Biosynthesis and Industrial Production of Androsteroids
Abstract
:1. Introduction
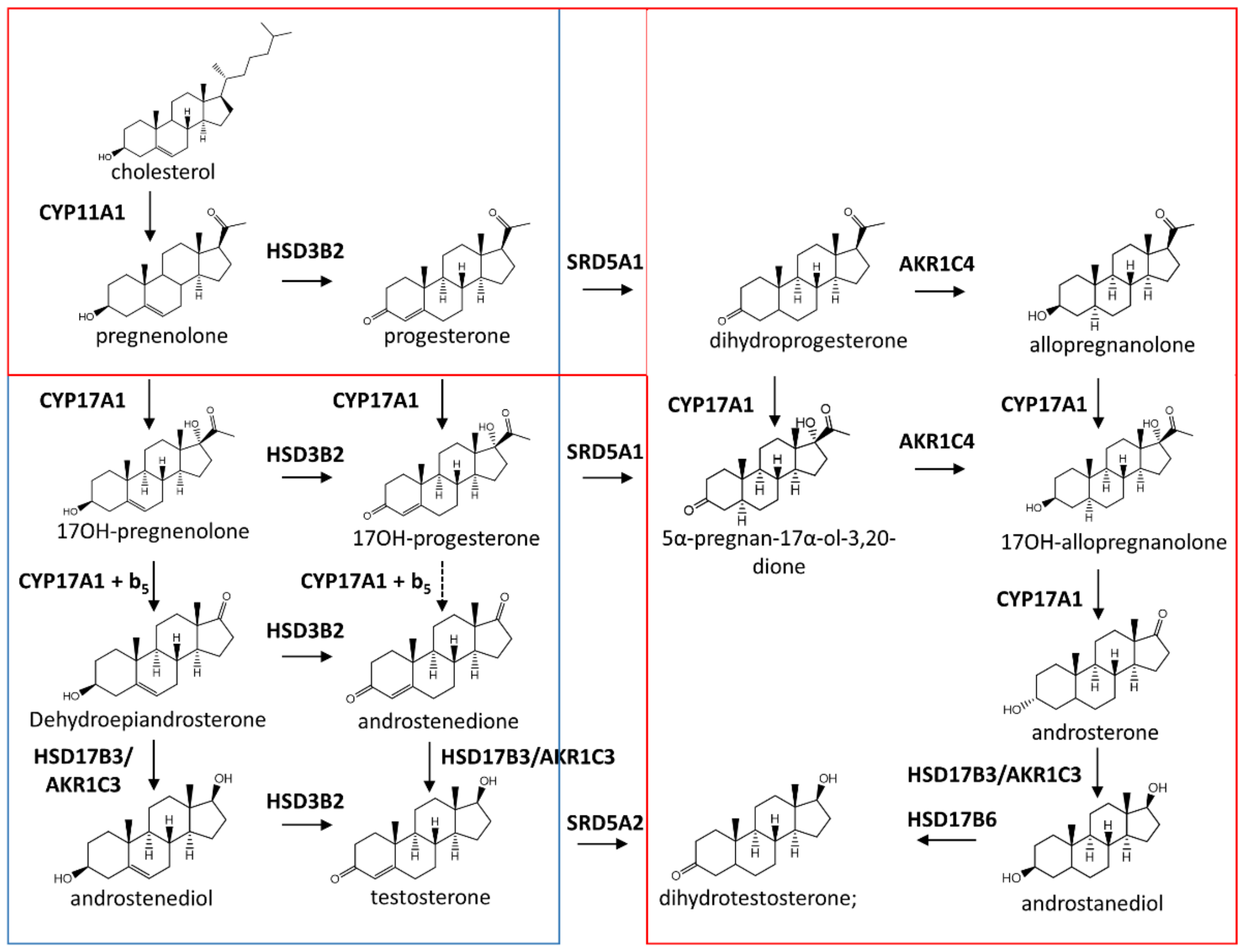
2. Biosynthesis of Steroid Hormones in Mammals
3. Heterologous Production of Androsteroids
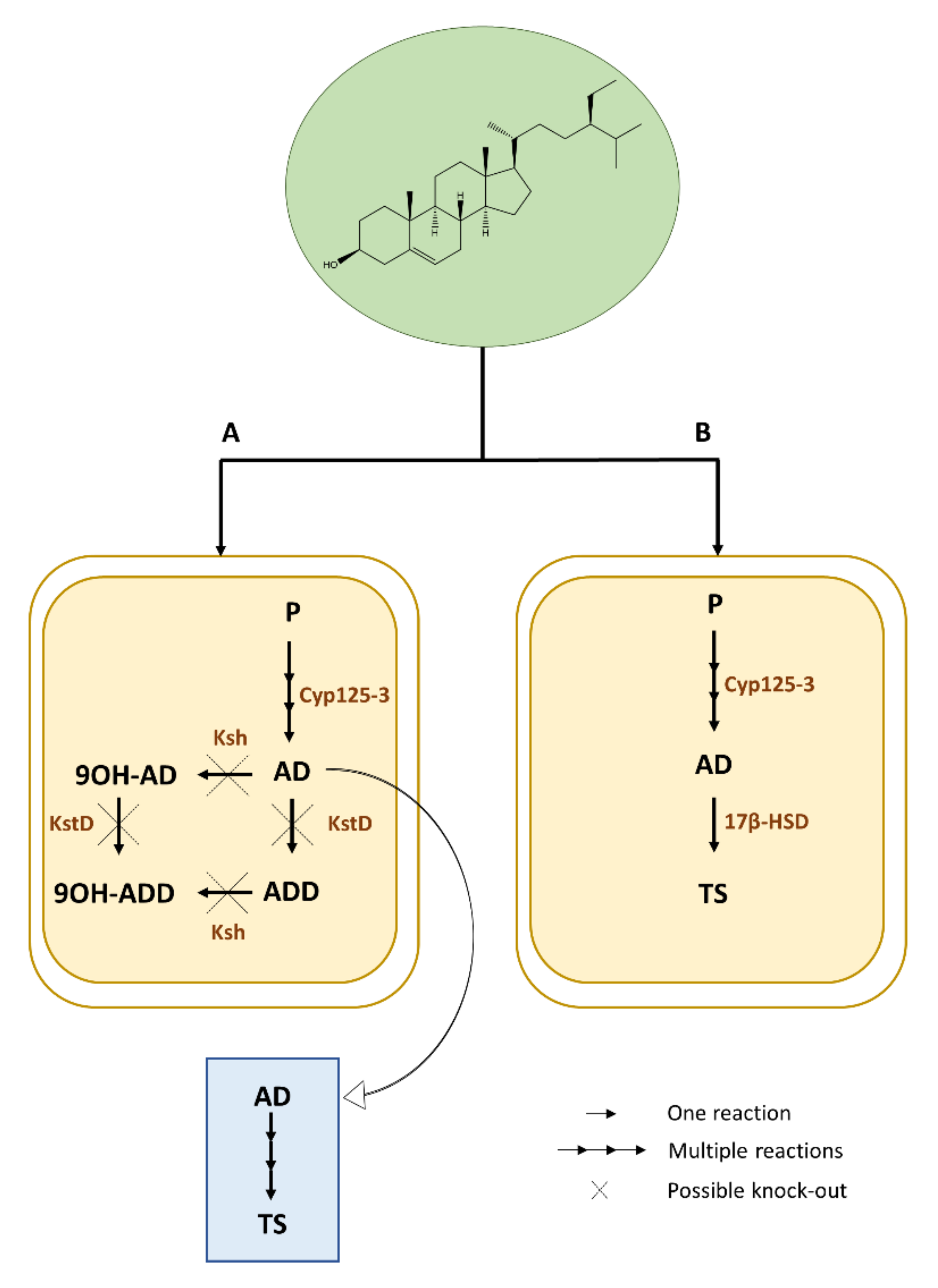
4. Microbial Biotransformation for Production of Steroids
5. Recent Developments in Microbial Biotransformation
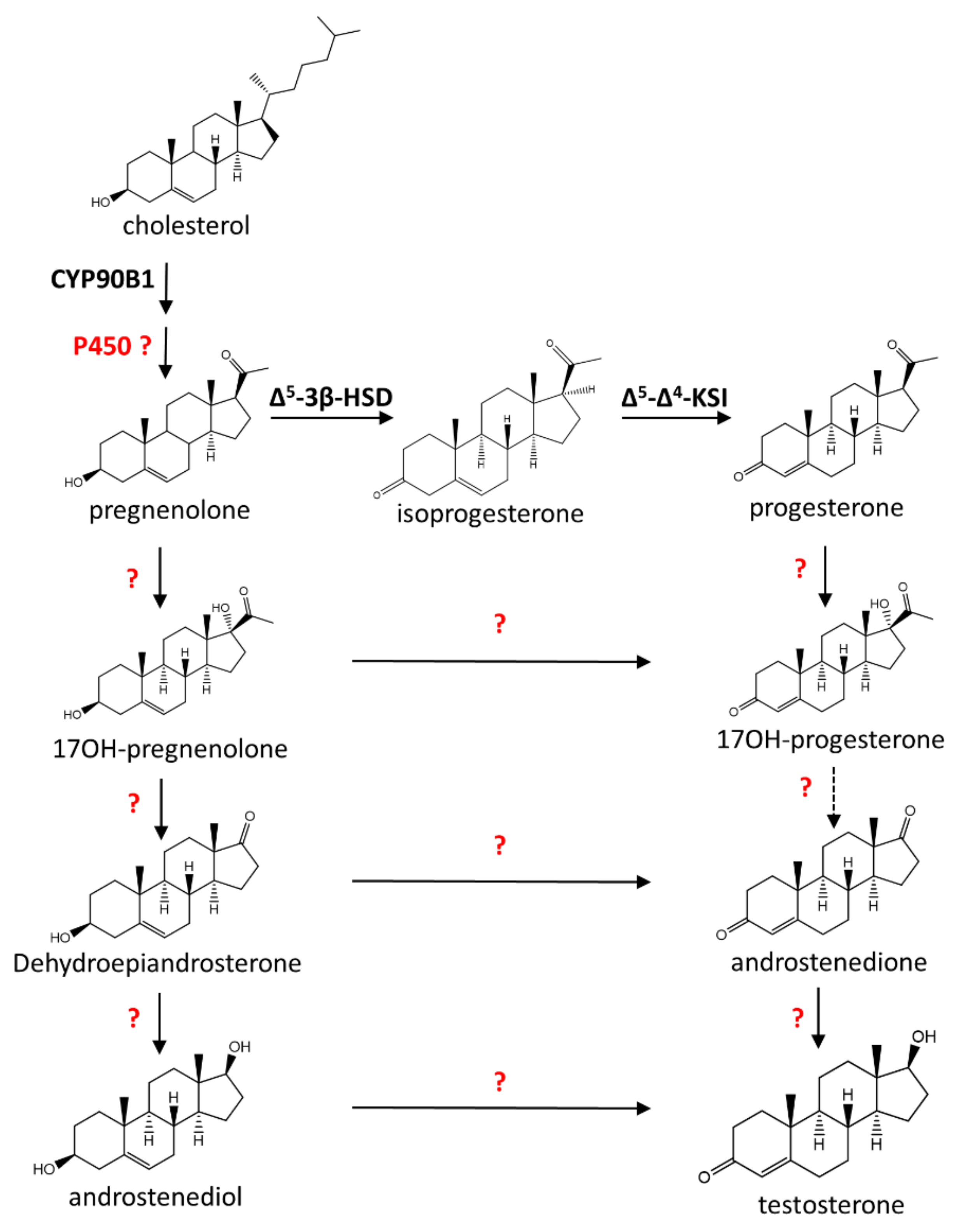
6. Biosynthesis of Androsteroids in Plants
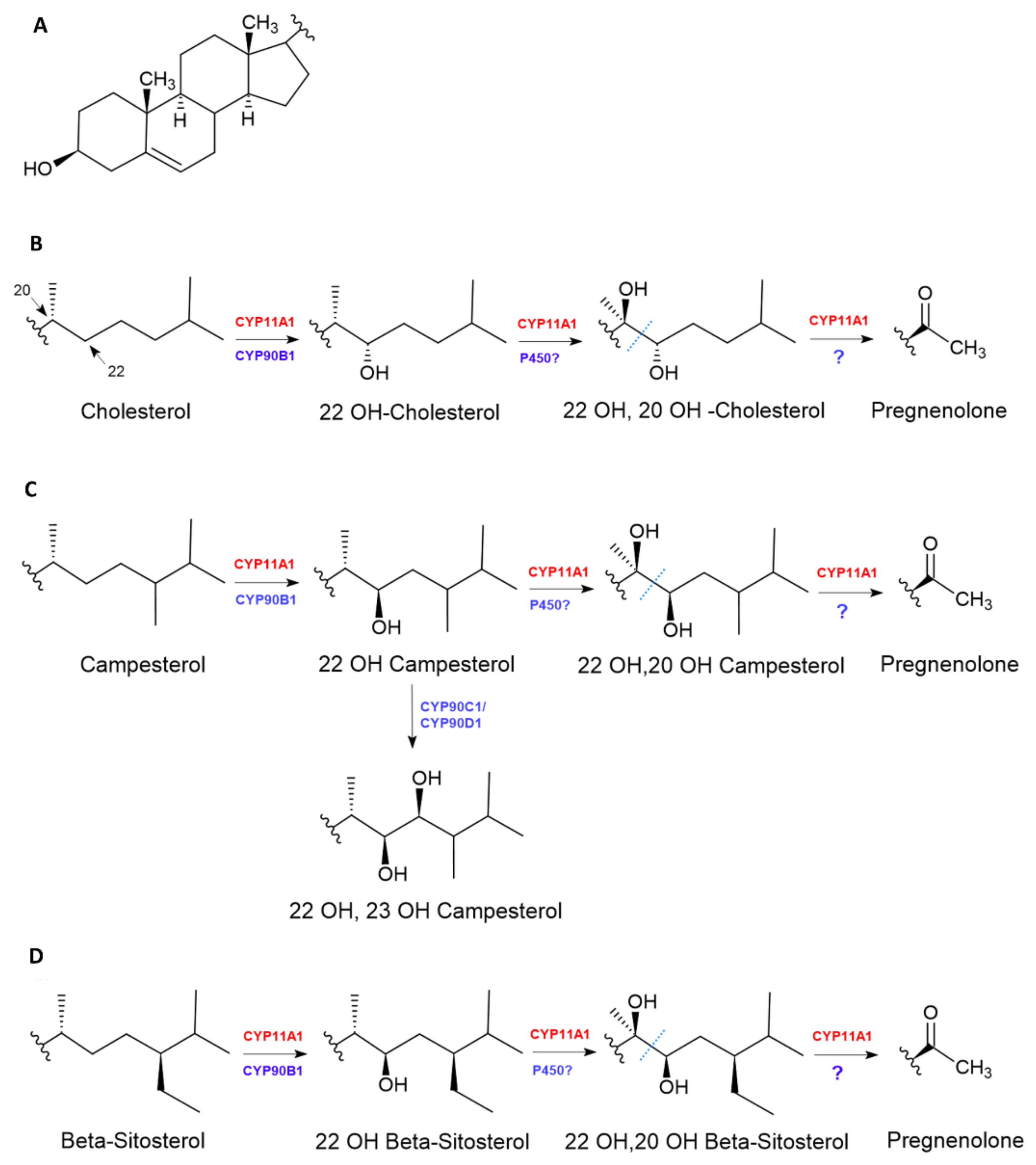
7. Biosynthesis of Pregnenolone and Progesterone in Plants
Author Contributions
Funding
Conflicts of Interest
References
- Pelley, J.W. Elsevier’s integrated biochemistry. Elsevier’s Integr. Biochem. 2006. [Google Scholar] [CrossRef]
- Holst, J.P.; Soldin, O.P.; Guo, T.; Soldin, S.J. Steroid hormones: Relevance and measurement in the clinical laboratory. Clin. Lab. Med. 2004. [Google Scholar] [CrossRef] [Green Version]
- Augustyn, A. Aldosterone|Definition, Structure, Function, & Facts|Britannica.com; Encyclopaedia Britannica: 2018. Available online: https://www.britannica.com/science/aldosterone (accessed on 4 August 2020).
- Miller, W.L. The syndrome of 17,20 lyase deficiency. J. Clin. Endocrinol. Metab. 2012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Becker, K.L.; Bilezikian, J.P.; Bremmer, W.J.; Hung, W.; Kahn, C.R.; Loriaux, D.L.; Nylen, E.S.; Rebar, R.W.; Robertson, G.L.; Snider, R.H. Principles and Practice of Endocrinology and Metabolism, on CD-ROM, 3rd ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2002. [Google Scholar]
- Ravel, R. Clinical Laboratory Medicine, 6th ed.; Mosby-Year Book, Inc.: St. Louis, MI, USA, 1995. [Google Scholar]
- Miller, W.L. Molecular Biology of Steroid Hormone Synthesis. Endocr. Rev. 1988, 9, 295–318. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.L.; Auchus, R.J. The Molecular Biology, Biochemistry, and Physiology of Human Steroidogenesis and Its Disorders. Endocr. Rev. 2011, 32, 81–151. [Google Scholar] [CrossRef] [Green Version]
- Dufort, I.; Rheault, P.; Huang, X.F.; Soucy, P.; Luu-The, V. Characteristics of a highly labile human type 5 17beta-hydroxysteroid dehydrogenase. Endocrinology 1999, 140, 568–574. [Google Scholar] [CrossRef] [PubMed]
- Luu-The, V.; Dufort, I.; Pelletier, G.; Labrie, F. Type 5 17beta-hydroxysteroid dehydrogenase: Its role in the formation of androgens in women. Mol. Cell. Endocrinol. 2001, 171, 77–82. [Google Scholar] [CrossRef]
- Auchus, R.J.; Lee, T.C.; Miller, W.L. Cytochrome b5 Augments the 17,20-Lyase Activity of Human P450c17 without Direct Electron Transfer. J. Biol. Chem. 1998, 273, 3158–3165. [Google Scholar] [CrossRef] [Green Version]
- Geuns, J.M.C. Steroid hormones and plant growth and development. Phytochemistry 1978, 17, 1–14. [Google Scholar] [CrossRef]
- Tarkowská, D. Plants are Capable of Synthesizing Animal Steroid Hormones. Molecules 2019, 24, 2585. [Google Scholar] [CrossRef] [Green Version]
- Tong, W.-Y.; Dong, X. Microbial Biotransformation: Recent Developments on Steroid Drugs. Recent Patents Biotechnol. 2009, 3, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Bureik, M.; Bernhardt, R. Steroid Hydroxylation: Microbial Steroid Biotransformations Using Cytochrome P450 Enzymes. In Modern Biooxidation; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2007; pp. 155–176. [Google Scholar] [CrossRef]
- Herráiz, I. Chemical Pathways of Corticosteroids, Industrial Synthesis from Sapogenins. In Microbial Steroids: Methods and Protocols; Barredo, J.-L., Herráiz, I., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 15–27. [Google Scholar] [CrossRef]
- Fernandes, P.; Cabral, J.M.S. Phytosterols: Applications and recovery methods. Bioresour. Technol. 2007, 98, 2335–2350. [Google Scholar] [CrossRef] [PubMed]
- Malaviya, A.; Gomes, J. Androstenedione production by biotransformation of phytosterols. Bioresour. Technol. 2008, 99, 6725–6737. [Google Scholar] [CrossRef] [PubMed]
- Donova, M.V.; Egorova, O.V. Microbial steroid transformations: Current state and prospects. Appl. Microbiol. Biotechnol. 2012, 94, 1423–1447. [Google Scholar] [CrossRef] [PubMed]
- Brzostek, A.; Dziadek, B.; Rumijowska-Galewicz, A.; Pawelczyk, J.; Dziadek, J. Cholesterol oxidase is required for virulence of Mycobacterium tuberculosis. FEMS Microbiol. Lett. 2007, 275, 106–112. [Google Scholar] [CrossRef] [Green Version]
- Brzostek, A.; Śliwiński, T.; Rumijowska-Galewicz, A.; Korycka-Machała, M.; Dziadek, J. Identification and targeted disruption of the gene encoding the main 3-ketosteroid dehydrogenase in Mycobacterium smegmatis. Microbiology 2005, 151, 2393–2402. [Google Scholar] [CrossRef] [Green Version]
- van der Geize, R.; Hessels, G.I.; van Gerwen, R.; Vrijbloed, J.W.; van der Meijden, P.; Dijkhuizen, L. Targeted Disruption of the kstD Gene Encoding a 3-Ketosteroid Δ1-Dehydrogenase Isoenzyme of Rhodococcus erythropolis Strain SQ1. Appl. Environ. Microbiol. 2000, 66, 2029–2036. [Google Scholar] [CrossRef] [Green Version]
- Fernández-Cabezón, L.; Galán, B.; García, J.L. New Insights on Steroid Biotechnology. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Wei, W.; Wang, F.; Fan, S.; Wei, D. Inactivation and Augmentation of the Primary 3-Ketosteroid-Δ1- Dehydrogenase in Mycobacterium neoaurum NwIB-01: Biotransformation of Soybean Phytosterols to 4-Androstene- 3,17-Dione or 1,4-Androstadiene-3,17-Dione. Appl. Environ. Microbiol. 2010, 76, 4578–4582. [Google Scholar] [CrossRef] [Green Version]
- Fernández-Cabezón, L.; Galán, B.; García, J.L. Engineering Mycobacterium smegmatis for testosterone production. Microb. Biotechnol. 2016, 10, 151–161. [Google Scholar] [CrossRef] [Green Version]
- Su, L.; Shen, Y.; Xia, M.; Shang, Z.; Xu, S.; An, X.; Wang, M. Overexpression of cytochrome p450 125 in Mycobacterium: A rational strategy in the promotion of phytosterol biotransformation. J. Ind. Microbiol. Biotechnol. 2018, 45, 857–867. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhang, Y.; Shen, Y.; Zhang, X.; Zhang, Z.; Xu, S.; Luo, J.; Xia, M.; Wang, M. Economical production of androstenedione and 9α-hydroxyandrostenedione using untreated cane molasses by recombinant mycobacteria. Bioresour. Technol. 2019, 290, 121750. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.-J.; Wang, L.; Liu, H.-H.; Liu, Y.-J.; Ren, Y.-H.; Wang, F.-Q.; Wei, D.-Z. Characterization and engineering control of the effects of reactive oxygen species on the conversion of sterols to steroid synthons in Mycobacterium neoaurum. Metab. Eng. 2019, 56, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Shao, M.; Zhang, X.; Rao, Z.; Xu, M.; Yang, T.; Li, H.; Xu, Z.; Yang, S. Efficient testosterone production by engineered Pichia pastoris co-expressing human 17β-hydroxysteroid dehydrogenase type 3 and Saccharomyces cerevisiae glucose 6-phosphate dehydrogenase with NADPH regeneration. Green Chem. 2016, 18, 1774–1784. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, X.; Wang, Y.; Han, Y.; Sun, J.; Shi, J.; Zhang, B. Identification, function, and application of 3-ketosteroid Δ1-dehydrogenase isozymes in Mycobacterium neoaurum DSM 1381 for the production of steroidic synthons. Microb. Cell Factories 2018, 17, 77. [Google Scholar] [CrossRef] [Green Version]
- Wongkittichote, P.; Ah Mew, N.; Chapman, K.A. Propionyl-CoA carboxylase—A review. Mol. Genet. Metab. 2017, 122, 145–152. [Google Scholar] [CrossRef]
- Su, L.; Shen, Y.; Zhang, W.; Gao, T.; Shang, Z.; Wang, M. Cofactor engineering to regulate NAD+/NADH ratio with its application to phytosterols biotransformation. Microb. Cell Factories 2017, 16. [Google Scholar] [CrossRef]
- Szentirmai, A. Microbial physiology of sidechain degradation of sterols. J. Ind. Microbiol. 1990, 6, 101–115. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, X.; Wang, X.; Wang, L.; Xia, M.; Luo, J.; Shen, Y.; Wang, M. Improving phytosterol biotransformation at low nitrogen levels by enhancing the methylcitrate cycle with transcriptional regulators PrpR and GlnR of Mycobacterium neoaurum. Microb. Cell Factories 2020, 19, 13. [Google Scholar] [CrossRef]
- Zhou, X. Efficient production of androstenedione by repeated batch fermentation in waste cooking oil media through regulating NAD+/NADH ratio and strengthening cell vitality of Mycobacterium neoaurum. Bioresour. Technol. 2019, 279, 209–217. [Google Scholar] [CrossRef]
- Tarkowská, D.; Strnad, M. Isoprenoid-derived plant signaling molecules: Biosynthesis and biological importance. Planta 2018. [Google Scholar] [CrossRef] [PubMed]
- Janeczko, A.; Skoczowski, A. Mammalian sex hormones in plants. Folia Histochem. Cytobiol. 2005. [Google Scholar] [CrossRef]
- Simons, R.G.; Grinwich, D.L. Immunoreactive detection of four mammalian steroids in plants. Can. J. Bot. 1989, 67, 288–296. [Google Scholar] [CrossRef]
- Ylstra, B.; Touraev, A.; Brinkmann, A.O.; Heberle-Bors, E.; Van Tunen, A.J. Steroid hormones stimulate germination and tube growth of in vitro matured tobacco pollen. Plant Physiol. 1995. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simerský, R.; Novák, O.; Morris, D.A.; Pouzar, V.; Strnad, M. Identification and quantification of several mammalian steroid hormones in plants by UPLC-MS/MS. J. Plant Growth Regul. 2009. [Google Scholar] [CrossRef]
- Šaden-Krehula, M.; Tajić, M.; Kolbah, D. Testosterone, epitestosterone and androstenedione in the pollen of scotch pine P. silvestris L. Experientia 1971. [Google Scholar] [CrossRef]
- Šaden-Krehula, M.; Tajić, M.; Kolbah, D. Sex hormones and corticosteroids in pollen of Pinus nigra. Phytochemistry 1979. [Google Scholar] [CrossRef]
- Yang, Z.H.; Tang, Y.; Cao, Z.X.; Tsao, T.H. Changes of the contents of testosterone-steroidal sex hormone in reproductive organs of Lilium davidii Duch. Acta Bot. Sin. 1994, 36, 215–220. [Google Scholar]
- Bennett, R.D.; Heftmann, E. Biosynthesis of pregnenolone from cholesterol in Haplopappus heterophyllus. Phytochemistry 1966. [Google Scholar] [CrossRef]
- Finsterbusch, A.; Lindemann, P.; Grimm, R.; Eckerskorn, C.; Luckner, M. Δ5-3β-hydroxysteroid dehydrogenase from Digitalis lanata Ehrh—A multifunctional enzyme in steroid metabolism? Planta 1999. [Google Scholar] [CrossRef]
- Shpakovski, G.V.; Spivak, S.G.; Berdichevets, I.N.; Babak, O.G.; Kubrak, S.V.; Kilchevsky, A.V.; Aralov, A.V.; Slovokhotov, I.Y.; Shpakovski, D.G.; Baranova, E.N.; et al. A key enzyme of animal steroidogenesis can function in plants enhancing their immunity and accelerating the processes of growth and development. BMC Plant Biol. 2017, 189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- .Spivak, S.G.; Berdichevets, I.N.; Litvinovskaya, R.P.; Drach, S.V.; Kartel, N.A.; Shpakovski, G.V. Some peculiarities of steroid metabolism in transgenic Nicotiana tabacum plants bearing the CYP11A1 cDNA of cytochrome P450SCC from the bovine adrenal cortex. Russ. J. Bioorg. Chem. 2010. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.T.; Li, W.; Kim, T.K.; Semak, I.; Wang, J.; Zjawiony, J.K.; Tuckey, R.C. Novel activities of CYP11A1 and their potential physiological significance. J. Steroid Biochem. Mol. Biol. 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strushkevich, N.; MacKenzie, F.; Cherkesova, T.; Grabovec, I.; Usanov, S.; Park, H.W. Structural basis for pregnenolone biosynthesis by the mitochondrial monooxygenase system. Proc. Natl. Acad. Sci. USA 2011. [Google Scholar] [CrossRef] [Green Version]
- Ohnishi, T.; Yokota, T.; Mizutani, M. Insights into the function and evolution of P450s in plant steroid metabolism. Phytochemistry 2009. [Google Scholar] [CrossRef]
- Fujita, S.; Ohnishi, T.; Watanabe, B.; Yokota, T.; Takatsuto, S.; Fujioka, S.; Yoshida, S.; Sakata, K.; Mizutani, M. Arabidopsis CYP90B1 catalyses the early C-22 hydroxylation of C 27, C28 and C29 sterols. Plant J. 2006. [Google Scholar] [CrossRef]
- Fujiyama, K.; Hino, T.; Kanadani, M.; Watanabe, B.; Jae Lee, H.; Mizutani, M.; Nagano, S. Structural insights into a key step of brassinosteroid biosynthesis and its inhibition. Nat. Plants 2019. [Google Scholar] [CrossRef]
| Substrate | Product | Organism | Process | Reference |
|---|---|---|---|---|
| Soybean phytosterols (stigmasterol, campesterol and β-sitosterol) | AD, ADD | Mycobacterium neoaurum NwIB-01 | KstD overexpression to increase production and purity of ADD | [24] |
| Androstenedione or cholesterol | TS | Mycobacterium smegmatis mc2155 | Comamonas testosteroni or Cochliobolus lunatus 17β-HSD heterologous overexpression | [25] |
| Phytosterols mixture: 51.7% of β-sitosterol, 27.2% of stigmasterol, 17.1% of campesterol, and 4.0% of brassicasterol | AD, ADD | Mycobacterium neoaurum TCCC 11978 | CYP125-3 overexpression | [26] |
| Phytosterols from untreated cane molasses | AD, 9OH-AD | Mycobacterium neoaurum TCCC 11978; Mycobacterium fortuitum TCCC 111744 | Co-expression of the PCC subunit beta and type II NADH dehydrogenase | [27] |
| Phytosterols | 9OH-AD, 4-HBC | Mycobacterium neoaurum ATCC 25795 | Neutralization of extra production of ROS through catalase overexpression and mycothiol or ergothioneine augmentation | [28] |
| Phytosterols | ADD | Mycobacterium neoaurum JC-12 | Increase NAD+ formation through heterologous expression of B. subtilis nicotinamide adenine dinucleotide oxidase and overexpression of catalase | [29] |
| Phytosterols | AD | Mycobacterium neoaurum TCCC 11978; Mycobacterium fortuitum TCCC 111744 | Type II NADH dehydrogenase overexpression, repeated batch fermentations | [27] |
| Androstenedione | TS | Rhodococcus ruber Chol-4 | Cochliobolus lunatus 17-ketosteroid reductase heterologous overexpression | [18] |
| Phytosterols | AD | Mycobacterium neoaurum TCCC 11978 | Repression of propionyl-CoA metabolism. 2-methylcitrate cycle pathway prpDBC overexpression and nitrogen transcription regulator GlnR deletion | [30] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batth, R.; Nicolle, C.; Cuciurean, I.S.; Simonsen, H.T. Biosynthesis and Industrial Production of Androsteroids. Plants 2020, 9, 1144. https://doi.org/10.3390/plants9091144
Batth R, Nicolle C, Cuciurean IS, Simonsen HT. Biosynthesis and Industrial Production of Androsteroids. Plants. 2020; 9(9):1144. https://doi.org/10.3390/plants9091144
Chicago/Turabian StyleBatth, Rituraj, Clément Nicolle, Ilenuta Simina Cuciurean, and Henrik Toft Simonsen. 2020. "Biosynthesis and Industrial Production of Androsteroids" Plants 9, no. 9: 1144. https://doi.org/10.3390/plants9091144
APA StyleBatth, R., Nicolle, C., Cuciurean, I. S., & Simonsen, H. T. (2020). Biosynthesis and Industrial Production of Androsteroids. Plants, 9(9), 1144. https://doi.org/10.3390/plants9091144







