Respiration Measurement in a Simulated Setting Incorporating the Internet of Things
Abstract
1. Introduction
1.1. The IoT in Healthcare
- Application layer: account hijacking, ransomware and brute force
- Middleware layer: cross-sire request forgery and scripting, session hijacking
- Network layer: eavesdropping, replay, access, denial of service
- Perception layer: device tampering
1.2. Respiration Rate
2. IoT for the Neonatal Intensive Care Unit (NICU)
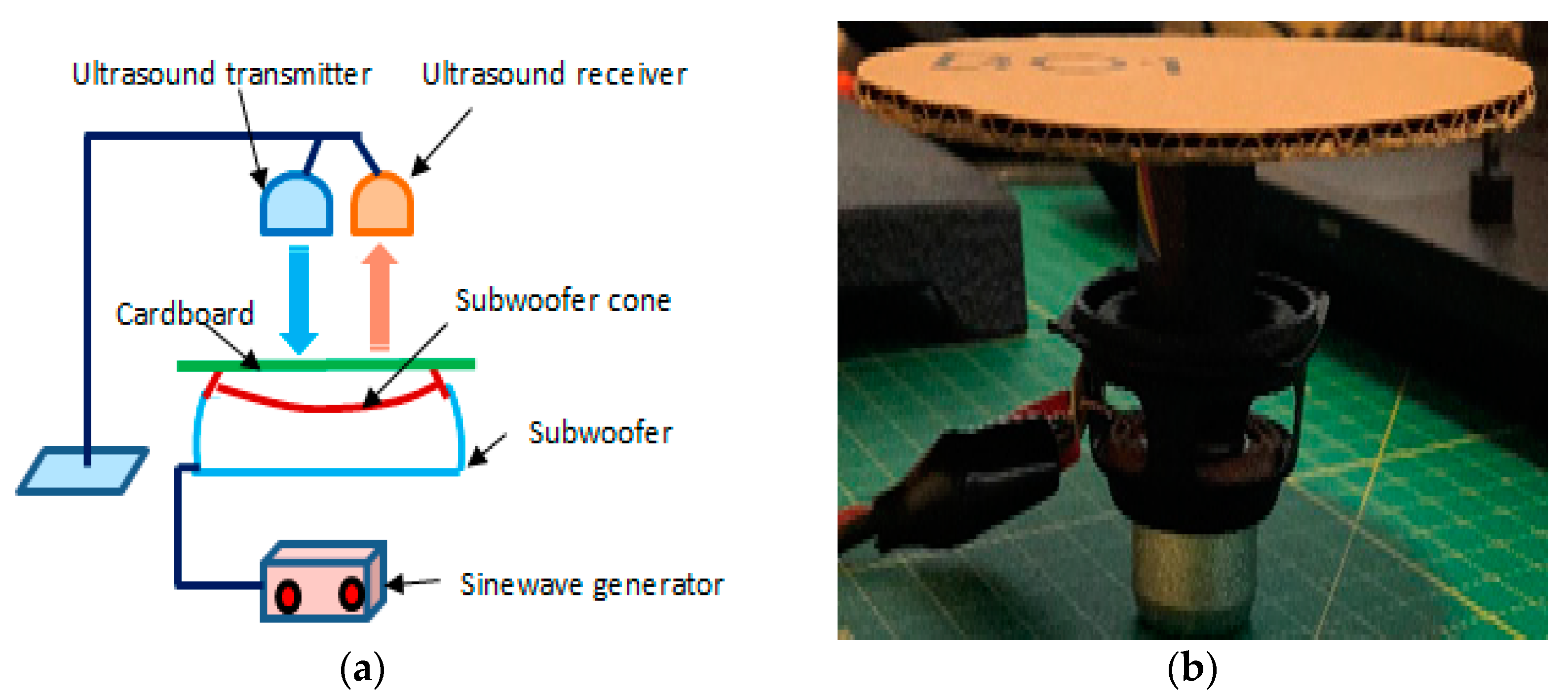
3. Set-Up for the Simulated Respiration Rate Measurement
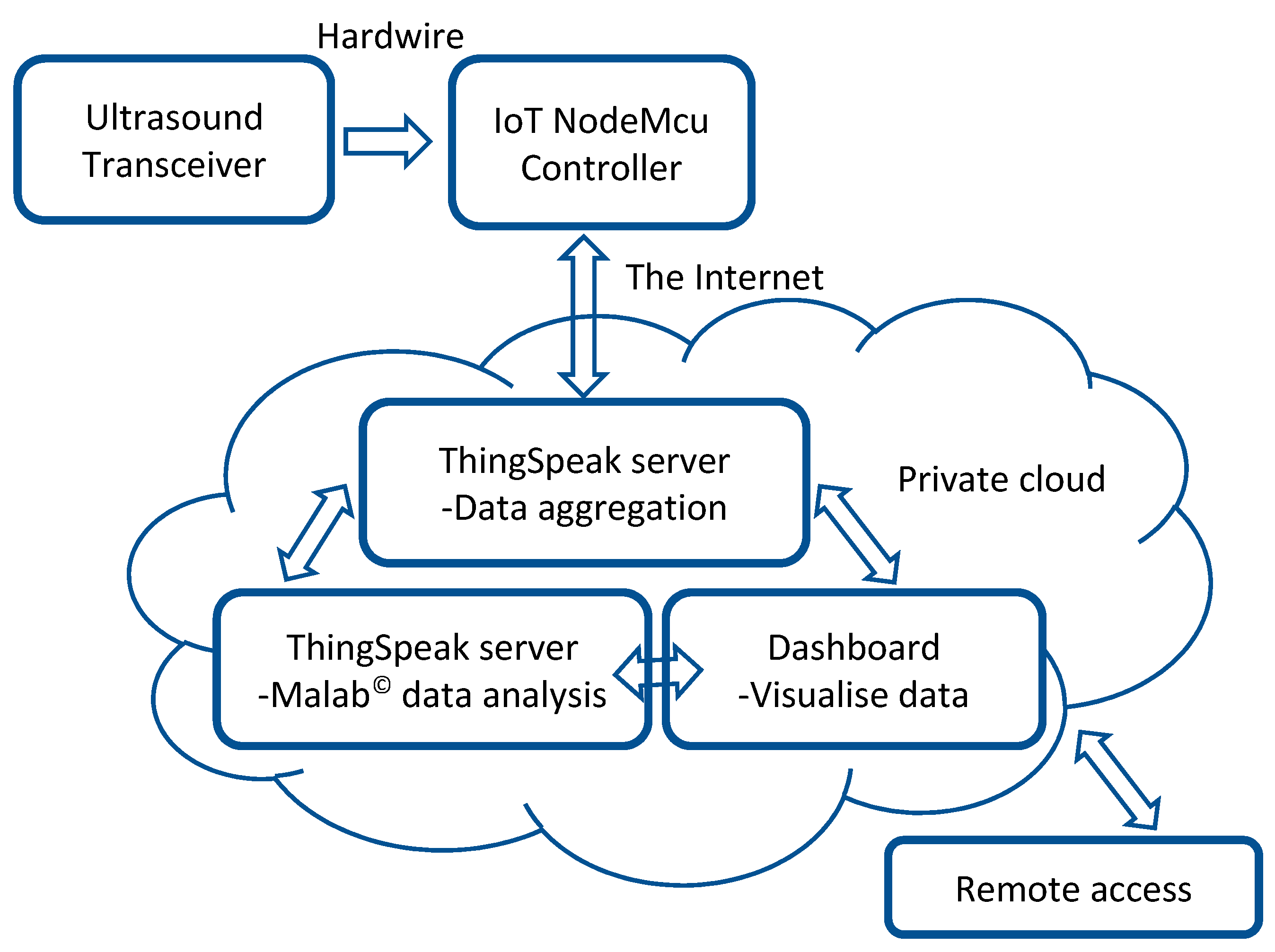
4. Integration of IoT into the Simulated Respiration Rate Measurement System
- Receiving the US signal reflected from the surface being monitored.
- Real-time wireless US data transmission to the ThingSpeak server.
- Data aggregation and analysis that included respiration rate calculation through the ThingSpeak server.
- Displaying the results through the ThingSpeak server with the aid of integrated Matlab© software.
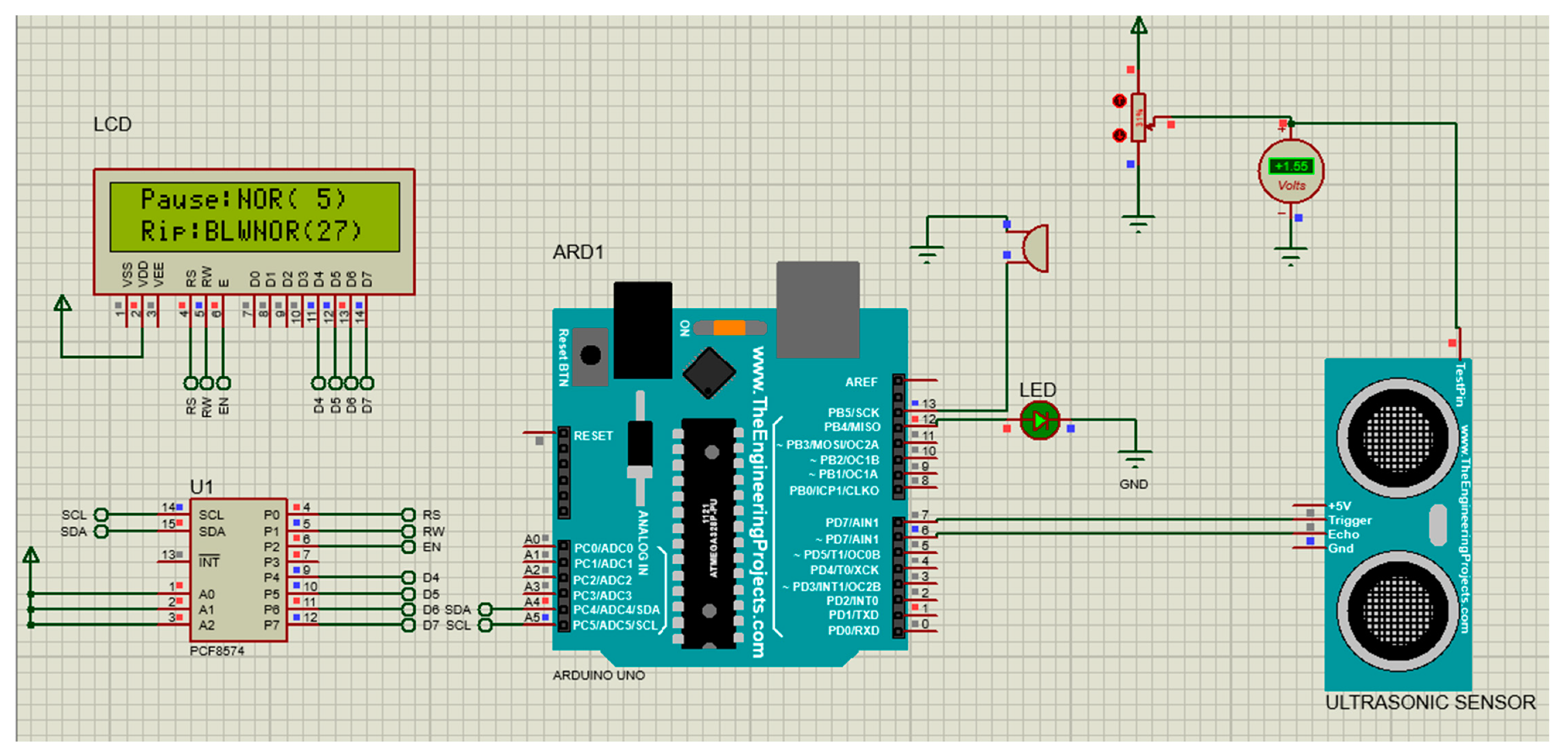
- US transceiver—The type of US transceiver was HC-SR04. This is a 4-pin module, whose pin names are supply voltage (Vcc), trigger, echo and ground. It provides 2 to 400 cm noncontact distance measurement. Its distance measurement accuracy can be 3 mm. The module contains US transmitter, receiver and control circuits. It operates by automatically sending eight 40 kHz signals for its measurements.
- ESP8266 NodeMCU—This is an IoT device that connects to an active wireless network. It was used to record the US signal and wirelessly transmit it to the ThingSpeak for processing over the cloud. NodeMCU connects an active wireless network for data transmission to the ThingSpeak. It is an open-source IoT platform. It has a firmware which runs on ESP8266, an integrated Wi-Fi chip, a power amplifier, power management modules, antenna switches and filters.
- ThingSpeak—This is an open-source web-based IoT analytics platform that allows users to aggregate data, analyzes them using web-integrated Matlab© and visualizes live data streams in the cloud. It allows users to create channels like YouTube or television [59]. The data can be analyzed and visualized using different ThingSpeak applications based on packages such as Matlab©. Matlab© was used for data analysis in this study. ThingSpeak has designed a secured system for user data protection. Each ThingSpeak channel has its own unique and secure read and write Application Programming Interface (API) keys.
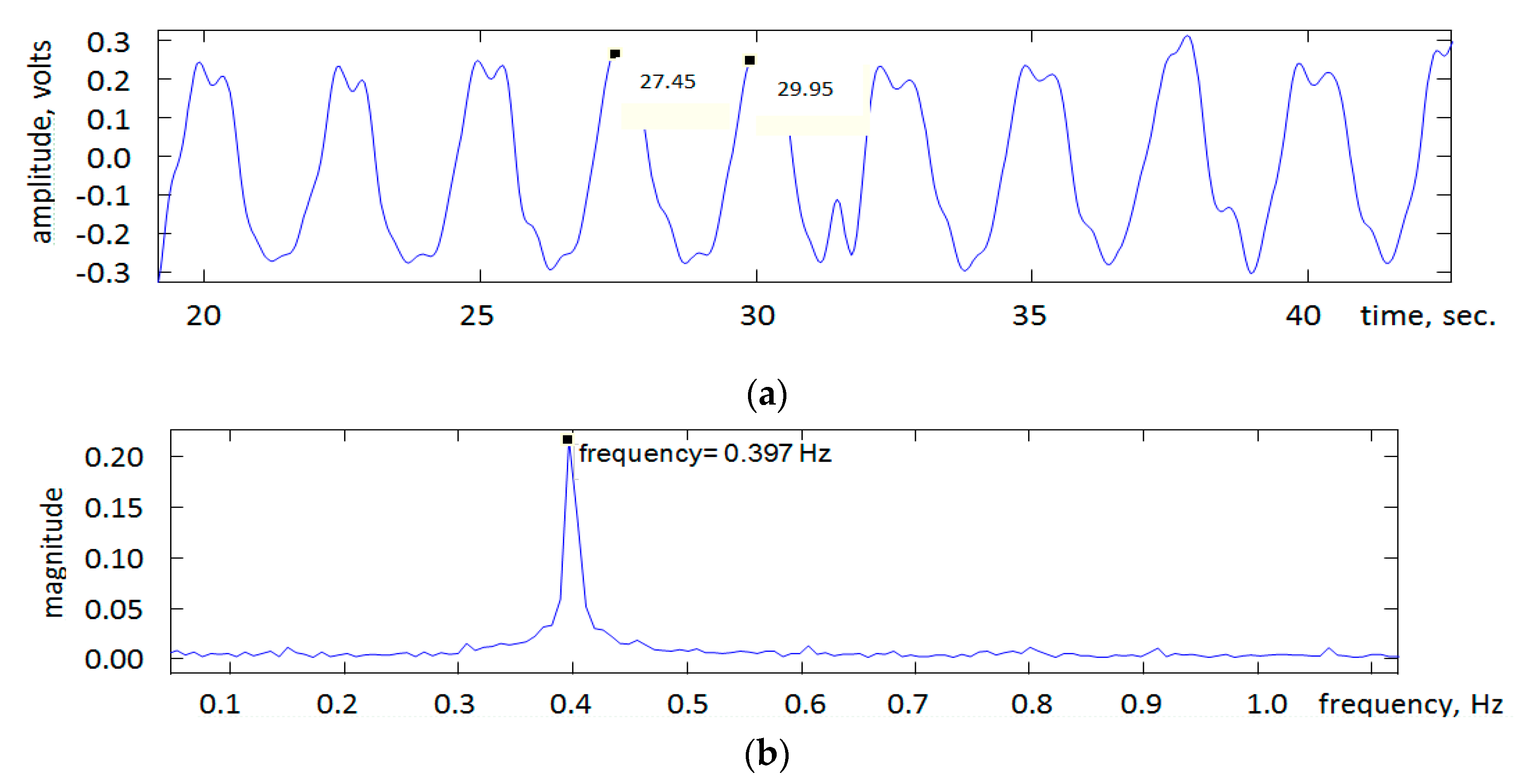
5. Results and Discussion
- Accuracy of the frequency setting on the signal generator.
- Accuracy of manually reading the frequency associated with the peak in the magnitude frequency spectrum.
- Level of accuracy provided by the US transceiver.
- Tracking: The US-based RR measurement method works while the baby’s chest (or abdomen if moving due to respiration) remains within the US field of view. If the baby moves, repositioning of the US transceiver will be needed. This repositioning requires the position of the baby to be tracked. A number of tracking algorithms were reported that may be considered. A relatively simple tracking approach is template matching. In this approach, the template of the section being tracked is segmented from the first image in the video and correlation is used to identify the section in the following images [60]. Another approach is the Kanade-Lucas-Tomasi (KLT) feature tracker [61] that looks for unique feature points in a region of interest and then tracks these points in subsequent images. The tracking algorithm requires a camera. An infrared thermal camera allows the baby to be viewed without reliance on background light, but it is more expensive than a visual camera.
- Incorporation into neonatal incubators: This requires design considerations that encompass the infants, medical practitioners and family members [62].
- Integration into smart hospital infrastructure: This will allow clinicians to be alerted if an anomaly in the child’s breathing is detected through warning messages sent to the clinicians’ digital devices [63].
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ibarra-Esquer, J.E.; González-Navarro, F.F.; Flores-Rios, B.L.; Burtseva, L.; Astorga-Vargas, M.A. Tracking the evolution of the Internet of Things concept across different application domains. Sensors 2017, 17, 1379. [Google Scholar] [CrossRef]
- Chen, S.; Xu, H.; Liu, D.; Hu, B.; Wang, H. A vision of IoT: Applications, challenges, and opportunities with china perspective. IEEE Internet Things J. 2014, 1, 349–359. [Google Scholar] [CrossRef]
- Kang, Y.-M.; Han, M.-R.; Han, K.-S.; Kim, J.-B. A study on the Internet of Things (IoT) applications. Int. J. Softw. Eng. Its Appl. 2015, 9, 117–126. [Google Scholar] [CrossRef]
- Kumar, S.; Tiwari, P.; Zymbler, M. Internet of Things is a revolutionary approach for future technology enhancement: A review. J. Big Data 2019, 6, 1–21. [Google Scholar] [CrossRef]
- Miorandi, D.; Sicari, S.; De Pellegrini, F.; Chlamtac, I. Internet of things: Vision, applications and research challenges. J. Ad Hoc Netw. 2012, 10, 1497–1516. [Google Scholar] [CrossRef]
- Joyia, J.G.; Liaqat, R.M.; Farooq, F.; Rehman, S. Internet of medical things (IOMT): Applications, benefits and future challenges in healthcare domain. J. Commun. 2017, 12, 240–247. [Google Scholar] [CrossRef]
- Dimitrov, D.V. Medical Internet of Things and Big Data in Healthcare. Health Inform. Res. 2016, 22, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Baker, S.B.; Xiang, W.; Atkinson, I. Internet of Things for Smart Healthcare: Technologies, Challenges, and Opportunities. IEEE Access 2017, 5, 26521–26544. [Google Scholar] [CrossRef]
- Maksimović, M.; Vujović, V.; Perišić, B. Do it yourself solution of Internet of Things healthcare system: Measuring body parameters and environmental parameters affecting health. J. Inf. Syst. Eng. Manag. 2016, 1, 25–39. [Google Scholar] [CrossRef]
- Liu, X.; Jia, M.; Zhang, X.; Lu, W. A novel multichannel Internet of things based on dynamic spectrum sharing in 5G communication. IEEE Internet Things J. 2018, 6, 5962–5970. [Google Scholar] [CrossRef]
- Brito, J.M. Technological trends for 5G networks influence of e-health and IoT applications. Int. J. Health Med. Commun. 2018, 9, 1–22. [Google Scholar] [CrossRef]
- Ahad, A.; Tahir, M.; Sheikh, M.A.; Ahmed, K.I.; Mughees, A.; Numani, A. Technologies trend towards 5G network for smart health-care using IoT: A review. Sensors 2020, 20, 4047. [Google Scholar] [CrossRef] [PubMed]
- Vijayalakshmi, A.W.; Arockiam, L. A secured architecture for IoT healthcare system. In Proceeding of the International Conference on Computer Networks, Big Data and IoT (ICCBI-2018). ICCBI 2018. Lecture Notes on Data Engineering and Communications Technologies; Pandian, A., Senjyu, T., Islam, S., Wang, H., Eds.; Springer International Publishing: Berlin, Germany, 2020; pp. 904–911. [Google Scholar] [CrossRef]
- Kelly, T.J.; Campbell, K.L.; Gong, E.; Scuffham, P. The Internet of Things: Impact and implications for health care delivery. J. Med. Internet Res. 2020, 22, e20135. [Google Scholar] [CrossRef] [PubMed]
- Darwish, A.; Hassanien, A.E.; Elhoseny, M.; Sangaiah, A.K.; Muhammad, K. The impact of the hybrid platform of internet of things and cloud computing on healthcare systems: Opportunities, challenges, and open problems. J. Ambient. Intell. Humaniz. Comput. 2019, 10, 4151–4166. [Google Scholar] [CrossRef]
- Miller, D.D.; Brown, E.W. Artificial Intelligence in Medical Practice: The Question to the Answer? Am. J. Med. 2018, 131, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.; Chircu, A. IoT and AI in healthcare: A systematic literature review. Issues Inf. Syst. 2018, 19, 33–41. [Google Scholar]
- Uslu, B.Ç.; Okay, E.; Dursun, E. Analysis of factors affecting IoT-based smart hospital design. J. Cloud Comput. 2020, 9, 1–23. [Google Scholar] [CrossRef]
- Salama, A.; Saatchi, R. Evaluation of wirelessly transmitted video quality using a modular fuzzy logic system. Technologies 2019, 7, 67. [Google Scholar] [CrossRef]
- Salama, A.; Saatchi, R.; Burke, D. Fuzzy logic and regression approaches for adaptive sampling of multimedia traffic in wireless computer networks. Technologies 2018, 6, 24. [Google Scholar] [CrossRef]
- Salama, A.; Saatchi, R. Probabilistic classification of quality of service in wireless computer networks. ICT Express 2019, 5, 155–162. [Google Scholar] [CrossRef]
- Dogman, A.; Saatchi, R.; Al-Khayatt, S. Quality of service using a combination of fuzzy c-means and regression model. Int. J. Electron. Electr. Eng. 2012, 6, 58–65. [Google Scholar]
- Dogman, A.; Saatchi, R.; Al-Khayatt, S. Collaborative scheme to improve QOS in WLAN-wired networks. Mediterr. J. Comput. Netw. 2014, 10, 170–180. [Google Scholar]
- Zhang, H.; Li, J.; Wen, B.; Xun, Y.; Liu, J. Connecting intelligent things in smart hospitals using NB-IoT. IEEE Internet Things J. 2018, 5, 1550–1560. [Google Scholar] [CrossRef]
- Beyene, Y.D.; Jantti, R.; Ruttik, K.; Iraji, S. On the performance of narrow-band Internet of Things (NB-IoT). In Proceedings of the 2017 IEEE Wireless Communications and Networking Conference (WCNC), San Francisco, CA, USA, 19–22 March 2017; pp. 1–6. [Google Scholar]
- Zeadally, S.; Siddiqui, F.; Baig, Z.; Ibrahim, A. Smart Healthcare challenges and potential solutions using internet of things (IoT) and big data analysis. PSU Res. Rev. 2020, 4, 93–109. [Google Scholar]
- Atzori, L.; Iera, A.; Morabito, G. The internet of things: A survey. Comput. Netw. 2010, 54, 2787–2805. [Google Scholar] [CrossRef]
- Islam, S.M.R.; Kwak, D.; Kabir, H.; Hossain, M.; Kwak, K.-S. The internet of things for health care: A comprehensive survey. IEEE Access 2015, 3, 678–708. [Google Scholar] [CrossRef]
- Nasiri, S.; Sadoughi, F.; Tadayon, M.H.; Dehnad, A. Security requirements of internet of things-based healthcare system: A survey. Acta Inform. Med. 2019, 27, 253–258. [Google Scholar] [CrossRef]
- Tawalbeh, L.; Muheidat, F.; Tawalbeh, M.; Quwaider, M. IoT Privacy and security: Challenges and solutions. Appl. Sci. 2020, 10, 4102. [Google Scholar] [CrossRef]
- Eken, C.; Eken, H. Security threats and recommendation in IoT Healthcare. In Proceedings of the 9th ERROSIM and the 57th SIMs Oulu Finland, 12–16 September 2016; pp. 369–374. [Google Scholar] [CrossRef]
- Chen, K.; Zhang, S.; Li, Z.; Zhang, Y.; Beng, Q.; Ray, S.; Jin, Y. Internet-of-things security and vulnerabilities: Taxonomy, challenges, and practice. J. Hardw. Syst. Secur. 2018, 2, 97–110. [Google Scholar] [CrossRef]
- Kandasamy, K.; Srinivas, S.; Achuthan, K.; Rangan, V.P. IoT cyber risks: A holistic analysis of cyber risk assessment frameworks, risk vectors, and risk ranking process. EURASIP J. Inf. Secur. 2020, 8, 1–18. [Google Scholar] [CrossRef]
- Darwish, S.; Nouretdinov, I.; Wolthusen, S.D. Towards composable threat assessment for medical IoT (MIoT). Procedia Comput. Sci. 2017, 113, 627–632. [Google Scholar] [CrossRef]
- Maple, C. Security and privacy in the Internet of Things. J. Cyber Policy 2017, 2, 155–184. [Google Scholar] [CrossRef]
- Suhardi, A.R. A survey of security aspects for internet of things in healthcare. In Information Science and Applications (ICISA); Part of the Lecture Notes in Electrical Engineering book series (LNEE, Volume 376); Publisher Springer, 2016; pp. 1237–1247. [Google Scholar] [CrossRef]
- Alsubaei, F.; Abuhussein, A.; Shiva, S. Security and privacy in the Internet of Medical Things: Taxonomy and risk assessment. In Proceedings of the IEEE International Workshop on Networks of Sensors, Wearable, and Medical Devices, Singapore, 9 October 2017. [Google Scholar]
- Daw, W.; Kaur, R.; Delaney, M.; Elphick, H.E. Respiratory rate is an early predictor of clinical deterioration in children. Pediatr. Pulmonol. 2020, 55, 2041–2049. [Google Scholar] [CrossRef]
- Daw, W.; Kingshott, R.; Saatchi, R.; Durke, D.; Holloway, A.; Travis, J.; Evans, R.; Jones, A.; Hughes, B.; Elphick, H. Medical devices for measuring respiratory rate in children: A review. J. Adv. Biomed. Eng. Technol. 2016, 3, 21–27. [Google Scholar] [CrossRef]
- Al-Khalidi, F.Q.; Saatchi, R.; Burke, D.; Elphick, H.; Tan, S. Respiration rate monitoring methods: A review. Pediatr. Pulmonol. 2011, 46, 523–529. [Google Scholar] [CrossRef]
- Cretikos, M.A.; Bellomo, R.; Hillman, K.; Chen, J.; Finfer, S.; Flabouris, A. Respiratory rate: The neglected vital sign. Med. J. Aust. 2008, 188, 657–659. [Google Scholar] [CrossRef]
- Marjanovic, N.; Mimoz, O.; Guenezan, J. An easy and accurate respiratory rate monitor is necessary. J. Clin. Monit. 2020, 34, 221–222. [Google Scholar] [CrossRef] [PubMed]
- Hogan, J. Why don’t nurses monitor the respiratory rates of patients? Br. J. Nurs. 2006, 15, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Philip, K.; Richardson, R.; Cohen, M. Staff perceptions of respiratory rate measurement in a general hospital. Br. J. Nurs. 2013, 22, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C. Respiratory rate: Why measurement and recording are crucial. Nurs. Times 2018, 114, 23–24. [Google Scholar]
- Rolfe, S. The importance of respiratory rate monitoring. Br. J. Nurs. 2019, 28, 504–508. [Google Scholar] [CrossRef] [PubMed]
- BaHammam, A. Comparison of nasal prong pressure and thermistor measurements for detecting respiratory events during sleep. Respiration 2004, 71, 385–390. [Google Scholar] [CrossRef]
- Elphick, H.; Alkali, A.; Kingshott, R.; Burke, D.; Saatchi, R. Exploratory study to evaluate respiratory rate using a thermal imaging camera. Respiration 2019, 97, 205–212. [Google Scholar] [CrossRef]
- Alkali, A.; Saatchi, R.; Elphick, H.; Burke, D. Thermal image processing for real-time noncontact respiration rate monitoring. IET Circuits Devices Syst. 2017, 11, 142–148. [Google Scholar] [CrossRef]
- Al-Kalidi, F.; Elphick, H.; Saatchi, R.; Burke, D. Respiratory rate measurement in children using a thermal camera. Int. J. Sci. Eng. Res. 2015, 6, 1748–1756. [Google Scholar]
- Chu, M.; Nguyen, T.; Pandey, V.; Zhou, Y.; Pham, H.N.; Bar-Yoseph, R.; Radom-Aizik, S.; Jain, R.; Cooper, D.M.; Khine, M. Respiration rate and volume measurements using wearable strain sensors. NPJ Digit. Med. 2019, 2, 8. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.S.; Saatchi, R.; Elphick, H.; Burke, D. Real-time vision based respiration monitoring system. In Proceedings of the 2010 7th International Symposium on Communication Systems, Networks & Digital Signal Processing (CSNDSP 2010), Newcastle Upon Tyne, UK, 21–23 July 2010; pp. 770–774. [Google Scholar]
- Harer, M.; Moreno, M.A. What parents need to know about the neonatal intensive care unit. JAMA Pediatr. 2019, 173, 508. [Google Scholar] [CrossRef]
- van Vonderen, J.J.; Roest, A.A.W.; Siew, M.L.; Walther, F.J.; Hooper, S.B.; Te Pas, A.B. Measuring physiological changes during the transition to life after birth. Neonatology 2014, 105, 230–242. [Google Scholar] [CrossRef]
- Villarroel, M.; Chaichulee, S.; Jorge, J.; Davis, S.; Green, G.; Arteta, C.; Zisserman, A.; McCormick, K.; Watkinson, P.; Tarassenko, L. Non-contact physiological monitoring of preterm infants in the neonatal intensive care unit. NPJ Digit. Med. 2019, 2, 128. [Google Scholar] [CrossRef]
- Barson, C.; Saatchi, R.; Godbole, P.; Ramlakhan, S. Infrared thermal imaging to detect inflammatory intra-abdominal pathology in infants. WSEAS Trans. Boil. Biomed. 2020, 17, 82–98. [Google Scholar] [CrossRef]
- Gadekar, C.; Vaze, V.M. Context aware computing: IoT for neonatal health monitoring. Adv. Comput. Sci. Technol. 2017, 10, 53–62. [Google Scholar]
- Shakunthala, M.; Jasmin Banu, R.; Deepika, L.; Indu, L. Neonatal healthcare monitoring in incubator using IoT. Int. J. Electr. Electron. Data Commun. 2018, 6, 59–64. [Google Scholar]
- Pasha, S. Thingspeak based sensing and monitoring system for IoT with Matlab Analysis. Int. J. New Technol. Res. (IJNTR) 2016, 2, 19–23. [Google Scholar]
- Mei, X.; Ling, H. Robust visual tracking and vehicle classification via sparse representation. IEEE Trans. Pattern Anal. Mach. Learn. 2011, 33, 2259–2272. [Google Scholar]
- Zivkovic, Z.; van der Heijden, F. Improving the selection of feature points for tracking. Pattern Anal. Appl. 2004, 7, 1–17. [Google Scholar] [CrossRef]
- Ferris, T.K.; Shepley, M.M. The design of neonatal incubators: A systems-oriented, human-centered approach. J. Perinatol. 2013, 33, S24–S31. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Yang, W.; Le Grange, J.M.; Wang, P.; Huang, W.; Ye, Z. Smart healthcare: Making medical care more intelligent. Glob. Health J. 2019, 3, 61–65. [Google Scholar] [CrossRef]
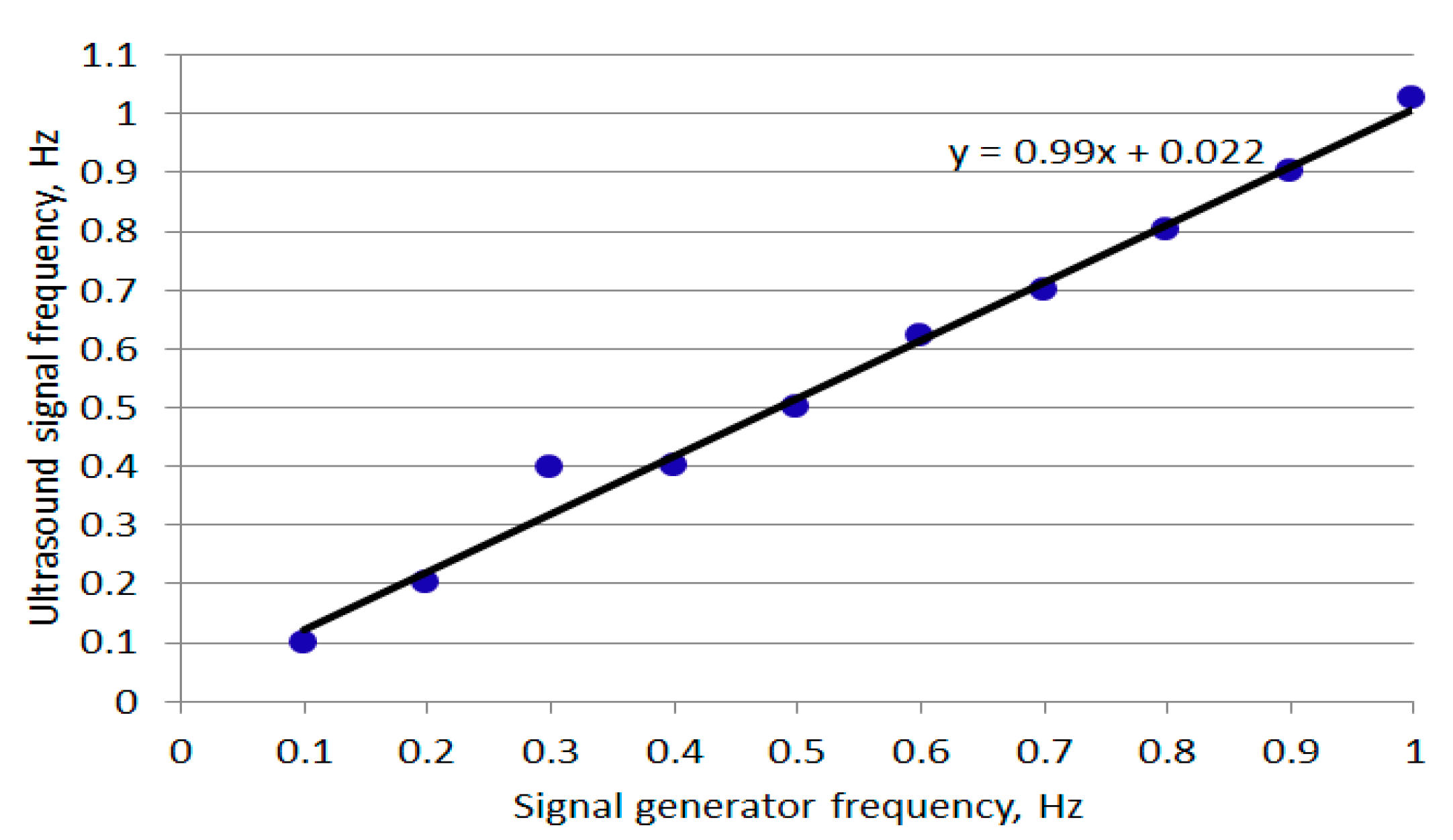
| Signal Generator Frequency, Hz, (S) | Ultrasound Signal Frequency, Hz, (U) | S-U |
|---|---|---|
| 0.1 | 0.100 | 0.000 |
| 0.2 | 0.201 | 0.001 |
| 0.3 | 0.397 | 0.097 |
| 0.4 | 0.399 | −0.001 |
| 0.5 | 0.500 | 0.000 |
| 0.6 | 0.622 | 0.022 |
| 0.7 | 0.699 | −0.001 |
| 0.8 | 0.801 | 0.001 |
| 0.9 | 0.901 | 0.001 |
| 1.0 | 1.026 | 0.026 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdulqader, T.; Saatchi, R.; Elphick, H. Respiration Measurement in a Simulated Setting Incorporating the Internet of Things. Technologies 2021, 9, 30. https://doi.org/10.3390/technologies9020030
Abdulqader T, Saatchi R, Elphick H. Respiration Measurement in a Simulated Setting Incorporating the Internet of Things. Technologies. 2021; 9(2):30. https://doi.org/10.3390/technologies9020030
Chicago/Turabian StyleAbdulqader, Tareq, Reza Saatchi, and Heather Elphick. 2021. "Respiration Measurement in a Simulated Setting Incorporating the Internet of Things" Technologies 9, no. 2: 30. https://doi.org/10.3390/technologies9020030
APA StyleAbdulqader, T., Saatchi, R., & Elphick, H. (2021). Respiration Measurement in a Simulated Setting Incorporating the Internet of Things. Technologies, 9(2), 30. https://doi.org/10.3390/technologies9020030






