Proteomics in the Study of Bacterial Keratitis
Abstract
:1. Introduction
2. Staphylococcus Aureus
3. Pseudomonas Aeruginosa
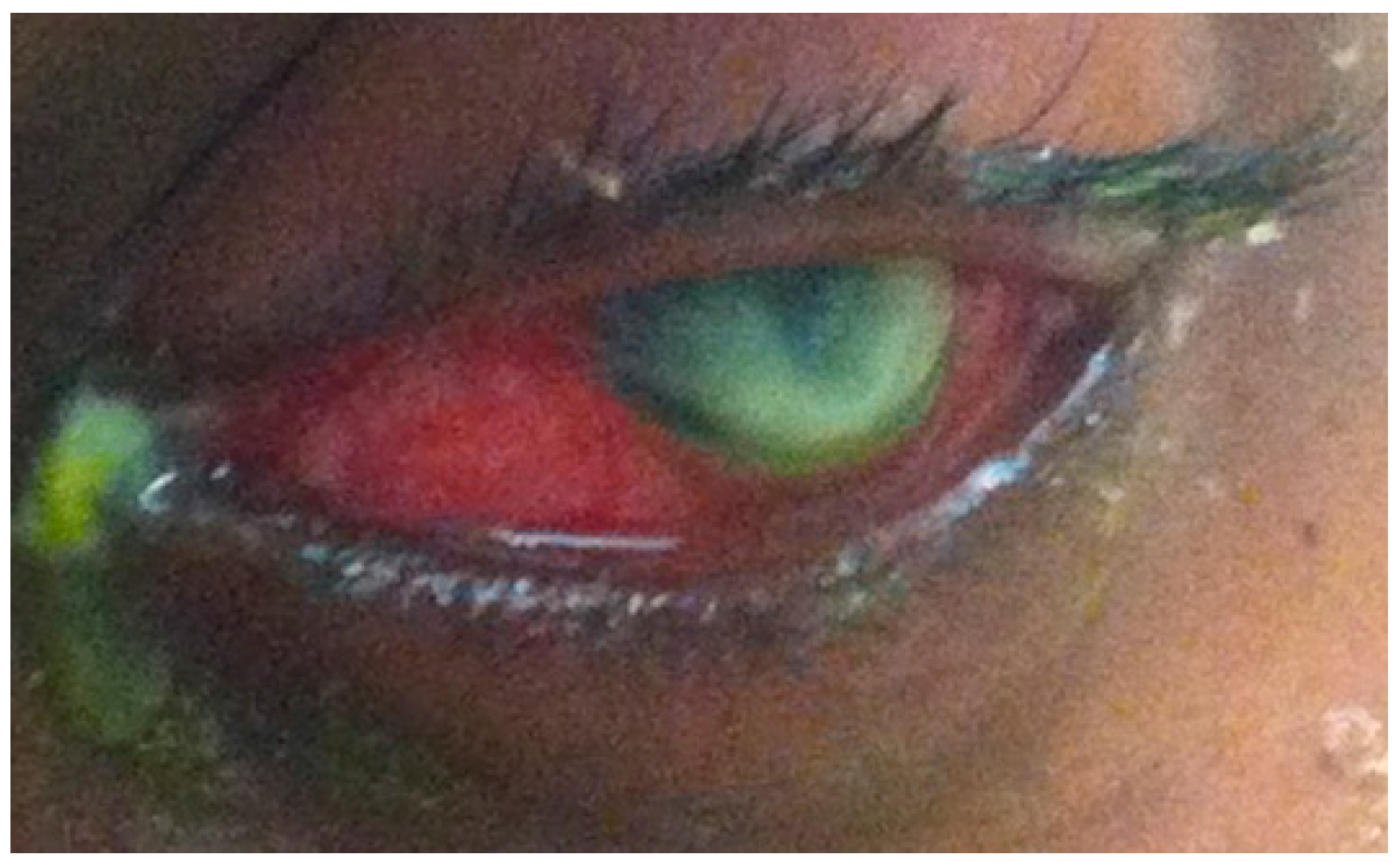
4. Streptococcus Pneumoniae
5. Serratia Species
6. Other Studies
6.1. Host Response Studies
6.2. Contact Lens Studies
7. Concluding Remarks and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Keay, L.; Edwards, K.; Naduvilath, T.; Taylor, H.R.; Snibson, G.R.; Forde, K.; Stapleton, F. Microbial keratitis predisposing factors and morbidity. Ophthalmology 2006, 113, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Al-Mujaini, A.; Al-Kharusi, N.; Thakral, A.; Wali, U.K. Bacterial keratitis: Perspective on epidemiology, clinico-pathogenesis, diagnosis and treatment. Sultan Qaboos Univ. Med. J. 2009, 9, 184–195. [Google Scholar] [PubMed]
- Parmar, P.; Salman, A.; Kalavathy, C.M.; Kaliamurthy, J.; Thomas, P.A.; Jesudasan, C.A. Microbial keratitis at extremes of age. Cornea 2006, 25, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Willcox, M.D. Pseudomonas aeruginosa infection and inflammation during contact lens wear: A review. Optom. Vis. Sci. 2007, 84, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Mah-Sadorra, J.H.; Najjar, D.M.; Rapuano, C.J.; Laibson, P.R.; Cohen, E.J. Serratia corneal ulcers: A retrospective clinical study. Cornea 2005, 24, 793–800. [Google Scholar] [CrossRef] [PubMed]
- American academy of ophthalmology cornea/external disease panel—preferred practice pattern guidelines, bacterial keratitis, Limited Revision. American Academy of Ophthalmology: San Francisco, CA, USA, 2011; Available online: http://one.aao.org/CE/PracticeGuidelines/PPP.aspx (accessed on 8 October 2015).
- Wilhelmus, K.R. Review of clinical experience with microbial keratitis associated with contact lenses. CLAO J. 1987, 13, 211–214. [Google Scholar] [PubMed]
- Jones, D.B. Pathogenesis of bacterial and fungal keratitis. Trans. Ophthalmol. Soc. U.K. 1978, 98, 367–371. [Google Scholar] [PubMed]
- Baker, N.R.; Minor, V.; Deal, C.; Shahrabadi, M.S.; Simpson, D.A.; Woods, D.E. Pseudomonas aeruginosa exoenzyme s is an adhesion. Infect. Immun. 1991, 59, 2859–2863. [Google Scholar] [PubMed]
- Reichert, R.; Stern, G. Quantitative adherence of bacteria to human corneal epithelial cells. Arch. Ophthalmol. 1984, 102, 1394–1395. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, F.; Bruttin, O.; Zografos, L.; Guex-Crosier, Y. Bacterial keratitis: A prospective clinical and microbiological study. Br. J. Ophthalmol. 2001, 85, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Otri, A.M.; Fares, U.; Al-Aqaba, M.A.; Miri, A.; Faraj, L.A.; Said, D.G.; Maharajan, S.; Dua, H.S. Profile of sight-threatening infectious keratitis: A prospective study. Acta Ophthalmol. 2012, 91, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Alexandrakis, G.; Alfonso, E.C.; Miller, D. Shifting trends in bacterial keratitis in south florida and emerging resistance to fluoroquinolones. Ophthalmology 2000, 107, 1497–1502. [Google Scholar] [CrossRef]
- Lichtinger, A.; Yeung, S.N.; Kim, P.; Amiran, M.D.; Iovieno, A.; Elbaz, U.; Ku, J.Y.; Wolff, R.; Rootman, D.S.; Slomovic, A.R. Shifting trends in bacterial keratitis in toronto: An 11-year review. Ophthalmology 2012, 119, 1785–1790. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, R.J.; Callegan, M.C.; Moreau, J.M.; Green, L.C.; Foster, T.J.; Hartford, O.M.; Engel, L.S.; Hill, J.M. Specific roles of α-toxin and β-toxin during Staphylococcus aureus corneal infection. Infect. Immun. 1997, 65, 1571–1578. [Google Scholar] [PubMed]
- Moreira, B.M.; Daum, R.S. Antimicrobial resistance in staphylococci. Pediatr. Clin. N. Am. 1995, 42, 619–648. [Google Scholar]
- Patti, J.M.; Allen, B.L.; McGavin, M.J.; Hook, M. Mscramm-mediated adherence of microorganisms to host tissues. Annu. Rev. Microbiol. 1994, 48, 585–617. [Google Scholar] [CrossRef] [PubMed]
- Jett, B.D.; Gilmore, M.S. Internalization of Staphylococcus aureus by human corneal epithelial cells: Role of bacterial fibronectin-binding protein and host cell factors. Infect. Immun. 2002, 70, 4697–4700. [Google Scholar] [CrossRef] [PubMed]
- Rhem, M.N.; Lech, E.M.; Patti, J.M.; McDevitt, D.; Hook, M.; Jones, D.B.; Wilhelmus, K.R. The collagen-binding adhesin is a virulence factor in Staphylococcus aureus keratitis. Infect. Immun. 2000, 68, 3776–3779. [Google Scholar] [CrossRef] [PubMed]
- Dajcs, J.J.; Thibodeaux, B.A.; Girgis, D.O.; O’Callaghan, R.J. Corneal virulence of Staphylococcus aureus in an experimental model of keratitis. DNA Cell Biol. 2002, 21, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Callegan, M.C.; Engel, L.S.; Hill, J.M.; O’Callaghan, R.J. Corneal virulence of Staphylococcus aureus: Roles of α-toxin and protein A in pathogenesis. Infect. Immun. 1994, 62, 2478–2482. [Google Scholar] [PubMed]
- Hume, E.B.; Cole, N.; Khan, S.; Garthwaite, L.L.; Aliwarga, Y.; Schubert, T.L.; Willcox, M.D. A Staphylococcus aureus mouse keratitis topical infection model: Cytokine balance in different strains of mice. Immunol. Cell Biol. 2005, 83, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Antonio, B.; Reyes-Grajeda, J.P.; Mariana, O.; Verónica, R.A.; Carolina, G.; Luz, L.N.; Herlinda, M.; Victor, M.B. Proteomic analysis of the interaction fusarium solani-Staphylococcus aureus isolated from human keratitis in presence of antimicrobial agents. Investig. Ophthalmol. Vis. Sci. 2014, 55, 392. [Google Scholar]
- Rattanatam, T.; Heng, W.J.; Rapuano, C.J.; Laibson, P.R.; Cohen, E.J. Trends in contact lens-related corneal ulcers. Cornea 2001, 20, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Schornack, M.M.; Faia, L.J.; Griepentrog, G.J. Pseudomonas keratitis associated with daily wear of silicone hydrogel contact lenses. Eye Contact Lens 2008, 34, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Green, M.; Apel, A.; Stapleton, F. Risk factors and causative organisms in microbial keratitis. Cornea 2008, 27, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Khatri, S.; Lass, J.H.; Heinzel, F.P.; Petroll, W.M.; Gomez, J.; Diaconu, E.; Kalsow, C.M.; Pearlman, E. Regulation of endotoxin-induced keratitis by PECAM-1, MIP-2, and toll-like receptor 4. Investig. Ophthalmol. Vis. Sci. 2002, 43, 2278–2284. [Google Scholar]
- Forster, R.K. Conrad berens lecture. The management of infectious keratitis as we approach the 21st century. CLAO J. 1998, 24, 175–180. [Google Scholar] [PubMed]
- Varaprasathan, G.; Miller, K.; Lietman, T.; Whitcher, J.P.; Cevallos, V.; Okumoto, M.; Margolis, T.P.; Yinghui, M.; Cunningham, E.T., Jr. Trends in the etiology of infectious corneal ulcers at the f. I. Proctor foundation. Cornea 2004, 23, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, F.; Keay, L.; Edwards, K.; Naduvilath, T.; Brian, G.; Jacobs, R. Studies of contact lens-related microbial keratitis in australia and new zealand: New learnings. Eye Contact Lens 2007, 33, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Doig, P.; Todd, T.; Sastry, P.A.; Lee, K.K.; Hodges, R.S.; Paranchych, W.; Irvin, R.T. Role of pili in adhesion of Pseudomonas aeruginosa to human respiratory epithelial cells. Infect. Immun. 1988, 56, 1641–1646. [Google Scholar] [PubMed]
- Holder, I.A.; Naglich, J.G. Experimental studies of the pathogenesis of infections due to Pseudomonas aeruginosa: Immunization using divalent flagella preparations. J. Trauma 1986, 26, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Hobden, J.A. Pseudomonas aeruginosa proteases and corneal virulence. DNA Cell Biol. 2002, 21, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Twining, S.S.; Kirschner, S.E.; Mahnke, L.A.; Frank, D.W. Effect of Pseudomonas aeruginosa elastase, alkaline protease, and exotoxin a on corneal proteinases and proteins. Investig. Ophthalmol. Vis. Sci. 1993, 34, 2699–2712. [Google Scholar]
- Iglewski, B.H.; Liu, P.V.; Kabat, D. Mechanism of action of Pseudomonas aeruginosa exotoxin aiadenosine diphosphate-ribosylation of mammalian elongation factor 2 in vitro and in vivo. Infect. Immun. 1977, 15, 138–144. [Google Scholar] [PubMed]
- Cryz, S.J., Jr.; Pitt, T.L.; Furer, E.; Germanier, R. Role of lipopolysaccharide in virulence of Pseudomonas aeruginosa. Infect. Immun. 1984, 44, 508–513. [Google Scholar] [PubMed]
- Engel, L.S.; Hill, J.M.; Caballero, A.R.; Green, L.C.; O’Callaghan, R.J. Protease iv, a unique extracellular protease and virulence factor from Pseudomonas aeruginosa. J. Biol. Chem. 1998, 273, 16792–16797. [Google Scholar] [CrossRef] [PubMed]
- Marquart, M.E.; Caballero, A.R.; Chomnawang, M.; Thibodeaux, B.A.; Twining, S.S.; O’Callaghan, R.J. Identification of a novel secreted protease from Pseudomonas aeruginosa that causes corneal erosions. Investig. Ophthalmol. Vis. Sci. 2005, 46, 3761–3768. [Google Scholar] [CrossRef] [PubMed]
- Woods, D.E.; Straus, D.C.; Johanson, W.G., Jr.; Bass, J.A. Role of fibronectin in the prevention of adherence of Pseudomonas aeruginosa to buccal cells. J. Infect. Dis. 1981, 143, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Hazlett, L.D.; Moon, M.M.; Strejc, M.; Berk, R.S. Evidence for n-acetylmannosamine as an ocular receptor for P. aeruginosa adherence to scarified cornea. Investig. Ophthalmol. Vis. Sci. 1987, 28, 1978–1985. [Google Scholar]
- Stern, G.A.; Weitzenkorn, D.; Valenti, J. Adherence of Pseudomonas aeruginosa to the mouse cornea. Epithelial v stromal adherence. Arch. Ophthalmol. 1982, 100, 1956–1958. [Google Scholar] [CrossRef] [PubMed]
- Hazlett, L.D.; Moon, M.; Berk, R.S. In vivo identification of sialic acid as the ocular receptor for Pseudomonas aeruginosa. Infect. Immun. 1986, 51, 687–689. [Google Scholar] [PubMed]
- Rudner, X.L.; Zheng, Z.; Berk, R.S.; Irvin, R.T.; Hazlett, L.D. Corneal epithelial glycoproteins exhibit Pseudomonas aeruginosa pilus binding activity. Investig. Ophthalmol. Vis. Sci. 1992, 33, 2185–2193. [Google Scholar]
- Karthikeyan, R.S.; Priya, J.L.; Leal, S.M., Jr.; Toska, J.; Rietsch, A.; Prajna, V.; Pearlman, E.; Lalitha, P. Host response and bacterial virulence factor expression in Pseudomonas aeruginosa and Streptococcus pneumoniae corneal ulcers. PLoS ONE 2013, 8, e64867. [Google Scholar] [CrossRef] [PubMed]
- Hazlett, L.D.; Rudner, X.L.; McClellan, S.A.; Barrett, R.P.; Lighvani, S. Role of IL-12 and IFN-γ in Pseudomonas aeruginosa corneal infection. Investig. Ophthalmol. Vis. Sci. 2002, 43, 419–424. [Google Scholar]
- Kernacki, K.A.; Berk, R.S. Characterization of the inflammatory response induced by corneal infection with Pseudomonas aeruginosa. J. Ocul. Pharmacol. 1994, 10, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Cole, N.; Krockenberger, M.; Stapleton, F.; Khan, S.; Hume, E.; Husband, A.J.; Willcox, M. Experimental Pseudomonas aeruginosa keratitis in interleukin-10 gene knockout mice. Infect. Immun. 2003, 71, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Cole, N.; Bao, S.; Willcox, M.; Husband, A.J. Expression of interleukin-6 in the cornea in response to infection with different strains of Pseudomonas aeruginosa. Infect. Immun. 1999, 67, 2497–2502. [Google Scholar] [PubMed]
- Kernacki, K.A.; Goebel, D.J.; Poosch, M.S.; Hazlett, L.D. Early cytokine and chemokine gene expression during Pseudomonas aeruginosa corneal infection in mice. Infect. Immun. 1998, 66, 376–379. [Google Scholar] [PubMed]
- O’Brien, T.P. Management of bacterial keratitis: Beyond exorcism towards consideration of organism and host factors. Eye 2003, 17, 957–974. [Google Scholar] [CrossRef] [PubMed]
- Sack, R.; Sathe, S.; Beaton, A.R.; McNamara, N.; Fleiszig, S.; Ni, M. Protein array characterization of bioactive proteins secreted by immortalized human corneal epithelium in response to pseudomonas constituents. Curr. Eye Res. 2009, 34, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Sewell, A.; Dunmire, J.; Wehmann, M.; Rowe, T.; Bouhenni, R. Proteomic analysis of keratitis-associated Pseudomonas aeruginosa. Mol. Vis. 2014, 20, 1182–1191. [Google Scholar] [PubMed]
- Boonpasart, S.; Kasetsuwan, N.; Puangsricharern, V.; Pariyakanok, L.; Jittpoonkusol, T. Infectious keratitis at king chulalongkorn memorial hospital: A 12-year retrospective study of 391 cases. J. Med. Assoc. Thail. 2002, 85 (Suppl. 1), S217–S230. [Google Scholar]
- Lalitha, P.; Srinivasan, M.; Manikandan, P.; Bharathi, M.J.; Rajaraman, R.; Ravindran, M.; Cevallos, V.; Oldenburg, C.E.; Ray, K.J.; Toutain-Kidd, C.M.; et al. Relationship of in vitro susceptibility to moxifloxacin and in vivo clinical outcome in bacterial keratitis. Clin. Infect. Dis. 2012, 54, 1381–1387. [Google Scholar] [CrossRef] [PubMed]
- Norina, T.J.; Raihan, S.; Bakiah, S.; Ezanee, M.; Liza-Sharmini, A.T.; Wan Hazzabah, W.H. Microbial keratitis: Aetiological diagnosis and clinical features in patients admitted to hospital universiti sains malaysia. Singap. Med. J. 2008, 49, 67–71. [Google Scholar]
- Ramesh, S.; Ramakrishnan, R.; Bharathi, M.J.; Amuthan, M.; Viswanathan, S. Prevalence of bacterial pathogens causing ocular infections in south india. Indian J. Pathol. Microbiol. 2010, 53, 281–286. [Google Scholar] [PubMed]
- Shalchi, Z.; Gurbaxani, A.; Baker, M.; Nash, J. Antibiotic resistance in microbial keratitis: Ten-year experience of corneal scrapes in the United Kingdom. Ophthalmology 2011, 118, 2161–2165. [Google Scholar] [CrossRef] [PubMed]
- Wagoner, M.D.; Al-Swailem, S.A.; Sutphin, J.E.; Zimmerman, M.B. Bacterial keratitis after penetrating keratoplasty: Incidence, microbiological profile, graft survival, and visual outcome. Ophthalmology 2007, 114, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Al Otaibi, A.G.; Allam, K.; Damri, A.J.; Shamri, A.A.; Kalantan, H.; Mousa, A. Childhood microbial keratitis. Oman J. Ophthalmol. 2012, 5, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Bashir, G.; Shah, A.; Thokar, M.A.; Rashid, S.; Shakeel, S. Bacterial and fungal profile of corneal ulcers--a prospective study. Indian J. Pathol. Microbiol. 2005, 48, 273–277. [Google Scholar] [PubMed]
- Bharathi, M.J.; Ramakrishnan, R.; Vasu, S.; Palaniappan, R. Aetiological diagnosis of microbial keratitis in south India—A study of 1618 cases. Indian J. Med. Microbiol. 2002, 20, 19–24. [Google Scholar] [PubMed]
- Bharathi, M.J.; Ramakrishnan, R.; Vasu, S.; Meenakshi, R.; Shivkumar, C.; Palaniappan, R. Epidemiology of bacterial keratitis in a referral centre in south india. Indian J. Med. Microbiol. 2003, 21, 239–245. [Google Scholar] [PubMed]
- Bharathi, M.J.; Ramakrishnan, R.; Meenakshi, R.; Padmavathy, S.; Shivakumar, C.; Srinivasan, M. Microbial keratitis in south india: Influence of risk factors, climate, and geographical variation. Ophthalmic Epidemiol. 2007, 14, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Feilmeier, M.R.; Sivaraman, K.R.; Oliva, M.; Tabin, G.C.; Gurung, R. Etiologic diagnosis of corneal ulceration at a tertiary eye center in kathmandu, nepal. Cornea 2010, 29, 1380–1385. [Google Scholar] [CrossRef] [PubMed]
- Deorukhkar, S.; Katiyar, R.; Saini, S. Epidemiological features and laboratory results of bacterial and fungal keratitis: A five-year study at a rural tertiary-care hospital in western maharashtra, india. Singap. Med. J. 2012, 53, 264–267. [Google Scholar]
- Mulet, M.E.; Perez-Santonja, J.J.; Ferrer, C.; Alio, J.L. Microbial keratitis after intrastromal corneal ring segment implantation. J. Refract. Surg. 2010, 26, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Cosar, C.B.; Cohen, E.J.; Rapuano, C.J.; Laibson, P.R. Clear corneal wound infection after phacoemulsification. Arch. Ophthalmol. 2001, 119, 1755–1759. [Google Scholar] [CrossRef] [PubMed]
- Lifshitz, T.; Levy, J.; Raiskup, F.; Klemperer, I.; Frucht-Pery, J. Two cases of pneumococcal keratitis following myopic lasik. J. Refract. Surg. 2005, 21, 498–501. [Google Scholar] [PubMed]
- Nubile, M.; Carpineto, P.; Lanzini, M.; Ciancaglini, M.; Zuppardi, E.; Mastropasqua, L. Multilayer amniotic membrane transplantation for bacterial keratitis with corneal perforation after hyperopic photorefractive keratectomy: Case report and literature review. J. Cataract Refract. Surg. 2007, 33, 1636–1640. [Google Scholar] [CrossRef] [PubMed]
- Griffith, F. The significance of pneumococcal types. J. Hyg. 1928, 27, 113–159. [Google Scholar] [CrossRef] [PubMed]
- Avery, O.T.; Macleod, C.M.; McCarty, M. Studies on the chemical nature of the substance inducing transformation of pneumococcal types: Induction of transformation by a desoxyribonucleic acid fraction isolated from pneumococcus type III. J. Exp. Med. 1944, 79, 137–158. [Google Scholar] [CrossRef] [PubMed]
- Norcross, E.W.; Tullos, N.A.; Taylor, S.D.; Sanders, M.E.; Marquart, M.E. Assessment of Streptococcus pneumoniae capsule in conjunctivitis and keratitis in vivo neuraminidase activity increases in nonencapsulated pneumococci following conjunctival infection. Curr. Eye Res. 2010, 35, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Reed, J.M.; O’Callaghan, R.J.; Girgis, D.O.; McCormick, C.C.; Caballero, A.R.; Marquart, M.E. Ocular virulence of capsule-deficient Streptococcus pneumoniae in a rabbit keratitis model. Investig. Ophthalmol. Vis. Sci. 2005, 46, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.K.; Hobden, J.A.; Hagenah, M.; O’Callaghan, R.J.; Hill, J.M.; Chen, S. The role of pneumolysin in ocular infections with Streptococcus pneumoniae. Curr. Eye Res. 1990, 9, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.K.; Allen, J.H. Ocular toxin of the pneumococcus. Am. J. Ophthalmol. 1971, 72, 175–180. [Google Scholar] [CrossRef]
- Johnson, M.K.; Allen, J.H. The role of cytolysin in pneumococcal ocular infection. Am. J. Ophthalmol. 1975, 80, 518–521. [Google Scholar] [CrossRef]
- Paton, J.C.; Rowan-Kelly, B.; Ferrante, A. Activation of human complement by the pneumococcal toxin pneumolysin. Infect. Immun. 1984, 43, 1085–1087. [Google Scholar] [PubMed]
- Alcantara, R.B.; Preheim, L.C.; Gentry-Nielsen, M.J. Pneumolysin-induced complement depletion during experimental pneumococcal bacteremia. Infect. Immun. 2001, 69, 3569–3575. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kelly, S.J.; Jedrzejas, M.J. Structure and molecular mechanism of a functional form of pneumolysin: A cholesterol-dependent cytolysin from Streptococcus pneumoniae. J. Struct. Biol. 2000, 132, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Karmakar, M.; Katsnelson, M.; Malak, H.A.; Greene, N.G.; Howell, S.J.; Hise, A.G.; Camilli, A.; Kadioglu, A.; Dubyak, G.R.; Pearlman, E. Neutrophil IL-1β processing induced by pneumolysin is mediated by the NLRP3/ASC inflammasome and Caspase-1 activation and is dependent on K+ efflux. J. Immunol. 2015, 194, 1763–1775. [Google Scholar] [PubMed]
- Taylor, S.D.; Sanders, M.E.; Tullos, N.A.; Stray, S.J.; Norcross, E.W.; McDaniel, L.S.; Marquart, M.E. The cholesterol-dependent cytolysin pneumolysin from Streptococcus pneumoniae binds to lipid raft microdomains in human corneal epithelial cells. PLoS ONE 2013, 8, e61300. [Google Scholar] [CrossRef] [PubMed]
- Green, S.N.; Sanders, M.; Moore, Q.C., III; Norcross, E.W.; Monds, K.S.; Caballero, A.R.; McDaniel, L.S.; Robinson, S.A.; Onwubiko, C.; O’Callaghan, R.J.; et al. Protection from Streptococcus pneumoniae keratitis by passive immunization with pneumolysin antiserum. Investig. Ophthalmol. Vis. Sci. 2008, 49, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Hejazi, A.; Falkiner, F.R. Serratia marcescens. J. Med. Microbiol. 1997, 46, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Parment, P.A. The role of Serratia marcescens in soft contact lens associated ocular infections. A review. Acta Ophthalmol. Scand. 1997, 75, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Maeda, H.; Takata, K.; Kamata, R.; Okamura, R. Purification and characterization of four proteases from a clinical isolate of Serratia marcescens kums 3958. J. Bacteriol. 1984, 157, 225–232. [Google Scholar] [PubMed]
- Yonemura, K.; Matsumoto, K.; Maeda, H. Isolation and characterization of nucleases from a clinical isolate of Serratia marcescens kums 3958. J. Biochem. 1983, 93, 1287–1295. [Google Scholar] [PubMed]
- Kamata, R.; Matsumoto, K.; Okamura, R.; Yamamoto, T.; Maeda, H. The serratial 56K protease as a major pathogenic factor in serratial keratitis. Clinical and experimental study. Ophthalmology 1985, 92, 1452–1459. [Google Scholar] [CrossRef]
- Kamata, R.; Yamamoto, T.; Matsumoto, K.; Maeda, H. A serratial protease causes vascular permeability reaction by activation of the hageman factor-dependent pathway in guinea pigs. Infect. Immun. 1985, 48, 747–753. [Google Scholar] [PubMed]
- Matsumoto, K.; Yamamoto, T.; Kamata, R.; Maeda, H. Pathogenesis of serratial infection: Activation of the hageman factor-prekallikrein cascade by serratial protease. J. Biochem. 1984, 96, 739–749. [Google Scholar] [PubMed]
- Bhaskar, A.; Upgade, A.; Kavitha, P. Characterization of keratitis disease causing 56k cysteine protease encoding gene from Serratia marcescens. J. Proteom. Bioinform. 2012, 5–10. [Google Scholar] [CrossRef]
- Zhou, R.; Zhang, R.; Sun, Y.; Platt, S.; Szczotka-Flynn, L.; Pearlman, E. Innate immune regulation of Serratia marcescens-induced corneal inflammation and infection. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7382–7388. [Google Scholar] [CrossRef] [PubMed]
- Preston, M.J.; Berk, J.M.; Hazlett, L.D.; Berk, R.S. Serum antibody response to Pseudomonas aeruginosa antigens during corneal infection. Infect. Immun. 1991, 59, 1984–1990. [Google Scholar] [PubMed]
- Hazlett, L.D. Corneal response to Pseudomonas aeruginosa infection. Prog. Retin. Eye Res. 2004, 23, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Hazlett, L.D.; McClellan, S.A.; Barrett, R.P.; Liu, J.; Zhang, Y.; Lighvani, S. Spantide I decreases type I cytokines, enhances IL-10, and reduces corneal perforation in susceptible mice after Pseudomonas aeruginosa infection. Investig. Ophthalmol. Vis. Sci. 2007, 48, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.C.; Heinzel, F.P.; Diaconu, E.; Sun, Y.; Hise, A.G.; Golenbock, D.; Lass, J.H.; Pearlman, E. Activation of Toll-like Receptor (TLR)2, TLR4, and TLR9 in the Mammalian Cornea Induces MyD88-Dependent Corneal Inflammation. Investig. Ophthalmol. Vis. Sci. 2005, 46, 589–595. [Google Scholar] [CrossRef] [PubMed]
- McClellan, S.A.; Huang, X.; Barrett, R.P.; van Rooijen, N.; Hazlett, L.D. Macrophages restrict Pseudomonas aeruginosa growth, regulate polymorphonuclear neutrophil influx, and balance pro- and anti-inflammatory cytokines in BALB/c mice. J. Immunol. 2003, 170, 5219–5227. [Google Scholar] [CrossRef] [PubMed]
- McClellan, S.A.; Huang, X.; Barrett, R.P.; Lighvani, S.; Zhang, Y.; Richiert, D.; Hazlett, L.D. Matrix metalloproteinase-9 amplifies the immune response to Pseudomonas aeruginosa corneal infection. Investig. Ophthalmol. Vis. Sci. 2006, 47, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.L.; Willcox, M.D.; Lloyd, A.; Wakefield, D.; Thakur, A. Regulatory role of IL-1β in the expression of IL-6 and IL-8 in human corneal epithelial cells during Pseudomonas aeruginosa colonization. Clin. Exp. Ophthalmol. 2001, 29, 171–174. [Google Scholar] [CrossRef]
- Li, C.; McClellan, S.A.; Barrett, R.; Hazlett, L.D. Interleukin 17 regulates Mer tyrosine kinase-positive cells in Pseudomonas aeruginosa keratitis. Investig. Ophthalmol. Vis. Sci. 2014, 55, 6886–6900. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Zhang, J.; Yu, F.S. Innate immune response of corneal epithelial cells to Staphylococcus aureus infection: Role of peptidoglycan in stimulating proinflammatory cytokine secretion. Investig. Ophthalmol. Vis. Sci. 2004, 45, 3513–3522. [Google Scholar] [CrossRef] [PubMed]
- Gowda, R.N.; Redfern, R.; Frikeche, J.; Pinglay, S.; Foster, J.W.; Lema, C.; Cope, L.; Chakravarti, S. Functions of peptidoglycan recognition proteins (Pglyrps) at the ocular surface: Bacterial keratitis in gene-targeted mice deficient in Pglyrp-2, -3 and -4. PLoS ONE 2015, 10, e0137129. [Google Scholar] [CrossRef] [PubMed]
- Shao, H.; Scott, S.G.; Nakata, C.; Hamad, A.R.; Chakravarti, S. Extracellular matrix protein lumican promotes clearance and resolution of Pseudomonas aeruginosa keratitis in a mouse model. PLoS ONE 2013, 8, e54765. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.C.; Vergnes, J.P.; Brown, S.I. O-glycosidic linkage in glycoprotein isolates from human ocular mucus. Exp. Eye Res. 1983, 37, 533–541. [Google Scholar] [CrossRef]
- Chao, C.C.; Butala, S.M.; Herp, A. Studies on the isolation and composition of human ocular mucin. Exp. Eye Res. 1988, 47, 185–196. [Google Scholar] [CrossRef]
- Green-Church, K.B.; Nichols, J.J. Mass spectrometry-based proteomic analyses of contact lens deposition. Mol. Vis. 2008, 14, 291–297. [Google Scholar] [PubMed]
- Zhao, Z.; Wei, X.; Aliwarga, Y.; Carnt, N.A.; Garrett, Q.; Willcox, M.D. Proteomic analysis of protein deposits on worn daily wear silicone hydrogel contact lenses. Mol. Vis. 2008, 14, 2016–2024. [Google Scholar] [PubMed]
- Stenson, S. Superior limbic keratoconjunctivitis associated with soft contact lens wear. Arch. Ophthalmol. 1983, 101, 402–404. [Google Scholar] [CrossRef] [PubMed]
- Omali, N.B.; Subbaraman, L.N.; Coles-Brennan, C.; Fadli, Z.; Jones, L.W. Biological and clinical implications of lysozyme deposition on soft contact lenses. Optom. Vis. Sci. 2015, 92, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Cash, P. Proteomics in medical microbiology. Electrophoresis 2000, 21, 1187–1201. [Google Scholar] [CrossRef]
- Grandi, G. Antibacterial vaccine design using genomics and proteomics. Trends Biotechnol. 2001, 19, 181–188. [Google Scholar] [CrossRef]
- Hare, N.J.; Solis, N.; Harmer, C.; Marzook, N.B.; Rose, B.; Harbour, C.; Crossett, B.; Manos, J.; Cordwell, S.J. Proteomic profiling of Pseudomonas aeruginosa AES-1R, PAO1 and PA14 reveals potential virulence determinants associated with a transmissible cystic fibrosis-associated strain. BMC Microbiol. 2012, 12, 16. [Google Scholar] [CrossRef] [PubMed]
- Ananthi, S.; Chitra, T.; Bini, R.; Prajna, N.V.; Lalitha, P.; Dharmalingam, K. Comparative analysis of the tear protein profile in mycotic keratitis patients. Mol. Vis. 2008, 14, 500–507. [Google Scholar] [PubMed]
- Ananthi, S.; Venkatesh Prajna, N.; Lalitha, P.; Valarnila, M.; Dharmalingam, K. Pathogen induced changes in the protein profile of human tears from fusarium keratitis patients. PLoS ONE 2013, 8, e53018. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Lu, Q.; Xue, P.; Zhang, H.; Dong, Z.; Yang, F.; Wang, N. Proteomic analysis of aqueous humor from patients with myopia. Mol. Vis. 2008, 14, 370–377. [Google Scholar] [PubMed]
- Niwa, T. Biomarker discovery for kidney diseases by mass spectrometry. J. Chromatogr. B 2008, 870, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Davidsson, P.; Sjogren, M. Proteome studies of CSF in ad patients. Mech. Ageing Dev. 2006, 127, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, P.R.; Verma, M.; Zhao, Y.; Srivastava, S. Proteomics for cancer biomarker discovery. Clin. Chem. 2002, 48, 1160–1169. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouhenni, R.; Dunmire, J.; Rowe, T.; Bates, J. Proteomics in the Study of Bacterial Keratitis. Proteomes 2015, 3, 496-511. https://doi.org/10.3390/proteomes3040496
Bouhenni R, Dunmire J, Rowe T, Bates J. Proteomics in the Study of Bacterial Keratitis. Proteomes. 2015; 3(4):496-511. https://doi.org/10.3390/proteomes3040496
Chicago/Turabian StyleBouhenni, Rachida, Jeffrey Dunmire, Theresa Rowe, and James Bates. 2015. "Proteomics in the Study of Bacterial Keratitis" Proteomes 3, no. 4: 496-511. https://doi.org/10.3390/proteomes3040496
APA StyleBouhenni, R., Dunmire, J., Rowe, T., & Bates, J. (2015). Proteomics in the Study of Bacterial Keratitis. Proteomes, 3(4), 496-511. https://doi.org/10.3390/proteomes3040496




