Testing Doses and Treatment Timelines of Anti-Angiogenic Drug Bevacizumab Numerically as a Single-Agent for the Treatment of Ovarian Cancer
Abstract
1. Introduction
2. Methodology
- Three different doses of Bevacizumab 5, 10, and 15 mg/kg are administered every Monday and Thursday, with the day count starting one day after the surgery. They are compared against the control (without anti-angiogenic intervention). These doses are also tested for the three day treatment option, i.e., Monday, Wednesday, and Friday. The administration rate is given aswhere D is the administered dose (for example, 15 mg/kg) and are the injection days (, , etc.).
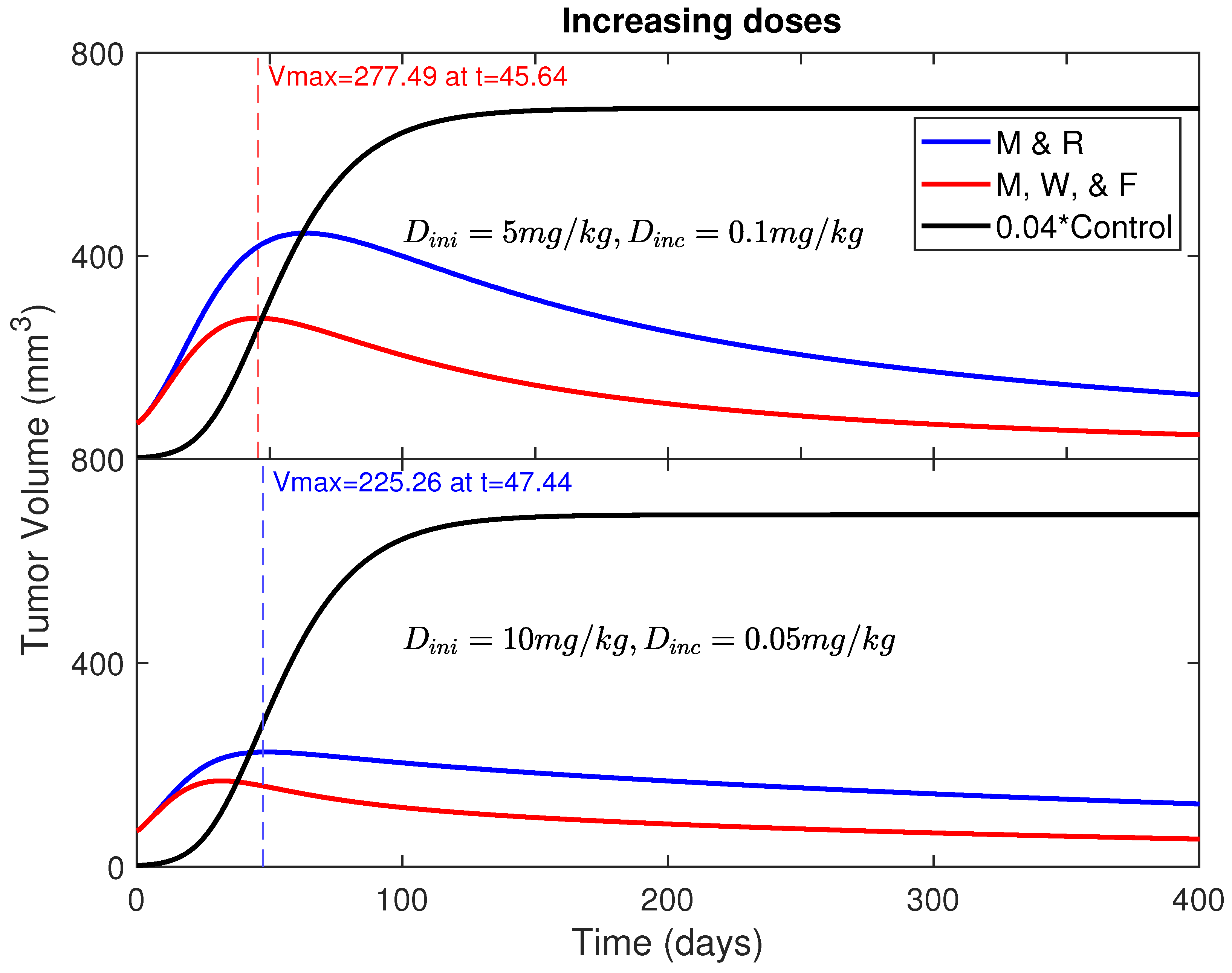
- Start with a moderate dose of 5 mg/kg or 10 mg/kg and increase the dose slightly by an equal amount after every treatment for the period of 400 days.where is the initial dose (for example, 5 mg/kg) given on day 1 () and (5.5 mg/kg for dose increment of 0.5 mg/kg) is the second dose given on Day 4 (), and so on.
3. Results
- Initial dose mg/kg with an increment of mg/kg
- Initial dose mg/kg with an increment of mg/kg
4. Conclusions and Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mayo Clinic. Ovarian Cancer. Available online: https://www.mayoclinic.org/diseases-conditions/ovarian-cancer/symptoms-causes/syc-20375941 (accessed on 31 August 2021).
- American Cancer Society. Key Statistics for Ovarian Cancer. Available online: https://www.cancer.org/cancer/ovarian-cancer/about/key-statistics.html (accessed on 12 January 2022).
- American Cancer Society. Survival Rates for Ovarian Cancer. Available online: https://www.cancer.org/cancer/ovarian-cancer/detection-diagnosis-staging/survival-rates.html (accessed on 1 March 2022).
- Conteduca, V.; Koph, B.; Burgio, S.L.; Bianchi, E.; Amadori, D.; De Giorgi, U. The emerging role of anti-angiogenic therapy in ovarian cancer (review). Int. J. Oncol. 2014, 44, 1417–1424. [Google Scholar] [CrossRef]
- Bhugwandass, C.S.; Pijnenborg, J.M.A.; Pijlman, B.; Ezendam, N.P.M. Effect of Chemotherapy on Health-Related Quality of Life among Early-Stage Ovarian Cancer Survivors: A Study from the Population-Based Profiles Registry. Curr. Oncol. 2016, 23, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Boyd, L.R.; Muggia, F.M. Carboplatin/paclitaxel induction in ovarian cancer: The finer points. Oncology (Williston Park) 2018, 32, 418–424. [Google Scholar] [PubMed]
- Cancer Research UK. Drugs that Block Cancer Blood Vessel Growth (Anti-Angiogenics). Available online: https://www.cancerresearchuk.org/about-cancer/cancer-in-general/treatment/targeted-cancer-drugs/types/anti-angiogenics (accessed on 13 January 2021).
- Kazazi-Hyseni, F.; Beijnen, J.H.; Schellens, J.H.M. Bevacizumab. Oncologist 2010, 15, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Avastin@Bevacizumab. Ovarian Cancer: Avastin Dosing and Usage. Available online: https://www.avastin.com/hcp/ovar/dosing-usage.html (accessed on 13 October 2022).
- Hall, M.; Bertelli, G.; Li, L.; Green, C.; Chan, S.; Yeoh, C.C.; Hasan, J.; Jones, R.; Ograbek, A.; Perren, T.J. Role of front-line bevacizumab in advanced ovarian cancer: The OSCAR study. Int. J. Gynecol. Cancer 2020, 30, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Kurnit, K.C.; Fleming, G.F.; Lengyel, E. Updates and new options in advanced epithelial ovarian cancer treatment. Obstet. Gynecol. 2021, 137, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Berton, D.; Floquet, A.; Lescaut, W.; Baron, G.; Kaminsky, M.-C.; Toussaint, P.; Largillier, R.; Savoye, A.-M.; Alexandre, J.; Delbaldo, C.; et al. Real-World Experience of Bevacizumab as First-Line Treatment for Ovarian Cancer: The GINECO ENCOURAGE Cohort of 468 French Patients. Front. Pharmacol. 2021, 12, 2315. [Google Scholar] [CrossRef] [PubMed]
- Emile, G.; Chauvenet, L.; Tigaud, J.M.; Chidiac, J.; Pujade Lauraine, E.; Alexandre, J. A clinical experience of single agent bevacizumab in relapsing ovarian cancer. Gynecol. Oncol. 2013, 129, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Poleszczuk, J.; Hahnfeldt, P.; Enderling, H. Therapeutic Implications from Sensitivity Analysis of Tumor Angiogenesis Models. PLoS ONE 2015, 3, e0120007. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Chemotherapy Safety. Available online: https://www.cancer.org/treatment/treatments-and-side-effects/treatment-types/chemotherapy/chemotherapy-safety.htm (accessed on 22 November 2019).
- Hahnfeldt, P.; Panigrahy, D.; Folkman, J.; Hlatky, L. Tumor development under angiogenic signaling: A dynamical theory of tumor growth, treatment response, and postvascular dormancy. Cancer Res. 2015, 59, 4770–4775. [Google Scholar]
- Bonate, P.L. Pharmacokinetic-Pharmacodynamic Modelidg and Simulation; Springer: New York, NY, USA, 2011. [Google Scholar]

| Symbol | Description | Units | Control (No Angiogenic Drug) | Model Value |
|---|---|---|---|---|
| e | drug impact | day conc | 0.4755 | |
| clearance rate | day | 0.0799 | ||
| growth rate | day | 0.0741 | 0.0741 | |
| loss of vessels | day | 0.0021 | 0.0021 | |
| b | stimulation | day | 1.3383 | 1.3383 |
| d | inhibition | day | 0.002 | 0.002 |
| initial V | mm | 71.2553 | 71.2553 | |
| initial K | mm | 71.6675 | 71.6675 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khatiwada, D.R.; Wallace, M. Testing Doses and Treatment Timelines of Anti-Angiogenic Drug Bevacizumab Numerically as a Single-Agent for the Treatment of Ovarian Cancer. Mathematics 2023, 11, 358. https://doi.org/10.3390/math11020358
Khatiwada DR, Wallace M. Testing Doses and Treatment Timelines of Anti-Angiogenic Drug Bevacizumab Numerically as a Single-Agent for the Treatment of Ovarian Cancer. Mathematics. 2023; 11(2):358. https://doi.org/10.3390/math11020358
Chicago/Turabian StyleKhatiwada, Dharma Raj, and Miana Wallace. 2023. "Testing Doses and Treatment Timelines of Anti-Angiogenic Drug Bevacizumab Numerically as a Single-Agent for the Treatment of Ovarian Cancer" Mathematics 11, no. 2: 358. https://doi.org/10.3390/math11020358
APA StyleKhatiwada, D. R., & Wallace, M. (2023). Testing Doses and Treatment Timelines of Anti-Angiogenic Drug Bevacizumab Numerically as a Single-Agent for the Treatment of Ovarian Cancer. Mathematics, 11(2), 358. https://doi.org/10.3390/math11020358








