Optimising Early Childhood Educational Settings for Health Using Nature-Based Solutions: The Microbiome Aspect
Abstract
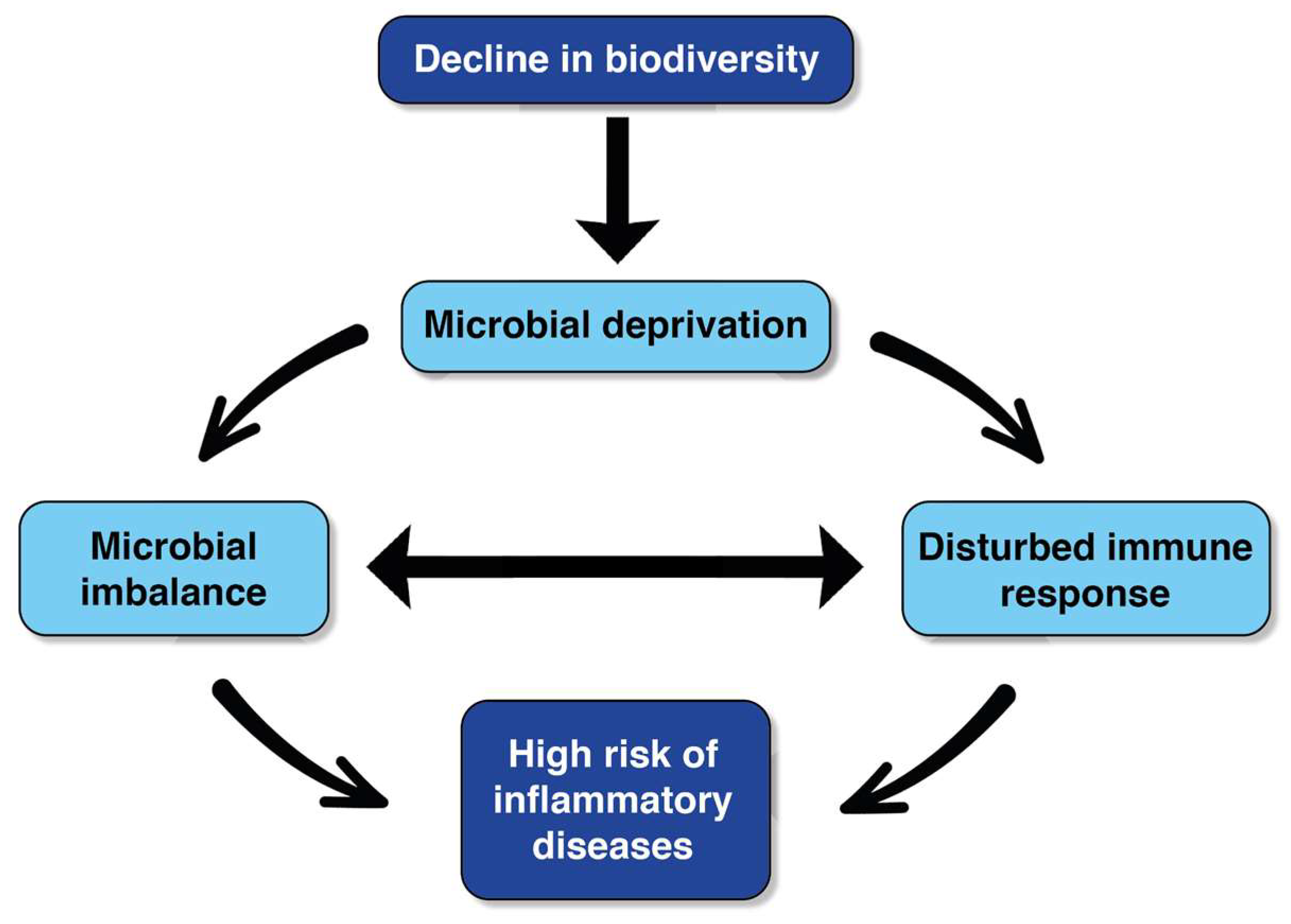
1. Introduction
2. The Microbiome-Inspired Green Infrastructure (MIGI) Context
3. Microbiome-Centric Considerations
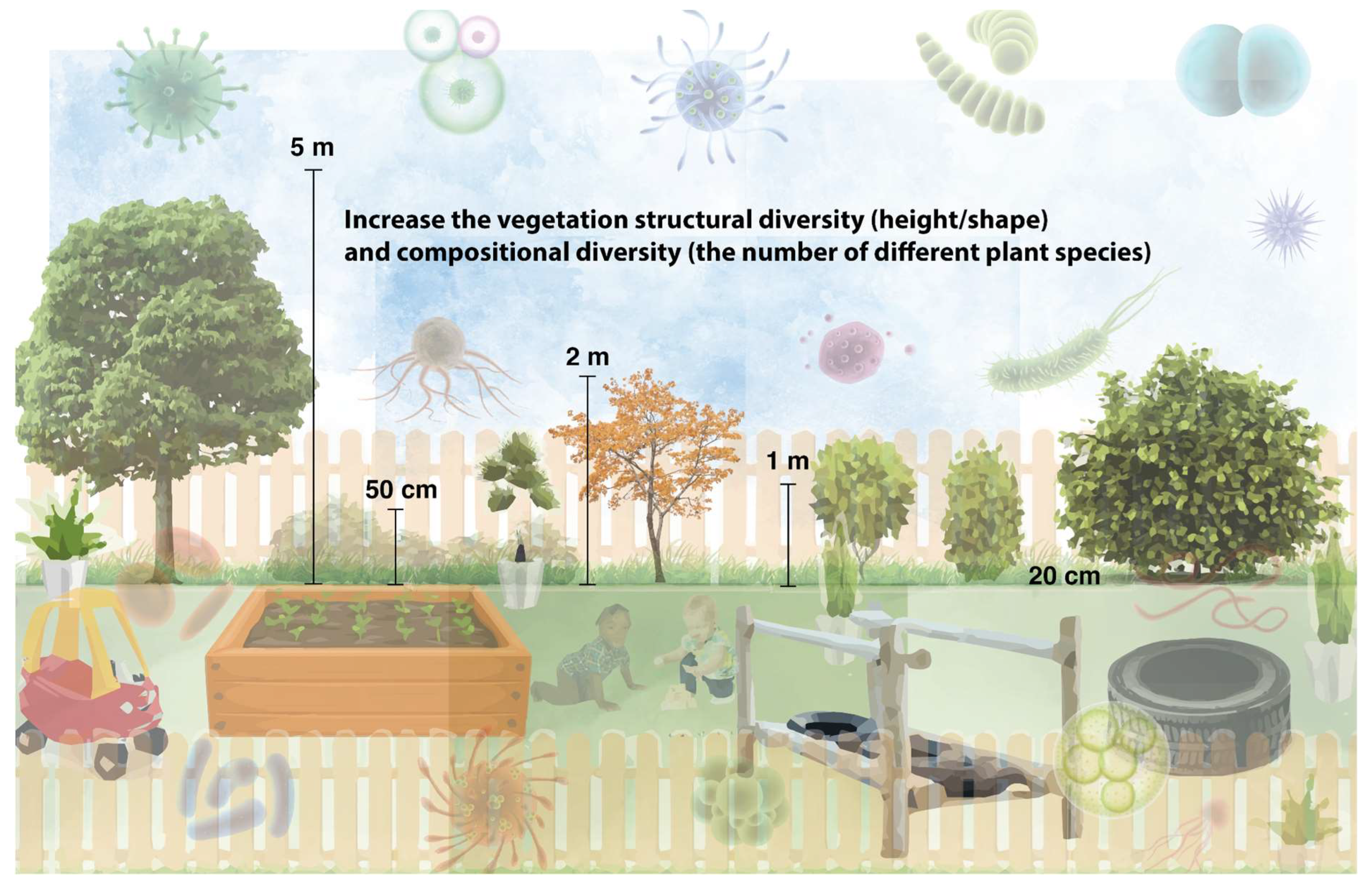
3.1. Increase the Diversity and Structural Complexity of Vegetation
3.2. Consult Green Barrier Planting Designs to Reduce Pollution
3.3. Consider the Use of Microbial Inoculants in Sand Pits
3.4. Create an Undulating Topography in the Outdoor Environment
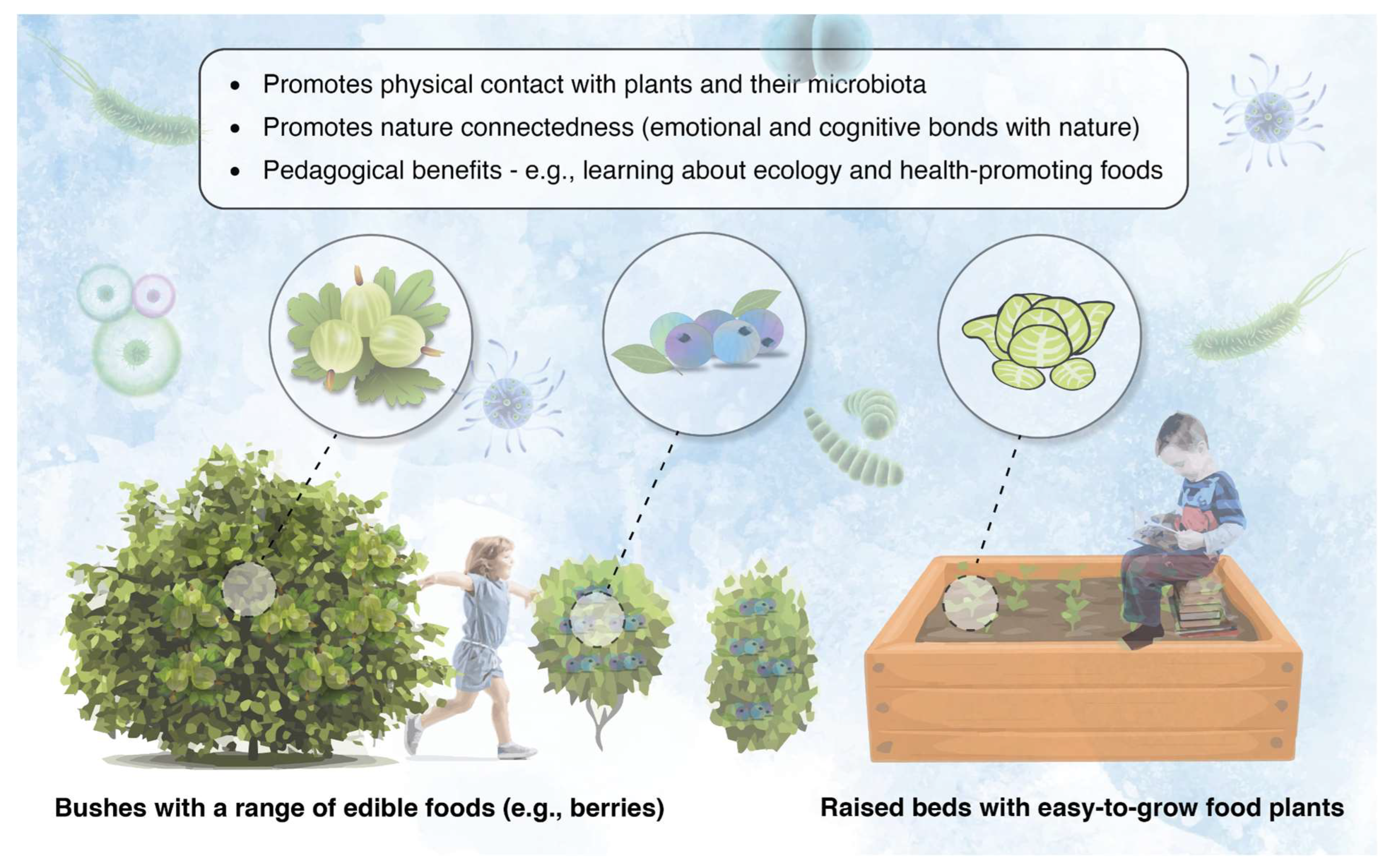
3.5. Include Foraging and Plant Cultivation Opportunities
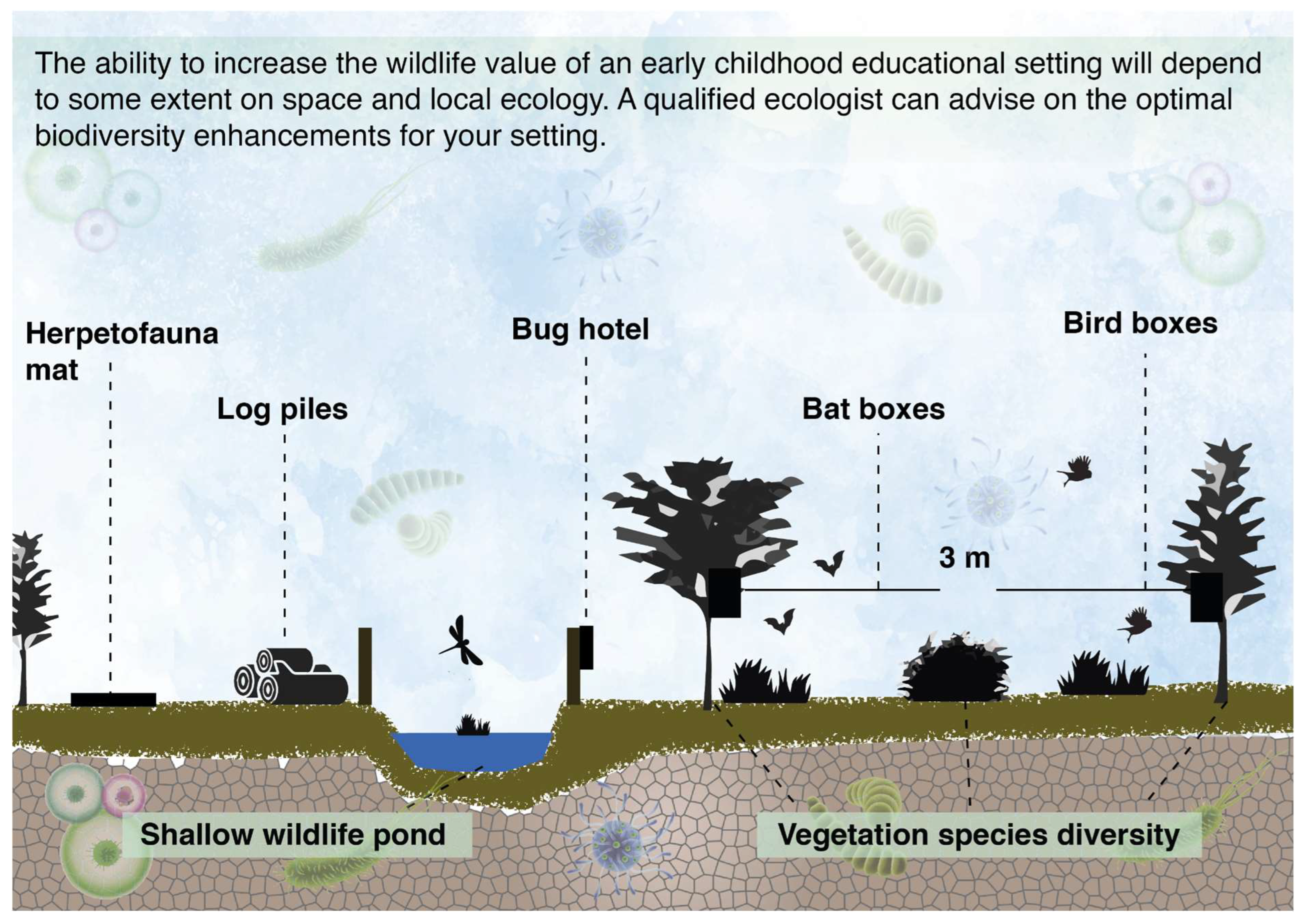
3.6. Increase the Wildlife Value of a Site
3.7. Avoid the Use of Harsh Pesticides, Fertilisers, and Artificial Grass
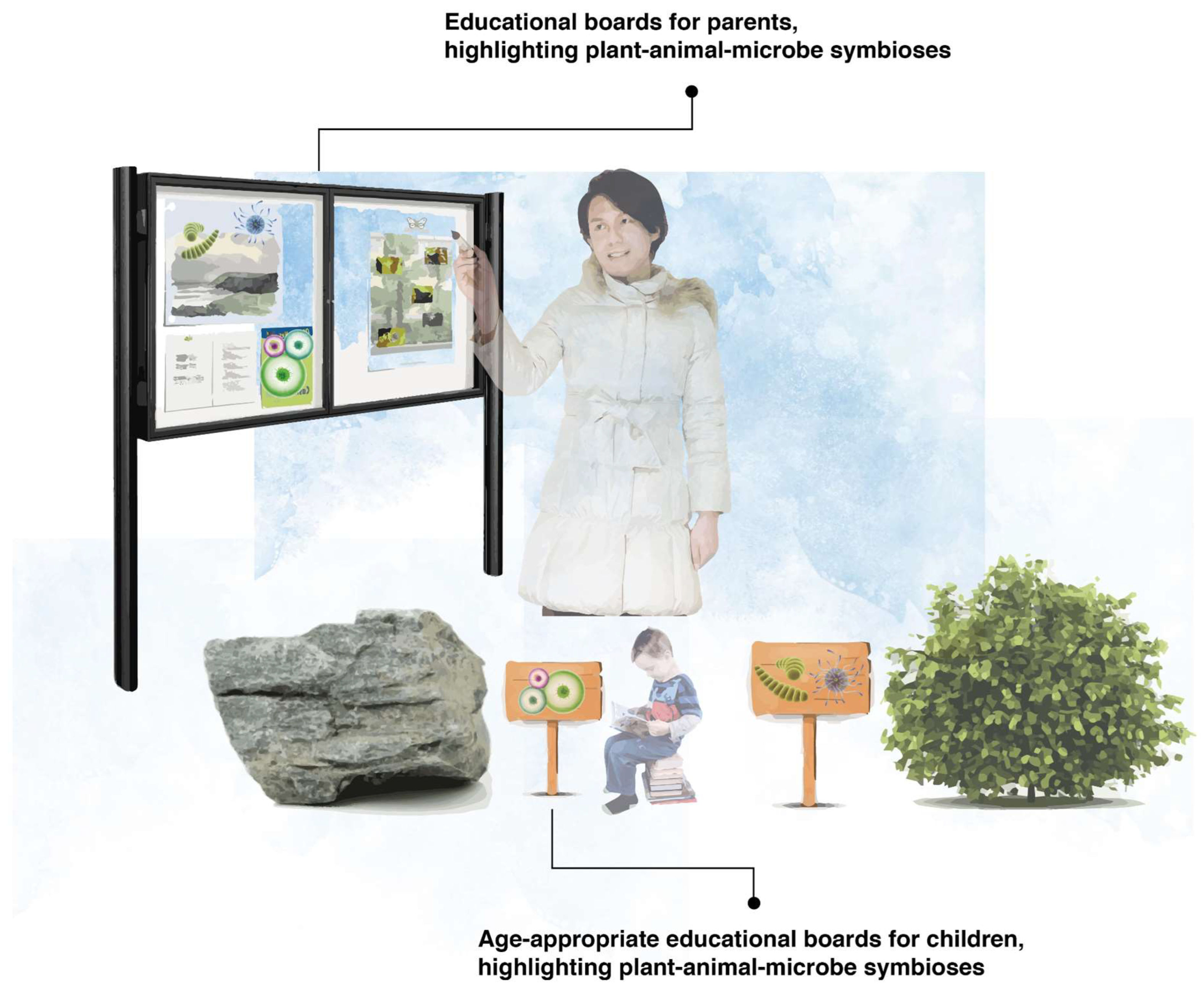
3.8. Educational Interpretation Boards to Make the Invisible Visible
4. Naturing the Indoor Environment and Future Possibilities
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Britto, P.R.; Lye, S.J.; Proulx, K.; Yousafzai, A.K.; Matthews, S.G.; Vaivada, T.; Perez-Escamilla, R.; Rao, N.; Ip, P.; Fernald, L.C.H.; et al. Nurturing Care: Promoting Early Childhood Development. Lancet 2017, 389, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Venetsanou, F.; Kambas, A. Environmental Factors Affecting Preschoolers’ Motor Development. Early Child. Educ. J. 2010, 37, 319–327. [Google Scholar] [CrossRef]
- McDonald, L.; Wardle, J.; Llewellyn, C.H.; van Jaarsveld, C.H.M.; Fisher, A. Predictors of Shorter Sleep in Early Childhood. Sleep Med. 2014, 15, 536–540. [Google Scholar] [CrossRef]
- Silventoinen, K.; Rokholm, B.; Kaprio, J.; Sørensen, T.I.A. The Genetic and Environmental Influences on Childhood Obesity: A Systematic Review of Twin and Adoption Studies. Int. J. Obes. 2010, 34, 29–40. [Google Scholar] [CrossRef]
- Liao, J.; Zhang, B.; Xia, W.; Cao, Z.; Zhang, Y.; Liang, S.; Hu, K.; Xu, S.; Li, Y. Residential Exposure to Green Space and Early Childhood Neurodevelopment. Environ. Int. 2019, 128, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Bruno, A.; Fumagalli, S.; Ghisleni, G.; Labra, M. The Microbiome of the Built Environment: The Nexus for Urban Regeneration for the Cities of Tomorrow. Microorganisms 2022, 10, 2311. [Google Scholar] [CrossRef]
- Roslund, M.I.; Parajuli, A.; Hui, N.; Puhakka, R.; Grönroos, M.; Soininen, L.; Nurminen, N.; Oikarinen, S.; Cinek, O.; Kramná, L.; et al. A Placebo-Controlled Double-Blinded Test of the Biodiversity Hypothesis of Immune-Mediated Diseases: Environmental Microbial Diversity Elicits Changes in Cytokines and Increase in T Regulatory Cells in Young Children. Ecotoxicol. Environ. Saf. 2022, 242, 113900. [Google Scholar] [CrossRef] [PubMed]
- Relman, D.A. The Human Microbiome: Ecosystem Resilience and Health. Nutr. Rev. 2012, 70, S2–S9. [Google Scholar] [CrossRef]
- Haahtela, T. A Biodiversity Hypothesis. Allergy 2019, 74, 1445–1456. [Google Scholar] [CrossRef]
- Robertson, R.C.; Manges, A.R.; Finlay, B.B.; Prendergast, A.J. The Human Microbiome and Child Growth – First 1000 Days and Beyond. Trends Microbiol. 2019, 27, 131–147. [Google Scholar] [CrossRef]
- Williams, C.L.; Garcia-Reyero, N.; Martyniuk, C.J.; Tubbs, C.W.; Bisesi, J.H. Regulation of Endocrine Systems by the Microbiome: Perspectives from Comparative Animal Models. Gen. Comp. Endocrinol. 2020, 292, 113437. [Google Scholar] [CrossRef] [PubMed]
- Renz, H.; Skevaki, C. Early Life Microbial Exposures and Allergy Risks: Opportunities for Prevention. Nat. Rev. Immunol. 2021, 21, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Parajuli, A.; Grönroos, M.; Siter, N.; Puhakka, R.; Vari, H.K.; Roslund, M.I.; Jumpponen, A.; Nurminen, N.; Laitinen, O.H.; Hyöty, H.; et al. Urbanization Reduces Transfer of Diverse Environmental Microbiota Indoors. Front. Microbiol. 2018, 9, 84. [Google Scholar] [CrossRef]
- Childcare and Early Years Survey of Parents, Reporting Year. 2021. Available online: https://explore-education-statistics.service.gov.uk/find-statistics/childcare-and-early-years-survey-of-parents (accessed on 1 December 2022).
- Early Childhood Education. Available online: https://www.unicef.org/india/what-we-do/early-childhood-education (accessed on 4 December 2022).
- Ansari, A.; Gottfried, M.A. Early Childhood Educational Settings and School Absenteeism for Children With Disabilities. AERA Open 2018, 4. [Google Scholar] [CrossRef]
- Roslund, M.I.; Puhakka, R.; Grönroos, M.; Nurminen, N.; Oikarinen, S.; Gazali, A.M.; Cinek, O.; Kramná, L.; Siter, N.; Vari, H.K.; et al. Biodiversity Intervention Enhances Immune Regulation and Health-Associated Commensal Microbiota among Daycare Children. Sci. Adv. 2020, 6, eaba2578. [Google Scholar] [CrossRef]
- Haahtela, T.; Holgate, S.; Pawankar, R.; Akdis, C.A.; Benjaponpitak, S.; Caraballo, L.; Demain, J.; Portnoy, J.; von Hertzen, L. The Biodiversity Hypothesis and Allergic Disease: World Allergy Organization Position Statement. World Allergy Organ. J. 2013, 6, 3. [Google Scholar] [CrossRef]
- Roslund, M.I.; Puhakka, R.; Nurminen, N.; Oikarinen, S.; Siter, N.; Grönroos, M.; Cinek, O.; Kramná, L.; Jumpponen, A.; Laitinen, O.H.; et al. Long-Term Biodiversity Intervention Shapes Health-Associated Commensal Microbiota among Urban Day-Care Children. Environ. Int. 2021, 157, 106811. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.M.; Watkins, H.; Man, I.; Liddicoat, C.; Cameron, R.; Parker, B.; Cruz, M.; Meagher, L. Microbiome-Inspired Green Infrastructure: A bioscience roadmap for urban ecosystem health. Archit. Res. Q. 2021, 25, 292–303. [Google Scholar] [CrossRef]
- Rai, S.; Singh, D.K.; Kumar, A. Microbial, Environmental and Anthropogenic Factors Influencing the Indoor Microbiome of the Built Environment. J. Basic Microbiol. 2021, 61, 267–292. [Google Scholar] [CrossRef]
- Robinson, J.M.; Cando-Dumancela, C.; Antwis, R.E.; Cameron, R.; Liddicoat, C.; Poudel, R.; Weinstein, P.; Breed, M.F. Exposure to Airborne Bacteria Depends upon Vertical Stratification and Vegetation Complexity. Sci. Rep. 2021, 11, 9516. [Google Scholar] [CrossRef]
- Li, H.; Wu, Z.-F.; Yang, X.-R.; An, X.-L.; Ren, Y.; Su, J.-Q. Urban Greenness and Plant Species Are Key Factors in Shaping Air Microbiomes and Reducing Airborne Pathogens. Environ. Int. 2021, 153, 106539. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Chi, L.; Mahbub, R.; Bian, X.; Tu, P.; Ru, H.; Lu, K. Multi-Omics Reveals That Lead Exposure Disturbs Gut Microbiome Development, Key Metabolites, and Metabolic Pathways. Chem. Res. Toxicol. 2017, 30, 996–1005. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Kwon, H.; Ha, M.; Lim, J.-A.; Lim, M.; Yoo, S.-J.; Paik, K. How Does Low Socioecon. Status Increase Blood Lead Levelsin KoreanChildren? IJERPH 2018, 15, 1488. [Google Scholar] [CrossRef] [PubMed]
- Varshney, S.; Gora, A.H.; Siriyappagouder, P.; Kiron, V.; Olsvik, P.A. Toxicological Effects of 6PPD and 6PPD Quinone in Zebrafish Larvae. J. Hazard. Mater. 2022, 424, 127623. [Google Scholar] [CrossRef]
- Fang, L.; Fang, C.; Di, S.; Yu, Y.; Wang, C.; Wang, X.; Jin, Y. Oral Exposure to Tire Rubber-Derived Contaminant 6ppd and 6ppd-Quinone Induced Hepatotoxicity in Mice. Sci. Total. Environ. 2023, 869, 161836. [Google Scholar] [CrossRef]
- Muller, R.; Muller, E. Air Pollution and Cigarette Equivalence. Berkeley Earth. 2015. Available online: https://berkeleyearth.org/air-pollution-and-cigarette-equivalence/ (accessed on 19 December 2022).
- Peled, R. Air pollution exposure: Who is at high risk? Atmos. Environ. 2011, 45, 1781–1785. [Google Scholar] [CrossRef]
- Redondo Bermúdez, M.d.C.; Kanai, J.M.; Astbury, J.; Fabio, V.; Jorgensen, A. Green Fences for Buenos Aires: Implementing Green Infrastructure for (More than) Air Quality. Sustainability 2022, 14, 4129. [Google Scholar] [CrossRef]
- Hui, N.; Grönroos, M.; Roslund, M.I.; Parajuli, A.; Vari, H.K.; Soininen, L.; Laitinen, O.H.; Sinkkonen, A. The ADELE Research Group Diverse Environmental Microbiota as a Tool to Augment Biodiversity in Urban Landscaping Materials. Front. Microbiol. 2019, 10, 536. [Google Scholar] [CrossRef]
- Qiu, Z.; Egidi, E.; Liu, H.; Kaur, S.; Singh, B.K. New Frontiers in Agriculture Productivity: Optimised Microbial Inoculants and in Situ Microbiome Engineering. Biotechnol. Adv. 2019, 37, 107371. [Google Scholar] [CrossRef]
- Dadzie, F.A.; Moles, A.T.; Erickson, T.E.; Slavich, E.; Muñoz-Rojas, M. Native Bacteria and Cyanobacteria Can Influence Seedling Emergence and Growth of Native Plants Used in Dryland Restoration. J. Appl. Ecol. 2022, 59, 2983–2992. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, R.; Chen, S.; Qi, G.; He, Z.; Zhao, X. Microbial Taxa and Functional Genes Shift in Degraded Soil with Bacterial Wilt. Sci. Rep. 2017, 7, 39911. [Google Scholar] [CrossRef]
- Roesch, L.F.W.; Fulthorpe, R.R.; Riva, A.; Casella, G.; Hadwin, A.K.M.; Kent, A.D.; Daroub, S.H.; Camargo, F.A.O.; Farmerie, W.G.; Triplett, E.W. Pyrosequencing Enumerates and Contrasts Soil Microbial Diversity. ISME J. 2007, 1, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.M.; Cando-Dumancela, C.; Liddicoat, C.; Weinstein, P.; Cameron, R.; Breed, M.F. Vertical Stratification in Urban Green Space Aerobiomes. Env. Health Perspect. 2020, 128, 117008. [Google Scholar] [CrossRef] [PubMed]
- Fjørtoft, I.; Sageie, J. The Natural Environment as a Playground for Children. Landsc. Urban Plan. 2000, 48, 83–97. [Google Scholar] [CrossRef]
- The Shocking State of Biodiversity: Education and Mass Extinction. Available online: https://www.earlham.ac.uk/articles/shocking-state-biodiversity-education-and-mass-extinction (accessed on 10 December 2022).
- Parsley, K.M. Plant Awareness Disparity: A Case for Renaming Plant Blindness. Plants People Planet 2020, 2, 598–601. [Google Scholar] [CrossRef]
- Barrable, A. The Case for Nature Connectedness as a Distinct Goal of Early Childhood Education. Int. J. Early Child. 2019, 6, 59–70. [Google Scholar]
- Grönroos, M.; Parajuli, A.; Laitinen, O.H.; Roslund, M.I.; Vari, H.K.; Hyöty, H.; Puhakka, R.; Sinkkonen, A. Short-term Direct Contact with Soil and Plant Materials Leads to an Immediate Increase in Diversity of Skin Microbiota. MicrobiologyOpen 2019, 8, e00645. [Google Scholar] [CrossRef]
- Cavicchioli, R.; Ripple, W.J.; Timmis, K.N.; Azam, F.; Bakken, L.R.; Baylis, M.; Behrenfeld, M.J.; Boetius, A.; Boyd, P.W.; Classen, A.T.; et al. Scientists’ Warning to Humanity: Microorganisms and Climate Change. Nat. Rev. Microbiol. 2019, 17, 569–586. [Google Scholar] [CrossRef]
- Edlinger, A.; Garland, G.; Hartman, K.; Banerjee, S.; Degrune, F.; García-Palacios, P.; Hallin, S.; Valzano-Held, A.; Herzog, C.; Jansa, J.; et al. Agricultural Management and Pesticide Use Reduce the Functioning of Beneficial Plant Symbionts. Nat. Ecol. Evol. 2022, 6, 1145–1154. [Google Scholar] [CrossRef]
- Barrable, A.; Booth, D.; Adams, D.; Beauchamp, G. Enhancing Nature Connection and Positive Affect in Children through Mindful Engagement with Natural Environments. Int. J. Environ. Res. Public Health 2021, 18, 4785. [Google Scholar] [CrossRef]
- Martin, L.; White, M.P.; Hunt, A.; Richardson, M.; Pahl, S.; Burt, J. Nature Contact, Nature Connectedness and Associations with Health, Wellbeing and pro-Environmental Behaviours. J. Environ. Psychol. 2020, 68, 101389. [Google Scholar] [CrossRef]
- Samal, K.; Kar, S.; Trivedi, S. Ecological Floating Bed (EFB) for Decontamination of Polluted Water Bodies: Design, Mechanism and Performance. J. Environ. Manag. 2019, 251, 109550. [Google Scholar] [CrossRef] [PubMed]
- Maraci, Ö.; Corsini, M.; Antonatou-Papaioannou, A.; Jünemann, S.; Sudyka, J.; Di Lecce, I.; Caspers, B.A.; Szulkin, M. Changes to the Gut Microbiota of a Wild Juvenile Passerine in a Multidimensional Urban Mosaic. Sci. Rep. 2022, 12, 6872. [Google Scholar] [CrossRef] [PubMed]
- Berg, M.; Koskella, B. Nutrient- and Dose-Dependent Microbiome-Mediated Protection against a Plant Pathogen. Curr. Biol. 2018, 28, 2487–2492.e3. [Google Scholar] [CrossRef] [PubMed]
- Daisley, B.A.; Chernyshova, A.M.; Thompson, G.J.; Allen-Vercoe, E. Deteriorating Microbiomes in Agriculture—the Unintended Effects of Pesticides on Microbial Life. MRR 2022, 1, 6. [Google Scholar] [CrossRef]
- Dechartres, J.; Pawluski, J.L.; Gueguen, M.M.; Jablaoui, A.; Maguin, E.; Rhimi, M.; Charlier, T.D. Glyphosate and glyphosate-based herbicide exposure during the peripartum period affects maternal brain plasticity, maternal behaviour and microbiome. Journ Neuroend 2019, 31, e12731. [Google Scholar] [CrossRef]
- Ruuskanen, S.; Rainio, M.J.; Gómez-Gallego, C.; Selenius, O.; Salminen, S.; Collado, M.C.; Saikkonen, K.; Saloniemi, I.; Helander, M. Glyphosate-Based Herbicides Influence Antioxidants, Reproductive Hormones and Gut Microbiome but Not Reproduction: A Long-Term Experiment in an Avian Model. Environ. Pollut. 2020, 266, 115108. [Google Scholar] [CrossRef]
- Puigbò, P.; Leino, L.I.; Rainio, M.J.; Saikkonen, K.; Saloniemi, I.; Helander, M. Does Glyphosate Affect Hum. Microbiota? Life 2022, 12, 707. [Google Scholar] [CrossRef]
- Watterson, A. Artificial Turf: Contested Terrains for Precautionary Public Health with Particular Reference to Europe? Int. J. Environ. Res. Public Health 2017, 14, 1050. [Google Scholar] [CrossRef]
- Keller, M.; Turco, R.F.; Gray, M.B.; Sigler, V. The Fate of Methicillin-Resistant Staphylococcus Aureus in a Synthetic Turf System. Sport. Health 2020, 12, 263–270. [Google Scholar] [CrossRef]
- Mutiara, M.M.; Rachmawati, E.; Sunkar, A. Effectivity Assessment of Interpretive Signs for Biodiversity Conservation. IOP Conf. Ser. Earth Environ. Sci. 2021, 739, 012066. [Google Scholar] [CrossRef]
- Robinson, J.M.; Cameron, R.; Jorgensen, A. Germaphobia! Does Our Relationship With and Knowledge of Biodiversity Affect Our Attitudes Toward Microbes? Front. Psychol. 2021, 12, 678752. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Deng, Q. The Basic Roles of Indoor Plants in Human Health and Comfort. Env. Sci. Pollut. Res. 2018, 25, 36087–36101. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, T.; Ahmad, D.; Serey, N.; Jouhara, H. Review of Ventilation Strategies to Reduce the Risk of Disease Transmission in High Occupancy Buildings. Int. J. Thermofluids 2020, 7–8, 100045. [Google Scholar] [CrossRef]
- Benjamin, D. Adaptation: Bio-Receptive Materials with a New Outlook on Performativity and Sustainability. In The Routledge Companion to Paradigms of Performativity in Design and Architecture; Routledge: Abingdon, UK, 2019; pp. 362–369. [Google Scholar]
- Keniger, L.E.; Gaston, K.J.; Irvine, K.N.; Fuller, R.A. What are the Benefits of Interacting with Nature? Int. J. Environ. Res. Public Health 2013, 10, 913–935. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.F.; Kuo, F.E. Is contact with nature important for healthy child development? State of the evidence. In Children and Their Environments: Learning, Using and Designing Spaces; Spencer, C., Blades, M., Eds.; Cambridge University Press: Cambridge, UK, 2006; pp. 124–158. [Google Scholar] [CrossRef]
- Dadvand, P.; Nieuwenhuijsen, M.J.; Esnaola, M.; Forns, J.; Basagaña, X.; Alvarez-Pedrerol, M.; Rivas, I.; López-Vicente, M.; De Castro Pascual, M.; Su, J.; et al. Green Spaces and Cognitive Development in Primary Schoolchildren. Proc. Natl. Acad. Sci. USA 2015, 112, 7937–7942. [Google Scholar] [CrossRef]
- Richardson, E.A.; Pearce, J.; Shortt, N.K.; Mitchell, R. The role of public and private natural space in children’s social, emotional and behavioural development in Scotland: A longitudinal study. Environ. Res. 2017, 158, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Barrable, A.; Booth, D. Nature Connection in Early Childhood: A Quantitative Cross-Sectional Study. Sustainability 2020, 12, 375. [Google Scholar] [CrossRef]
- Sandseter, E.B.H. Affordances for Risky Play in Preschool: The Importance of Features in the Play Environment. Early Child. Educ. J. 2009, 36, 439–446. [Google Scholar] [CrossRef]
- Skelton, K.R.; Lowe, C.; Zaltz, D.A.; Benjamin-Neelon, S.E. Garden-Based Interventions and Early Childhood Health: An Umbrella Review. Int. J. Behav. Nutr. Phys. Act. 2020, 17, 121. [Google Scholar] [CrossRef]
- Early Childhood Development and Health. Available online: https://www.unicef.org/serbia/en/early-childhood-development-and-health (accessed on 10 December 2022).
- Nagle, G.A.; Usry, L.R. Using Public Health Strategies to Shape Early Childhood Policy. Am. J. Orthopsychiatry 2016, 86, 171–178. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Population Health and Public Health Practice; Committee on Applying Neurobiological and Socio-Behavioral Sciences from Prenatal Through Early Childhood Development: A Health Equity Approach; Negussie, Y.; Geller, A.; DeVoe, J.E. (Eds.) Promoting Health Equity Through Early Care and Education; National Academies Press (US): Washington, DC, USA, 2019. [Google Scholar]
- Woolner, P. Building Schools for the Future through a participatory design process: Exploring the issues and investigating ways forward. BERA 2009. Available online: https://www.ncl.ac.uk/media/wwwnclacuk/cflat/files/Woolner2009Building%20Schools%20for%20the%20Future%20through%20a%20participatory%20design%20process.pdf (accessed on 15 February 2023).
- Building Better Schools: Investing in Scotland’s Future; ANNEX E Sources of Further Information. Available online: http://www.gov.scot/publications/building-better-schools-investing-scotlands-future/pages/14/ (accessed on 10 December 2022).
- Matwiejczyk, L.; Mehta, K.; Scott, J.; Tonkin, E.; Coveney, J. Characteristics of Effective Interventions Promoting Healthy Eating for Pre-Schoolers in Childcare Settings: An Umbrella Review. Nutrients 2018, 10, 293. [Google Scholar] [CrossRef] [PubMed]
- One Health Basics | One Health | CDC. Available online: https://www.cdc.gov/onehealth/basics/index.html (accessed on 10 December 2022).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robinson, J.M.; Barrable, A. Optimising Early Childhood Educational Settings for Health Using Nature-Based Solutions: The Microbiome Aspect. Educ. Sci. 2023, 13, 211. https://doi.org/10.3390/educsci13020211
Robinson JM, Barrable A. Optimising Early Childhood Educational Settings for Health Using Nature-Based Solutions: The Microbiome Aspect. Education Sciences. 2023; 13(2):211. https://doi.org/10.3390/educsci13020211
Chicago/Turabian StyleRobinson, Jake M., and Alexia Barrable. 2023. "Optimising Early Childhood Educational Settings for Health Using Nature-Based Solutions: The Microbiome Aspect" Education Sciences 13, no. 2: 211. https://doi.org/10.3390/educsci13020211
APA StyleRobinson, J. M., & Barrable, A. (2023). Optimising Early Childhood Educational Settings for Health Using Nature-Based Solutions: The Microbiome Aspect. Education Sciences, 13(2), 211. https://doi.org/10.3390/educsci13020211








