Emergency Contraception Counseling in California Community Pharmacies: A Mystery Caller Study
Abstract
:1. Introduction
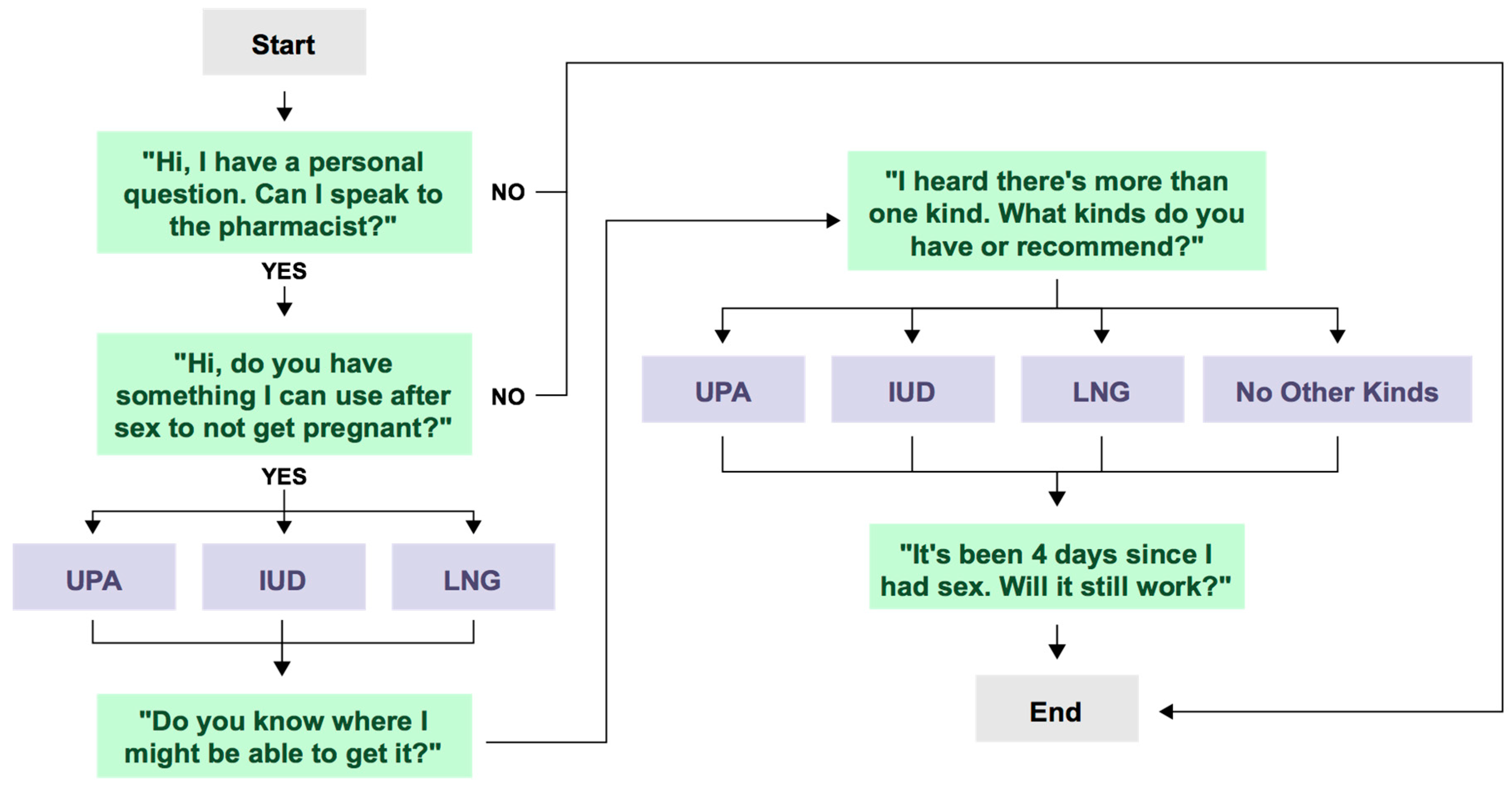
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Batur, P.; Cleland, K.; McNamara, M.; Wu, J.; Pickle, S.; EC Survey Group. Emergency contraception: A multispecialty survey of clinician knowledge and practices. Contraception 2015, 93, 145–152. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists. Committee Opinion No. 542: Access to emergency contraception. Obstet. Gynecol. 2012, 120, 1250–1253. [Google Scholar]
- Rafie, S.; Stone, R.H.; Wilkinson, T.A.; Borgelt, L.M.; El-Ibiary, S.Y.; Ragland, D. Role of the community pharmacist in emergency contraception counseling and delivery in the United States: Current trends and future prospects. Integr. Pharm. Res. Pract. 2017, 6, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Lalitkumar, P.; Berger, C.; Gemzell-Danielsson, K. Emergency contraception. Best Pract. Res. Clin. Endocrinol. Metab. 2013, 27, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Cleland, K.; Zhu, H.; Goldstuck, N.; Cheng, L.; Trussell, J. The efficacy of intrauterine devices for emergency contraception: A systematic review of 35 years of experience. Hum. Reprod. 2012, 27, 1994–2000. [Google Scholar] [CrossRef] [PubMed]
- Glasier, A.F.; Cameron, S.T.; Fine, P.M.; Logan, S.J.; Casale, W.; Van Horn, J.; Sogor, L.; Blithe, D.L.; Scherrer, B.; Mathe, H.; et al. Ulipristal acetate versus levonorgestrel for emergency contraception: A randomised non-inferiority trial and meta-analysis. Lancet 2010, 375, 555–562. [Google Scholar] [CrossRef]
- Kapp, N.; Abitbol, J.; Mathé, H.; Scherrer, B.; Guillard, H.; Gainer, E.; Ulmann, A. Effect of body weight and BMI on the efficacy of levonorgestrel emergency contraception. Contraception 2014, 91, 97–104. [Google Scholar] [CrossRef] [PubMed]
- American Society for Emergency Contraception. Providing Ongoing Hormonal Contraception after Use of Emergency Contraceptive Pills. September 2016. Available online: http://americansocietyforec.org/uploads/3/4/5/6/34568220/asec_fact_sheet-_hormonal_contraception_after_ec.pdf (accessed on 10 February 2019).
- California Board of Pharmacy. Emergency CONTRACEPTION Protocol. Available online: https://www.pharmacy.ca.gov/publications/ec_protocol.pdf (accessed on 28 March 2019).
- Brant, A.; White, K.; St Marie, P. Pharmacy availability of ulipristal acetate emergency contraception: An audit study. Contraception 2014, 90, 338–339. [Google Scholar] [CrossRef]
- Bullock, H.; Steele, S.; Kurata, N.; Tschann, M.; Elia, J.; Kaneshiro, B.; Salcedo, J. Pharmacy access to ulipristal acetate in Hawaii: Is a prescription enough? Contraception 2016, 93, 452–454. [Google Scholar] [CrossRef] [PubMed]
- Shigesato, M.; Elia, J.; Tschann, M.; Bullock, H.; Hurwitz, E.; Wu, Y.Y.; Salcedo, J. Pharmacy access to ulipristal acetate in major cities throughout the United States. Contraception 2018, 97, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, T.A.; Clark, P.; Rafie, S.; Carroll, A.E.; Miller, E. Access to emergency contraception after removal of age restrictions. Pediatrics 2017, 140, e20164262. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, T.A.; Rafie, S.; Clark, P.D.; Carroll, A.E.; Miller, E. Evaluating community pharmacy responses about levonorgestrel emergency contraception by mystery caller characteristics. J. Adolesc. Health 2018, 63, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Uysal, J.; Tavrow, P.; Hsu, R.; Alterman, A. Availability and accessibility of emergency contraception to adolescent callers in pharmacies in four Southwestern states. J. Adolesc. Health 2019, 64, 219–225. [Google Scholar] [CrossRef] [PubMed]
| Method(s) Discussed Initially | n (%) | Additional Method(s) Discussed after Probing | n (%) |
|---|---|---|---|
| Levonorgestrel and Ulipristal acetate | 4 (1.7%) | Levonorgestrel | – |
| Ulipristal acetate | – | ||
| Copper IUD | – | ||
| Unspecified | – | ||
| None | 4 (1.7%) | ||
| Levonorgestrel | 211 (88.3%) | Levonorgestrel and Ulipristal acetate | – |
| Ulipristal acetate | 48 (20.1%) | ||
| Copper IUD | 1 (0.4%) | ||
| Unspecified | – | ||
| None | 162 (67.8%) | ||
| Ulipristal acetate | 1 (0.4%) | Levonorgestrel and Ulipristal acetate | – |
| Levonorgestrel | – | ||
| Copper IUD | – | ||
| Unspecified | – | ||
| None | 1 (0.4%) | ||
| Copper IUD * | 0 | Levonorgestrel and Ulipristal acetate | – |
| Levonorgestrel | – | ||
| Ulipristal acetate | – | ||
| Unspecified | – | ||
| None | – | ||
| Unspecified ** | 4 (1.7%) | Levonorgestrel and Ulipristal acetate | 1 (0.4%) |
| Levonorgestrel | 1 (0.4%) | ||
| Ulipristal acetate | – | ||
| Copper IUD | – | ||
| None | 2 (0.8%) | ||
| None *** | 19 (7.9%) | Levonorgestrel and Ulipristal acetate | – |
| Levonorgestrel | – | ||
| Ulipristal acetate | – | ||
| Copper IUD | – | ||
| Unspecified | – | ||
| None | 19 (7.9%) |
| San Diego | San Francisco | p-Value | Chain | Independent | p-Value | |
|---|---|---|---|---|---|---|
| (n = 127) | (n = 112) | (n = 197) | (n = 42) | |||
| LNG Discussed | 113 (89%) | 103 (92%) | p = 0.434 | 191 (96.9%) | 25 (59.5%) | p < 0.001 * |
| UPA Discussed | 24 (18.9%) | 30 (26.8%) | p = 0.146 | 47 (23.9%) | 7 (16.7%) | p = 0.312 |
| LNG Available | 111 (87.4%) | 103 (92%) | p = 0.250 | 190 (96.4%) | 24 (57.1%) | p < 0.001 * |
| UPA Available | 10 (7.9%) | 13 (11.6%) | p = 0.329 | 22 (11.1%) | 1 (2.4%) | p = 0.080 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ditmars, L.; Rafie, S.; Kashou, G.; Cleland, K.; Bayer, L.; Wilkinson, T.A. Emergency Contraception Counseling in California Community Pharmacies: A Mystery Caller Study. Pharmacy 2019, 7, 38. https://doi.org/10.3390/pharmacy7020038
Ditmars L, Rafie S, Kashou G, Cleland K, Bayer L, Wilkinson TA. Emergency Contraception Counseling in California Community Pharmacies: A Mystery Caller Study. Pharmacy. 2019; 7(2):38. https://doi.org/10.3390/pharmacy7020038
Chicago/Turabian StyleDitmars, Lindsay, Sally Rafie, Gellan Kashou, Kelly Cleland, Lisa Bayer, and Tracey A. Wilkinson. 2019. "Emergency Contraception Counseling in California Community Pharmacies: A Mystery Caller Study" Pharmacy 7, no. 2: 38. https://doi.org/10.3390/pharmacy7020038
APA StyleDitmars, L., Rafie, S., Kashou, G., Cleland, K., Bayer, L., & Wilkinson, T. A. (2019). Emergency Contraception Counseling in California Community Pharmacies: A Mystery Caller Study. Pharmacy, 7(2), 38. https://doi.org/10.3390/pharmacy7020038






