status of citrus Huanglongbing in China. Trop. Plant Pathol. 2020, 45, 279–284. [Google Scholar] [CrossRef]
- Bové, J.M.; Rogers, M.E. Huanglongbing-control workshop: Summary. Acta. Hort 2015, 1065, 869–889. [Google Scholar] [CrossRef]
- Fan, G.; Liu, B.; Wu, R.; Li, T.; Cai, Z.; Ke, C. Three Decades of Research on Citrus Huanglongbing in China. Fujian J. Agric. Sci. 2009, 24, 183–190. (In Chinese) [Google Scholar]
- Halbert, S.E.; Manjunath, K.L. Asian citrus psyllids (Sternorrhyncha: Psyllidae) and greening disease of citrus: A literature review and assessment of risk in Florida. Fla. Entomol. 2004, 87, 330–353. [Google Scholar] [CrossRef]
- Gottwald, T.R. Current epidemiological understanding of citrus huanglongbing. Annu. Rev. Phytopathol. 2010, 48, 119–139. [Google Scholar] [CrossRef]
- Hung, T.; Hung, S.; Chen, C.; Hsu, M.; Su, H. Detection by PCR of Candidatus Liberibacter asiaticus, the bacterium causing citrus huanglongbing in vector psyllids: Application to the study of vector–pathogen relationships. Plant Pathol. 2010, 53, 96–102. [Google Scholar] [CrossRef]
- Liu, S.; Ye, J. Control Technologies for Citrus Huanglongbing (HLB) and Asian Citrus Psyllid (Diaphorina citri). Xiandai Hortic. 2016, 7, 130. (In Chinese) [Google Scholar]
- Huang, R.; Xie, L. Emerging Strategies for Huanglongbing (HLB) Control in Citrus. Chin. Hortic. Abstr. 2014, 9, 205–206. (In Chinese) [Google Scholar]
- Vilamiu, R.G.d.; Ternes, S.; Braga, G.A.; Laranjeira, F.F. A model for Huanglongbing spread between citrus plants including delay times and human intervention. AIP Conf. Proc. Am. Inst. Phys. 2012, 1479, 2315–2319. [Google Scholar]
- Zhang, F.; Qiu, Z.; Huang, A.; Zhao, X. Optimal control and cost-effectiveness analysis of a Huanglongbing model with comprehensive interventions. Appl. Math. Model. 2021, 90, 719–741. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, S.; Chen, D.; Liu, B. Modeling the transmission dynamics and optimal control strategy for Huanglongbing. Mathematics 2024, 12, 2648. [Google Scholar] [CrossRef]
- Wang, J.; Feng, F.; Guo, Z.; Lv, H.; Wang, J. Threshold Dynamics of a Vector-Borne Epidemic Model for Huanglongbing with Impulsive Control. Appl. Math. 2019, 10, 196–211. [Google Scholar] [CrossRef]
- Chiyaka, C.; Singer, B.H.; Halbert, S.E.; Morris, J.G., Jr.; van Bruggen, A.H. Modeling huanglongbing transmission within a citrus tree. Proc. Natl. Acad. Sci. USA 2012, 109, 12213–12218. [Google Scholar] [CrossRef]
- Vilamiu, R.G.d.; Ternes, S.; Laranjeira, F.F.; de C. Santos, T.T. Modelling the effect of an alternative host population on the spread of citrus Huanglongbing. AIP Conf. Proc. Am. Inst. Phys. 2013, 1558, 2504–2508. [Google Scholar]
- Wang, J.; Gao, S.; Luo, Y.; Xie, D. Threshold dynamics of a huanglongbing model with logistic growth in periodic environments. Abstr. Appl. Anal. 2014, 2014, 841367. [Google Scholar] [CrossRef]
- Li, D.; Wang, E. Research on monitoring and prevention and control techniques for citrus Huanglongbing (HLB) epidemic. J. Zhejiang Agric. Sci. 2012, 2012, 73–76. (In Chinese) [Google Scholar]
- Zhang, F.; Qiu, Z.; Zhong, B.; Feng, T.; Huang, A. Modeling Citrus Huanglongbing transmission within an orchard and its optimal control. Math. Biosci. Eng. 2020, 17, 2048–2069. [Google Scholar] [CrossRef]
- Inoue, H.; Ohnishi, J.; Ito, T.; Tomimura, K.; Miyata, S.; Iwanami, T.; Ashihara, W. Enhanced proliferation and efficient transmission of Candidatus Liberibacter asiaticus by adult Diaphorina citri after acquisition feeding in the nymphal stage. Ann. Appl. Biol. 2009, 155, 29–36. [Google Scholar] [CrossRef]
- Mingxue, D. Formming Process and Basis and Technological Points of the Theory Emphasis on Control Citrus Psylla for Integrated Control Huanglongbing. Chin. Agric. Sci. Bull. 2009, 25, 358–363. (In Chinese) [Google Scholar]
- Li, T.; Ke, C. Detection of the bearing rate of Liberobacter asiaticum, in citrus psylla and its host plant Murraya paniculata by nested PCR. Acta Phytotaxon. Sin. 2002, 29, 31–35. [Google Scholar]
- Chen, G.; Deng, M.; Tang, M.; Chen, T.; Tan, Y. Dynamic observation of overwintering adult citrus psyllid population dynamics during winter-spring seasons. South China Fruits 2010, 39, 36–38. (In Chinese) [Google Scholar]
- Deng, M.; Chen, G.; Tang, M.; Chen, T. Recent advances in citrus Huanglongbing research. South. Hortic. 2006, 17, 49–51. (In Chinese) [Google Scholar]
- Gao, S.; Tu, Y.; Wang, J. Basic reproductive number for a general hybrid epidemic model. Adv. Differ. Equ. 2018, 2018, 310. [Google Scholar] [CrossRef]
- Van den Driessche, P.; Watmough, J. Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 2002, 180, 29–48. [Google Scholar] [CrossRef]
- Yang, Y.; Xiao, Y. Threshold dynamics for compartmental epidemic models with impulses. Nonlinear Anal. Real World Appl. 2012, 13, 224–234. [Google Scholar] [CrossRef]
- Gao, S.; Teng, Z.; Xie, D. Analysis of a delayed SIR epidemic model with pulse vaccination. Chaos Solitons Fractals 2009, 40, 1004–1011. [Google Scholar] [CrossRef]

| Parameters | Description | Value | Unit | References |
|---|---|---|---|---|
| Carrying capacity of citrus trees | 2 | − | Assumed | |
| Carrying capacity of psyllids | 20 | − | Assumed | |
| Replanting rate of citrus trees | [20] | |||
| The transition rate from to | [16] | |||
| System period | 1 | year | [19] | |
| q | Number of pulses per system period | 4 | [19] | |
| b | Feeding rate of psyllids on citrus trees | 0.1–10 | Assumed | |
| Probability of disease transmission from infected psyllids to citrus trees | [17] | |||
| Probability of disease transmission from infected psyllids to citrus tree | 0.037–0.07 | [17] | ||
| The pathogen acquisition rate when feeding on infected trees | [21] | |||
| The proportionality coefficient of the pathogen acquisition rate of relative to | [17,21] | |||
| Natural mortality rate of citrus trees | 0.00275–0.004167 | [22] | ||
| Disease-induced mortality rate of citrus trees | 0.016667–0.027775 | [23] | ||
| r | Psyllid birth rate | 3.78327–33.526137 | [22,24] | |
| Natural mortality rate of psyllids | 0.1169825–0.95052 | [25] | ||
| The removal rates of infected plants at impulsive time | − | Assumed | ||
| The removal rates of infected plants at impulsive time | − | Assumed | ||
| The killing rates of psyllids at impulsive time | − | Assumed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, F.; Luo, Y.; Zhang, Y.; Gao, S. High-Dimensional Modeling of Huanglongbing Dynamics with Time-Varying Impulsive Control. Mathematics 2025, 13, 1546. https://doi.org/10.3390/math13101546
Xie F, Luo Y, Zhang Y, Gao S. High-Dimensional Modeling of Huanglongbing Dynamics with Time-Varying Impulsive Control. Mathematics. 2025; 13(10):1546. https://doi.org/10.3390/math13101546
Chicago/Turabian StyleXie, Feiping, Youquan Luo, Yan Zhang, and Shujing Gao. 2025. "High-Dimensional Modeling of Huanglongbing Dynamics with Time-Varying Impulsive Control" Mathematics 13, no. 10: 1546. https://doi.org/10.3390/math13101546
APA StyleXie, F., Luo, Y., Zhang, Y., & Gao, S. (2025). High-Dimensional Modeling of Huanglongbing Dynamics with Time-Varying Impulsive Control. Mathematics, 13(10), 1546. https://doi.org/10.3390/math13101546





