Smart Sensing System for Early Detection of Bone Loss: Current Status and Future Possibilities
Abstract
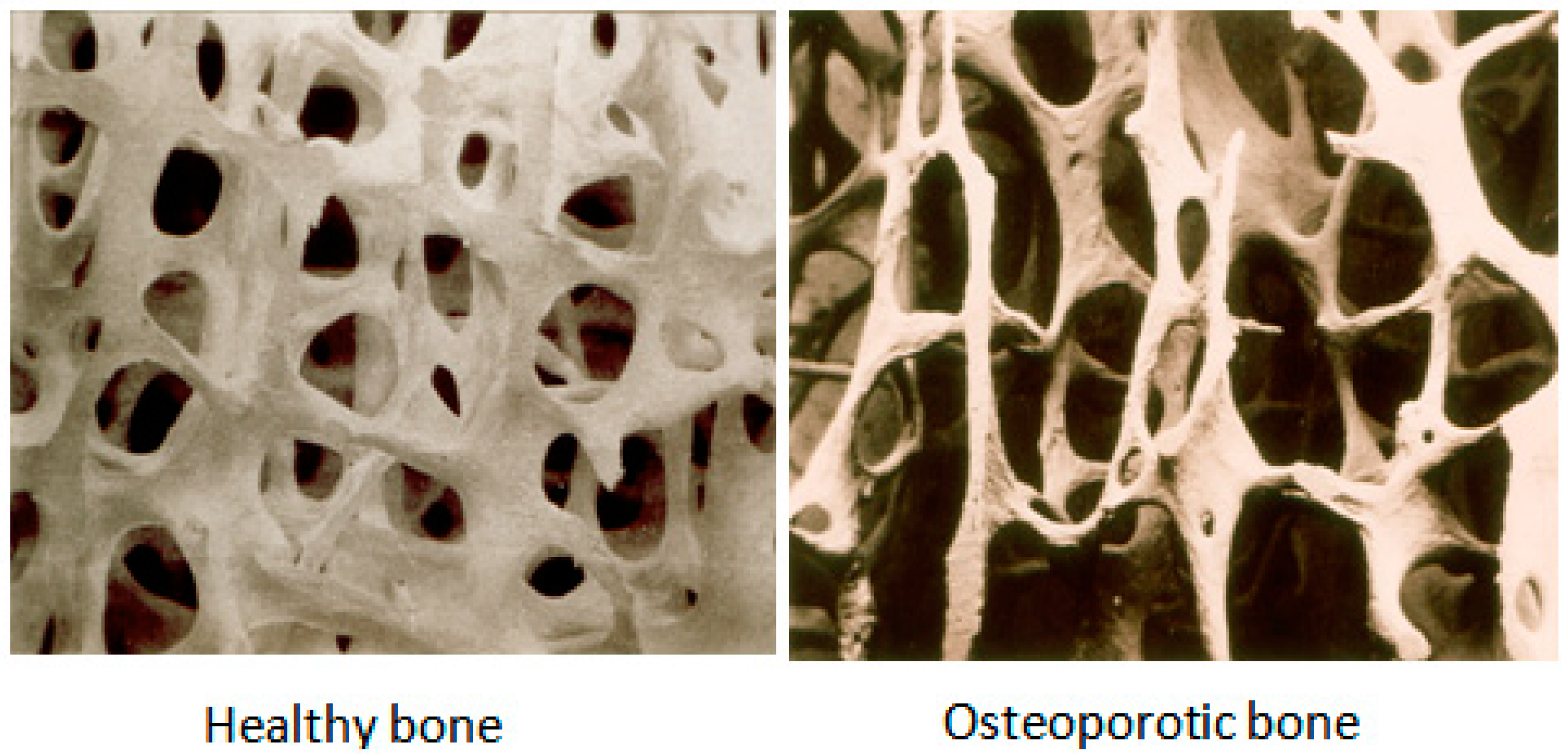
:1. Introduction
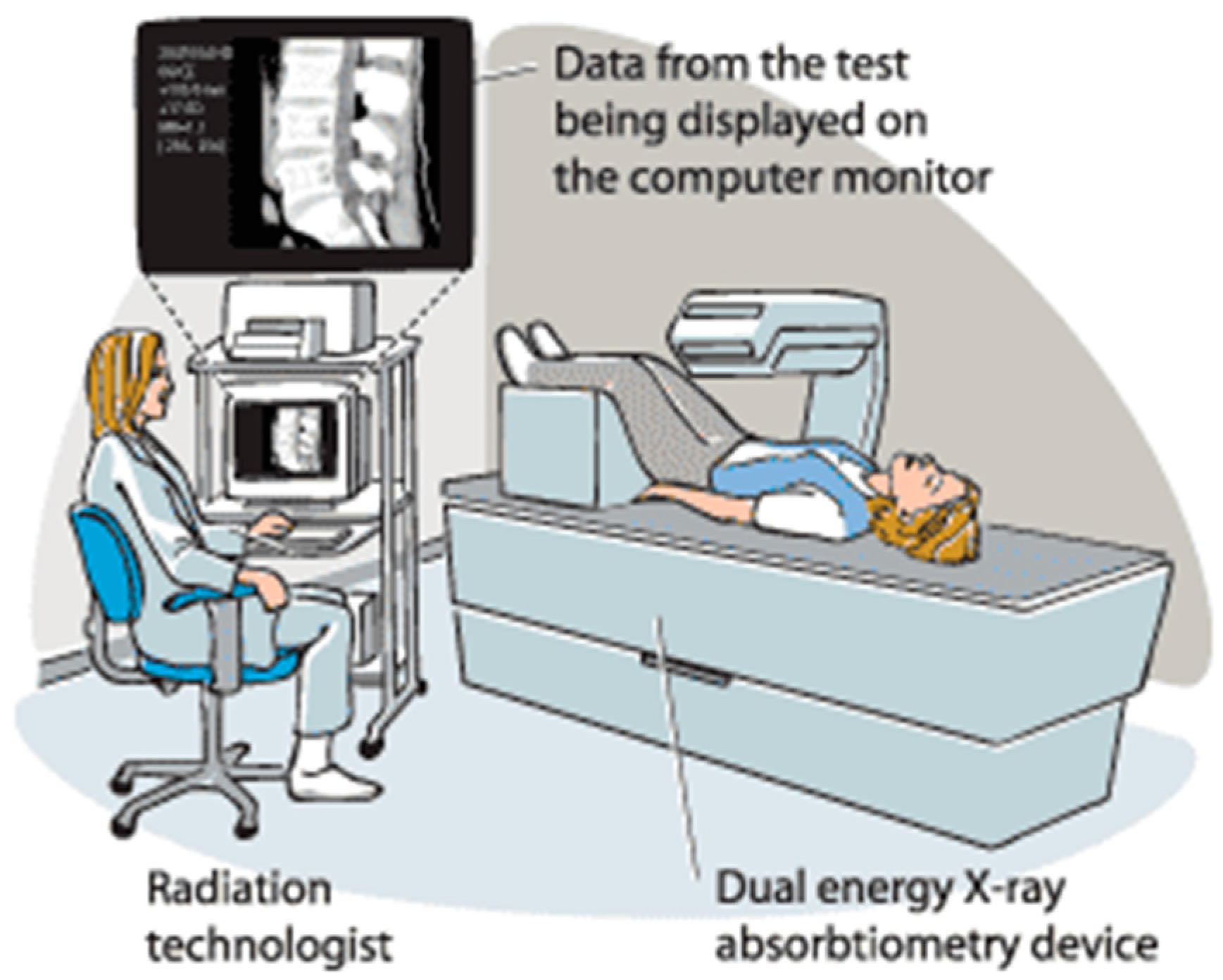
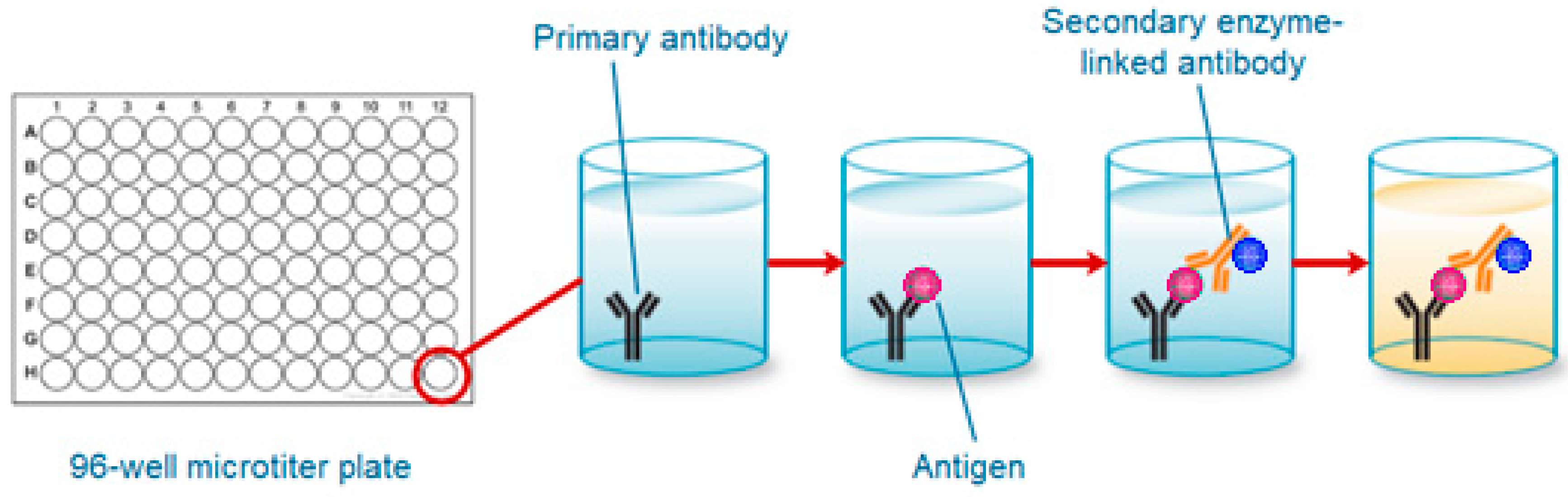
2. Current Methodologies
3. Interdigital Sensor Based Measurement
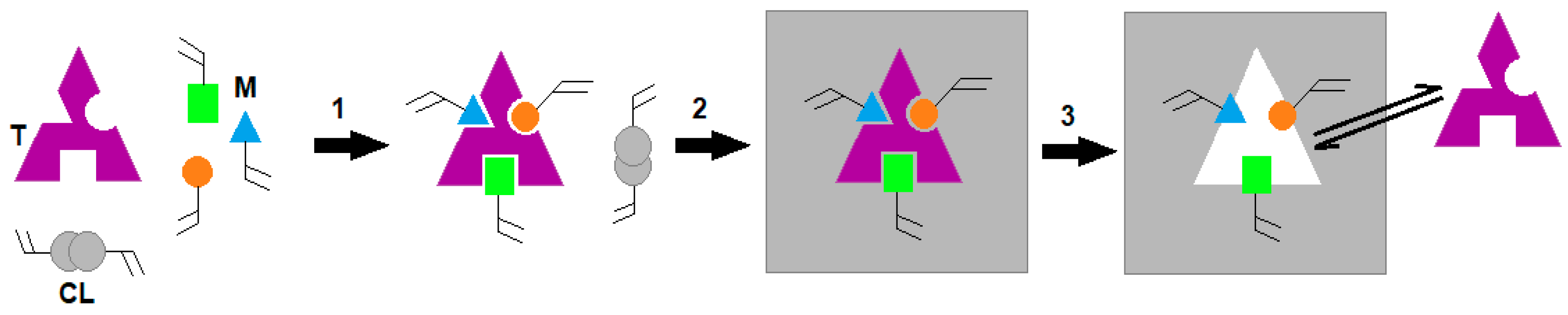
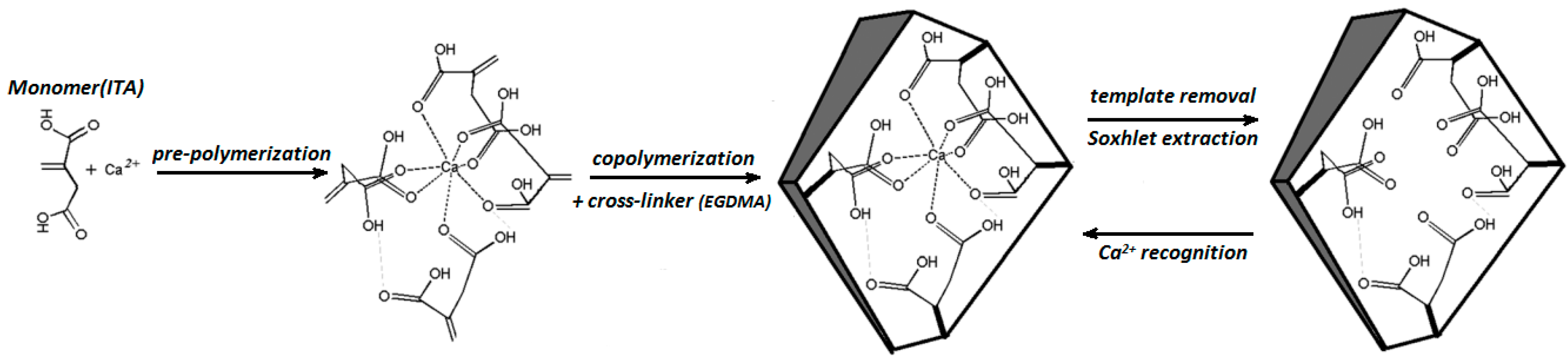
3.1. Synthesis of Artificial Antibodies for CTx-I and Functionalization of the Sensor
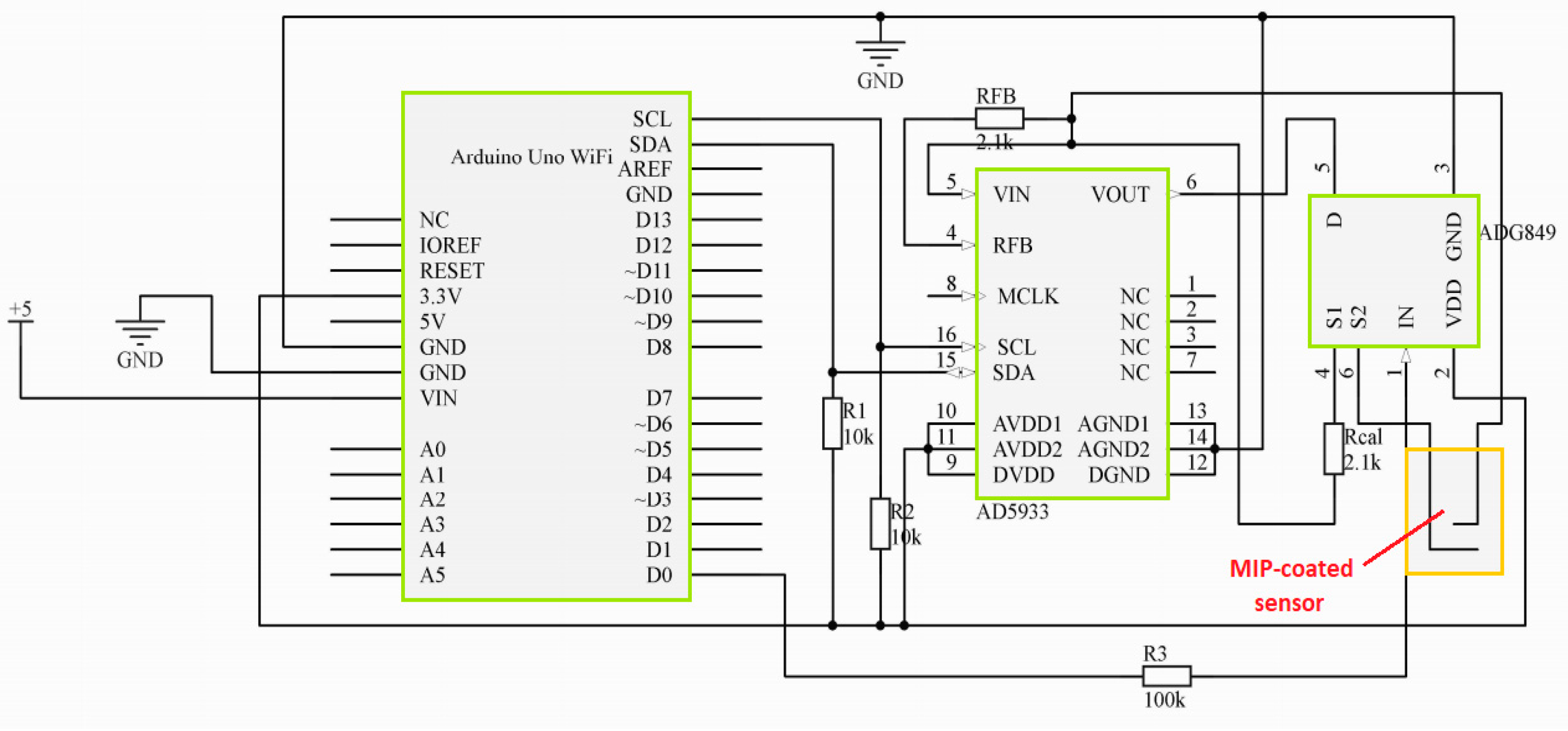
3.2. Development of a Microcontroller-Based System for Impedance Monitoring
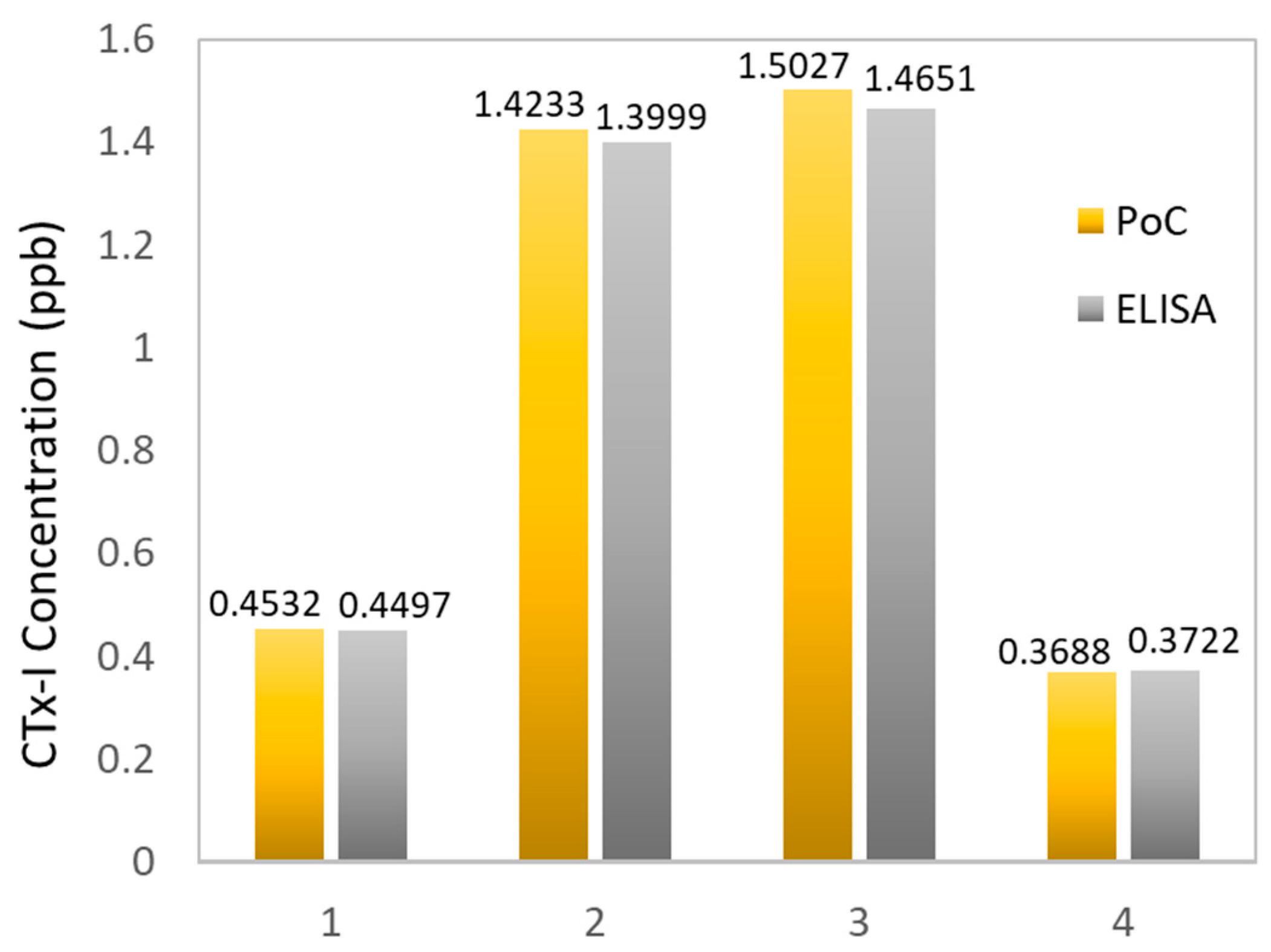
3.3. Development of the Calibration Curve and Real Sample Measurement
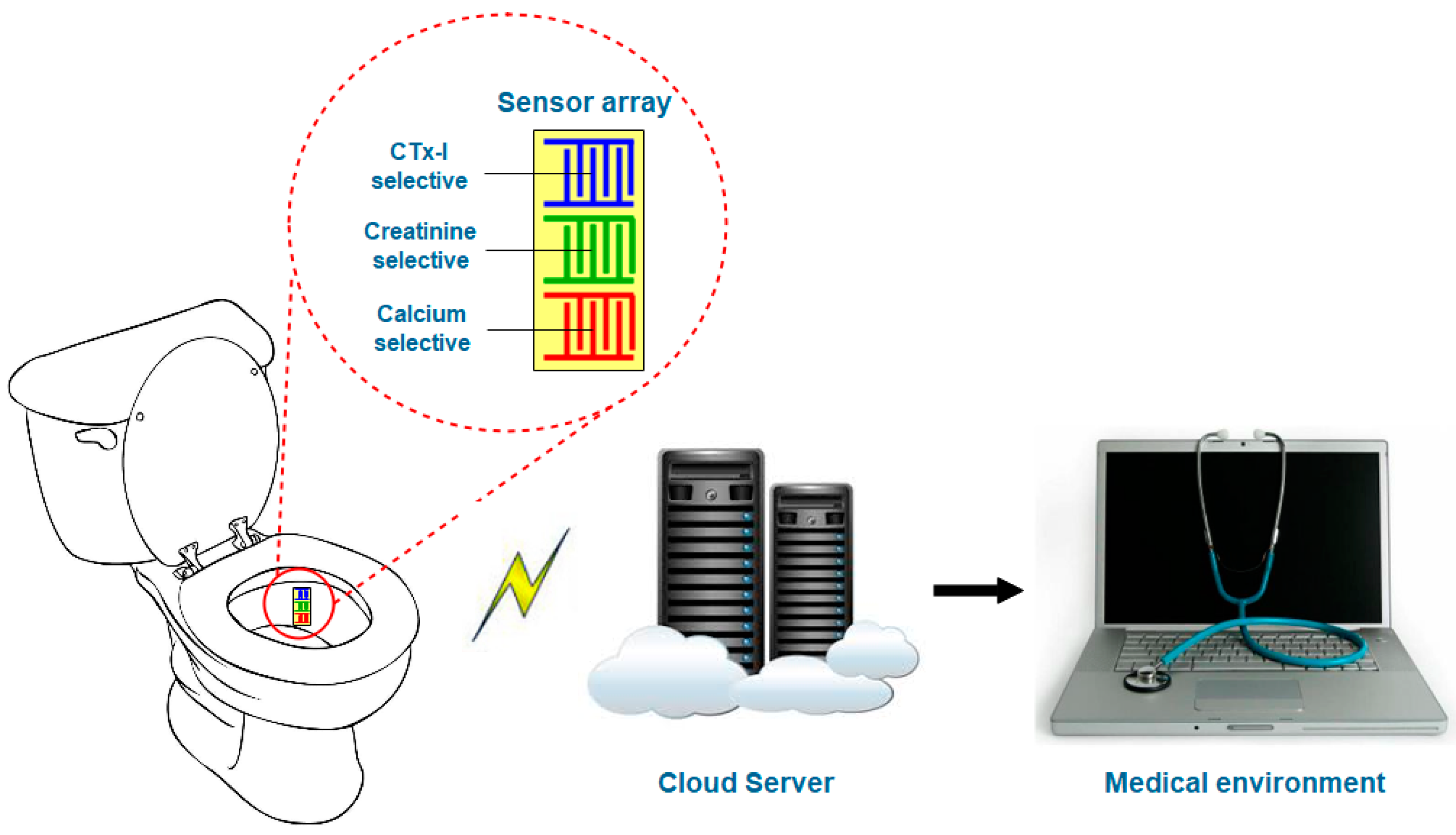
3.4. Future Possibilities
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Rachner, T.D.; Khosla, S.; Hofbauer, L.C. Osteoporosis: Now and the future. Lancet 2011, 377, 1276–1287. [Google Scholar] [CrossRef]
- Oden, A.; McCloskey, E.; Kanis, J.; Harvey, N.; Johansson, H. Burden of high fracture probability worldwide: Secular increases 2010–2040. Osteoporos. Int. 2015, 26, 2243–2248. [Google Scholar] [CrossRef] [PubMed]
- Seibel, M.J.; Robins, S.P.; Bilezikian, J.P. Dynamics of Bone and Cartilage Metabolism: Principles and Clinical Applications; Academic Press: Cambridge, MA, USA, 2006. [Google Scholar]
- Afsarimanesh, N.; Zia, A.I.; Mukhopadhyay, S.C.; Kruger, M.; Yu, P.-L.; Kosel, J.; Kovacs, Z. Smart sensing system for the prognostic monitoring of bone health. Sensors 2016, 16, 976. [Google Scholar] [CrossRef] [PubMed]
- Afsarimanesh, N.; Mukhopadhyay, S.C.; Marlena, K. Biosensors for the measurement of C-terminal telopeptide of type I collagen (CTX-I). J. Osteoporos. Phys. Act. 2017, 5. [Google Scholar] [CrossRef]
- Srivastava, A.; MacFarlane, G.; Srivastava, V.; Mohan, S.; Baylink, D. A new monoclonal antibody ELISA for detection and characterization of C-telopeptide fragments of type I collagen in urine. Calcif. Tissue Int. 2001, 69, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Osteoporosis in RA. Available online: https://www.nras.org.uk/osteoporosis-in-ra (accessed on 23 February 2018).
- Kanis, J.A.; Melton, L.J.; Christiansen, C.; Johnston, C.C.; Khaltaev, N. The diagnosis of osteoporosis. J. Bone Miner. Res. 1994, 9, 1137–1141. [Google Scholar] [CrossRef] [PubMed]
- Bone Mineral Density Scan. Available online: http://www.medbroadcast.com/procedure/getprocedure/bone-mineral-density-scan (accessed on 23 February 2018).
- Schuit, S.; Van der Klift, M.; Weel, A.; De Laet, C.; Burger, H.; Seeman, E.; Hofman, A.; Uitterlinden, A.; Van Leeuwen, J.; Pols, H. Fracture incidence and association with bone mineral density in elderly men and women: The rotterdam study. Bone 2004, 34, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Eastell, R.; Barton, I.; Hannon, R.; Chines, A.; Garnero, P.; Delmas, P. Relationship of early changes in bone resorption to the reduction in fracture risk with risedronate. J. Bone Miner. Res. 2003, 18, 1051–1056. [Google Scholar] [CrossRef] [PubMed]
- Aydin, S. A short history, principles, and types of ELISA, and our laboratory experience with peptide/protein analyses using ELISA. Peptides 2015, 72, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Lequin, R.M. Enzyme immunoassay (eia)/enzyme-linked immunosorbent assay (ELISA). Clin. Chem. 2005, 51, 2415–2418. [Google Scholar] [CrossRef] [PubMed]
- Engvall, E. The ELISA, enzyme-linked immunosorbent assay. Clin. Chem. 2010, 56, 319–320. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, M.; Patil, M.; Epur, R.; Yun, Y.; Shanov, V.; Schulz, M.; Heineman, W.R.; Datta, M.K.; Kumta, P.N. Gold-coated carbon nanotube electrode arrays: Immunosensors for impedimetric detection of bone biomarkers. Biosens. Bioelectron. 2016, 77, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.-H.; Bhattacharya, A.; Watts, N.B.; Schulz, M.J. A label-free electronic biosensor for detection of bone turnover markers. Sensors 2009, 9, 7957–7969. [Google Scholar] [CrossRef] [PubMed]
- Mamishev, A.V.; Sundara-Rajan, K.; Fumin, Y.; Yanqing, D.; Zahn, M. Interdigital sensors and transducers. Proc. IEEE 2004, 92, 808–845. [Google Scholar] [CrossRef]
- Rahman, M.S.A.; Mukhopadhyay, S.C.; Yu, P.-L. Novel Sensors for Food Inspection: Modelling, Fabrication and Experimentation; Springer: Basel, Switzerland, 2014. [Google Scholar]
- Zia, A.I.; Rahman, M.S.A.; Mukhopadhyay, S.C.; Yu, P.-L.; Al-Bahadly, I.H.; Gooneratne, C.P.; Kosel, J.; Liao, T.-S. Technique for rapid detection of phthalates in water and beverages. J. Food Eng. 2013, 116, 515–523. [Google Scholar] [CrossRef]
- Haupt, K.; Linares, A.V.; Bompart, M.; Bui, B.T.S. Molecularly imprinted polymers. In Molecular Imprinting; Haupt, K., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 1–28. [Google Scholar]
- Whitcombe, M.J.; Kirsch, N.; Nicholls, I.A. Molecular imprinting science and technology: A survey of the literature for the years 2004–2011. J. Mol. Recognit. 2014, 27, 297–401. [Google Scholar] [PubMed]
- Rechichi, A.; Cristallini, C.; Vitale, U.; Ciardelli, G.; Barbani, N.; Vozzi, G.; Giusti, P. New biomedical devices with selective peptide recognition properties. Part 1: Characterization and cytotoxicity of molecularly imprinted polymers. J. Cell. Mol. Med. 2007, 11, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- Afsarimanesh, N.; Alahi, M.; Mukhopadhyay, S.; Kruger, M.; Yu, P.-L. Development of molecular imprinted polymer interdigital sensor for C-terminal telopeptide of type I collagen. In Proceedings of the 2016 10th International Conference on Sensing Technology (ICST), Nanjing, China, 11–13 November 2016; pp. 1–5. [Google Scholar]
- Afsarimanesh, N.; Mukhopadhyay, S.C.; Kruger, M. Molecularly imprinted polymer-based electrochemical biosensor for bone loss detection. IEEE Trans. Biomed. Eng. 2017. [Google Scholar] [CrossRef] [PubMed]
- Devices, A. AD5933: Impedance Analyzer. Available online: http://www.analog.com/media/en/technical-documentation/data-sheets/AD5933.pdf (accessed on 4 September 2017).
- Mankar, J.; Darode, C.; Trivedi, K.; Kanoje, M.; Shahare, P. Review of I2C protocol. Int. J. 2014, 2, 474–479. [Google Scholar]
- Thingspeak. Available online: https://thingspeak.com/ (accessed on 26 August 2017).
- Website, A. Ciao. Available online: https://www.arduino.cc/en/Reference/Ciao (accessed on 26 August 2017).
- Urine Cartilaps. Available online: http://peramed.com/peramed/docs/AC-10F1_EN.pdf (accessed on 23 February 2018).
- Khadro, B.; Betatache, A.; Sanglar, C.; Bonhommé, A.; Errachid, A.; Jaffrezic-Renault, N. Molecularly imprinted polymers (MIP) based electrochemical sensor for detection of urea and creatinine. Sens. Lett. 2011, 9, 2261–2264. [Google Scholar] [CrossRef]
- Lee, M.-H.; Tsai, T.-C.; Thomas, J.L.; Lin, H.-Y. Recognition of creatinine by poly (ethylene-co-vinylalcohol) molecular imprinting membrane. Desalination 2008, 234, 126–133. [Google Scholar] [CrossRef]
- Khashayar, P.; Aghaei Meybodi, H.; Amoabediny, G.; Larijani, B. Biochemical markers of bone turnover and their role in osteoporosis diagnosis: A narrative review. Recent Pat. Endocr. Metab. Immune Drug Discov. 2015, 9, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Petra, S.; Gerald, A. Multiplexed point-of-care testing-xpoct. Trends Biotechnol. 2017, 35, 728–742. [Google Scholar]
- Piraino, F.; Volpetti, F.; Watson, C.; Maerkl, S.J. A digital–analog microfluidic platform for patient-centric multiplexed biomarker diagnostics of ultralow volume samples. ACS Nano 2016, 10, 1699–1710. [Google Scholar] [CrossRef] [PubMed]
- Piraino, F. Diagnostic Devices with Microfluidics; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Branger, C.; Meouche, W.; Margaillan, A. Recent advances on ion-imprinted polymers. React. Funct. Polym. 2013, 73, 859–875. [Google Scholar] [CrossRef]
- Alizadeh, T.; Shamkhali, A.N.; Hanifehpour, Y.; Joo, S.W. A Ca2+ selective membrane electrode based on calcium-imprinted polymeric nanoparticles. New J. Chem. 2016, 40, 8479–8487. [Google Scholar] [CrossRef]

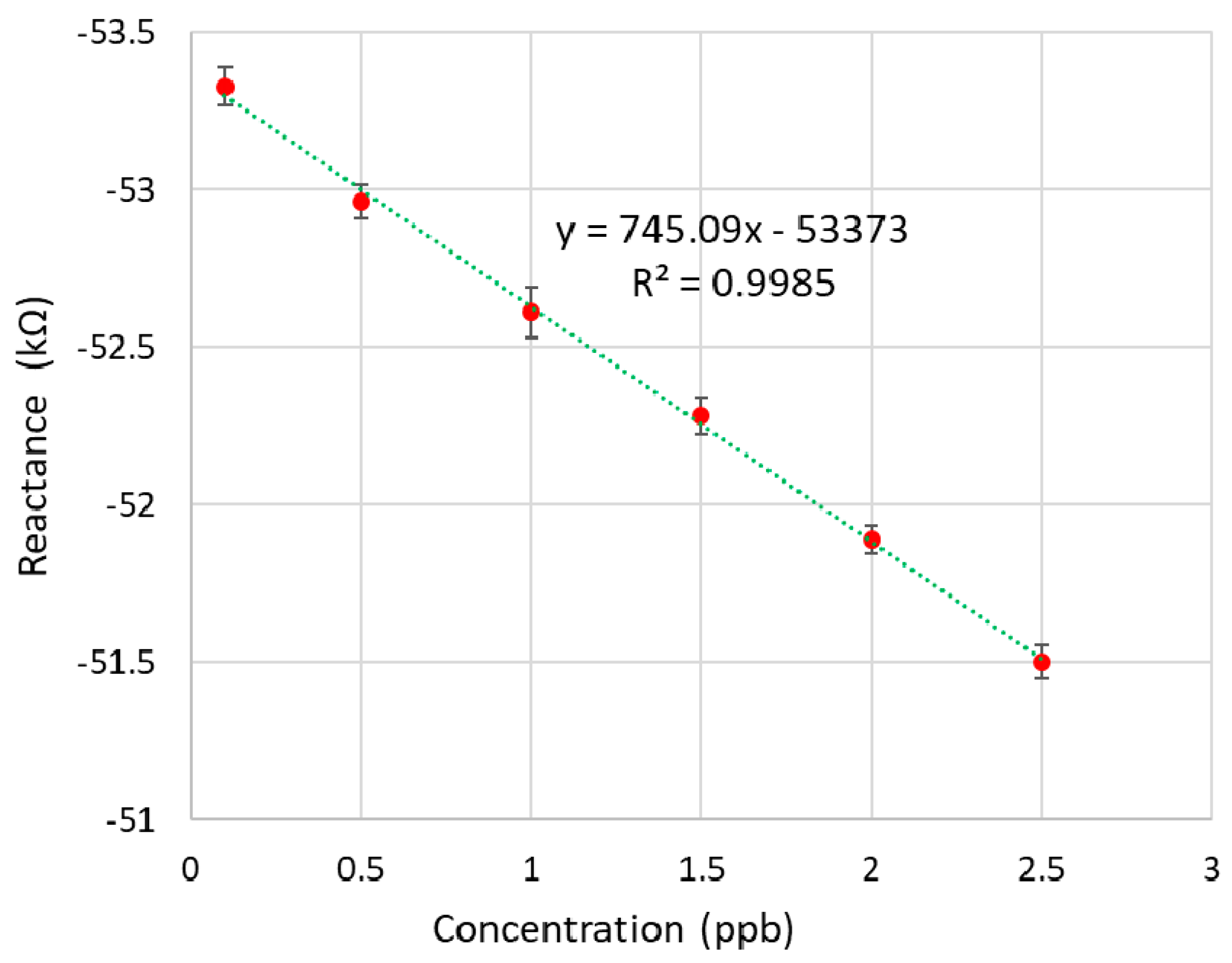

| CTx-I Concentration | Reactance (kΩ) * | Standard Deviation (Ω) |
|---|---|---|
| 0.1 | −53.36 | 60.02 |
| 0.5 | −52.94 | 53.61 |
| 1 | −52.60 | 79.34 |
| 1.5 | −52.25 | 58.34 |
| 2 | −51.87 | 44.75 |
| 2.5 | −51.53 | 54.33 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Afsarimanesh, N.; Alahi, M.E.E.; Mukhopadhyay, S.C.; Kruger, M. Smart Sensing System for Early Detection of Bone Loss: Current Status and Future Possibilities. J. Sens. Actuator Netw. 2018, 7, 10. https://doi.org/10.3390/jsan7010010
Afsarimanesh N, Alahi MEE, Mukhopadhyay SC, Kruger M. Smart Sensing System for Early Detection of Bone Loss: Current Status and Future Possibilities. Journal of Sensor and Actuator Networks. 2018; 7(1):10. https://doi.org/10.3390/jsan7010010
Chicago/Turabian StyleAfsarimanesh, Nasrin, Md Eshrat E Alahi, Subhas Chandra Mukhopadhyay, and Marlena Kruger. 2018. "Smart Sensing System for Early Detection of Bone Loss: Current Status and Future Possibilities" Journal of Sensor and Actuator Networks 7, no. 1: 10. https://doi.org/10.3390/jsan7010010
APA StyleAfsarimanesh, N., Alahi, M. E. E., Mukhopadhyay, S. C., & Kruger, M. (2018). Smart Sensing System for Early Detection of Bone Loss: Current Status and Future Possibilities. Journal of Sensor and Actuator Networks, 7(1), 10. https://doi.org/10.3390/jsan7010010








