Diabetes Screening Through Community Pharmacies in England: A Cost-Effectiveness Study
Abstract
1. Introduction
2. Aim
3. Methods
3.1. Ethics Approval
3.2. Patient and Public Involvement
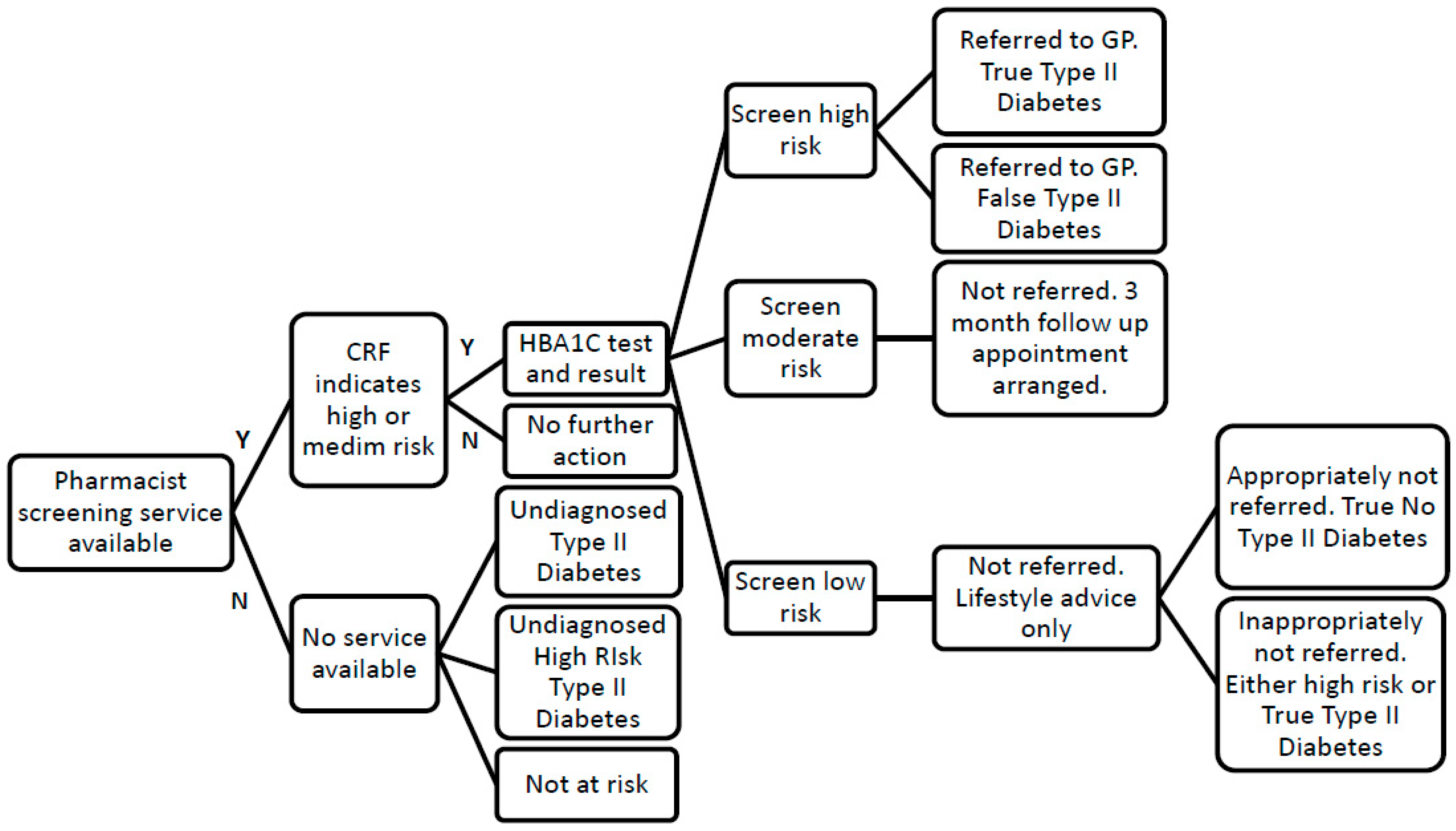
3.3. Service Data
- Completion of standardised type 2 diabetes screening customer service record form (CRF), consisting of a valid and reliable tool for diabetes screening [13] and questions required by the UK government for screening purposes, by the individual with support from a healthcare assistant (HCA) to estimate their risk level for diabetes.
- Signposting to further services as appropriate, e.g., weight-management, smoking cessation for individuals identified at low risk (score less than 16).
- Offering an HbA1c non-fasting finger prick blood test by the pharmacist for individuals determined to be at moderate or high risk of diabetes (score greater than 15)
- Providing a follow up pharmacist appointment in three months to individuals identified as being at high risk of diabetes via the HbA1C test (between 6.1% (43mmol/mol) and 6.4% (47mmol/mol)).
- Advising individuals identified as being likely to have type 2 diabetes via the HbA1C test (≥6.5% (48mmol/mol)) to see their GP for further testing and appropriate treatment.
- Communication of all HbA1c test results to the individual’s GP with their consent.
- Pharmacy location
- Age group, gender and ethnicity of patient
- Risk assessment score derived from the customer report form [22]
- HbA1c result
3.4. Effectiveness Modelling
3.5. Comparator
3.6. Costs of Pharmacy Led Screening and Cost-Effectiveness
3.7. Sensitivity Analysis
4. Results
5. Discussion
6. Strengths and Limitations
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Diabetes UK. Diabetes: Facts and Stats 2015 [updated May 2015]. Available online: https://www.mrc.ac.uk/documents/pdf/diabetes-uk-facts-and-stats-june-2015/ (accessed on 20 March 2019).
- Hex, N.; Bartlett, C.; Wright, D.; Taylor, M.; Varley, D. Estimating the current and future costs of Type 1 and Type 2 diabetes in the UK, including direct health costs and indirect societal and productivity costs. Diabet. Med. 2012, 29, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Diabetes UK. Diagnostic Criteria for Diabetes. Available online: https://www.diabetes.org.uk/Professionals/Position-statements-reports/Diagnosis-ongoing-management-monitoring/New_diagnostic_criteria_for_diabetes/ (accessed on 20 March 2019).
- National Institute for Health and Clinical Excellence. Preventing Type 2 Diabetes: Risk Identification and Interventions for Individuals at High Risk; National Institute for Health and Clinical Excellence: London, UK, 2012. [Google Scholar]
- Gillett, M.; Brennan, A.; Watson, P.; Khunti, K.; Davies, M.; Mostafa, S.; Gray, L.J. The cost-effectiveness of testing strategies for type 2 diabetes: A modelling study. Health Technol. Assess. (Winchester, England) 2015, 19, 1–80. [Google Scholar] [CrossRef]
- Stevens, J.W.; Khunti, K.; Harvey, R.; Johnson, M.; Preston, L.; Woods, H.B.; Davies, M.; Goyder, E. Preventing the progression to type 2 diabetes mellitus in adults at high risk: A systematic review and network meta-analysis of lifestyle, pharmacological and surgical interventions. Diabetes Res. Clin. Pract. 2015, 107, 320–331. [Google Scholar] [CrossRef] [PubMed]
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet (London, England) 1998, 352, 837–853. [Google Scholar] [CrossRef]
- Gillies, C.L.; Lambert, P.C.; Abrams, K.R.; Sutton, A.J.; Cooper, N.J.; Hsu, R.T.; Davies, M.J.; Khunti, K. Different strategies for screening and prevention of type 2 diabetes in adults: Cost effectiveness analysis. Br. Med. J. 2008, 336, 1180–1185. [Google Scholar] [CrossRef] [PubMed]
- Spigt, M.; Rikkers, A.; Doornbos, M.; Wouters, E.; Spitz, I.; Van Amelsvoort, L.; Zwietering, P. The effect of screening on the prevalence of diagnosed type 2 diabetes in primary care. Scand. J. Prim Health Care 2009, 27, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Simmons, R.K.; Hennings, S.H.; Wareham, N.J.; Griffin, S.J. How much does screening bring forward the diagnosis of type 2 diabetes and reduce complications? Twelve year follow-up of the Ely cohort. Diabetologia 2012, 55, 1651–1659. [Google Scholar] [CrossRef] [PubMed]
- Schaufler, T.; Wolff, M. Cost effectiveness of preventive screening programmes for type 2 diabetes mellitus in Germany (Structured abstract). Appl. Health Econom. Health Policy 2010, 8, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Hoerger, T.J.; Hicks, K.A.; Sorensen, S.W.; Herman, W.H.; Ratner, R.E.; Ackermann, R.T.; Zhang, P.; Engelgau, M.M. Cost-effectiveness of screening for pre-diabetes among overweight and obese US adults (Structured abstract). Diabetes Care 2007, 30, 2874–2879. [Google Scholar] [CrossRef] [PubMed]
- Gray, L.J.; Taub, N.A.; Khunti, K.; Gardiner, E.; Hiles, S.; Webb, D.R.; Srinivasan, B.T.; Davies, M.J. The Leicester Risk Assessment score for detecting undiagnosed Type 2 diabetes and impaired glucose regulation for use in a multiethnic UK setting. Diabet. Med. 2010, 27, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Krass, I.; Mitchell, B.; Clarke, P.; Brillant, M.; Dienaar, R.; Hughes, J.; Lau, P.; Peterson, G.; Stewart, K.; Taylor, S.; et al. Pharmacy diabetes care program: Analysis of two screening methods for undiagnosed type 2 diabetes in Australian community pharmacy. Diabetes Res. Clin. Pract. 2007, 75, 339–347. [Google Scholar] [CrossRef]
- Twigg, M.J.; Wright, D.J.; Thornley, T.; Haynes, L. Community pharmacy type 2 diabetes risk assessment: Demographics and risk results. Int. J. Pharm. Pract. 2015, 23, 80–82. [Google Scholar] [CrossRef] [PubMed]
- de Graaf, G.; Postmus, D.; Bakker, S.J.; Buskens, E. Design of stepwise screening for prediabetes and type 2 diabetes based on costs and cases detected. J. Clin. Epidemiol. 2015, 69, 1010–1018. [Google Scholar] [CrossRef] [PubMed]
- Jackson, B.R. The dangers of false-positive and false-negative test results: False-positive results as a function of pretest probability. Clin. Lab. Med. 2008, 28, 305–319. [Google Scholar] [CrossRef] [PubMed]
- van den Donk, M.; Sandbaek, A.; Borch-Johnsen, K.; Lauritzen, T.; Simmons, R.K.; Wareham, N.J.; Griffin, S.J.; Davies, M.J.; Khunti, K.; Rutten, G.E. Screening for type 2 diabetes. Lessons from the ADDITION-Europe study. Diabet. Med. 2011, 28, 1416–1424. [Google Scholar] [CrossRef] [PubMed]
- Willis, A.; Rivers, P.; Gray, L.J.; Davies, M.; Khunti, K. The effectiveness of screening for diabetes and cardiovascular disease risk factors in a community pharmacy setting. PLoS ONE 2014, 9, e91157. [Google Scholar] [CrossRef] [PubMed]
- Dhippayom, T.; Fuangchan, A.; Tunpichart, S.; Chaiyakunapruk, N. Opportunistic screening and health promotion for type 2 diabetes: An expanding public health role for the community pharmacist. J. Public Health (Oxf.) 2013, 35, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Fikri-Benbrahim, N.; Martínez-Martínez, F.; Saéz-Benito, L.; Luque, B.S.; Corpas, J.P.; Moullin, J.C.; Sabater-Hernández, D. Assessment of a screening protocol for type 2 diabetes in community pharmacy. The DiabNow Study. Diabetes Res. Clin. Pract. 2015, 108, e49–e52. [Google Scholar] [CrossRef] [PubMed]
- Tankova, T.; Chakarova, N.; Dakovska, L.; Atanassova, I. Assessment of HbA1c as a diagnostic tool in diabetes and prediabetes. Acta Diabetol. 2012, 49, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Webb, D.R.; Khunti, K.; Srinivasan, B.; Gray, L.; Jarvis, J.; Hiles, S. ADDITION Leicester: Prevalence of impaired glucose regulation (IGR) and screen detected Type 2 diabetes in a mixed ethnic UK population. Diabet. Med. 2009, 26 (Suppl. 1), 3. [Google Scholar]
- Wright, D.; Twigg, M.J.; Barton, G.; Thornley, T.; Kerr, C. An evluation of a multi-site community pharmacy-based chronic obstructive pulmonary disease support service. Int. J. Pharm. Pract. 2015, 23, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Drummond, M.F.; Sculpher, M.J.; Torrance, G.W.; O’Brien, B.J.; Stoddart, G.L. Methods for the Economic Evaluation of Health Care Programmes, 3rd ed.; Oxford University Press: New York, NY, USA, 2005. [Google Scholar]
- Curtis, L.; Burns, A. Unit Costs of Health and Social Care; Personal Social Services Research Unit., University of Kent: Canterbury, UK, 2017. [Google Scholar]
- Webb, D.R.; Gray, L.J.; Khunti, K.; Srinivasan, B.; Taub, N.; Campbell, S.; Barnett, J.; Farooqi, A.; Echouffo-Tcheugui, J.B.; Griffin, S.J.; et al. Screening for diabetes using an oral glucose tolerance test within a western multi-ethnic population identifies modifiable cardiovascular risk: The ADDITION-Leicester study. Diabetologia 2011, 54, 2237–2246. [Google Scholar] [CrossRef] [PubMed]
- Khunti, K.; Gillies, C.L.; Dallosso, H.; Brady, E.M.; Gray, L.J.; Kilgallen, G.; Willis, A.; Zafar, A.; Davies, M.J. Assessment of response rates and yields for Two opportunistic Tools for Early detection of Non-diabetic hyperglycaemia and Diabetes (ATTEND). A randomised controlled trial and cost-effectiveness analysis. Diabetes Res. Clin. Pract. 2016, 118, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Simoens, S.; De Coster, S.; Lenie, J.; Hayen, V.; Laekeman, G. Detecting pre-diabetes and the role of the pharmacist. Pharm. Pract. 2011, 9, 88–92. [Google Scholar] [CrossRef]
- Thomas, C.; Sadler, S.; Breeze, P.; Squires, H.; Gillett, M.; Brennan, A. Assessing the potential return on investment of the proposed UK NHS diabetes prevention programme in different population subgroups: An economic evaluation. BMJ Open 2017, 7, e014953. [Google Scholar] [CrossRef]
| Demographic | Proportion | ||
|---|---|---|---|
| Normal Glucose Tolerance # | High Risk of Diabetes | Type 2 Diabetes | |
| White (Leicester) [23] | 0.955 | 0.017 | 0.028 |
| Ethnic minorities (Leicester) [23] | 0.931 | 0.024 | 0.045 |
| White (Surrey) [1,5] | 0.909 | 0.074 | 0.017 |
| Ethnic minorities (Surrey) [1,5] | 0.919 | 0.024 * | 0.057 |
| Demographic | Leicester (n = 172) No. (%) | Surrey (n = 164) No. (%) |
|---|---|---|
| Female | 99 (57.2) | 110 (67.1) |
| White European | 98 (57) | 106 (64.6) |
| Age group | ||
| <50 | 93 (54.1) | 109 (66.5) |
| 50–59 | 27 (15.7) | 25 (15.2) |
| 60–69 | 22 (12.8) | 21 (12.8) |
| 70+ | 30 (17.4) | 9 (5.5) |
| CRF Test Results | (n = 172) | (n = 164) |
| Low risk (<16) | 108 (62.8) | 124 (76.6) |
| Medium to Hhgh risk (16+) | 64 (37.1) | 40 (24.4) |
| Available HbA1c Test Results | (n = 33) * | (n = 31) * |
| Suspected diabetes ≥6.5% (48mmol/mol) | 5 | 3 |
| High risk of-diabetes 6.1% (43mmol/mol) to 6.4% (47mmol/mol) | 5 | 5 |
| Unlikely to have diabetes ≤6% (42mmol/mol) | 23 | 23 |
| Predicted HbA1c Test Results | ||
| Suspected diabetes ≥6.5% (48mmol/mol) | 9 # (5.2) | 4 # (2.4) |
| High risk of diabetes 6.1% (43mmol/mol) to 6.4% (47mmol/mol) | 9 # (5.2) | 6 # (3.9) |
| Unlikely to have diabetes ≤6% (42mmol/mol) | 45 # (26.2) | 30 # (18.1) |
| White Leicester | White Surrey | Ethnic Minority Leicester | Ethnic Minority Surrey | |
|---|---|---|---|---|
| Numbers (Proportions) | ||||
| CRF Questionnaire | ||||
| Total numbers | 97 | 134 | 75 | 30 |
| Screen High/Moderate Risk | 53.9 (0.556) | 76.4 (0.57) | 41.9 (0.558) | 16.8 (0.558) |
| True type 2 diabetes (T2D) | 0.33 (0.006) | 0.28 (0.004) | 0.41 (0.01) | 0.21 (0.013) |
| True high risk of diabetes | 1.34 (0.025) | 8.03 (0.105) | 1.46 (0.035) | 0.58 (0.035) |
| False positives | 52.2 (0.969) | 68.1 (0.891) | 40 (0.955) | 16 (0.953) |
| Screen Low Risk | 43.1 (0.444) | 57.6 (0.43) | 33.1 (0.442) | 13.2 (0.442) |
| True Negatives | 42.7 (0.991) | 55.7 (0.966) | 32.7 (0.987) | 13.1 (0.986) |
| False Negatives – True T2D | 0.08 (0.002) | 0.07 (0.001) | 0.1 (0.003) | 0.05 (0.004) |
| False Negatives – High risk of diabetes | 0.31 (0.007) | 1.88 (0.033) | 0.34 (0.01) | 0.14 (0.01) |
| Assume all high/moderate risk from CRF receive HbA1c | ||||
| Numbers | 53.9 (0.556) | 76.4 (0.57) | 41.9 (0.558) | 16.8 (0.558) |
| HbA1c blood test +ve | 16.1 (0.298) | 23.9 (0.313) | 12.7 (0.303) | 5.1 (0.304) |
| Screen High Risk | 4.46 (0.278) | 5.68 (0.238) | 3.55 (0.281) | 1.46 (0.286) |
| TrueT2D | 0.29 (0.02) | 0.24 (0.01) | 0.36 (0.03) | 0.18 (0.04) |
| False Positives | 4.18 (0.26) | 5.44 (0.23) | 3.2 (0.25) | 1.28 (0.25) |
| Screen Moderate Risk | 11.6 (0.722) | 18.2 (0.762) | 9.1 (0.719) | 3.6 (0.714) |
| True high risk of-diabetes | 0.65 (0.041) | 3.94 (0.71) | 0.71 (0.29) | 0.29 (0) |
| False Positives | 11 (0.682) | 14.3 (8.4) | 8.4 (3.4) | 3.4 (0) |
| Cost Item | Total Cost | Cost Per Test | Assumption |
|---|---|---|---|
| Costs of providing CRF screening | |||
| Training (annual equivalent cost) | £55 | £0.74 | 73 people screened per year in each practice |
| Total marketing (including leaflets, posters, hanging cards) | £155 | £2.11 | |
| Printing: service/clinical/customer feedback forms | £26 | £0.35 | |
| Healthcare assistant (HCA) | £235 | £3.23 | |
| Total cost of CRF | £471 | £6.44 | |
| Cost of HbA1c test | 40.9 out of 73 individuals will receive HbA1c testing | ||
| HbA1c Analyser (annual equivalent cost) | £328 | £8.02 | |
| Internal quality control, annual (6 samples) | £72 | £1.76 | |
| External quality assurance, annual (6 samples) | £216 | £5.29 | |
| Ecoloc bins collection annual fee | £67 | £1.64 | |
| Pharmacist | £748 | £18.29 | |
| Consumables | £190 | £4.64 | |
| Total for HbA1c | £1621 | £39.64 | |
| Average cost per person screened in pharmacy | £28.65 | ||
| Costs 2017 £ Sterling | Leicester | Surrey | Total |
|---|---|---|---|
| Costs of CRF screening | £1109 | £1057 | £2166 |
| Costs of HbA1c test | £3795 | £3691 | £7486 |
| Total costs of Screening | £4903 | £4748 | £9651 |
| Number appropriately referred | 0.642 | 0.420 | 1.062 |
| Cost per appropriately referred individual | £7638 | £11,297 | £9086 |
| Cost per appropriately referred individual assuming 60% attend medical practice | £12,730 | £18,828 | £15,142 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wright, D.; Little, R.; Turner, D.; Thornley, T. Diabetes Screening Through Community Pharmacies in England: A Cost-Effectiveness Study. Pharmacy 2019, 7, 30. https://doi.org/10.3390/pharmacy7010030
Wright D, Little R, Turner D, Thornley T. Diabetes Screening Through Community Pharmacies in England: A Cost-Effectiveness Study. Pharmacy. 2019; 7(1):30. https://doi.org/10.3390/pharmacy7010030
Chicago/Turabian StyleWright, David, Richard Little, David Turner, and Tracey Thornley. 2019. "Diabetes Screening Through Community Pharmacies in England: A Cost-Effectiveness Study" Pharmacy 7, no. 1: 30. https://doi.org/10.3390/pharmacy7010030
APA StyleWright, D., Little, R., Turner, D., & Thornley, T. (2019). Diabetes Screening Through Community Pharmacies in England: A Cost-Effectiveness Study. Pharmacy, 7(1), 30. https://doi.org/10.3390/pharmacy7010030






