Potential Risks of Plant Invasions in Protected Areas of Sri Lanka under Climate Change with Special Reference to Threatened Vertebrates
Abstract
1. Introduction
2. Methods
2.1. Study Area
2.2. Risk Assessments of Multiple IAPS into Protected Areas
2.3. Risk Assessments of Individual IAPS into Protected Areas
2.4. Potential Risks of IAPS on Threatened Vertebrates
3. Results
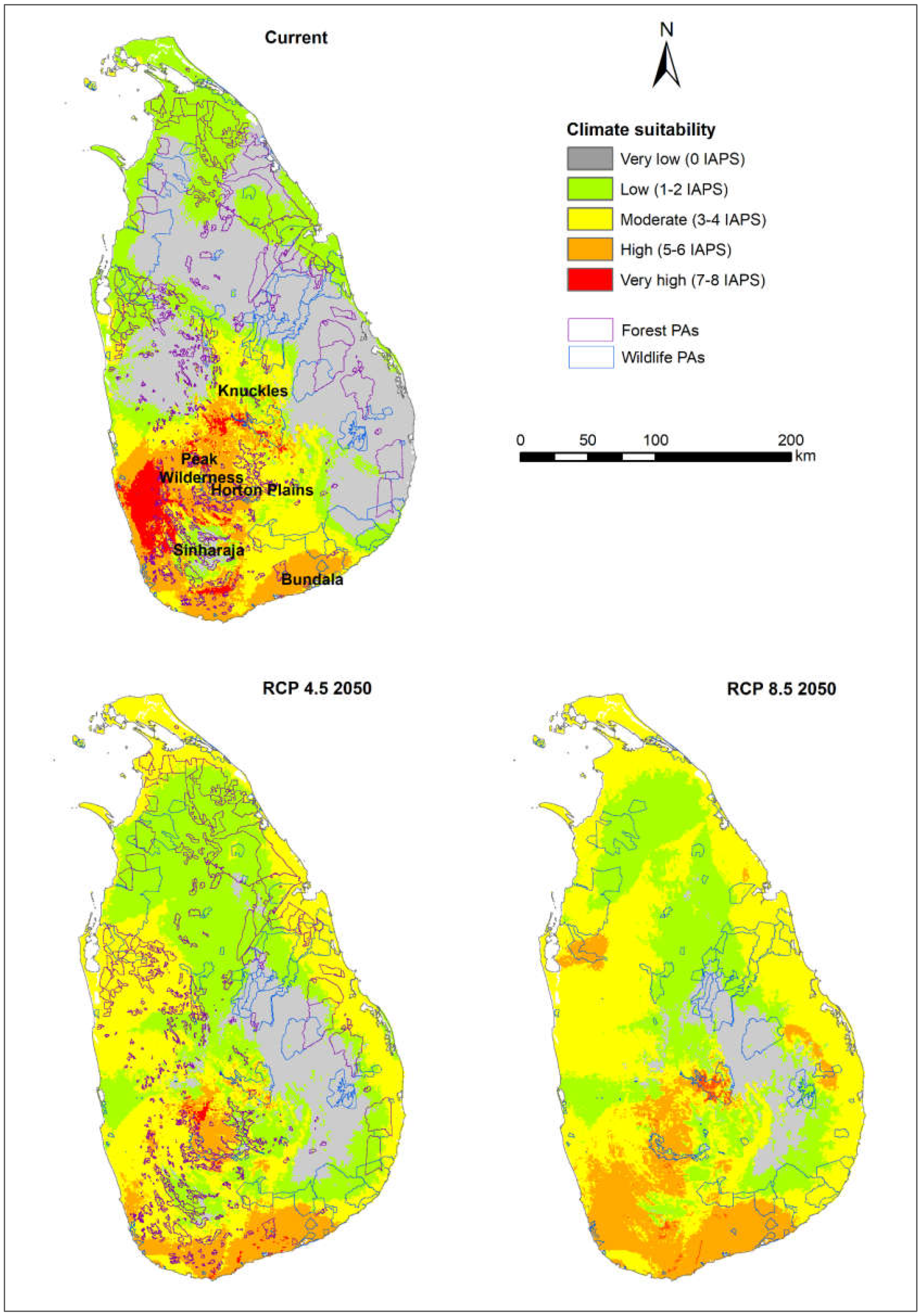
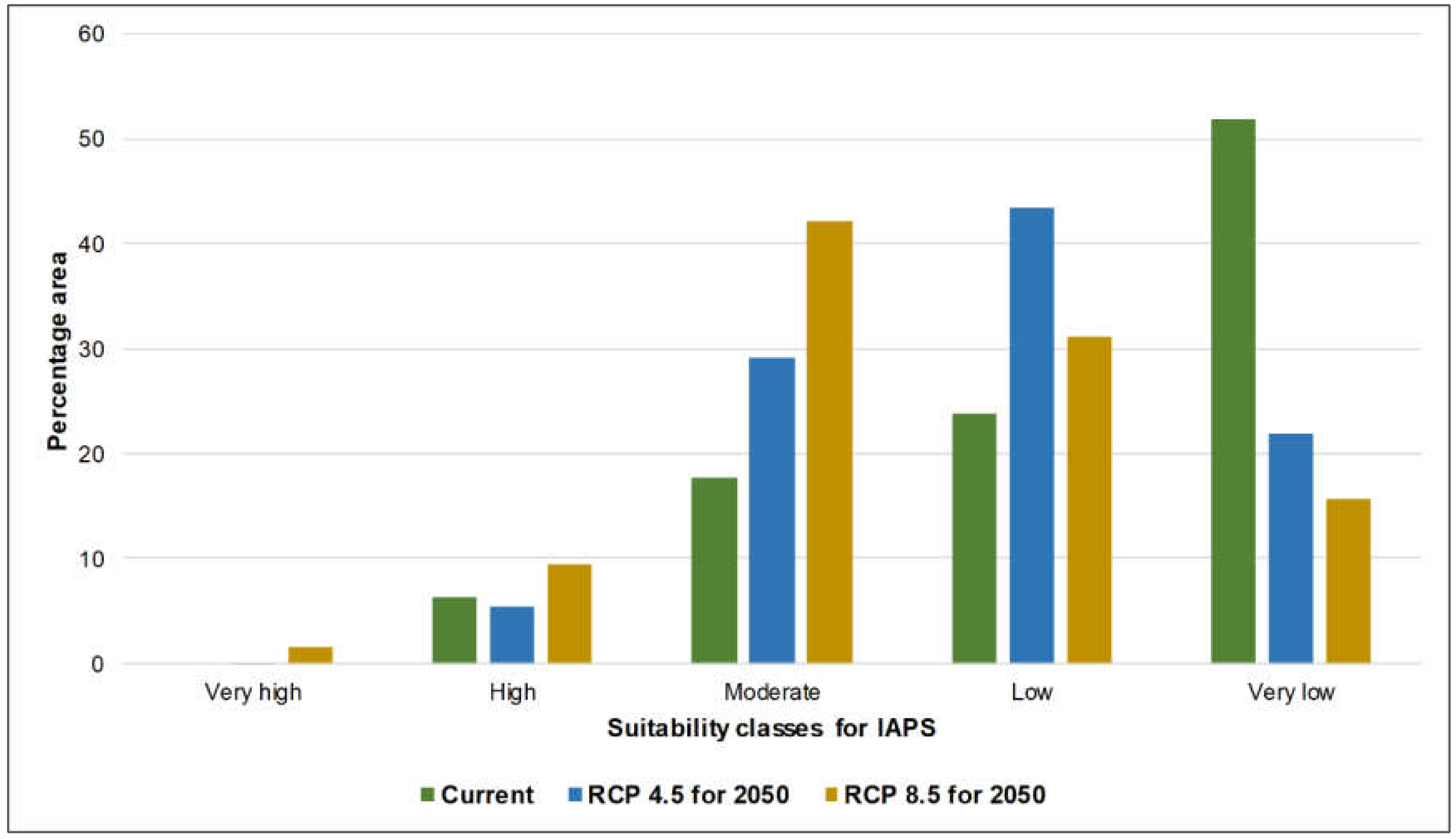
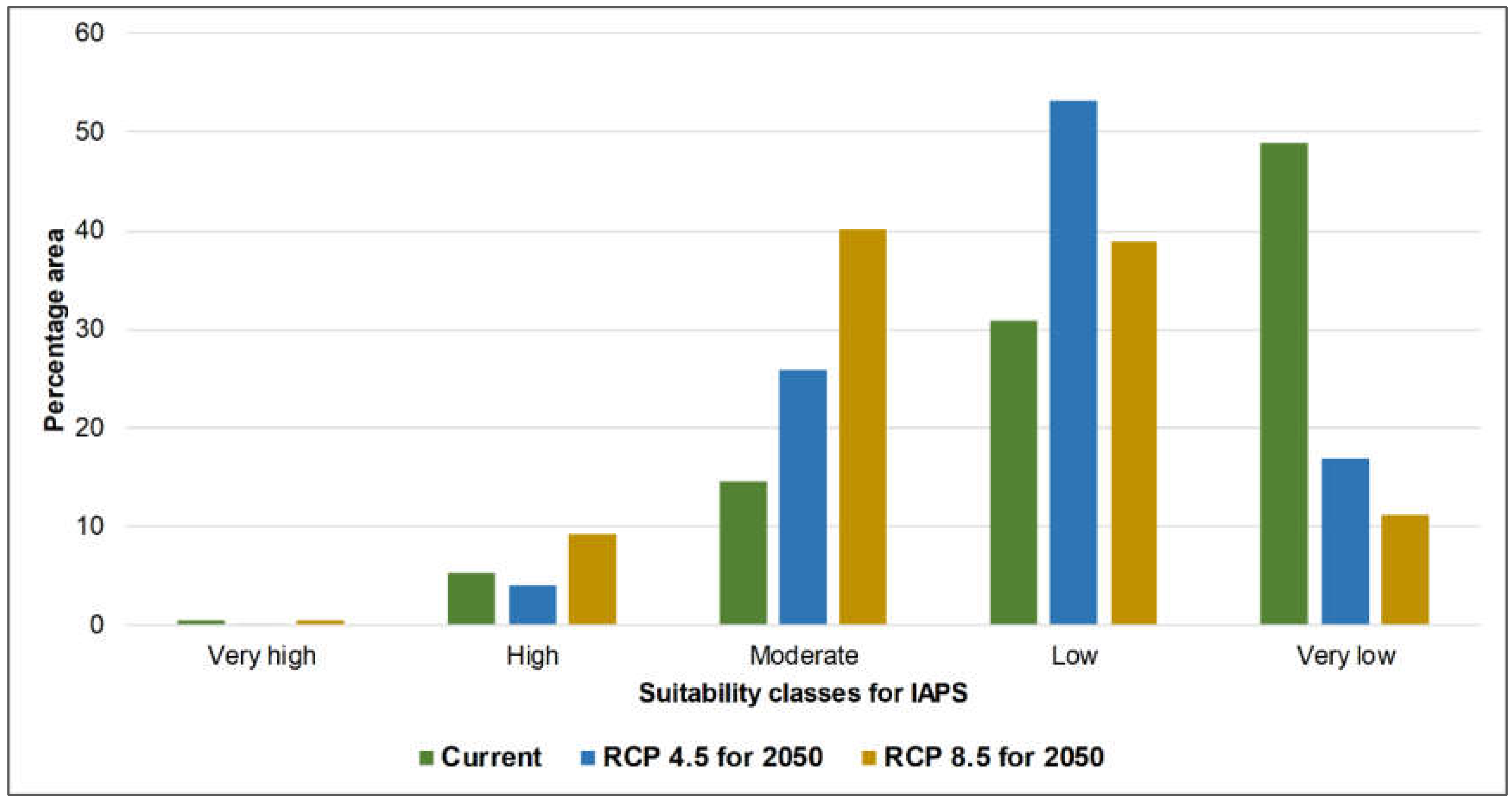
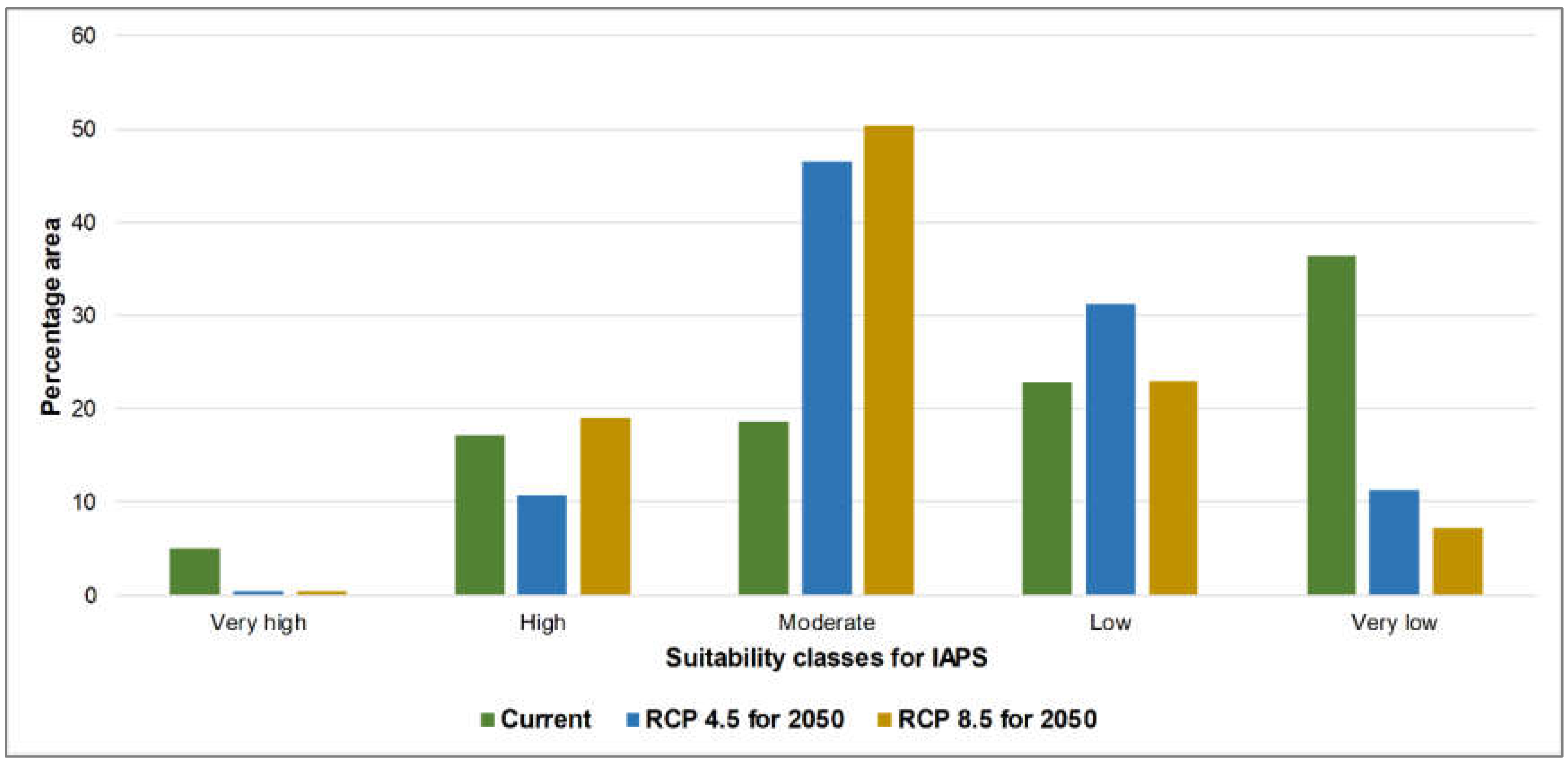
3.1. Risk Assessments of Multiple IAPS into Protected Areas
3.2. Risk Assessments of Individual IAPS into Protected Areas
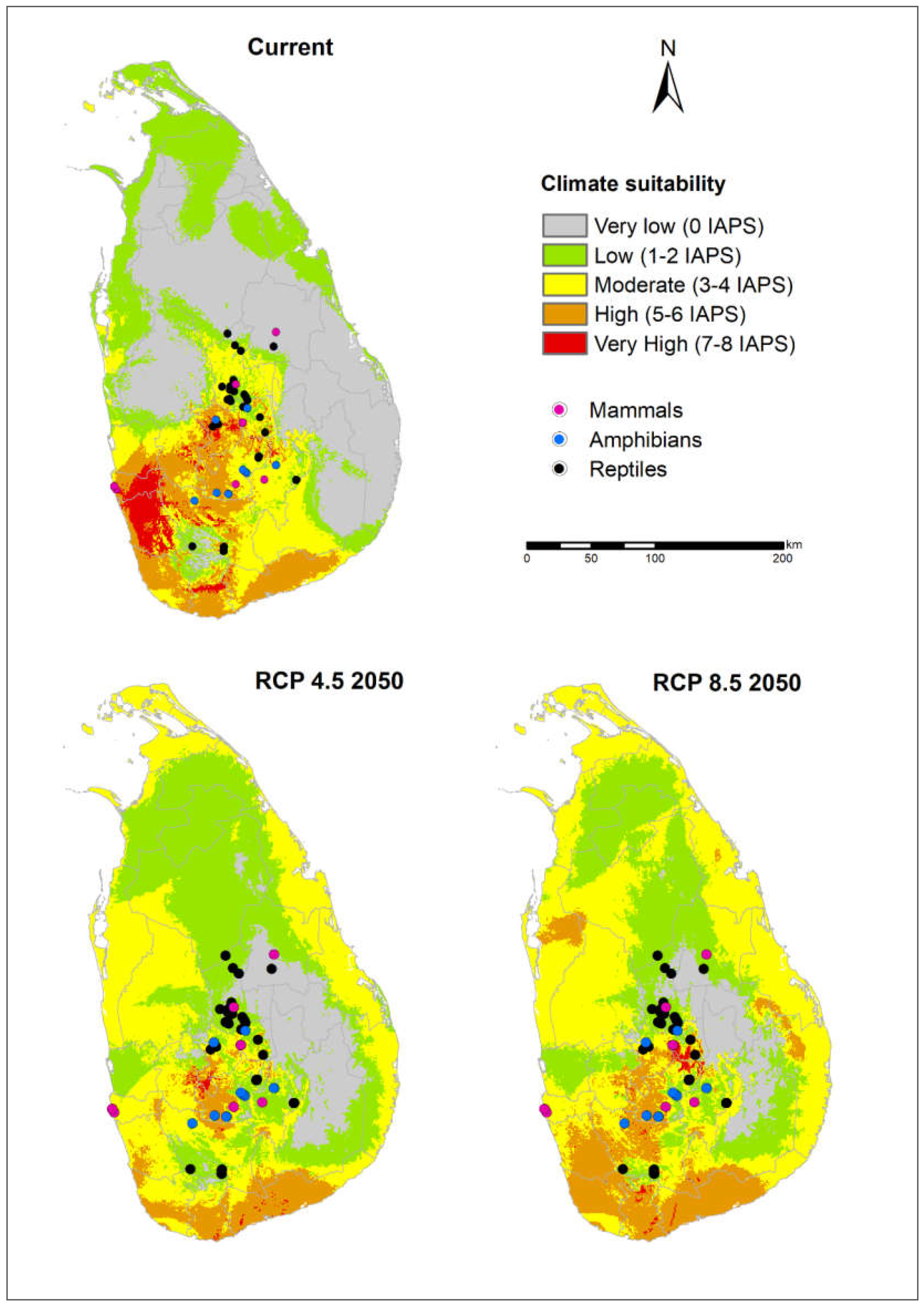
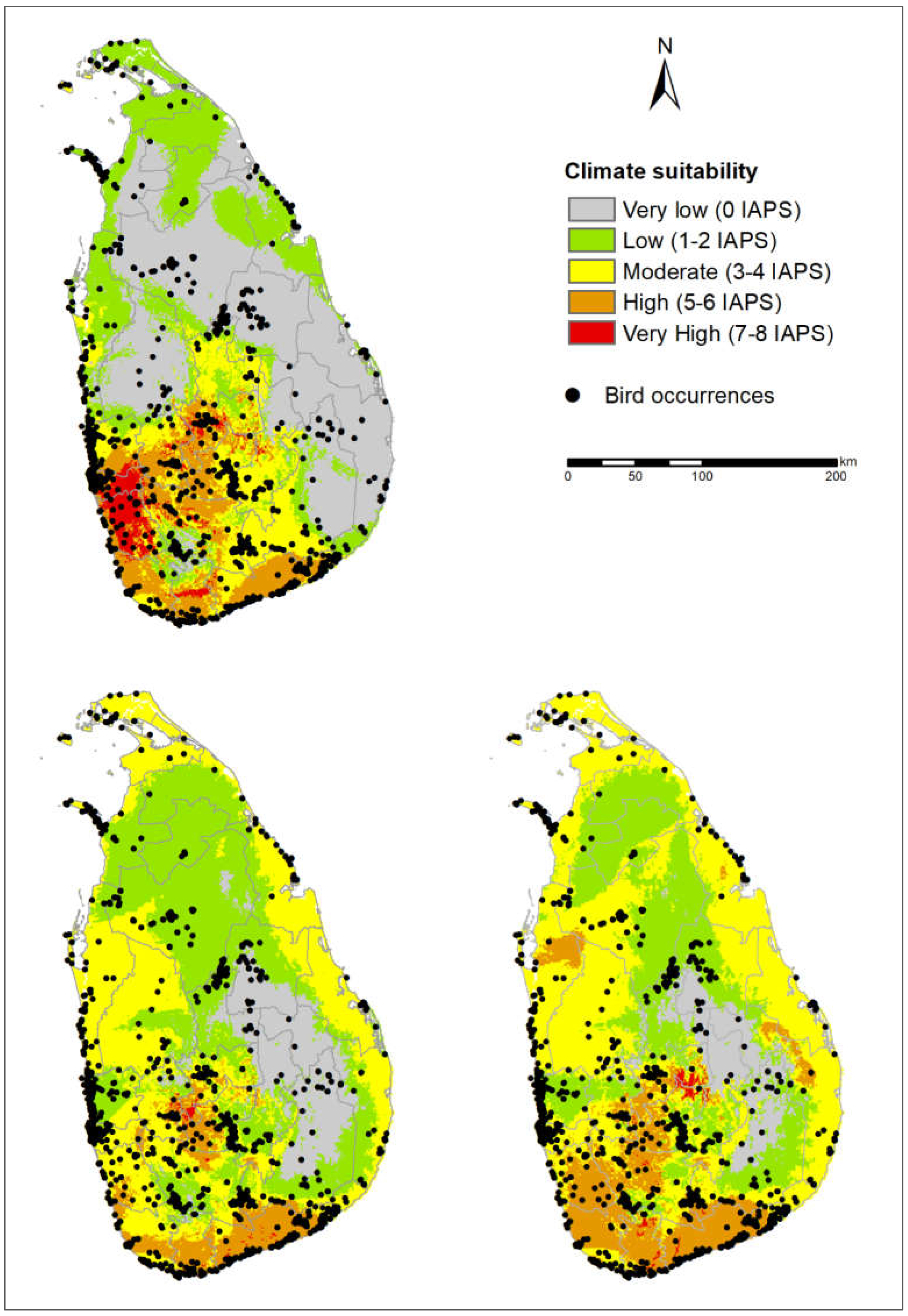
3.3. Potential Risks of IAPS on Threatened Vertebrates
4. Discussion
4.1. Risk Assessments of Multiple IAPS into Protected Areas
4.2. Risk Assessments of Individual IAPS into Protected Areas
4.3. Potential Risks of IAPS on Threatened Vertebrates
4.4. Vertebrate-Rich Protected Areas Vulnerable to Multiple IAPS Invasion
4.5. Limitations of the Study
4.6. Recommendations for Future Studies
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rands, M.R.; Adams, W.M.; Bennun, L.; Butchart, S.H.; Clements, A.; Coomes, D.; Entwistle, A.; Hodge, I.; Kapos, V.; Scharlemann, J.P. Biodiversity conservation: Challenges beyond 2010. Science 2010, 329, 1298–1303. [Google Scholar] [CrossRef]
- Secretariat of the Convention on Biological Diversity. Global Biodiversity Outlook 4; Secretariat of the Convention on Biological Diversity: Montréal, QC, Canada, 2014; p. 155.
- IPCC. Managing the Risks of Extreme Events and Disasters to Advance Climate Change Adaptation. A Special Report of Working Groups I and II of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2012; p. 582. [Google Scholar]
- Kumar, L.; Tehrany, M.S. Climate change impacts on the threatened terrestrial vertebrates of the Pacific Islands. Sci. Rep. 2017, 7, 5030. [Google Scholar] [CrossRef] [PubMed]
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Synthesis; Island Press: Washington, DC, USA, 2005. [Google Scholar]
- IPCC. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Kariyawasam, C.S.; Kumar, L.; Ratnayake, S.S. Invasive Plants Distribution Modeling: A Tool for Tropical Biodiversity Conservation with Special Reference to Sri Lanka. Trop. Conserv. Sci. 2019, 12, 1–12. [Google Scholar] [CrossRef]
- Achard, F.; Eva, H.D.; Stibig, H.-J.; Mayaux, P.; Gallego, J.; Richards, T.; Malingreau, J.-P. Determination of deforestation rates of the world’s humid tropical forests. Science 2002, 297, 999–1002. [Google Scholar] [CrossRef] [PubMed]
- Lobell, D.B.; Burke, M.B.; Tebaldi, C.; Mastrandrea, M.D.; Falcon, W.P.; Naylor, R.L. Prioritizing climate change adaptation needs for food security in 2030. Science 2008, 319, 607–610. [Google Scholar] [CrossRef] [PubMed]
- Paini, D.R.; Sheppard, A.W.; Cook, D.C.; De Barro, P.J.; Worner, S.P.; Thomas, M.B. Global threat to agriculture from invasive species. Proc. Natl. Acad. Sci. USA 2016, 113, 7575–7579. [Google Scholar] [CrossRef]
- Leadley, P.; Pereira, H.M.; Alkemade, R.; Fernandez-Manjarrés, J.F.; Proença, V.; Scharlemann, J.P.W.; Walpole, M.J. Biodiversity Scenarios: Projections of 21st Century Change in Biodiversity and Associated Ecosystem Services; Secretariat of the Convention on Biological Diversity: Montreal, QC, Canada, 2010; p. 132.
- Fandohan, A.B.; Oduor, A.M.; Sodé, A.I.; Wu, L.; Cuni-Sanchez, A.; Assédé, E.; Gouwakinnou, G.N. Modeling vulnerability of protected areas to invasion by Chromolaena odorata under current and future climates. Ecosyst. Health Sustain. 2015, 1, 1–12. [Google Scholar] [CrossRef]
- Taylor, S.; Kumar, L. Potential distribution of an invasive species under climate change scenarios using CLIMEX and soil drainage: A case study of Lantana camara L. in Queensland, Australia. J. Environ. Manag. 2013, 114, 414–422. [Google Scholar] [CrossRef]
- Bellard, C.; Jeschke, J.M.; Leroy, B.; Mace, G.M. Insights from modeling studies on how climate change affects invasive alien species geography. Ecol. Evol. 2018, 8, 5688–5700. [Google Scholar] [CrossRef]
- McNeely, J.A. Strangers in our midst: The problem of invasive alien species. Environment 2004, 46, 16. [Google Scholar] [CrossRef]
- Bellard, C.; Cassey, P.; Blackburn, T.M. Alien species as a driver of recent extinctions. Biol. Lett. 2016, 12, 20150623. [Google Scholar] [CrossRef] [PubMed]
- Foxcroft, L.C.; Pyšek, P.; Richardson, D.M.; Pergl, J.; Hulme, P.E. The bottom line: Impacts of alien plant invasions in protected areas. In Plant Invasions in Protected Areas; Springer: Dordrecht, The Netherlands, 2013; pp. 19–41. [Google Scholar]
- Pyšek, P.; Jarošík, V.; Hulme, P.E.; Pergl, J.; Hejda, M.; Schaffner, U.; Vilà, M. A global assessment of invasive plant impacts on resident species, communities and ecosystems: The interaction of impact measures, invading species’ traits and environment. Glob. Chang. Biol. 2012, 18, 1725–1737. [Google Scholar] [CrossRef]
- Dudley, N. Guidelines for Applying Protected Area Management Categories; IUCN: Gland, Switzerland, 2008. [Google Scholar]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Da Fonseca, G.A.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853. [Google Scholar] [CrossRef]
- UNEP-WCMC IUCN and NGS. Protected Planet Report 2018. 2018. Available online: https://livereport.protectedplanet.net/pdf/Protected_Planet_Report_2018.pdf (accessed on 10 August 2019).
- Pauchard, A.; Alaback, P.B. Influence of elevation, land use, and landscape context on patterns of alien plant invasions along roadsides in protected areas of South-Central Chile. Conserv. Biol. 2004, 18, 238–248. [Google Scholar] [CrossRef]
- Mack, R.N.; Simberloff, D.; Mark Lonsdale, W.; Evans, H.; Clout, M.; Bazzaz, F.A. Biotic invasions: Causes, epidemiology, global consequences, and control. Ecol. Appl. 2000, 10, 689–710. [Google Scholar] [CrossRef]
- Foxcroft, L.C.; Pyšek, P.; Richardson, D.M.; Genovesi, P. Plant Invasions in Protected Areas: Patterns, Problems and Challenges; Springer: Berlin, Germany, 2013; Volume 7. [Google Scholar]
- Wilson, K.A.; Underwood, E.C.; Morrison, S.A.; Klausmeyer, K.R.; Murdoch, W.W.; Reyers, B.; Wardell-Johnson, G.; Marquet, P.A.; Rundel, P.W.; McBride, M.F. Conserving biodiversity efficiently: What to do, where, and when. PLoS Biol. 2007, 5, e223. [Google Scholar] [CrossRef]
- Franklin, J.F. Preserving biodiversity: Species, ecosystems, or landscapes? Ecol. Appl. 1993, 3, 202–205. [Google Scholar] [CrossRef]
- Weerakoon, D. Analysis of Faunal Groups. In The National Red List 2012 of Sri Lanka: Conservation Status of the Fauna and Flora; Weerakoon, D.K., Wijesundara, S., Eds.; Ministry of Environment: Colombo, Sri Lanka, 2012; pp. 145–147. [Google Scholar]
- Cumberlidge, N.; Ng, P.K.; Yeo, D.C.; Magalhães, C.; Campos, M.R.; Alvarez, F.; Naruse, T.; Daniels, S.R.; Esser, L.J.; Attipoe, F.Y. Freshwater crabs and the biodiversity crisis: Importance, threats, status, and conservation challenges. Biol. Conserv. 2009, 142, 1665–1673. [Google Scholar] [CrossRef]
- Kariyawasam, C.S.; Kumar, L.; Ratnayake, R.S.S. Invasive Plant Species Establishment and Range Dynamics in Sri Lanka under Climate Change. Entropy 2019, 21, 571. [Google Scholar] [CrossRef]
- Phillips, S.; Anderson, R.P.; Schapire, R.E. Maximum entropy modelling of species geographic distributions. Ecol. Model. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Elith, J.; Graham, C.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A.; et al. Novel methods improve prediction of species’ distributions from occurrence data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Thibaud, E.; Petitpierre, B.; Broennimann, O.; Davison, A.C.; Guisan, A. Measuring the relative effect of factors affecting species distribution model predictions. Methods Ecol. Evol. 2014, 5, 947–955. [Google Scholar] [CrossRef]
- Lamsal, P.; Kumar, L.; Aryal, A.; Atreya, K. Invasive alien plant species dynamics in the Himalayan region under climate change. Ambio 2018, 47, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Suzuki, T.; O’ishi, R.; Komuro, Y.; Watanabe, S.; Emori, S.; Takemura, T.; Chikira, M.; Ogura, T.; Sekiguchi, M. Improved climate simulation by MIROC5: Mean states, variability, and climate sensitivity. J. Clim. 2010, 23, 6312–6335. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Thuiller, W.; Richardson, D.M.; PYŠEK, P.; Midgley, G.F.; Hughes, G.O.; Rouget, M. Niche-based modelling as a tool for predicting the risk of alien plant invasions at a global scale. Glob. Chang. Biol. 2005, 11, 2234–2250. [Google Scholar] [CrossRef]
- Adhikari, D.; Tiwary, R.; Barik, S.K. Modelling Hotspots for Invasive Alien Plants in India. PLoS ONE 2015, 10, e0134665. [Google Scholar] [CrossRef]
- Leroy, B.; Delsol, R.; Hugueny, B.; Meynard, C.N.; Barhoumi, C.; Barbet-Massin, M.; Bellard, C. Without quality presence–absence data, discrimination metrics such as TSS can be misleading measures of model performance. J. Biogeogr. 2018, 45, 1994–2002. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M. Modeling of species distributions with Maxent: New extensions and a comprehensive evaluation. Ecography 2008, 31, 161–175. [Google Scholar] [CrossRef]
- Elith, J.; Kearney, M.; Phillips, S. The art of modelling range-shifting species. Methods Ecol. Evol. 2010, 1, 330–342. [Google Scholar] [CrossRef]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Allouche, O.; Tsoar, A.; Kadmon, R. Assessing the accuracy of species distribution models: Prevalence, kappa and the true skill statistic (TSS). J. Appl. Ecol. 2006, 43, 1223–1232. [Google Scholar] [CrossRef]
- Shabani, F.; Ahmadi, M.; Peters, K.J.; Haberle, S.; Champreux, A.; Saltré, F.; Bradshaw, C.J. Climate-driven shifts in the distribution of koala-browse species from the Last Interglacial to the near future. Ecography 2019, 42, 1587–1599. [Google Scholar] [CrossRef]
- Shabani, F.; Kumar, L.; Ahmadi, M. A comparison of absolute performance of different correlative and mechanistic species distribution models in an independent area. Ecol. Evol. 2016, 6, 5973–5986. [Google Scholar] [CrossRef]
- Liu, C.; Newell, G.; White, M. On the selection of thresholds for predicting species occurrence with presence-only data. Ecol. Evol. 2016, 6, 337–348. [Google Scholar] [CrossRef]
- Liu, C.; White, M.; Newell, G. Selecting thresholds for the prediction of species occurrence with presence-only data. J. Biogeogr. 2013, 40, 778–789. [Google Scholar] [CrossRef]
- Shrestha, U.B.; Shrestha, B.B. Climate change amplifies plant invasion hotspots in Nepal. Divers. Distrib. 2019, 25, 1599–1612. [Google Scholar] [CrossRef]
- MoFE. Biodiversity Conservation in Sri Lanka: A Framework for Action; Ministry of Forestry and Environment: Battaramulla, Sri Lanka, 1999.
- Gunawardene, N.R.; Daniels, D.A.; Gunatilleke, I.; Gunatilleke, C.; Karunakaran, P.; Nayak, G.K.; Prasad, S.; Puyravaud, P.; Ramesh, B.; Subramanian, K. A brief overview of the Western Ghats–Sri Lanka biodiversity hotspot. Curr. Sci. 2007, 93, 1567–1572. [Google Scholar]
- MOE. The National Red List 2012 of Sri Lanka; Conservation Status of the Fauna and Flora; Ministry of Environment: Colombo, Sri Lanka, 2012; p. 476.
- GBIF. GBIF Home Page. Available online: https://www.gbif.org (accessed on 21 September 2019).
- Allison, S.D.; Vitousek, P.M. Rapid nutrient cycling in leaf litter from invasive plants in Hawai’i. Oecologia 2004, 141, 612–619. [Google Scholar] [CrossRef]
- Wijesinghe, M.R.; Brooke, M.d.L. Impact of habitat disturbance on the distribution of endemic species of small mammals and birds in a tropical rain forest in Sri Lanka. J. Trop. Ecol. 2005, 21, 661–668. [Google Scholar] [CrossRef]
- Surasinghe, T.D. Conservation overview of herpetofauna of Sinharaja man and biosphere reserve of Sri Lanka. Zoos’Print J. 2007, 22, 2535–2538. [Google Scholar] [CrossRef]
- Diez, J.M.; D’Antonio, C.M.; Dukes, J.S.; Grosholz, E.D.; Olden, J.D.; Sorte, C.J.; Blumenthal, D.M.; Bradley, B.A.; Early, R.; Ibáñez, I. Will extreme climatic events facilitate biological invasions? Front. Ecol. Environ. 2012, 10, 249–257. [Google Scholar] [CrossRef]
- Gallardo, B.; Aldridge, D.C.; González-Moreno, P.; Pergl, J.; Pizarro, M.; Pyšek, P.; Thuiller, W.; Yesson, C.; Vilà, M. Protected areas offer refuge from invasive species spreading under climate change. Glob. Chang. Biol. 2017, 23, 5331–5343. [Google Scholar] [CrossRef] [PubMed]
- Thompson, K.; Hodgson, J.G.; Rich, T.C. Native and alien invasive plants: More of the same? Ecography 1995, 18, 390–402. [Google Scholar] [CrossRef]
- Daehler, C.C. Performance comparisons of co-occurring native and alien invasive plants: Implications for conservation and restoration. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 183–211. [Google Scholar] [CrossRef]
- Bellard, C.; Thuiller, W.; Leroy, B.; Genovesi, P.; Bakkenes, M.; Courchamp, F. Will climate change promote future invasions? Glob. Chang. Biol. 2013, 19, 3740–3748. [Google Scholar] [CrossRef]
- Waser, A.M.; Splinter, W.; Van der Meer, J. Indirect effects of invasive species affecting the population structure of an ecosystem engineer. Ecosphere 2015, 6, 1–12. [Google Scholar] [CrossRef]
- Taylor, S.; Kumar, L. Global climate change impacts on pacific islands terrestrial biodiversity: A review. Trop. Conserv. Sci. 2016, 9, 203–223. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Walker, L.R. Biological invasion by Myrica faya in Hawaii: Plant demography, nitrogen fixation, ecosystem effects. Ecol. Monogr. 1989, 59, 247–265. [Google Scholar] [CrossRef]
- Buckingham, L. Threats to wild cats across international borders. Biodiversity 2019, 20, 41–42. [Google Scholar] [CrossRef]
- De Silva, S.; Webber, C.E.; Weerathunga, U.; Pushpakumara, T.; Weerakoon, D.K.; Wittemyer, G. Demographic variables for wild Asian elephants using longitudinal observations. PLoS ONE 2013, 8, e82788. [Google Scholar] [CrossRef] [PubMed]
- Bachen, D.A.; Litt, A.R.; Gower, C.N. Simulating cheatgrass (Bromus tectorum) invasion decreases access to food resources for small mammals in sagebrush steppe. Biol. Invasions 2018, 20, 2301–2311. [Google Scholar] [CrossRef]
- Bahir, M.M.; Surasinghe, T.D. A conservation assessment of the Sri Lankan Agamidae (Reptilia: Sauria). Raffles Bull. Zool. 2005, 12, 407–412. [Google Scholar]
- Wilson, G.; Gruber, M.A.; Lester, P.J. Foraging relationships between elephants and Lantana camara invasion in Mudumalai Tiger Reserve, India. Biotropica 2014, 46, 194–201. [Google Scholar] [CrossRef]
- Sampson, C.; Leimgruber, P.; Tonkyn, D.; Pastorini, J.; Janaka, H.; Sotherden, E.; Fernando, P. Effects of illegal grazing and invasive Lantana camara on Asian elephant habitat use. Biol. Conserv. 2018, 220, 50–59. [Google Scholar] [CrossRef]
- Regmi, S.; Chalise, M.K. Food Habit and Conservation Threats of Wild Water Buffalo. Nature Khabar. 2019. Available online: http://naturekhabar.com/en/archives/12250 (accessed on 28 August 2019).
- Ullah, H.; Nagelkerken, I.; Goldenberg, S.U.; Fordham, D.A. Climate change could drive marine food web collapse through altered trophic flows and cyanobacterial proliferation. PLoS Biol. 2018, 16, e2003446. [Google Scholar] [CrossRef] [PubMed]
- Schirmel, J.; Bundschuh, M.; Entling, M.H.; Kowarik, I.; Buchholz, S. Impacts of invasive plants on resident animals across ecosystems, taxa, and feeding types: A global assessment. Glob. Chang. Biol. 2016, 22, 594–603. [Google Scholar] [CrossRef]
- Spatz, D.R.; Zilliacus, K.M.; Holmes, N.D.; Butchart, S.H.; Genovesi, P.; Ceballos, G.; Tershy, B.R.; Croll, D.A. Globally threatened vertebrates on islands with invasive species. Sci. Adv. 2017, 3, e1603080. [Google Scholar] [CrossRef]
- Bellard, C.; Rysman, J.-F.; Leroy, B.; Claud, C.; Mace, G.M. A global picture of biological invasion threat on islands. Nat. Ecol. Evol. 2017, 1, 1862–1869. [Google Scholar] [CrossRef]
- Hetem, R.S.; Fuller, A.; Maloney, S.K.; Mitchell, D. Responses of large mammals to climate change. Temperature 2014, 1, 115–127. [Google Scholar] [CrossRef]
- Bickford, D.; Howard, S.D.; Ng, D.J.; Sheridan, J.A. Impacts of climate change on the amphibians and reptiles of Southeast Asia. Biodivers. Conserv. 2010, 19, 1043–1062. [Google Scholar] [CrossRef]
- Gunatilleke, C.V.S.; Gunatilleke, I.A.U.N.; Ethugala, A.U.K.; Esufali, S. Ecology of Sinharaja Rain Forest and the Forest Dynamics Plot in Sri Lanka’s Natural World Heritage Site; WHT Publications: Colombo, Sri Lanka, 2004. [Google Scholar]
- ME&RE. Sri Lanka’s Fifth National Report to the Convention on Biological Diversity; Ministry of Environment & Renewable Energy: Colombo, Sri Lanka, 2014; p. 128.
- DWC. Biodiversity Baseline Survey: Horton Plains National Park; Sri Lanka Protected Areas Management and Wildlife Conservation Project (PAM&WCP/CONSULT/02/BDBS); Department of Wildlife Conservation: Colombo, Sri Lanka, 2007; p. 40.
- Karunarathna, D.M.S.S.; Peabotuwage, P.I.K.; Perera, B.N.H.; Karunatilaka, H.M.A. Second known locality of the Critically Endangered Adenomus dasi Manamendra-Arachchi & Pethiyagoda, 1998 (Bufonidae) from Samanala Nature Reserve, Sri Lanka. Frog Leg 2012, 18, 21. [Google Scholar]
- DWC. Biodiversity Baseline Survey: Bundala National Park; Sri Lanka Protected Areas Management and Wildlife Conservation Project (PAM&WCP/CONSULT/02/BDBS); Department of Wildlife Conservation, Ministry of Environment and Natural Resources: Colombo, Sri Lanka, 2008; p. 46.
- Araújo, M.B.; Anderson, R.P.; Barbosa, A.M.; Beale, C.M.; Dormann, C.F.; Early, R.; Garcia, R.A.; Guisan, A.; Maiorano, L.; Naimi, B. Standards for distribution models in biodiversity assessments. Sci. Adv. 2019, 5, eaat4858. [Google Scholar] [CrossRef] [PubMed]
- Guillera-Arroita, G.; Lahoz-Monfort, J.J.; Elith, J. Maxent is not a presence–absence method: A comment on Thibaud et al. Methods Ecol. Evol. 2014, 5, 1192–1197. [Google Scholar] [CrossRef]
- Masters, G.; Norgrove, L. Climate Change and Invasive Alien Species; CABI Working Paper 1; CABI: Wallingford, UK, 2010; p. 30. [Google Scholar]
- Webber, B.L.; Yates, C.J.; Le Maitre, D.C.; Scott, J.K.; Kriticos, D.J.; Ota, N.; McNeill, A.; Le Roux, J.J.; Midgley, G.F. Modelling horses for novel climate courses: Insights from projecting potential distributions of native and alien Australian acacias with correlative and mechanistic models. Divers. Distrib. 2011, 17, 978–1000. [Google Scholar] [CrossRef]
- Gallardo, B.; Aldridge, D.C.; Frid, C. The ‘dirty dozen’: Socio-economic factors amplify the invasion potential of 12 high-risk aquatic invasive species in Great Britain and Ireland. J. Appl. Ecol. 2013, 50, 757–766. [Google Scholar] [CrossRef]
- Jiménez-Valverde, A.; Peterson, A.T.; Soberón, J.; Overton, J.; Aragón, P.; Lobo, J.M. Use of niche models in invasive species risk assessments. Biol. Invasions 2011, 13, 2785–2797. [Google Scholar] [CrossRef]
- Soberón, J. Grinnellian and Eltonian niches and geographic distributions of species. Ecol. Lett. 2007, 10, 1115–1123. [Google Scholar] [CrossRef]
- Brancatelli, G.I.; Zalba, S.M. Vector analysis: A tool for preventing the introduction of invasive alien species into protected areas. Nat. Conserv. 2018, 24, 43. [Google Scholar] [CrossRef]
- Foxcroft, L.C.; Spear, D.; Van Wilgen, N.J.; McGeoch, M.A. Assessing the association between pathways of alien plant invaders and their impacts in protected areas. NeoBiota 2019, 43, 1. [Google Scholar] [CrossRef]
- Pyšek, P.; Jarošík, V.; Pergl, J. Alien plants introduced by different pathways differ in invasion success: Unintentional introductions as a threat to natural areas. PLoS ONE 2011, 6, e24890. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-J.; Wan, J.-Z.; Qu, H.; Zhang, Z.-X. Modelling plant invasion pathways in protected areas under climate change: Implication for invasion management. Web Ecol. 2017, 17, 69–77. [Google Scholar] [CrossRef]
- Bradley, B.A.; Blumenthal, D.M.; Wilcove, D.S.; Ziska, L.H. Predicting plant invasions in an era of global change. Trends Ecol. Evol. 2010, 25, 310–318. [Google Scholar] [CrossRef] [PubMed]
- Standish, R.J.; Robertson, A.W.; Williams, P.A. The impact of an invasive weed Tradescantia fluminensis on native forest regeneration. J. Appl. Ecol. 2001, 38, 1253–1263. [Google Scholar] [CrossRef]
- Elith, J.; Leathwick, J. Predicting species distributions from museum and herbarium records using multiresponse models fitted with multivariate adaptive regression splines. Divers. Distrib. 2007, 13, 265–275. [Google Scholar] [CrossRef]
- Sekercioglu, C.H. Increasing awareness of avian ecological function. Trends Ecol. Evol. 2006, 21, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Stoner, K.E.; Riba-Hernández, P.; Vulinec, K.; Lambert, J.E. The role of mammals in creating and modifying seedshadows in tropical forests and some possible consequences of their elimination. Biotropica 2007, 39, 316–327. [Google Scholar] [CrossRef]
- Ratnayake, R.S.S.; Kumar, L.; Kariyawasam, C.S. Neglected and Underutilized Fruit Species in Sri Lanka: Prioritisation and Understanding the Potential Distribution under Climate Change. Agronomy 2020, 10, 34. [Google Scholar] [CrossRef]
- Kogo, B.K.; Kumar, L.; Koech, R.; Kariyawasam, C.S. Modelling climate suitability for rainfed Maize cultivation in Kenya using a Maximum Entropy (MaxENT) approach. Agronomy 2019, 9, 727. [Google Scholar] [CrossRef]
- Lavoie, C. The impact of invasive knotweed species (Reynoutria spp.) on the environment: Review and research perspectives. Biol. Invasions 2017, 19, 2319–2337. [Google Scholar] [CrossRef]
- Hulme, P.E.; Pyšek, P.; Pergl, J.; Jarošík, V.; Schaffner, U.; Vilà, M. Greater focus needed on alien plant impacts in protected areas. Conserv. Lett. 2014, 7, 459–466. [Google Scholar] [CrossRef]
| Taxonomic Group | Total Species | Total Threatened | National Conservation Status | ||
|---|---|---|---|---|---|
| CR | EN | VU | |||
| Amphibians | 111 (95) | 73 (71) | 34 (34) | 28 (27) | 10 (9) |
| Freshwater fish | 91 (50) | 45 (39) | 19 (16) | 19 (17) | 5 (4) |
| Reptiles | 211 (124) | 107 (87) | 38 (36) | 50 (39) | 18 (11) |
| Birds | 240 (27) | 67 (18) | 18 | 18 (7) | 31 (11) |
| Mammals (excluding marine) | 95 (21) | 53 (18) | 13 (06) | 25 (08) | 15 (04) |
| Total | 748 (317) | 345 (233) | 122 (92) | 140 (98) | 79 (39) |
| Percentage Area | ||||
|---|---|---|---|---|
| Classification | Area | Current | RCP 4.5_2050 | RCP 8.5_2050 |
| Very high | A | 0.48 | 0.06 | 1.63 |
| B | 0.41 | 0.15 | 0.52 | |
| C | 5.07 | 0.37 | 0.44 | |
| High | A | 6.27 | 5.36 | 9.41 |
| B | 5.19 | 4.08 | 9.17 | |
| C | 17.18 | 10.77 | 18.91 | |
| Moderate | A | 17.59 | 29.28 | 42.2 |
| B | 14.49 | 25.88 | 40.26 | |
| C | 18.57 | 46.45 | 50.3 | |
| Low | A | 23.87 | 43.4 | 31.13 |
| B | 30.95 | 53.12 | 38.96 | |
| C | 22.76 | 31.22 | 23.09 | |
| Very low | A | 51.8 | 21.89 | 15.63 |
| B | 48.97 | 16.77 | 11.09 | |
| C | 36.42 | 11.18 | 7.26 | |
| No | Species | Current | RCP 4.5 2050 | RCP 8.5 2050 | ||
|---|---|---|---|---|---|---|
| Area km2 | Percentage Change | Area km2 | Percentage Change | |||
| 1 | Alstonia macrophylla | 1185 | 1605 | 35 | 1867 | 58 |
| 2 | Annona glabra | 322 | 1354 | 320 | 2012 | 525 |
| 3 | Austroeupatorium inulifolium | 1391 | 967 | −30 | 817 | −41 |
| 4 | Clidemia hirta | 1480 | 925 | −38 | 1045 | −29 |
| 5 | Dillenia suffruticosa | 410 | 874 | 113 | 1096 | 167 |
| 6 | Lantana camara | 5238 | 2884 | −45 | 3816 | −27 |
| 7 | Leucaena leucocephala | 3277 | 3233 | −1 | 5740 | 75 |
| 8 | Mimosa pigra | 1827 | 6809 | 273 | 9484 | 419 |
| 9 | Opuntia dillenii | 3159 | 14,047 | 345 | 16,874 | 434 |
| 10 | Panicum maximum | 5747 | 2015 | −65 | 3035 | −47 |
| 11 | Parthenium hysterophorus | 2559 | 12,267 | 379 | 13,595 | 431 |
| 12 | Prosopis juliflora | 1441 | 1115 | −23 | 3680 | 155 |
| 13 | Sphagneticola trilobata | 2729 | 1512 | −45 | 2157 | −21 |
| 14 | Ulex europaeus | 344 | 0 | −100 | 0 | −100 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kariyawasam, C.S.; Kumar, L.; Ratnayake, S.S. Potential Risks of Plant Invasions in Protected Areas of Sri Lanka under Climate Change with Special Reference to Threatened Vertebrates. Climate 2020, 8, 51. https://doi.org/10.3390/cli8040051
Kariyawasam CS, Kumar L, Ratnayake SS. Potential Risks of Plant Invasions in Protected Areas of Sri Lanka under Climate Change with Special Reference to Threatened Vertebrates. Climate. 2020; 8(4):51. https://doi.org/10.3390/cli8040051
Chicago/Turabian StyleKariyawasam, Champika S., Lalit Kumar, and Sujith S. Ratnayake. 2020. "Potential Risks of Plant Invasions in Protected Areas of Sri Lanka under Climate Change with Special Reference to Threatened Vertebrates" Climate 8, no. 4: 51. https://doi.org/10.3390/cli8040051
APA StyleKariyawasam, C. S., Kumar, L., & Ratnayake, S. S. (2020). Potential Risks of Plant Invasions in Protected Areas of Sri Lanka under Climate Change with Special Reference to Threatened Vertebrates. Climate, 8(4), 51. https://doi.org/10.3390/cli8040051





