Use of Non-Destructive Measurements to Identify Cucurbit Species (Cucurbita maxima and Cucurbita moschata) Tolerant to Waterlogged Conditions
Abstract
:1. Introduction
2. Results
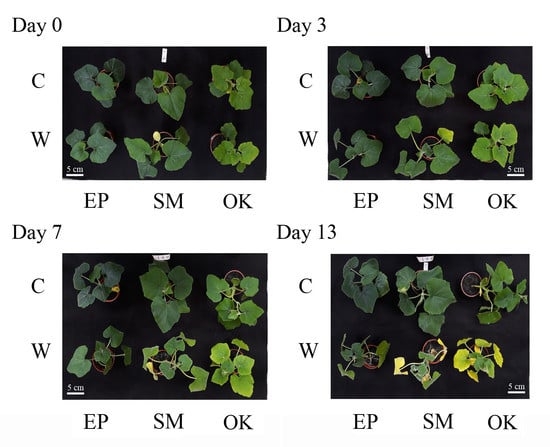
2.1. Waterlogging Effects on Growth Traits in Squashes
2.2. Physiological Responses of Squash Plants to Waterlogging Stress
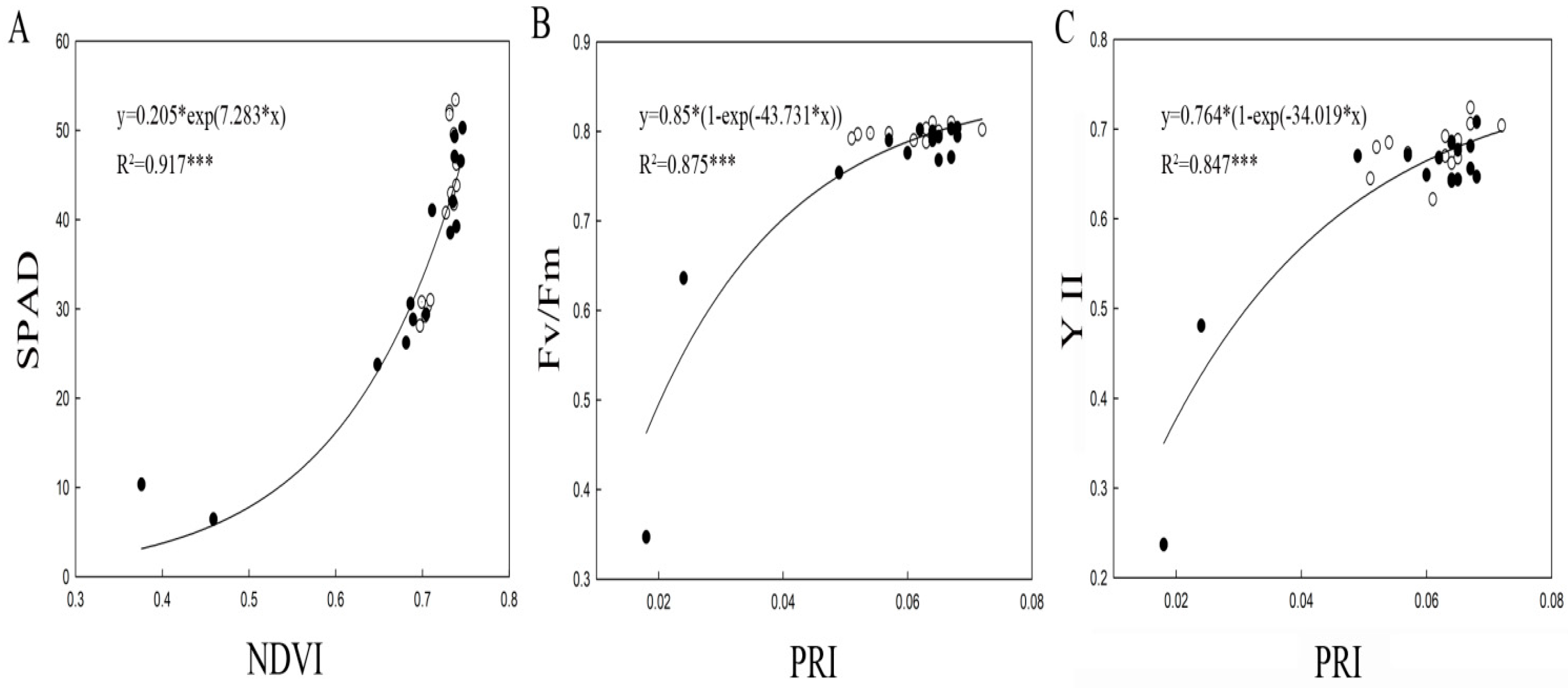
2.3. Relationships Among SPAD, ChlF, and Spectral Reflectance Variables in Squashes
3. Discussion
4. Materials and Methods
4.1. Plant Materials, Cultivation, and Waterlogging Treatments
4.2. Plant Growth Measurements
4.3. Relative Water Content (RWC)
4.4. Determination of Electrolyte leakage (EL), Chlorophyll Content (CC), Chlorophyll Fluorescence (ChlF), and Spectral Reflectance
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Henriques, F.; Guine, R.; Barroca, M.J. Chemical properties of pumpkin dried by different methods. Croat. J. Food Technol. Biotechnol. Nutr. 2012, 7, 98–105. [Google Scholar]
- Ara, N.; Korakot, N.; Lv, W.; Yang, J.; Hu, Z.; Zhang, M. Antioxidant enzymatic activities and gene expression associated with heat tolerance in the stems and roots of two cucurbit species (“Cucurbita maxima” and “Cucurbita moschata”) and their interspecific inbred line “Maxchata”. Int. J. Mol. Sci. 2013, 14, 24008–24028. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chiang, C.M.; Chen, C.C.; Chen, S.P.; Lin, K.H.; Chen, L.R.; Su, Y.H.; Yen, H.C. Overexpression of the ascorbate peroxidase gene from eggplant and sponge gourd enhances flood tolerance in transgenic Arabidopsis. J. Plant Res. 2017, 130, 373–386. [Google Scholar] [CrossRef] [PubMed]
- Aydinli, G.; Kurtar, E.S.; Mennan, S. Screening of Cucurbita maxima and Cucurbita moschata genotypes for resistance against Meloidogyne arenaria, M. incognita, M. javanica, and M. luci. J. Nematol. 2019, 51, e2019–e2057. [Google Scholar] [CrossRef] [Green Version]
- Biswas, J.; Kalra, N. Effect of waterlogging and submergence on crop physiology and growth of different crops and its remedies: Bangladesh Perspectives. Saudi J. Eng. Technol. 2018, 3, 315–329. [Google Scholar]
- Zhou, W.; Chen, F.; Meng, Y.; Chandrasekaran, U.; Luo, X.; Yang, W.; Shu, K. Plant waterlogging/flooding stress responses: From seed germination to maturation. Plant Physiol. Biochem. 2020, 148, 228–236. [Google Scholar] [CrossRef]
- Huynh, N.L.; Van Toai, T.; Streeter, J.; Banowetz, G. Regulation of flooding tolerance of SAG12: Ipt Arabidopsis plants by cytokinin. J. Exp. Bot. 2005, 56, 1397–1407. [Google Scholar] [CrossRef] [Green Version]
- Fukao, T.; Bailey-Serres, J. Ethylene—A key regulator of submergence responses in rice. Plant Sci. 2008, 175, 43–51. [Google Scholar] [CrossRef]
- Schwarz, D.; Rouphael, Y.; Colla, G.; Venema, J.H. Grafting as a tool to improve tolerance of vegetables to abiotic stresses: Thermal stress, water stress and organic pollutants. Sci. Hortic. 2010, 127, 162–171. [Google Scholar] [CrossRef]
- Djanaguiraman, M.; Prasad, P.V.; Seppanen, M. Selenium protects sorghum leaves from oxidative damage under high temperature stress by enhancing antioxidant defense system. Plant Physiol. Biochem. 2010, 48, 999–1007. [Google Scholar] [CrossRef]
- Skotnica, J.; Matoušková, M.; Nauš, J. Thermo luminescence and fluorescence study of changes in Photosystem II photochemistry in desiccating barley leaves. Photosynth. Res. 2000, 65, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K.; Rinderle, U. The role of chlorophyll fluorescence in the detection of stress conditions in plants. CRC Crit. Rev. Anal. Chem. 1998, 19, S29–S85. [Google Scholar] [CrossRef]
- Levizou, E.; Drilias, P.; Psaras, G.K.; Maneta, Y. Nondestructiveassessment of leaf chemistry and physiology through spectral reflectancemeasurements may be misleading when changes in trichome density cooccur. New Phytol. 2005, 165, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Ballester, C.; Zarco-Tejada, P.J.; Nicola, E.; Alarco, J.J.; Fereres, E.; Intrigliolo, D.; Gonzalez-Dugo, V. Evaluating the performance of xanthophyll, chlorophyll and structure-sensitive spectral indices to detect water stress in five fruit tree species. Precis. Agric. 2018, 19, 178–193. [Google Scholar] [CrossRef]
- Yudina, L.; Ekaterina, S.; Ekaterina, G.; Vladimir, N.; Vladimir, V.; Vladimir, S. A light-induced decrease in the photochemical reflectance index (PRI) can be used to estimate the energy-dependent component of non-photochemical quenching under heat stress and soil drought in pea, wheat, and pumpkin. Photosynth. Res. 2020. [Google Scholar] [CrossRef]
- Gamon, J.A.; Kovalchuck, O.; Wong, C.Y.S.; Harris, A.; Garrity, S.R. Monitoring seasonal and diurnal changes in photosynthetic pigments with automated PRI and NDVI sensors. Biogeosciences 2015, 12, 4149–4159. [Google Scholar] [CrossRef] [Green Version]
- Hernández-Clemente, R.; Navarro-Cerrillo, R.; Morales, F.; Zarco-Tejada, P.J. Assessing structural effects on PRI for stress detection in conifer forests. Remote Sens. Environ. 2011, 115, 2360–2375. [Google Scholar] [CrossRef]
- Zheng, C.; Jiang, D.; Liu, F.; Dai, T.; Qi, J.; Cao, W. Effects of salt and waterlogging stresses and their combination on leaf photosynthesis, chloroplast ATP synthesis, and antioxidant capacity in wheat. Plant Sci. 2009, 176, 575–582. [Google Scholar] [CrossRef]
- Bailey-Serres, J.; Fukao, T.; Ronald, P.; Ismail, A.; Heuer, S.; Mackill, D. Submergence tolerant rice: SUB1′s Journey from landrace to modern cultivar. Rice 2010, 3, 138–147. [Google Scholar] [CrossRef] [Green Version]
- Ling, Q.; Huang, W.; Jarvis, P. Use of a SPAD-502 m to measure leaf chlorophyll concentration in Arabidopsis thaliana. Photosynth. Res. 2011, 107, 209–214. [Google Scholar] [CrossRef] [Green Version]
- Netto, A.T.; Campostrini, E.; Oliveira, J.G.; Bressan-Smith, R.E. Photosynthetic pigments, nitrogen, chlorophyll a fluorescence and SPAD-502 readings in coffee leaves. Sci. Hortic. 2005, 104, 199–209. [Google Scholar] [CrossRef]
- Liu, L.; Yang, X.; Zhou, H.; Liu, S.; Zhou, L.; Li, X.; Yang, J.; Wu, J. Relationship of root zone soil moisture with solar-induced chlorophyll fluorescence and vegetation indices in winter wheat: A comparative study based on continuous ground-measurements. Ecol. Indic. 2018, 90, 9–17. [Google Scholar] [CrossRef]
- Jiménez, J.D.l.C.; Cardoso, J.A.; Leiva, L.F.; Gil, J.; Forero, M.G.; Worthington, M.L.; Miles, J.W.; Rao, I.M. Non-destructive Phenotyping to Identify Brachiaria Hybrids Tolerant to Waterlogging Stress under Field Conditions. Front. Plant Sci. 2017, 8, 167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Zou, X.; Shi, J.; Hao, L. In vivo noninvasive detection ofchlorophyll distribution in cucumber (Cucumis sativus) leavesby indices based on hyperspectral imaging. Anal. Chim. Acta 2011, 706, 105–112. [Google Scholar]
- Camejo, D.; Jimenez, A.; Alarcon, J.J.; Torres, W.; Gomez, J.M.; Sevilla, F. Changes in photosynthetic parameters and antioxidant activities following heat-shock treatment in tomato plants. Funct. Plant Biol. 2006, 33, 177–187. [Google Scholar] [CrossRef]
- Diao, M.; Ma, L.; Wang, J.; Cui, J.; Fu, A.; Liu, H.Y. Selenium promotes the growth and photosynthesis of tomato seedlings under salt stress by enhancing chloroplast antioxidant defense system. J. Plant Growth Regul. 2014, 33, 671–682. [Google Scholar] [CrossRef]
- He, L.; Li, Y.; Bin, L.; Nanshan, D.; Guo, S. The effect of exogenous calcium oncucumber fruit quality, photosynthesis, chlorophyll fluorescence, and fast chlorophyll fluorescence during the fruiting period under hypoxic stress. BMC Plant Biol. 2018, 18, 180. [Google Scholar] [CrossRef]
- Pozo, A.D.; Méndez-Espinoza, A.M.; Romero-Bravo, S.; Garriga, M.; Estrada, F.; Alcaíno, M.; Camargo-Rodriguez, A.V.; Corke, F.M.K.; Doonan, J.H.; Lobos, G.A. Genotypic variations in leaf and whole-plant water use efficiencies are closely related in bread wheat genotypes under well-watered and water-limited conditions during grain filling. Sci. Rep. 2020, 10, 460. [Google Scholar] [CrossRef] [Green Version]
- Lin, K.H.; Kuo, W.S.; Chiang, C.M.; Hsiung, T.C.; Chiang, M.C.; Lo, H.F. Study of sponge gourd ascorbate peroxidase and winter squash superoxide dismutase under respective flooding and chilling stresses. Sci. Hortic. 2013, 162, 333–340. [Google Scholar] [CrossRef] [Green Version]
- Hoagland, D.R.; Arnon, D.I. The water-culture for growing plants without soil. Calif. Agric. Exp. Stat. Circ. 1950, 347, 32. [Google Scholar]
- Yamasaki, S.; Dillenburg, L.R. Measurements of leaf relative water content in Araucaria angustifolia. Rev. Bras. Fisiol. Veg. 1999, 11, 69–75. [Google Scholar]
- Huang, M.; Guo, Z. Responses of antioxidative system to chilling stress in two rice cultivars differing in sensitivity. Biol. Plant 2005, 49, 81–84. [Google Scholar] [CrossRef]
- Weng, J.H.; Jhaung, L.H.; Lin, R.J.; Chen, H.Y. Relationship between photochemical efficiency of photosystem II and the photochemical reflectance index of mango tree: Merging data from different illuminations, seasons and leaf colors. Tree Physiol. 2010, 30, 469–478. [Google Scholar]
- Jia, M.; Li, D.; Colombo, R.; Wang, Y.; Wang, X.; Cheng, T.; Zhu, Y.; Yao, X.; Xu, C.; Ouer, G.; et al. Quantifying Chlorophyll Fluorescence Parameters from Hyperspectral Reflectance at the Leaf Scale under Various Nitrogen Treatment Regimes in Winter Wheat. Remote Sens. 2019, 11, 2838. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Filella, I.; Garbulsky, M.F.; Penuelas, J. Affecting factors and recent improvements of the photochemical reflectance index (PRI) for remotely sensing foliar, canopy and ecosystemic radiation-use efficiencies. Remote Sens. 2016, 8, 677. [Google Scholar] [CrossRef] [Green Version]


| Recover | ||||||
|---|---|---|---|---|---|---|
| Phenotype | Tissue | Cultivar | C | W | C | W |
| Fresh weight (g) | shoot | EP | 11.01 ± 1.17 a | 9.07 ± 1.57 bc | 9.16 ± 1.04 bc | 8.98 ± 1.29 bc |
| SM | 9.63 ± 1.89 bc | 9.01 ± 1.68 bc | 7.40 ± 0.40 cd | 8.40 ± 1.81 bcd | ||
| OK | 10.87 ± 1.90 ab | 8.54 ± 1.02 bcd | 8.09 ± 1.63 bcd | 7.16 ± 1.16 d | ||
| root | EP | 1.82 ± 0.90 bc | 2.45 ± 0.61 bc | 1.55 ± 0.57 cd | 3.73 ± 0.35 a | |
| SM | 1.34 ± 0.76 cd | 2.37 ± 0.74 bc | 1.31 ± 0.24 cd | 3.16 ± 0.74 ab | ||
| OK | 1.41 ± 0.67 cd | 2.33 ± 0.16 bc | 1.12 ± 0.34 d | 1.62 ± 0.29 cd | ||
| Dry weight (g) | shoot | EP | 1.07 ± 0.28 cd | 1.53 ± 0.37 bc | 1.37 ± 0.42 bc | 1.79 ± 0.20 a |
| SM | 0.77 ± 0.29 d | 0.77 ± 0.41 d | 1.37 ± 0.34 bc | 1.60 ± 0.51 ab | ||
| OK | 0.84 ± 0.18 d | 1.21 ± 0.20 cd | 0.81 ± 0.27 d | 1.45 ± 0.42 bc | ||
| root | EP | 0.16 ± 0.06 bcd | 0.17 ± 0.03 bc | 0.13 ± 0.07 bcd | 0.27 ± 0.05 a | |
| SM | 0.09 ± 0.06 de | 0.19 ± 0.07 b | 0.12 ± 0.03 bcd | 0.28 ± 0.11 a | ||
| OK | 0.09 ± 0.05 de | 0.12 ± 0.03 bcd | 0.07 ± 0.03 e | 0.11 ± 0.04 cde | ||
| Dry matter (%) | shoot | EP | 9.56 ± 1.24 fg | 16.76 ± 2.99 cd | 18.45 ± 2.91 bc | 22.84 ± 2.86 a |
| SM | 8.14 ± 0.81 g | 16.87 ± 3.14 cd | 14.86 ± 2.72 de | 20.15 ± 2.56 a | ||
| OK | 7.73 ± 0.72 g | 13.45 ± 0.57 de | 10.06 ± 1.00 f | 14.54 ± 2.34 de | ||
| root | EP | 7.16 ± 1.30 b | 6.97 ± 0.73 bc | 8.53 ± 1.62 ab | 11.58 ± 1.69 a | |
| SM | 6.20 ± 1.39 bc | 7.85 ± 1.18 ab | 9.10 ± 1.07 ab | 7.36 ± 1.86 b | ||
| OK | 5.79 ± 0.75 c | 5.62 ± 1.12 c | 5.92 ± 1.54 c | 6.55 ± 1.10 bc | ||
| Root/shoot FW (%) | EP | 20.01 ± 3.51 c | 26.85 ± 2.71 b | 16.63 ± 4.35 cd | 42.16 ± 4.44 a | |
| SM | 14.61 ± 4.43 de | 27.72 ± 4.79 b | 17.80 ± 3.97 cd | 42.97 ± 4.32 a | ||
| OK | 12.83 ± 2.13 e | 24.95 ± 3.74 bc | 14.27 ± 3.40 de | 20.29 ± 2.16 c | ||
| Root/shoot DW (%) | EP | 14.68 ± 2.62 b | 11.49 ± 2.79 bc | 9.41 ± 2.73 cd | 19.33 ± 3.38 a | |
| SM | 11.14 ± 3.07 bc | 13.95 ± 3.77 bc | 8.69 ± 2.18 cd | 15.77 ± 3.21 b | ||
| OK | 9.68 ± 2.18 cd | 9.96 ± 1.61 bcd | 8.37 ± 2.49 cd | 8.02 ± 1.47 d | ||
| Duration (day) | |||||||
|---|---|---|---|---|---|---|---|
| Index | Cultivar | Treatment | 0 | 1 | 3 | 7 | 13 |
| SPAD | EP | C | 46.24 ± 3.27 bc | 49.58 ± 2.13 bc | 53.42 ± 4.07 a | 52.11 ± 2.02 a | 51.78 ± 4.27 ab |
| W | 46.54 ± 3.36 bc | 49.31 ± 2.72 bc | 50.27 ± 2.94 ab | 47.05 ± 2.03 bc | 41.04 ± 3.22 cd | ||
| SM | C | 42.14 ± 3.31 bcd | 41.74 ± 3.65 bcd | 43.86 ± 3.32 bc | 42.96 ± 2.05 bc | 40.77 ± 3.67 cd | |
| W | 42.02 ± 3.57 bcd | 39.23 ± 3.70 cde | 38.51 ± 2.62 cde | 30.59 ± 3.05 de | 10.35 ± 2.02 g | ||
| OK | C | 29.21 ± 3.51 de | 28.11 ± 3.16 de | 30.31 ± 3.87 de | 31.00 ± 4.09 de | 30.77 ± 2.09 de | |
| W | 29.41 ± 3.90 de | 28.81 ± 3.64 de | 26.20 ± 2.93 ef | 23.75 ± 2.55 f | 6.45 ± 1.16 h | ||
| Duration (day) | |||||||
|---|---|---|---|---|---|---|---|
| Index | Cultivar | Treatment | 0 | 1 | 3 | 7 | 13 |
| Fv/Fm | EP | C | 0.798 ± 0.006 ab | 0.798 ± 0.008 ab | 0.798 ± 0.009 ab | 0.797 ± 0.006 ab | 0.792 ± 0.012 ab |
| W | 0.795 ± 0.007 ab | 0.800 ± 0.014 a | 0.790 ± 0.013 ab | 0.768 ± 0.033 ab | 0.754 ± 0.042 b | ||
| SM | C | 0.794 ± 0.014 ab | 0.795 ± 0.006 ab | 0.800 ± 0.007 a | 0.803 ± 0.010 a | 0.790 ± 0.018 ab | |
| W | 0.794 ± 0.006 ab | 0.795 ± 0.009 ab | 0.771 ± 0.020 ab | 0.776 ± 0.023 ab | 0.636 ± 0.077 c | ||
| OK | C | 0.807 ± 0.007 a | 0.810 ± 0.007 a | 0.810 ± 0.006 a | 0.802 ± 0.007 a | 0.788 ± 0.025 ab | |
| W | 0.804 ± 0.007 a | 0.803 ± 0.007 a | 0.802 ± 0.009 a | 0.790 ± 0.014 ab | 0.347 ± 0.013 d | ||
| Y II | EP | C | 0.683 ± 0.055 bc | 0.673 ± 0.022 bc | 0.685 ± 0.042 bc | 0.680 ± 0.043 bc | 0.645 ± 0.026 c |
| W | 0.686 ± 0.044 bc | 0.642 ± 0.012 c | 0.671 ± 0.038 bc | 0.644 ± 0.058 c | 0.670 ± 0.055 bc | ||
| SM | C | 0.688 ± 0.034 bc | 0.662 ± 0.025 bc | 0.668 ± 0.032 bc | 0.670 ± 0.042 bc | 0.622 ± 0.064 c | |
| W | 0.677 ± 0.024 bc | 0.647 ± 0.014 c | 0.656 ± 0.061 c | 0.649 ± 0.048 c | 0.481 ± 0.070 d | ||
| OK | C | 0.706 ± 0.025 ab | 0.684 ± 0.017 bc | 0.724 ± 0.020 a | 0.704 ± 0.028 ab | 0.692 ± 0.019 bc | |
| W | 0.708 ± 0.028 ab | 0.681 ± 0.011 bc | 0.668 ± 0.030 bc | 0.644 ± 0.044 c | 0.237 ± 0.013 e | ||
| Duration (day) | |||||||
|---|---|---|---|---|---|---|---|
| Index | Cultivar | Treatment | 0 | 1 | 3 | 7 | 13 |
| NDVI | EP | C | 0.739 ± 0.009 ab | 0.736 ± 0.008 ab | 0.738 ± 0.007 ab | 0.731 ± 0.010 ab | 0.731 ± 0.015 ab |
| W | 0.744 ± 0.007 a | 0.737 ± 0.011 ab | 0.746 ± 0.014 a | 0.737 ± 0.011 ab | 0.711 ± 0.016 bc | ||
| SM | C | 0.734 ± 0.017 ab | 0.736 ± 0.008 ab | 0.739 ± 0.008 ab | 0.733 ± 0.017 ab | 0.727 ± 0.016 ab | |
| W | 0.735 ± 0.014 ab | 0.739 ± 0.011 ab | 0.732 ± 0.011 ab | 0.686 ± 0.021 de | 0.376 ± 0.017 h | ||
| OK | C | 0.703 ± 0.019 cd | 0.697 ± 0.025 de | 0.706 ± 0.014 cd | 0.709 ± 0.018 cd | 0.699 ± 0.020 de | |
| W | 0.704 ± 0.017 cd | 0.689 ± 0.020 de | 0.681 ± 0.039 de | 0.648 ± 0.021 f | 0.459 ± 0.080 g | ||
| PRI | EP | C | 0.065 ± 0.007 bc | 0.057 ± 0.008 c | 0.054 ± 0.010 c | 0.052 ± 0.007 c | 0.051 ± 0.010 cd |
| W | 0.064 ± 0.005 bc | 0.064 ± 0.005 bc | 0.057 ± 0.006 c | 0.065 ± 0.004 bc | 0.049 ± 0.004 d | ||
| SM | C | 0.065 ± 0.009 bc | 0.064 ± 0.004 bc | 0.065 ± 0.008 bc | 0.063 ± 0.007 bc | 0.061 ± 0.008 bc | |
| W | 0.065 ± 0.009 bc | 0.068 ± 0.006 bc | 0.067 ± 0.005 bc | 0.060 ± 0.006 bc | 0.024 ± 0.013 e | ||
| OK | C | 0.067 ± 0.004 bc | 0.064 ± 0.004 bc | 0.067 ± 0.006 bc | 0.072 ± 0.004 a | 0.063 ± 0.009 bc | |
| W | 0.068 ± 0.002 bc | 0.067 ± 0.003 bc | 0.062 ± 0.004 bc | 0.064 ± 0.004 bc | 0.018 ± 0.013 e | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, H.-H.; Lin, K.-H.; Huang, M.-Y.; Su, Y.-R. Use of Non-Destructive Measurements to Identify Cucurbit Species (Cucurbita maxima and Cucurbita moschata) Tolerant to Waterlogged Conditions. Plants 2020, 9, 1226. https://doi.org/10.3390/plants9091226
Lin H-H, Lin K-H, Huang M-Y, Su Y-R. Use of Non-Destructive Measurements to Identify Cucurbit Species (Cucurbita maxima and Cucurbita moschata) Tolerant to Waterlogged Conditions. Plants. 2020; 9(9):1226. https://doi.org/10.3390/plants9091226
Chicago/Turabian StyleLin, Hsin-Hung, Kuan-Hung Lin, Meng-Yuan Huang, and Yi-Ru Su. 2020. "Use of Non-Destructive Measurements to Identify Cucurbit Species (Cucurbita maxima and Cucurbita moschata) Tolerant to Waterlogged Conditions" Plants 9, no. 9: 1226. https://doi.org/10.3390/plants9091226