Modeling the Kinetics of the Singlet Oxygen Effect in Aqueous Solutions of Proteins Exposed to Thermal and Laser Radiation
Abstract
1. Introduction
2. Materials and Methods
2.1. Mathematical Algorithm
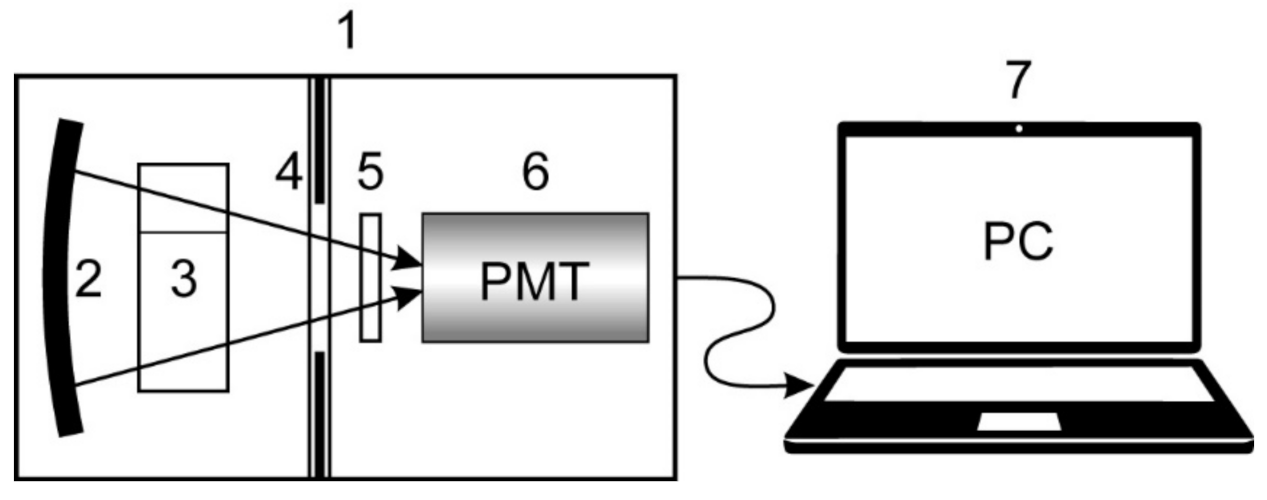
2.2. Physical Experiment Protocol
3. Results
3.1. ROS Reactions in Aqueous Protein Solutions
3.2. Kinetic Model
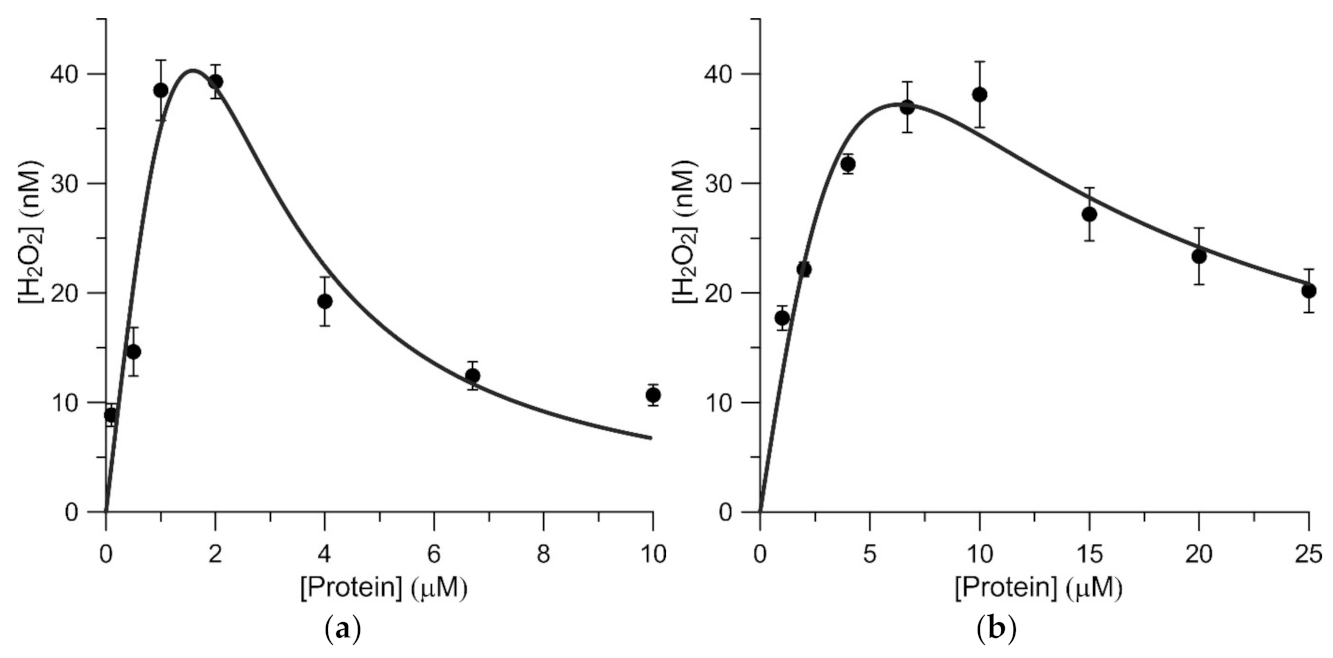
3.3. Stationary Dependences of the Hydrogen Peroxide Concentration on the Protein Concentration in Aqueous Solutions of BSA and BGG
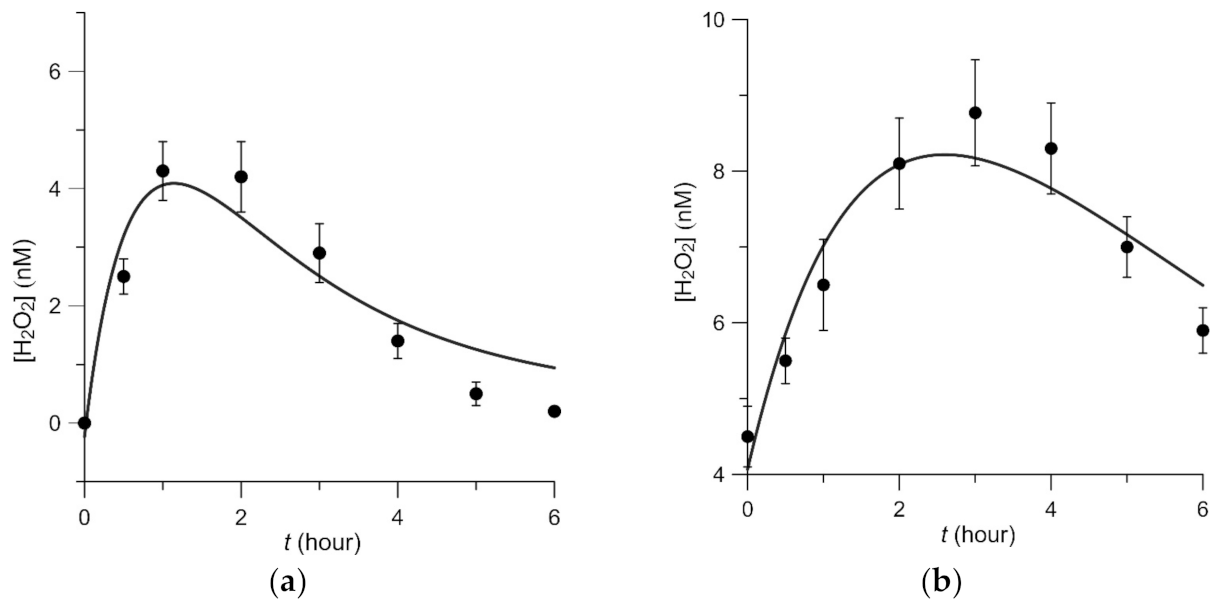
3.4. Dynamics of Hydrogen Peroxide Formation in Aqueous Solutions of BSA and BGG
3.5. Chemical Constants
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bitsouni, V.; Gialelis, N.; Stratis, I.G. Rigorous Analysis of the Quasi-Steady-State Assumption in Enzyme Kinetics. Mathematics 2022, 10, 1086. [Google Scholar] [CrossRef]
- Nikolov, S.G.; Wolkenhauer, O.; Nenov, M.; Vera, J. Detection and Analysis of Critical Dynamic Properties of Oligodendrocyte Differentiation. Mathematics 2022, 10, 2928. [Google Scholar] [CrossRef]
- Sukhinov, A.; Belova, Y.; Nikitina, A.; Sidoryakina, V. Sufficient Conditions for the Existence and Uniqueness of the Solution of the Dynamics of Biogeochemical Cycles in Coastal Systems Problem. Mathematics 2022, 10, 2092. [Google Scholar] [CrossRef]
- Snezhkina, A.V.; Kudryavtseva, A.V.; Kardymon, O.L.; Savvateeva, M.V.; Melnikova, N.V.; Krasnov, G.S.; Dmitriev, A.A. ROS generation and antioxidant defense systems in normal and malignant cells. Oxid. Med. Cell. Longev. 2019, 2019, 6175804. [Google Scholar] [CrossRef]
- Gapeyev, A.B.; Lukyanova, N.A.; Gudkov, S.V. Hydrogen peroxide induced by modulated electromagnetic radiation protects the cells from DNA damage. Cent. Eur. J. Biol. 2014, 9, 915–921. [Google Scholar] [CrossRef]
- Piskarev, I. The role of light emission of spark discharge in air in the generation of active species. High Energy Chem. 2019, 53, 92–93. [Google Scholar] [CrossRef]
- Filev, A.D.; Shmarina, G.V.; Ershova, E.S.; Veiko, N.N.; Martynov, A.V.; Borzikova, M.A.; Poletkina, A.A.; Dolgikh, O.A.; Veiko, V.P.; Bekker, A.A.; et al. Oxidized cell-free DNA role in the antioxidant defense mechanisms under stress. Oxid. Med. Cell. Longev. 2019, 2019, 1245749. [Google Scholar] [CrossRef]
- Bruskov, V.I.; Malakhova, L.V.; Masalimov, Z.K.; Chernikov, A.V. Heat-induced formation of reactive oxygen species and 8-oxoguanine, a biomarker of damage to DNA. Nucleic Acids Res. 2002, 30, 1354–1363. [Google Scholar] [CrossRef]
- Gudkov, S.V.; Bruskov, V.I.; Astashev, M.E.; Chernikov, A.V.; Yaguzhinsky, L.S.; Zakharov, S.D. Oxygen-dependent auto-oscillations of water luminescence triggered by the 1264 nm radiation. J. Phys. Chem. B 2011, 115, 7693–7698. [Google Scholar] [CrossRef]
- Zakharov, S.D.; Ivanov, A.V. Light-oxygen effect in cells and its potential applications in tumour therapy. Quantum Electron. 1999, 29, 1031. [Google Scholar] [CrossRef]
- Zakharov, S.D.; Ivanov, A.V.; Wolf, E.; Danilov, V.P.; Murina, T.M.; Nguen, K.; Novikov, E.; Panasenko, N.; Perov, S.; Skopinov, S. Structural rearrangements in the aqueous phase of cell suspensions and protein solutions induced by a light-oxygen effect. Quantum Electron. 2003, 33, 149. [Google Scholar] [CrossRef]
- Chin, K.K.; Trevithick-Sutton, C.C.; McCallum, J.; Jockusch, S.; Turro, N.J.; Scaiano, J.; Foote, C.S.; Garcia-Garibay, M.A. Quantitative determination of singlet oxygen generated by excited state aromatic amino acids, proteins, and immunoglobulins. J. Am. Chem. Soc. 2008, 130, 6912–6913. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Zhang, H. Targeting oxidative stress in disease: Promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021, 20, 689–709. [Google Scholar] [CrossRef] [PubMed]
- Pisoschi, A.M.; Pop, A.; Iordache, F.; Stanca, L.; Predoi, G.; Serban, A.I. Oxidative stress mitigation by antioxidants-an overview on their chemistry and influences on health status. Eur. J. Med. Chem. 2021, 209, 112891. [Google Scholar] [CrossRef] [PubMed]
- Brillo, V.; Chieregato, L.; Leanza, L.; Muccioli, S.; Costa, R. Mitochondrial dynamics, ROS, and cell signaling: A blended overview. Life 2021, 11, 332. [Google Scholar] [CrossRef]
- Ochoa, C.D.; Wu, R.F.; Terada, L.S. ROS signaling and ER stress in cardiovascular disease. Mol. Aspects Med. 2018, 63, 18–29. [Google Scholar] [CrossRef]
- Sharapov, M.; Novoselov, V. Catalytic and signaling role of peroxiredoxins in carcinogenesis. Biochem. 2019, 84, 79–100. [Google Scholar] [CrossRef]
- Nakamura, H.; Takada, K. Reactive oxygen species in cancer: Current findings and future directions. Cancer Sci. 2021, 112, 3945. [Google Scholar] [CrossRef]
- Vlasova, I.I. Peroxidase activity of human hemoproteins: Keeping the fire under control. Molecules 2018, 23, 2561. [Google Scholar] [CrossRef]
- Borisova-Mubarakshina, M.M.; Vetoshkina, D.V.; Ivanov, B.N. Antioxidant and signaling functions of the plastoquinone pool in higher plants. Physiol. Plant. 2019, 166, 181–198. [Google Scholar] [CrossRef]
- Smirnova, O.A.; Bartosch, B.; Zakirova, N.F.; Kochetkov, S.N.; Ivanov, A.V. Polyamine metabolism and oxidative protein folding in the ER as ROS-producing systems neglected in virology. Int. J. Mol. Sci. 2018, 19, 1219. [Google Scholar] [CrossRef] [PubMed]
- Chernov, A.; Reshetnikov, D.; Yu, A.K.; Manokhin, A.; Gudkov, S. Influence of wideband visible light with an padding red component on the functional state of mice embryos and embryonic stem cells. J. Photochem. Photobiol. 2018, 188, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Amaroli, A.; Pasquale, C.; Zekiy, A.; Utyuzh, A.; Benedicenti, S.; Signore, A.; Ravera, S. Photobiomodulation and oxidative stress: 980 nm diode laser light regulates mitochondrial activity and reactive oxygen species production. Oxid. Med. Cell. Longev. 2021, 2021, 6626286. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, V.; Karp, O.; Bruskov, V.; Andreev, S.; Bunkin, N.; Gudkov, S. Formation of long-lived reactive products in blood serum under heat treatment and low-intensity laser irradiation, their role in hydrogen peroxide generation and DNA damage. Indian J. Biochem. Biophys. 2019, 56, 214–223. [Google Scholar] [CrossRef]
- Levenberg, K. A method for the solution of certain non-linear problems in least squares. Q. Appl. Math. 1944, 2, 164–168. [Google Scholar] [CrossRef]
- Marquardt, D.W. An algorithm for least-squares estimation of nonlinear parameters. SIAM J. Appl. Math. 1963, 11, 431–441. [Google Scholar] [CrossRef]
- Nocedal, J.; Wright, S. Numerical Optimization; Springer Science and Business Media: New York, NY, USA, 2006. [Google Scholar]
- Hua, L.; Bo, L.; Tong, L.; Wang, M.; Fu, H.; Guo, R. Angular Acceleration Sensor Fault Diagnosis Based on LM-BP Neural Network. In Proceedings of the 37th Chinese Control Conference, Wuhan, China, 25–27 July 2018; pp. 6028–6032. [Google Scholar]
- Luo, G.; Zou, L.; Wang, Z.; Lv, C.; Ou, J.; Huang, Y. A novel kinematic parameters calibration method for industrial robot based on Levenberg-Marquardt and Differential Evolution hybrid algorithm. Robot. Comput. Integr. Manuf. 2021, 71, 102165. [Google Scholar] [CrossRef]
- Kumar, S.S.; Sowmya, R.; Shankar, B.M.; Lingaraj, N.; Sivakumar, S. Analysis of Connected Word Recognition systems using Levenberg Mar-quardt Algorithm for cockpit control in unmanned aircrafts. Mater. Today Proc. 2021, 37, 1813–1819. [Google Scholar] [CrossRef]
- Mahmoudabadi, Z.S.; Rashidi, A.; Yousefi, M. Synthesis of 2D-Porous MoS2 as a Nanocatalyst for Oxidative Desulfuriza-tion of Sour Gas Condensate: Process Parameters Optimization Based on the Levenberg–Marquardt Algorithm. J. Environ. Chem. Eng. 2021, 9, 105200. [Google Scholar] [CrossRef]
- Kudinov, I.A.; Pavlov, O.V.; Kholopov, I.S. Implementation of an algorithm for determining the spatial coordinates and the angular orientation of an object based on reference marks, using information from a single camera. Comput. Opt. 2015, 39, 413–419. [Google Scholar] [CrossRef]
- Fan, J. The modified Levenberg–Marquardt method for nonlinear equations with cubic convergence. Math. Comp. 2012, 81, 447–466. [Google Scholar] [CrossRef]
- Yang, X. A higher-order Levenberg–Marquardt method for nonlinear equations. Appl. Math. Comput. 2013, 219, 10682–10694. [Google Scholar] [CrossRef]
- Chen, L. A high-order modified Levenberg–Marquardt method for systems of nonlinear equations with fourth-order convergence. Appl. Math. Comput. 2016, 285, 79–93. [Google Scholar] [CrossRef]
- Derakhshandeh, S.Y.; Pourbagher, R.; Kargar, A. A novel fuzzy logic Levenberg-Marquardt method to solve the ill-conditioned power flow problem. Int. J. Electr. Power Energy Syst. 2018, 99, 299–308. [Google Scholar] [CrossRef]
- Shinkarenko, N.V.; Aleskovskii, V. Singlet oxygen: Methods of preparation and detection. Russ. Chem. Rev. 1981, 50, 220. [Google Scholar] [CrossRef]
- Davies, M.J. The oxidative environment and protein damage. Biochim. Biophys. Acta Proteins Proteom. 2005, 1703, 93–109. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J. Reactive species formed on proteins exposed to singlet oxygen. Photochem. Photobiol. Sci. 2004, 3, 17–25. [Google Scholar] [CrossRef]
- Hawkins, C.L.; Davies, M.J. Generation and propagation of radical reactions on proteins. Biochim. Biophys. Acta Bioenerg. 2001, 1504, 196–219. [Google Scholar] [CrossRef]
- Panchenkov, G.M.; Lebedev, V.P. Chemical Kinetics and Catalysis; Khimiya: Moscow, Russia, 1985. [Google Scholar]
- Takagi, J.; Ishigure, K. Thermal decomposition of hydrogen peroxide and its effect on reactor water monitoring of boiling water reactors. Nucl. Sci. Eng. 1985, 89, 177–186. [Google Scholar] [CrossRef]


| Protein | Exposure | a (L2·Micromole−2) | b | γ |
|---|---|---|---|---|
| BGG | Thermal (Figure 2a) | 5.17 | 23.33 | 2.45 |
| Laser (Figure 3a) | 36.45 | 315.14 | 2.23 | |
| BSA | Thermal (Figure 2b) | 3.32 | 76.52 | 1.81 |
| Laser (Figure 3b) | 69.84 | 125.78 | 1.43 |
| Protein | Exposure | (h−1) | (h−1) | (nmole·L−1) | (nmole·L−1·h−1) | |
|---|---|---|---|---|---|---|
| BGG | Thermal (Figure 4a) | 0.066 | 0.136 | 0.426 | 0.028 | 1.38·10−3 |
| Laser (Figure 5a) | 0.096 | 0.148 | 0.939 | 0.00023 | 4.86·10−4 | |
| BSA | Thermal (Figure 4b) | 0.029 | 0.163 | 0.354 | 0.033 | 1.34·10−3 |
| Laser (Figure 5b) | 0.044 | 0.295 | 0.396 | 0.00041 | 9.77·10−4 |
| Protein | Exposure | k2 | k3 | k4 | |
|---|---|---|---|---|---|
| BGG | Thermal | 4.6⋅10−6 | 4.4⋅105 | 5.9⋅106 | 2.7⋅10−5 |
| Laser | 1.07⋅10−6 | 2.6⋅106 | 9.8⋅106 | 8.5⋅10−5 | |
| BSA | Thermal | 6.8⋅10−7 | 0.87⋅105 | 5.6⋅105 | 1.3⋅10−5 |
| Laser | 1.1⋅10−7 | 2.8⋅105 | 1.9⋅106 | 3.5⋅10−6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shkirin, A.V.; Chirikov, S.N.; Suyazov, N.V.; Reut, V.E.; Grigorieva, D.V.; Gorudko, I.V.; Bruskov, V.I.; Gudkov, S.V. Modeling the Kinetics of the Singlet Oxygen Effect in Aqueous Solutions of Proteins Exposed to Thermal and Laser Radiation. Mathematics 2022, 10, 4295. https://doi.org/10.3390/math10224295
Shkirin AV, Chirikov SN, Suyazov NV, Reut VE, Grigorieva DV, Gorudko IV, Bruskov VI, Gudkov SV. Modeling the Kinetics of the Singlet Oxygen Effect in Aqueous Solutions of Proteins Exposed to Thermal and Laser Radiation. Mathematics. 2022; 10(22):4295. https://doi.org/10.3390/math10224295
Chicago/Turabian StyleShkirin, Alexey V., Sergey N. Chirikov, Nikolai V. Suyazov, Veronika E. Reut, Daria V. Grigorieva, Irina V. Gorudko, Vadim I. Bruskov, and Sergey V. Gudkov. 2022. "Modeling the Kinetics of the Singlet Oxygen Effect in Aqueous Solutions of Proteins Exposed to Thermal and Laser Radiation" Mathematics 10, no. 22: 4295. https://doi.org/10.3390/math10224295
APA StyleShkirin, A. V., Chirikov, S. N., Suyazov, N. V., Reut, V. E., Grigorieva, D. V., Gorudko, I. V., Bruskov, V. I., & Gudkov, S. V. (2022). Modeling the Kinetics of the Singlet Oxygen Effect in Aqueous Solutions of Proteins Exposed to Thermal and Laser Radiation. Mathematics, 10(22), 4295. https://doi.org/10.3390/math10224295







