Review of Reduced-Order Models for Homogeneous CO2 Nucleation in Supersonic and Hypersonic Expansion Flows
Abstract
1. Introduction
2. Nucleation Theory
2.1. Classical Nucleation Theory
2.2. Self-Consistent Classical Nucleation Theory
2.3. Mean-Field Kinetic Nucleation Theory
2.4. Semiphenomenological Nucleation Theory
2.5. Extended Modified Liquid Drop Dynamical Nucleation Theory
2.6. Semi-Empirical Density Gradient Theory
2.7. Scaled Nucleation Rate Model
2.8. Nonisothermal Nucleation
2.9. Vibrational Nonequilibrium
3. Results and Discussion
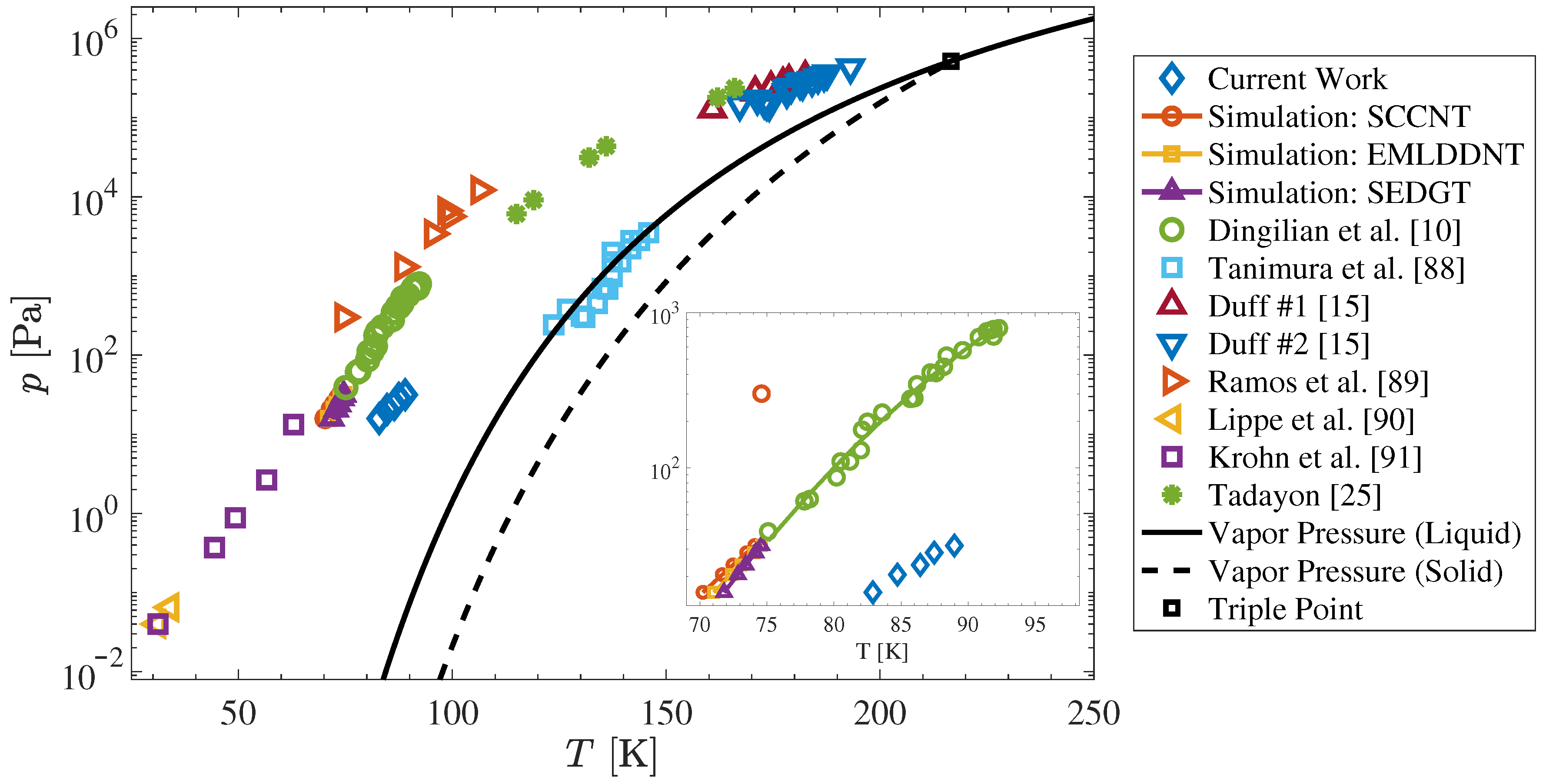
4. Experimental and Simulation Results
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| A | Temperature dependent variable in Equation (16) |
| b | Mean squared energy fluctation of impinging molecules |
| Second virial coefficient of the vapor | |
| c | Concentration (mole fraction) |
| Specific heat at constant volume per molecule of the vapor | |
| Specific heat at constant volume per molecule of the carrier gas | |
| d | Hard sphere diameter |
| e | Energy |
| Homogeneous free energy density | |
| Probability of cluster existing in EMLDDNT volume | |
| g | Density square gradient, , or condensate mass fraction |
| Condensate mass fraction with complete condensation | |
| h | Planck constant |
| Latent energy of phase change per molecule | |
| H | Unit step function |
| J | Steady state nucleation rate |
| Isothermal steady state nucleation rate | |
| Nonisothermal steady state nucleation rate | |
| Transient nucleation rate | |
| Boltzmann constant | |
| Mass per molecule | |
| M | Number of molecules |
| n | Number density of molecules |
| Number density of free molecules in the vapor | |
| Saturated liquid number density | |
| Number density of clusters with N molecules | |
| Equilibrium number density of clusters | |
| Saturated vapor number density | |
| N | Number of molecules in the cluster |
| Coordination number of the liquid | |
| Number of molecules in critical cluster | |
| Stagnation pressure or EMLDDNT pressure without cluster | |
| Vapor pressure within EMLDDNT volume | |
| Pressure of carrier gas | |
| Pressure of homogeneous fluid | |
| Hard sphere pressure | |
| SEDGT normal pressure | |
| Saturated vapor pressure | |
| Pressure of vapor | |
| SEDGT tangential pressure | |
| P | Average pressure in EMLDDNT volume |
| Total pressure within EMLDDNT volume | |
| q | Thermal energy released per condensing molecule |
| r | Radius of droplet or radius within droplet |
| Radius of molecule | |
| R | Radius of EMLDDNT volume or ideal gas constant |
| Saturated liquid surface area per molecule | |
| S | Saturation ratio, |
| T | Temperature |
| Critical point temperature | |
| Nondimensional temperature, | |
| Saturated liquid volume per molecule | |
| V | Volume |
| Z | Zel’dovich factor |
| Total integrated attractive potential | |
| Cluster evaporation rate | |
| Cluster impingement rate | |
| Impingement rate onto critical cluster | |
| Tolman length | |
| Total free energy within EMLDDNT closed volume | |
| Closed system free energy barrier of droplet with N molcules | |
| Free energy barrier of cluster with N molecules | |
| Free energy barrier of critical cluster | |
| Pressure difference between cluster and gas | |
| Residence time of freestream molecule | |
| SEDGT influence parameter correction factor, | |
| Nondimensional SEDGT influence parameter correction factor, | |
| Difference in chemical potential | |
| Difference in chemical potential of homogeneous fluid | |
| Hard sphere difference in chemical potential | |
| Lennard–Jones potential | |
| Packing fraction of hard spheres | |
| Isothermal compressibility of the liquid | |
| SEDGT influence parameter | |
| SEDGT infinite plane influence parameter | |
| Nondimensional SEDGT infinite plane influence parameter | |
| Nondimensional SEDGT influence parameter, | |
| Thermal de Broglie wavelength | |
| SNT variable (Equation (24)) | |
| Surface tension or Lennard-Jones zero energy distance | |
| Infinite plane surface tension | |
| Characteristic time for gas expansion | |
| Characteristic time for transient nucleation | |
| Inhomogeneous free energy density | |
| Eötvös constant |
Appendix A. Fluid Properties
References
- Lakebrink, M.T.; Bowcutt, K.G.; Winfree, T.; Huffman, C.C.; Juliano, T.J. Optimization of a Mach-6 Quiet Wind-Tunnel Nozzle. J. Spacecr. Rocket. 2018, 55, 315–321. [Google Scholar] [CrossRef]
- Daum, F.L.; Gyarmathy, G. Condensation of Air and Nitrogen in Hypersonic Wind Tunnels. Am. Inst. Aeronaut. Astronaut. 1967, 6, 458–465. [Google Scholar] [CrossRef]
- Faro, I.; Small, T.R.; Hill, F.K. The Supersaturation of Nitrogen in a Hypersonic Wind Tunnel. J. Appl. Phys. 1952, 23, 40–43. [Google Scholar] [CrossRef]
- Willmarth, W.W.; Nagamatsu, H.T. The Condensation of Nitrogen in a Hypersonic Nozzle. J. Appl. Phys. 1952, 23, 1089–1095. [Google Scholar] [CrossRef]
- Tans, P.; Keeling, R. Trends in Atmospheric Carbon Dioxide; NOAA Global Monitoring Laboratory. Available online: https://gml.noaa.gov/ccgg/trends/data.html (accessed on 1 November 2021).
- Lax, P.A.; Leonov, S.B. Semiempirical Model for Homogeneous Nitrogen Condensation in Hypersonic Wind Tunnels. AIAA J. 2020, 58, 4807–4818. [Google Scholar] [CrossRef]
- Goglia, G.L. Limit of Supersaturation of Nitrogen Vapor Expanding in a Nozzle. Ph.D. Thesis, University of Michigan, Ann Arbor, MI, USA, 1959. [Google Scholar]
- Zahoransky, R.A. Nitrogen Nucleation in an Unsteady Supersonic Flow Field. Z. Für Flugwiss. Und Weltraumforsch. 1986, 10, 34–37. [Google Scholar]
- Steinwandel, J. Homogeneous Condensation of Nitrogen in the Expansion Wave of a Cryogenic Shock Tube. Berichte Bunsenges. Phys. Chem. 1985, 89, 481–484. [Google Scholar] [CrossRef]
- Dingilian, K.K.; Halonen, R.; Tikkanen, V.; Reischl, B.; Vehkamäki, H.; Wyslouzil, B.E. Homogeneous nucleation of carbon dioxide in supersonic nozzles I: Experiments and classical theories. Phys. Chem. Chem. Phys. 2020, 22, 19282–19298. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.E. Nucleation and Growth of CO2 Ice Crystals in the Martian Atmosphere. Ph.D. Thesis, University of California Los Angeles, Los Angeles, CA, USA, 1999. [Google Scholar]
- Brown, K.W. Coherent Raman Spectroscopy of Non-Polar Molecules and Molecular Clusters. Ph.D. Thesis, Oregon State University, Corvallis, OR, USA, 1991. [Google Scholar]
- Mayer, S.G. Size Estimates of Molecular Clusters Using Elastic Light Scattering and CARS Spectroscopy. Ph.D. Thesis, Oregon State University, Corvallis, OR, USA, 1997. [Google Scholar]
- Erbland, P.J.; Rizzetta, D.P.; Miles, R.B. Numerical and Experimental Investigation of CO2 Condensate Behavior in Hypersonic Flow. In Proceedings of the 21st AIAA Aerodynamic Measurement Technology and Ground Testing Conference, Denver, CO, USA, 19–22 June 2000. Paper No. 2000-2379. [Google Scholar] [CrossRef]
- Duff, K.M. Non-Equilibrium Condensation of Carbon Dioxide in Supersonic Nozzles. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 1966. [Google Scholar]
- Ozawa, T.; Suzuki, T.; Fujita, K. Investigation of Condensation Effect in CO2 Hypersonic Rarefied Flows. In Proceedings of the 54th AIAA Aerospace Sciences Meeting, San Diego, CA, USA, 4–8 January 2000. Paper No. 2016-1729. [Google Scholar] [CrossRef]
- Kumar, R.; Li, Z.; Levin, D.A. Modeling of carbon dioxide condensation in the high pressure flows using the statistical BGK approach. Phys. Fluids 2011, 23, 052001. [Google Scholar] [CrossRef]
- Horsch, M.; Lin, Z.; Windmann, T.; Hasse, H.; Vrabec, J. The air pressure effect on the homogeneous nucleation of carbon dioxide by molecular simulation. Atmos. Res. 2011, 101, 519–526. [Google Scholar] [CrossRef]
- Horsch, M.; Vrabec, J.; Bernreuther, M.; Grottel, S.; Reina, G.; Wix, A.; Schaber, K.; Hasse, H. Homogeneous nucleation in supersaturated vapors of methane, ethane, and carbon dioxide predicted by brute force molecular dynamics. J. Chem. Phys. 2008, 128, 164510. [Google Scholar] [CrossRef]
- Li, Z.; Zhong, J.; Levin, D.A. Modeling of CO2 Homogeneous and Heterogeneous Condensation Plumes. J. Phys. Chem. C 2010, 114, 5276–5286. [Google Scholar] [CrossRef]
- Hale, B.N. Temperature dependence of homogeneous nucleation rates for water: Near equivalence of the empirical fit of Wölk and Strey, and the scaled nucleation model. J. Chem. Phys. 2005, 122, 204509. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.K.; Kawamura, K.; Tanaka, H.; Nakazawa, K. Tests of the homogeneous nucleation theory with molecular-dynamics simulations. I. Lennard-Jones molecules. J. Chem. Phys. 2005, 122, 184514. [Google Scholar] [CrossRef] [PubMed]
- Merikanto, J.; Zapadinsky, E.; Lauri, A.; Vehkamäki, H. Origin of the Failure of Classical Nucleation Theory: Incorrect Description of the Smallest Clusters. Phys. Rev. Lett. 2007, 98, 145702. [Google Scholar] [CrossRef] [PubMed]
- Holten, V.; Labetski, D.G.; van Dongen, M.E.H. Homogeneous nucleation of water between 200 and 240 K: New wave tube data and estimation of the Tolman length. J. Chem. Phys. 2005, 123, 104505. [Google Scholar] [CrossRef]
- Tadayon, P. Determination of Interfacial Tension from Optical Measurements of Nucleation Rates. Ph.D. Thesis, Oregon State University, Corvallis, OR, USA, 1998. [Google Scholar]
- Desgranges, C.; Delhommelle, J. Free energy calculations along entropic pathways. I. Homogeneous vapor-liquid nucleation for atomic and molecular systems. J. Chem. Phys. 2016, 145, 204112. [Google Scholar] [CrossRef]
- Kido, A.; Nakanishi, K. Molecular dynamics study of nucleation in supersaturated vapor of carbon dioxide. Fluid Phase Equilibria 1999, 158–160, 79–86. [Google Scholar] [CrossRef]
- Halonen, R.; Tikkanen, V.; Reischl, B.; Dingilian, K.K.; Wyslouzil, B.E.; Vehkamäki, H. Homogeneous nucleation of carbon dioxide in supersonic nozzles II: Molecular dynamics simulations and properties of nucleating clusters. Phys. Chem. Chem. Phys. 2021, 23, 4517–4529. [Google Scholar] [CrossRef]
- Iland, K. Experimente zur homogenen Keimbildung von Argon und Stickstoff. Ph.D. Thesis, Universität zu Köln, Köln, Germany, 2004. [Google Scholar]
- Jortner, J. Cluster size effects. Z. Phys. Atoms Mol. Clust. 1992, 24, 247–275. [Google Scholar] [CrossRef]
- Hoare, M.; Pal, P. Physical cluster mechanics: Statistical thermodynamics and nucleation theory for monatomic systems. Adv. Phys. 1975, 24, 645–678. [Google Scholar] [CrossRef]
- Hoare, M.; Pal, P. Physical cluster mechanics: Statics and energy surfaces for monatomic systems. Adv. Phys. 1971, 20, 161–196. [Google Scholar] [CrossRef]
- Torchet, G.; de Feraudy, M.; Boutin, A.; Fuchs, A.H. Structural transformation in (CO2)N clusters, N<100. J. Chem. Phys. 1996, 105, 3671–3678. [Google Scholar] [CrossRef]
- Maillet, J.B.; Boutin, A.; Fuchs, A.H. From molecular clusters to bulk matter. II. Crossover from icosahedral to crystalline structures in CO2 clusters. J. Chem. Phys. 1999, 111, 2095–2102. [Google Scholar] [CrossRef]
- Castleman, A.W.; Keesee, R.G. Clusters: Properties and Formation. Annu. Rev. Phys. Chem. 1986, 37, 525–550. [Google Scholar] [CrossRef]
- Maillet, J.B.; Boutin, A.; Fuchs, A.H. The Melting Phase Transition in Small Carbon Dioxide Clusters. Mol. Simul. 1997, 19, 285–299. [Google Scholar] [CrossRef]
- Rytkönen, A.; Valkealahti, S.; Manninen, M. Melting and evaporation of argon clusters. J. Chem. Phys. 1997, 106, 1888–1892. [Google Scholar] [CrossRef]
- Doye, J.P.K.; Calvo, F. Entropic effects on the structure of Lennard-Jones clusters. J. Chem. Phys. 2002, 116, 8307–8317. [Google Scholar] [CrossRef]
- Maillet, J.B.; Boutin, A.; Buttefey, S.; Calvo, F.; Fuchs, A.H. From molecular clusters to bulk matter. I. Structure and thermodynamics of small CO2, N2, and SF6 clusters. J. Chem. Phys. 1998, 109, 329–337. [Google Scholar] [CrossRef]
- Acevedo, A.J.; Caballero, L.M.; López, G.E. Phase transitions in molecular clusters. J. Chem. Phys. 1997, 106, 7257–7261. [Google Scholar] [CrossRef]
- Ford, I.J. Nucleation theorems, the statistical mechanics of molecular clusters, and a revision of classical nucleation theory. Phys. Rev. E 1997, 56, 5615–5629. [Google Scholar] [CrossRef]
- Ellerby, H.M. Distribution of density fluctuations in a molecular theory of vapor-phase nucleation. Phys. Rev. E 1994, 49, 4287–4297. [Google Scholar] [CrossRef]
- Vehkamäki, H. Classical Nucleation Theory in Multicomponent Systems; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar] [CrossRef]
- Kalikmanov, V.I. Nucleation Theory; Lecture Notes in Physics; Springer: Berlin/Heidelberg, Germany, 2013; Volume 860. [Google Scholar] [CrossRef]
- Abraham, F.F. Homogeneous Nucleation Theory: The Pretransition Theory of Vapor Condensation; Academic Press: Cambridge, MA, USA, 1974. [Google Scholar] [CrossRef]
- Friedlander, S.K. The Mechanics of Aerosols, 2nd ed.; Oxford University Press: Oxford, UK, 2000. [Google Scholar]
- Zel’dovich, J.B. K Teorii Obrazovaniya Novoy Fazy. Kavitatsiya. Zhurnal Eksperimental’noy Teor. Fiziki 1942, 12, 525–538. [Google Scholar]
- Lothe, J.; Pound, G.M. Statistical Mechanics of Nucleation. In Nucleation; Zettlemoyer, A.C., Ed.; Marcel Dekker: New York, NY, USA, 1969; Chapter 3; pp. 109–149. [Google Scholar]
- Wakeshima, H. Time Lag in the Self-Nucleation. J. Chem. Phys. 1954, 22, 1614–1615. [Google Scholar] [CrossRef]
- Wu, D.T. The time lag in nucleation theory. J. Chem. Phys. 1992, 97, 2644–2650. [Google Scholar] [CrossRef]
- Wedekind, J.; Hyvärinen, A.P.; Brus, D.; Reguera, D. Unraveling the “Pressure Effect” in Nucleation. Phys. Rev. Lett. 2008, 101, 125703. [Google Scholar] [CrossRef]
- Tolman, R.C. The Effect of Droplet Size on Surface Tension. J. Chem. Phys. 1949, 17, 333–337. [Google Scholar] [CrossRef]
- Rowlinson, J.S.; Widom, B. Molecular Theory of Capillarity, 1st ed.; International Series of Monographs on Chemistry; Oxford University Press: New York, NY, USA, 1982; Volume 8. [Google Scholar]
- Gránásy, L. Semiempirical van der Waals/Cahn-Hilliard theory: Size dependence of the Tolman length. J. Chem. Phys. 1998, 109, 9660–9663. [Google Scholar] [CrossRef]
- Koga, K.; Zeng, X.C. Validity of Tolman’s equation: How large should a droplet be? J. Chem. Phys. 1998, 109, 4063–4070. [Google Scholar] [CrossRef]
- Wang, X.S.; Zhu, R.Z. Relation between Tolman length and isothermal compressibility for simple liquids. Chin. Phys. B 2013, 22, 036801. [Google Scholar] [CrossRef][Green Version]
- Horsch, M.; Vrabec, J.; Hasse, H. Modification of the classical nucleation theory based on molecular simulation data for surface tension, critical nucleus size, and nucleation rate. Phys. Rev. E 2008, 78, 011603. [Google Scholar] [CrossRef] [PubMed]
- Girshick, S.L.; Chiu, C.P. Kinetic nucleation theory: A new expression for the rate of homogeneous nucleation from an ideal supersaturated vapor. J. Chem. Phys. 1990, 93, 1273–1277. [Google Scholar] [CrossRef]
- Wilemski, G. The Kelvin equation and self-consistent nucleation theory. J. Chem. Phys. 1995, 103, 1119–1126. [Google Scholar] [CrossRef]
- Courtney, W.G. Remarks on Homogeneous Nucleation. J. Chem. Phys. 1942, 35, 2249–2250. [Google Scholar] [CrossRef]
- Reguera, D.; Bowles, R.K.; Djikaev, Y.; Reiss, H. Phase transitions in systems small enough to be clusters. J. Chem. Phys. 2003, 118, 164720. [Google Scholar] [CrossRef]
- Kalikmanov, V.I. Mean-field kinetic nucleation theory. J. Chem. Phys. 2006, 124, 124505. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M.E. The theory of condensation and the critical point. Physics 1967, 3, 255–283. [Google Scholar] [CrossRef]
- Sinha, S.; Bhabhe, A.; Laksmono, H.; Wölk, J.; Strey, R.; Wyslouzil, B. Argon nucleation in a cryogenic supersonic nozzle. J. Chem. Phys. 2010, 132, 064304. [Google Scholar] [CrossRef]
- Laaksonen, A.; Ford, I.J.; Kulmala, M. Revised parametrization of the Dillmann-Meier theory of homogeneous nucleation. Phys. Rev. E 1994, 49, 5517–5524. [Google Scholar] [CrossRef]
- Delale, C.F.; Meier, G.E.A. A semiphenomenological droplet model of homogeneous nucleation from the vapor phase. J. Chem. Phys. 1993, 98, 9850–9858. [Google Scholar] [CrossRef]
- Dillmann, A.; Meier, G.E.A. A refined droplet approach to the problem of homogeneous nucleation from the vapor phase. J. Chem. Phys. 1991, 94, 3872–3884. [Google Scholar] [CrossRef]
- Reguera, D.; Reiss, H. Fusion of the Extended Modified Liquid Drop Model for Nucleation and Dynamical Nucleation Theory. Phys. Rev. Lett. 2004, 93, 165701. [Google Scholar] [CrossRef]
- Reguera, D.; Reiss, H. Extended Modified Liquid Drop-Dynamical Nucleation Theory (EMLD-DNT) Approach to Nucleation: A New Theory. J. Phys. Chem. B 2004, 108, 19831–19842. [Google Scholar] [CrossRef]
- Baidakov, V.G.; Boltachev, G.S. Extended version of the van der Waals capillarity theory. J. Chem. Phys. 2004, 121, 8594–8601. [Google Scholar] [CrossRef]
- Carnahan, N.F.; Starling, K.E. Equation of State for Nonattracting Rigid Spheres. J. Chem. Phys. 1969, 51, 635–636. [Google Scholar] [CrossRef]
- Hale, B.N. Application of a scaled homogeneous nucleation-rate formalism to experimental data at T≪Tc. Phys. Rev. A 1986, 33, 4156–4163. [Google Scholar] [CrossRef] [PubMed]
- Hale, B.N. The Scaling of Nucleation Rates. Metall. Trans. A 1992, 23, 1863–1868. [Google Scholar] [CrossRef]
- Palit, S.R. Thermodynamic Interpretation of the Eötvös Constant. Nature 1956, 177, 1180. [Google Scholar] [CrossRef]
- Wedekind, J.; Reguera, D.; Strey, R. Influence of thermostats and carrier gas on simulations of nucleation. J. Chem. Phys. 2007, 127, 064501. [Google Scholar] [CrossRef] [PubMed]
- Feder, J.; Russel, K.C.; Lothe, J.; Pound, G.M. Homogeneous nucleation and growth of droplets in vapours. Adv. Phys. 1966, 15, 111–178. [Google Scholar] [CrossRef]
- Makarov, G.; Petin, A. Disintegration of ArN, KrN, and (N2)N Clusters during Collisions with Highly Vibrationally Excited SF6 Molecules. J. Exp. Theor. Phys. 2014, 146, 398–405. [Google Scholar] [CrossRef]
- Sharma, S.P.; Ruffin, S.M.; Gillespie, W.D.; Meyer, S.A. Vibrational relaxation measurements in an expanding flow using spontaneous Raman scattering. J. Thermophys. Heat Transf. 1993, 7, 697–703. [Google Scholar] [CrossRef]
- Vincenti, W.G.; Kruger, C.H. Introduction to Physical Gas Dynamics; John Wiley and Sons: New York, NY, USA, 1967. [Google Scholar]
- Kobraei, H.R.; Anderson, B.R. Formation energies and concentrations of microclusters for homogeneous nucleation. J. Chem. Phys. 1988, 88, 4451–4459. [Google Scholar] [CrossRef]
- Balla, R.J.; Everhart, J.L. Rayleigh Scattering Density Measurements, Cluster Theory, and Nucleation Calculations at Mach 10. Am. Inst. Aeronaut. Astronaut. 2012, 50, 698–707. [Google Scholar] [CrossRef]
- Balla, R.J.; Rhode, M.N.; Everhart, J.L. Supersaturation Total Temperature, Pitot Pressure, and Rayleigh Scattering Measurements at Mach 10. Am. Inst. Aeronaut. Astronaut. 2014, 52, 1452–1465. [Google Scholar] [CrossRef]
- Elliott, S.; Hasegawa, M.; Sakaue, H.; Leonov, S. Shock-dominated flow control by plasma array: Pressure analysis including pressure-sensitive paint visualization. Exp. Therm. Fluid Sci. 2022, 131, 110522. [Google Scholar] [CrossRef]
- Houpt, A.; Hedlund, B.; Leonov, S.; Ombrello, T.; Carter, C. Quasi-DC electrical discharge characterization in a supersonic flow. Exp. Fluids 2017, 58, 1–17. [Google Scholar] [CrossRef]
- Hill, P.G. Condensation of Water Vapour during Supersonic Expansion in Nozzles. J. Fluid Mech. 1966, 25, 593–620. [Google Scholar] [CrossRef]
- Sivells, J.C. Aerodynamic design of axisymmetric hypersonic wind-tunnel nozzles. J. Spacecr. Rocket. 1970, 7, 1292–1299. [Google Scholar] [CrossRef]
- Gyarmathy, G. The Spherical Droplet in Gaseous Carrier Streams: Review and Synthesis. In Multiphase Science and Technology; Hewitt, G.F., Delhaye, J.M., Zuber, N., Eds.; McGraw-Hill: New York, NY, USA, 1982; Volume 1, Chapter 2; pp. 99–279. [Google Scholar]
- Tanimura, S.; Park, Y.; Amaya, A.; Modak, V.; Wyslouzil, B.E. Following heterogeneous nucleation of CO2 on H2O ice nanoparticles with microsecond resolution. RSC Adv. 2015, 5, 105537–105550. [Google Scholar] [CrossRef]
- Ramos, A.; Fernández, J.M.; Tejeda, G.; Montero, S. Quantitative study of cluster growth in free-jet expansions of CO2 by Rayleigh and Raman scattering. Phys. Rev. A 2005, 72, 053204. [Google Scholar] [CrossRef]
- Lippe, M.; Szczepaniak, U.; Hou, G.L.; Chakrabarty, S.; Ferreiro, J.J.; Chasovskikh, E.; Signorell, R. Infrared Spectroscopy and Mass Spectrometry of CO2 Clusters during Nucleation and Growth. J. Phys. Chem. A 2019, 123, 2426–2437. [Google Scholar] [CrossRef] [PubMed]
- Krohn, J.; Lippe, M.; Li, C.; Signorell, R. Carbon dioxide and propane nucleation: The emergence of a nucleation barrier. Phys. Chem. Chem. Phys. 2020, 22, 15986–15998. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y. Heterogeneous nucleation or homogeneous nucleation? J. Chem. Phys. 2000, 112, 9949–9955. [Google Scholar] [CrossRef]
- Anderson, J.D. Modern Compressible Flow: With Historical Perspective, 3rd ed.; McGraw-Hill: New York, NY, USA, 2003. [Google Scholar]
- Clifford, A.A.; Gray, P.; Platts, N. Lennard-Jones 12:6 parameters for ten small molecules. J. Chem. Soc. Faraday Trans. 1977, 73, 381–382. [Google Scholar] [CrossRef]
- Span, R.; Wagner, W. A New Equation of State for Carbon Dioxide Covering the Fluid Region from the Triple- Point Temperature to 1100 K at Pressures up to 800 MPa. J. Phys. Chem. Ref. Data 1996, 25, 1509–1596. [Google Scholar] [CrossRef]
- Herzberg, G. Molecular Spectra and Molecular Structure II. Infrared and Raman Spectra of Polyatomic Molecules; D. Van Nostrand Company: New York, NY, USA, 1945. [Google Scholar]
- Irikura, K.K. Experimental Vibrational Zero-Point Energies: Diatomic Molecules. J. Phys. Chem. Ref. Data 2007, 36, 389–397. [Google Scholar] [CrossRef]
- Jasper, J.J. The Surface Tension of Pure Liquid Compounds. J. Phys. Chem. Ref. Data 1972, 1, 841–1010. [Google Scholar] [CrossRef]
- Poling, B.E. Properties of Gases and Liquids, 5th ed.; McGraw-Hill Education: New York, NY, USA, 2001. [Google Scholar]
- Magee, J.W. Specific heats (Cv) of saturated and compressed liquid and vapor carbon dioxide. Int. J. Thermophys. 1986, 7, 1163–1182. [Google Scholar] [CrossRef]
- Acree, W.; Chickos, J.S. Phase Transition Enthalpy Measurements of Organic and Organometallic Compounds. Sublimation, Vaporization and Fusion Enthalpies from 1880 to 2015. Part 1. C1-C10. J. Phys. Chem. Ref. Data 2016, 45, 033101. [Google Scholar] [CrossRef]
- Huber, M.L.; Sykioti, E.A.; Assael, M.J.; Perkins, R.A. Reference Correlation of the Thermal Conductivity of Carbon Dioxide from the Triple Point to 1100 K and up to 200 MPa. J. Phys. Chem. Ref. Data 2016, 45, 013102. [Google Scholar] [CrossRef] [PubMed]
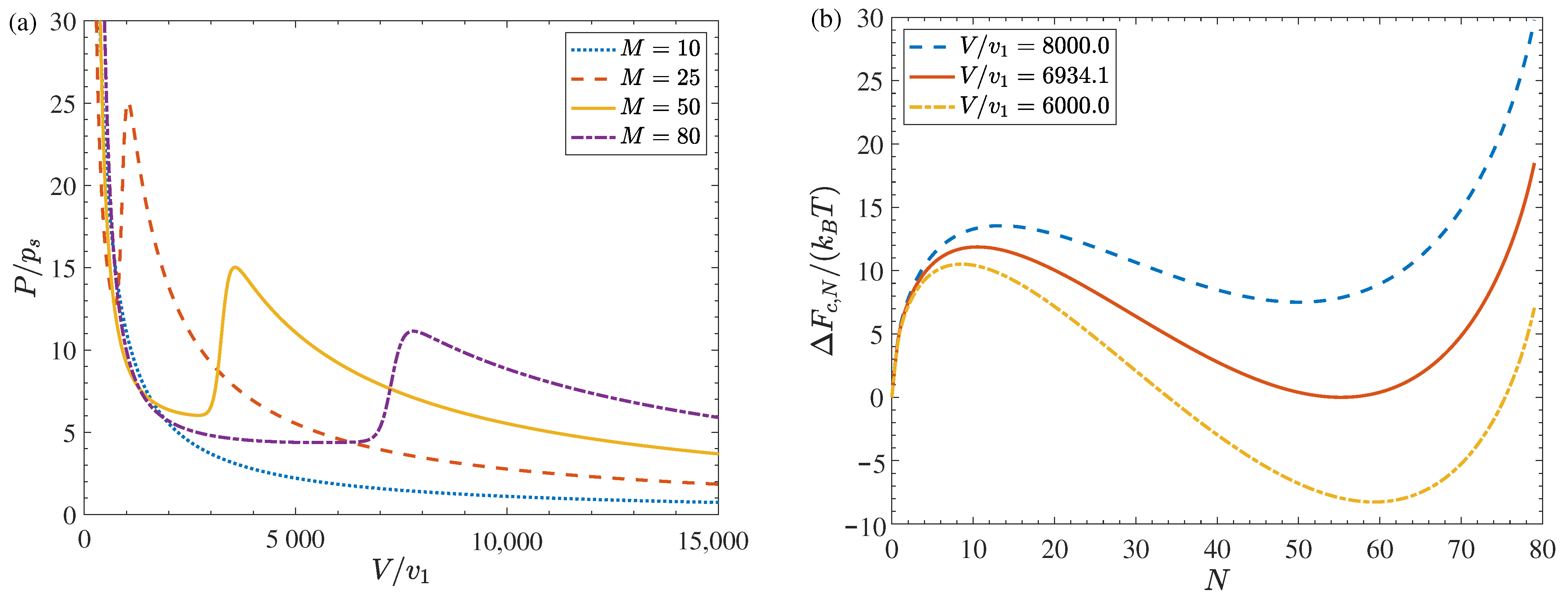
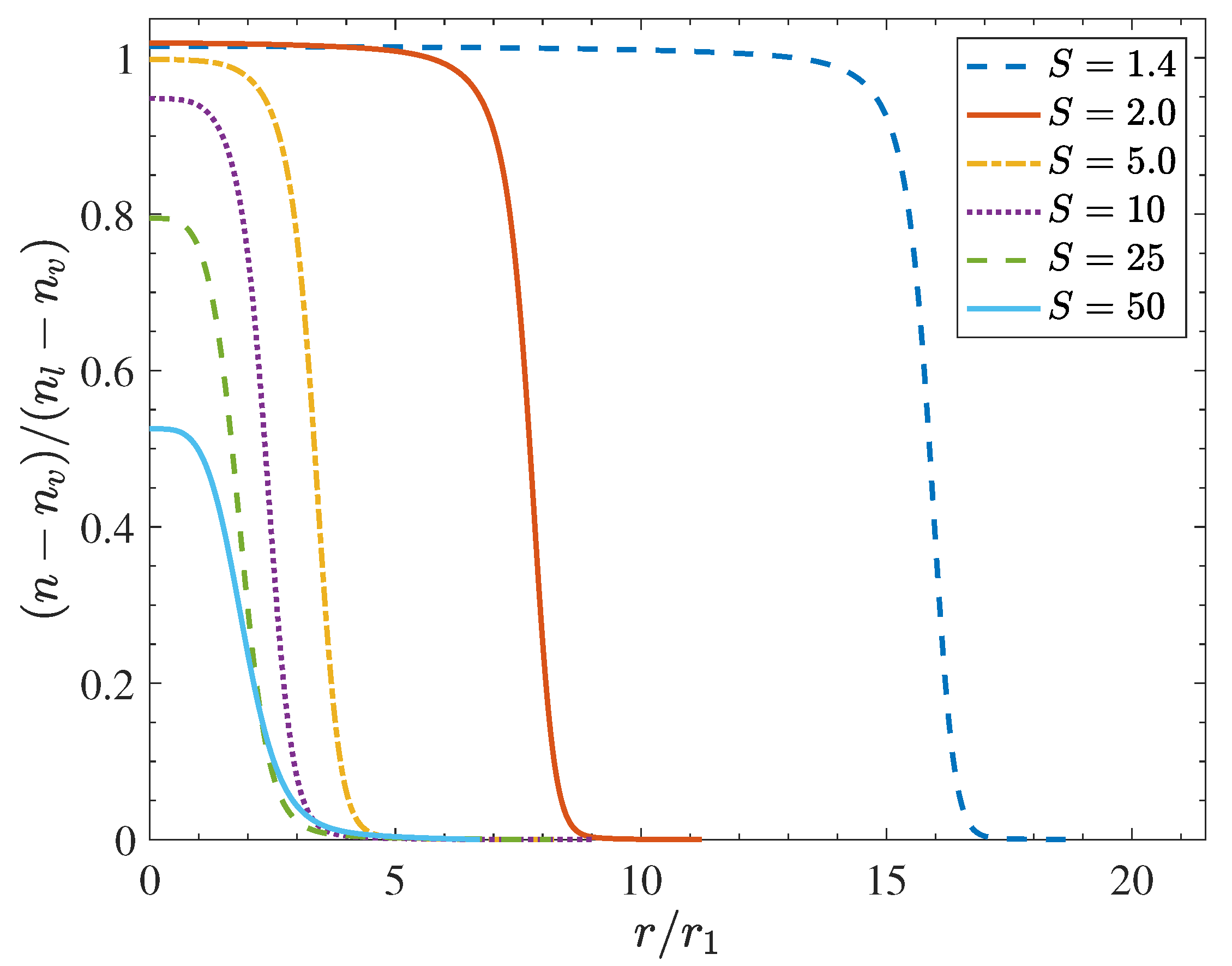
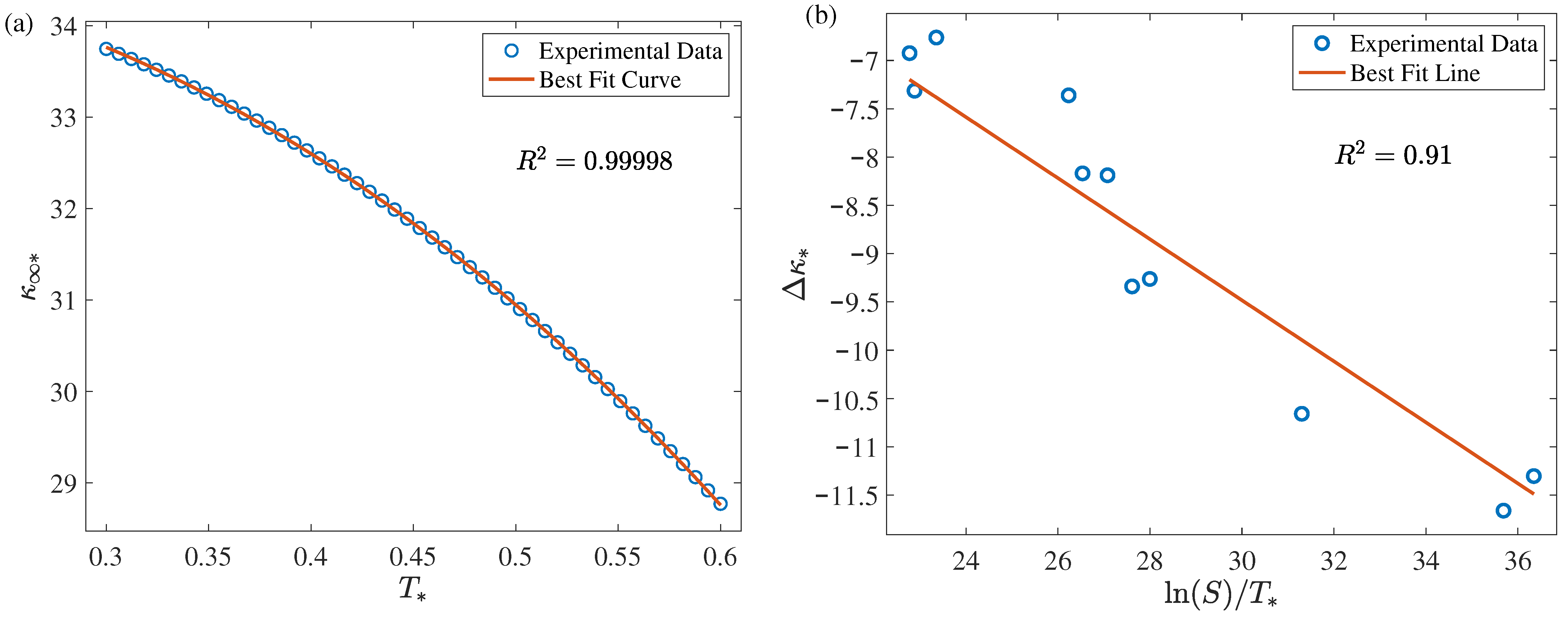

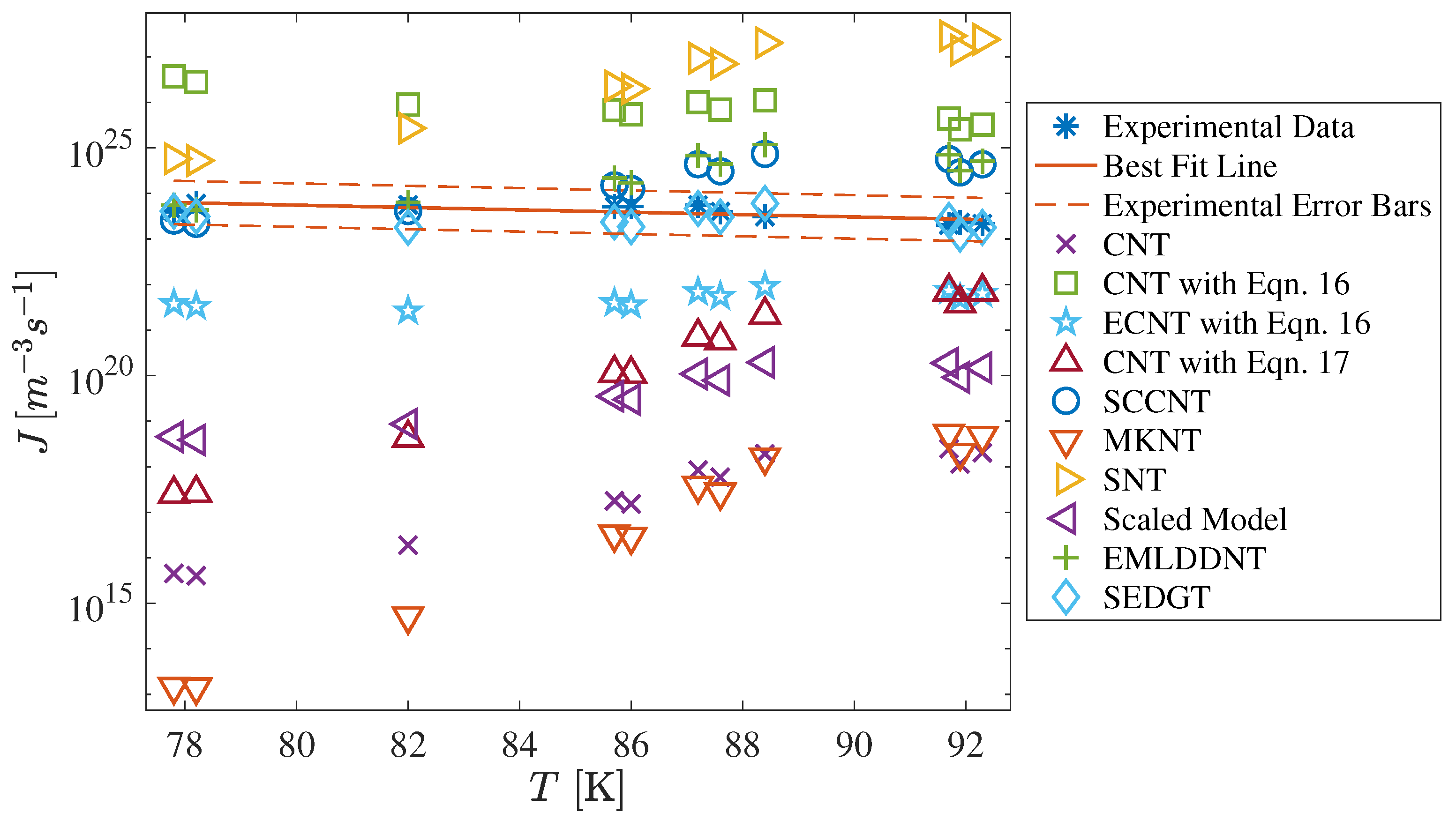
| Facility Type | Carrier | CO [%] | T [K] | [Pa] | S | Ref. |
|---|---|---|---|---|---|---|
| de Laval Nozzle | Air | 1.2 | 88–97 | 16–32 | 606–2.9 × | Present |
| Planar Nozzle | Ar | 2.0–39.3 | 75–92 | 39–793 | 2.3 × –6.1 × | [10] |
| Planar & de Laval | – | 100 | 161–193 | 1.3 × –4.3 × | 2.7–7.8 | [15] |
| Fixed Orifice Free Jet | He | 5–100 | 115–166 | 6.1 × –2.4 × | 9.3–146 | [25] |
| de Laval Nozzle | 2.4–25.2 | 124–146 | 240–3.5 × | 0.5–1.4 | [88] | |
| Fixed Orifice Free Jet | – | 100 | 75–106 | 301–1.2 × | 1.8 × –2.2 × | [89] |
| de Laval Nozzle | Ar + | 7 | 31–34 | 0.04–0.065 | 1.1 × –3.3 × | [90] |
| de Laval Nozzle | Ar + | 0.12–50 | 31–63 | 0.04–13 | 1.1 × –1.8 × | [91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lax, P.A.; Leonov, S.B. Review of Reduced-Order Models for Homogeneous CO2 Nucleation in Supersonic and Hypersonic Expansion Flows. Aerospace 2021, 8, 368. https://doi.org/10.3390/aerospace8120368
Lax PA, Leonov SB. Review of Reduced-Order Models for Homogeneous CO2 Nucleation in Supersonic and Hypersonic Expansion Flows. Aerospace. 2021; 8(12):368. https://doi.org/10.3390/aerospace8120368
Chicago/Turabian StyleLax, Philip A., and Sergey B. Leonov. 2021. "Review of Reduced-Order Models for Homogeneous CO2 Nucleation in Supersonic and Hypersonic Expansion Flows" Aerospace 8, no. 12: 368. https://doi.org/10.3390/aerospace8120368
APA StyleLax, P. A., & Leonov, S. B. (2021). Review of Reduced-Order Models for Homogeneous CO2 Nucleation in Supersonic and Hypersonic Expansion Flows. Aerospace, 8(12), 368. https://doi.org/10.3390/aerospace8120368







