Anti-Melanoma Activities and Phytochemical Compositions of Sorbus commixta Fruit Extracts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effects of S. commixta Fruit Extracts and Solvent Fractions on Human Melanoma SK-MEL-2 Cell
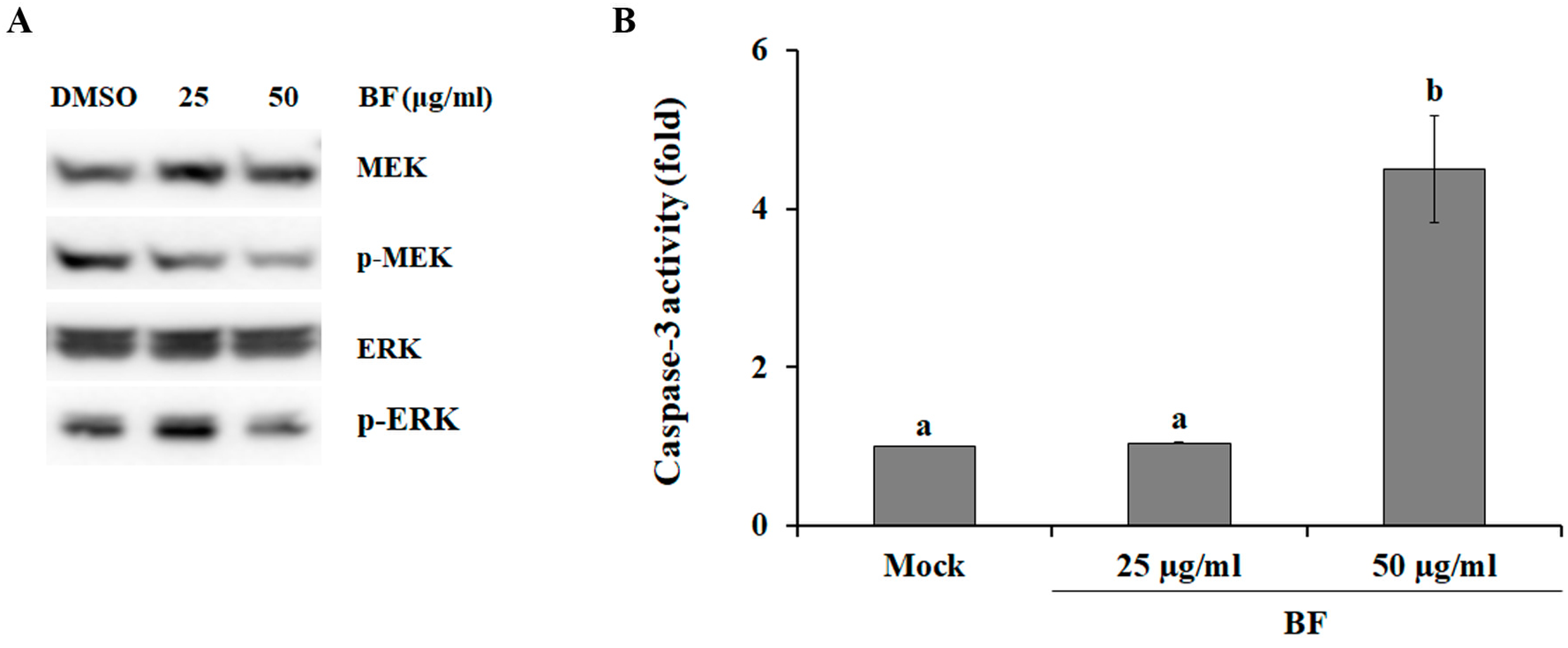
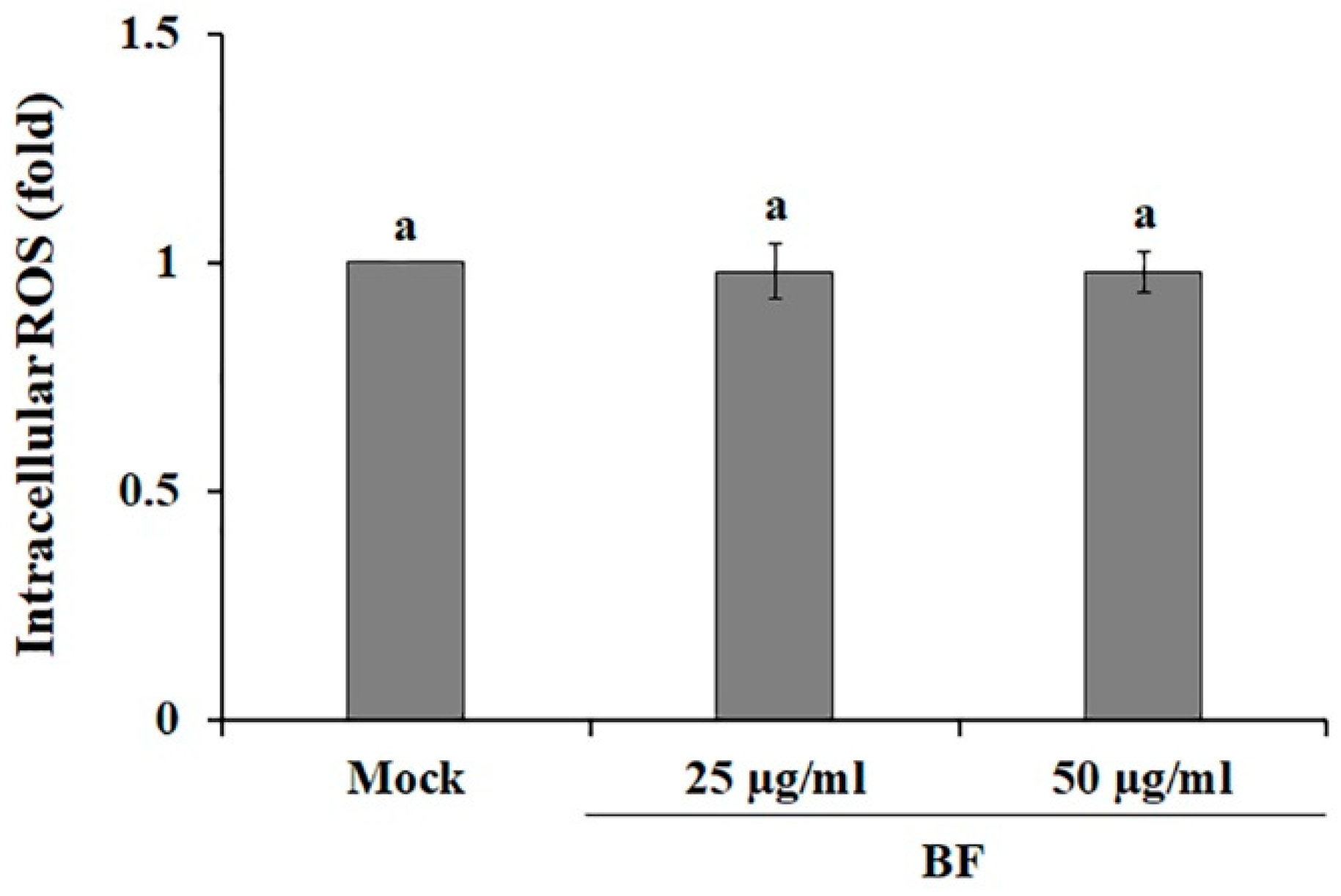
2.2. BF Blocks the MEK/ERK Signaling Pathway
3. Materials and Methods
3.1. Plant Materials and Sample Preparation
3.2. Cell Viability Assay
3.3. Western Blot
3.4. Caspase-3 Activity and Intracellular ROS Measurement
3.5. Determination of TPC and TFC
3.6. HPLC Analysis
3.7. Statistical Analyses
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics, 2016. CA. Cancer J. Clin. 2016, 66, 271–289. [Google Scholar] [CrossRef] [Green Version]
- Cummins, D.L.; Cummins, J.M.; Pantle, H.; Silverman, M.A.; Leonard, A.L.; Chanmugam, A. Cutaneous malignant melanoma. Mayo Clin. Proc. 2006, 81, 500–507. [Google Scholar] [CrossRef] [Green Version]
- Hussein, M.R.; Haemel, A.K.; Wood, G.S. Apoptosis and melanoma: Molecular mechanisms. J. Pathol. 2003, 199, 275–288. [Google Scholar] [CrossRef] [PubMed]
- Markovic, S.N.; Erickson, L.A.; Rao, R.D.; Weenig, R.H.; Pockaj, B.A.; Bardia, A.; Vachon, C.M.; Schild, S.E.; McWilliams, R.R.; Hand, J.L.; et al. Malignant melanoma in the 21st century, part 1: Epidemiology, risk factors, screening, prevention, and diagnosis. Mayo Clin. Proc. 2007, 82, 364–380. [Google Scholar] [CrossRef]
- Kato, M.; Liu, W.; Akhand, A.A.; Hossain, K.; Takeda, K.; Takahashi, M.; Nakashima, I. Ultraviolet radiation induces both full activation of Ret kinase and malignant melanocytic tumor promotion in RFP-RET-transgenic mice. J. Invest. Dermatol. 2000, 115, 1157–1158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Libra, M.; Malaponte, G.; Navolanic, P.M.; Gangemi, P.; Bevelacqua, V.; Proietti, L.; Bruni, B.; Stivala, F.; Mazzarino, M.C.; Travali, S.; et al. Analysis of BRAF mutation in primary and metastatic melanoma. Cell Cycle 2005, 4, 1382–1384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guevara-Canales, J.O.; Gutiérrez-Morales, M.M.; Sacsaquispe-Contreras, S.J.; Sánchez-Lihón, J.; Morales-Vadillo, R. Malignant melanoma of the oral cavity. Review of the literature and experience in a Peruvian Population. Med. Oral Patol. Oral Cir. Bucal 2012, 17, e206–e211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vikey, A.K.; Vikey, D. Primary malignant melanoma, of head and neck: A comprehensive review of literature. Oral Oncol. 2012, 2012, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Xiao, P.; Zheng, B.; Sun, J.; Yang, J. Biochanin a induces anticancer effects in SK-Mel-28 human malignant melanoma cells via induction of apoptosis, inhibition of cell invasion and modulation of Nf-κB and MAPK signaling pathways. Oncol. Lett. 2017, 14, 5989–5993. [Google Scholar] [CrossRef]
- Srivastava, V.; Negi, A.S.; Kumar, J.K.; Gupta, M.M.; Khanuja, S.P.S. Plant-based anticancer molecules: A chemical and biological profile of some important leads. Bioorganic Med. Chem. 2005, 13, 5892–5908. [Google Scholar] [CrossRef]
- Dar, K.B.; Bhat, A.H.; Amin, S.; Anees, S.; Zargar, M.A.; Masood, A. Herbal compounds as potential anticancer therapeutics: Current. Ann. Pharmacol. Pharm. 2017, 2, 1106. [Google Scholar]
- Lee, T.K.; Roh, H.S.; Yu, J.S.; Kwon, D.J.; Kim, S.Y.; Baek, K.H.; Kim, K.H. A novel cytotoxic activity of the fruit of Sorbus commixta against human lung cancer cells and isolation of the major constituents. J. Funct. Foods 2017, 30, 1–7. [Google Scholar] [CrossRef]
- Sohn, E.J.; Kang, D.G.; Mun, Y.J.; Woo, W.H.; Lee, H.S. Anti-atherogenic effects of the methanol extract of Sorbus cortex in atherogenic-diet rats. Biol. Pharm. Bull. 2005, 28, 1444–1449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, T.; Lee, Y.J.; Jang, H.J.; Kim, A.R.; Hong, S.; Kim, T.W.; Kim, M.Y.; Lee, J.; Lee, Y.G.; Cho, J.Y. Anti-inflammatory activity of Sorbus commixta water extract and its molecular inhibitory mechanism. J. Ethnopharmacol. 2011, 134, 493–500. [Google Scholar] [CrossRef]
- Bae, J.T.; Sim, G.S.; Kim, J.H.; Pyo, H.B.; Yun, J.W.; Lee, B.C. Antioxidative activity of the hydrolytic enzyme treated Sorbus commixta Hedl. and its inhibitory effect on matrix metalloproteinase-1 in UV irradiated human dermal fibroblasts. Arch. Pharm. Res. 2007, 30, 1116–1123. [Google Scholar] [CrossRef]
- Sohn, E.J.; Kang, D.G.; Choi, D.H.; Lee, A.S.; Mun, Y.J.; Woo, W.H.; Kim, J.S.; Lee, H.S. Effect of methanol extract of Sorbus cortex in a rat model of L-NAME-induced atherosclerosis. Biol. Pharm. Bull. 2005, 28, 1239–1243. [Google Scholar] [CrossRef] [Green Version]
- Yin, M.H.; Kang, D.G.; Choi, D.H.; Kwon, T.O.; Lee, H.S. Screening of vasorelaxant activity of some medicinal plants used in Oriental medicines. J. Ethnopharmacol. 2005, 99, 113–117. [Google Scholar] [CrossRef]
- Lee, S.-O.; Lee, H.W.; Lee, I.-S.; Im, H.G. The pharmacological potential of Sorbus commixta cortex on blood alcohol concentration and hepatic lipid peroxidation in acute alcohol-treated rats. J. Pharm. Pharmacol. 2006, 58, 685–893. [Google Scholar] [CrossRef]
- Shikov, A.N.; Pozharitskaya, O.N.; Makarov, V.G.; Wagner, H.; Verpoorte, R.; Heinrich, M. Medicinal Plants of the Russian Pharmacopoeia; Their history and applications. J. Ethnopharmacol. 2014, 154, 481–536. [Google Scholar] [CrossRef] [Green Version]
- Raudonis, R.; Raudone, L.; Gaivelyte, K.; Viškelis, P.; Janulis, V. Phenolic and antioxidant profiles of rowan (Sorbus L.) fruits. Nat. Prod. Res. 2014, 28, 1231–1240. [Google Scholar] [CrossRef]
- Zymone, K.; Raudone, L.; Raudonis, R.; Marksa, M.; Ivanauskas, L.; Janulis, V. Phytochemical profiling of fruit powders of twenty Sorbus L. Cultivars. Molecules 2018, 23, 2593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sołtys, A.; Galanty, A.; Podolak, I. Ethnopharmacologically important but underestimated genus Sorbus: A comprehensive review. Phytochem. Rev. 2020, 19, 491–526. [Google Scholar] [CrossRef]
- Che Sulaiman, I.S.; Basri, M.; Fard Masoumi, H.R.; Chee, W.J.; Ashari, S.E.; Ismail, M. Effects of temperature, time, and solvent ratio on the extraction of phenolic compounds and the anti-radical activity of Clinacanthus nutans Lindau leaves by response surface methodology. Chem. Cent. J. 2017, 11, 54. [Google Scholar] [CrossRef] [PubMed]
- Do, Q.D.; Angkawijaya, A.E.; Tran-Nguyen, P.L.; Huynh, L.H.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.H. Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. J. Food Drug Anal. 2014, 22, 296–302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nawaz, H.; Shi, J.; Mittal, G.S.; Kakuda, Y. Extraction of polyphenols from grape seeds and concentration by ultrafiltration. Sep. Purif. Technol. 2006, 48, 176–181. [Google Scholar] [CrossRef]
- Chew, K.K.; Khoo, M.Z.; Ng, S.Y.; Thoo, Y.Y.; Aida, W.M.W.; Ho, C.W. Effect of ethanol concentration, extraction time and extraction temperature on the recovery of phenolic compounds and antioxidant capacity of Orthosiphon stamineus extracts. Int. Food Res. J. 2011, 18, 1427–1435. [Google Scholar]
- Khoo, K.S.; Leong, H.Y.; Chew, K.W.; Lim, J.-W.; Ling, T.C.; Show, P.L.; Yen, H.-W. Liquid biphasic system: A recent bioseparation technology. Processes 2020, 8, 149. [Google Scholar] [CrossRef] [Green Version]
- Widyawati, P.S.; Budianta, T.D.W.; Kusuma, F.A.; Wijaya, E.L. Difference of solvent polarity to phytochemical content and antioxidant activity of Pluchea indicia less leaves extracts. Int. J. Pharmacogn. Phytochem. Res. 2014, 6, 850–855. [Google Scholar]
- Haminiuk, C.W.I.; Plata-Oviedo, M.S.V.; de Mattos, G.; Carpes, S.T.; Branco, I.G. Extraction and quantification of phenolic acids and flavonols from Eugenia pyriformis using different solvents. J. Food Sci. Technol. 2014, 51, 2862–2866. [Google Scholar] [CrossRef] [Green Version]
- Chua, L.S.; Lau, C.H.; Chew, C.Y.; Dawood, D.A.S. Solvent fractionation and acetone precipitation for crude saponins from eurycoma longifolia extract. Molecules 2019, 24, 1416. [Google Scholar] [CrossRef] [Green Version]
- Khazir, J.; Mir, B.A.; Pandita, M.; Pilcher, L.; Riley, D.; Chashoo, G. Design and synthesis of sulphonyl acetamide analogues of quinazoline as anticancer agents. Med. Chem. Res. 2020, 29, 916–925. [Google Scholar] [CrossRef]
- Gào, X.; Schöttker, B. Reduction-oxidation pathways involved in cancer development: A systematic review of literature reviews. Oncotarget 2017, 8, 51888–51906. [Google Scholar] [CrossRef] [Green Version]
- Afanas’ev, I. Reactive oxygen species signaling in cancer: Comparison with aging. Aging Dis. 2011, 2, 219–230. [Google Scholar] [PubMed]
- Hadi, S.M.; Asad, S.F.; Singh, S.; Ahmad, A. Putative mechanism for anticancer and apoptosis-inducing properties of plant-derived polyphenolic compounds. IUBMB Life 2000, 50, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.Y.; Hadi, S.M.; Mohammad, R.M.; Azmi, A.S. Prooxidant anticancer activity of plant-derived polyphenolic compounds: An underappreciated phenomenon. Funct. Foods Cancer Prev. Ther. 2020, 221–236. [Google Scholar] [CrossRef]
- Roberts, P.J.; Der, C.J. Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene 2007, 26, 3291–3310. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Pan, W.; Liu, S.; Shen, Z.; Xu, Y.; Hu, L. ERK/MAPK signalling pathway and tumorigenesis (Review). Exp. Ther. Med. 2020, 19, 1997–2007. [Google Scholar] [CrossRef] [Green Version]
- Julien, O.; Wells, J.A. Caspases and their substrates. Cell Death Differ. 2017, 9, 738–742. [Google Scholar] [CrossRef]
- O’Donovan, N.; Crown, J.; Stunell, H.; Hill, A.D.K.; McDermott, E.; O’Higgins, N.; Duffy, M.J. Caspase 3 in breast cancer. Clin. Cancer Res. 2003, 9, 738–742. [Google Scholar]
- Ponder, K.G.; Boise, L.H. The prodomain of caspase-3 regulates its own removal and caspase activation. Cell Death Discov. 2019, 28, 56. [Google Scholar] [CrossRef]
- Cheung, E.C.C.; Slack, R.S. Emerging role for ERK as a key regulator of neuronal apoptosis. Sci. STKE 2004, 2004, 45. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Li, J.; Han, J.; Hou, N.; Song, Y.; Dong, L. Chlorogenic acid enhances the effects of 5-fluorouracil in human hepatocellular carcinoma cells through the inhibition of extracellular signal-regulated kinases. Anti-Cancer Drugs 2015, 26, 540–546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caparica, R.; Júlio, A.; Araújo, M.E.M.; Baby, A.R.; Fonte, P.; Costa, J.G.; de Almeida, T.S. Anticancer activity of rutin and its combination with ionic liquids on renal cells. Biomolecules 2020, 10, 233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiaschi, T.; Chiarugi, P. Oxidative stress, tumor microenvironment, and metabolic reprogramming: A diabolic liaison. Int. J. Cell Biol. 2012, 2012, 762825. [Google Scholar] [CrossRef] [Green Version]
- Liu, D.; Qiu, X.; Xiong, X.; Chen, X.; Pan, F. Current updates on the role of reactive oxygen species in bladder cancer pathogenesis and therapeutics. Clin. Transl. Oncol. 2020, 22, 1687–1697. [Google Scholar] [CrossRef] [Green Version]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Santana-Gálvez, J.; Villela Castrejón, J.; Serna-Saldívar, S.O.; Jacobo-Velázquez, D.A. Anticancer potential of dihydrocaffeic acid: A chlorogenic acid metabolite. CYTA J. Food 2020, 18, 245–248. [Google Scholar] [CrossRef] [Green Version]
- Yoo, T.K.; Kim, J.S.; Hyun, T.K. Polyphenolic composition and anti-melanoma activity of white forsythia (Abeliophyllum distichum nakai) organ extracts. Plants 2020, 9, 757. [Google Scholar] [CrossRef]
- Jin, S.; Eom, S.H.; Kim, J.S.; Jo, I.H.; Hyun, T.K. Influence of ripening stages on phytochemical composition and bioavailability of ginseng berry (Panax ginseng C.A. Meyer). J. Appl. Bot. Food Qual. 2019, 92, 130–137. [Google Scholar] [CrossRef]

| Polyphenolic Compounds | Water Extract | 25% EtOH Extract | 50% EtOH Extract | 75% EtOH Extract | EtOH Extract |
|---|---|---|---|---|---|
| Total phenol content 1 | 50.27 ± 4.2 a | 55.71 ± 3.44 a | 60.02 ± 9.35 a | 64.36 ± 6.35 a | 67.98 ± 4.28 a |
| Total flavonoid content 2 | 2.49 ± 0.25 a | 3.53 ± 0.23 a | 4.91 ± 0.25 b | 6.09 ± 0.21 c | 8.65 ± 0.5 d |
| Chlorogenic acid 3 | 111.81 ± 2.54 a | 127.14 ± 1.56 b | 150.70 ± 3.40 c | 176.72 ± 4.58 d | 344.70 ± 7.18 e |
| Ferulic acid 3 | 0.14 ± 0.02 b | 0.09 ± 0.01 a | 0.06 ± 0.002 a | 0.08 ± 0.01 a | 0.13 ± 0.01 b |
| Hydroxybenzoic acid 3 | 0.13 ± 0.01 a | 0.13 ± 0.02 a | 0.10 ± 0.01 a | 0.10 ± 0.01 a | 0.20 ± 0.01 b |
| Protocatechuic acid 3 | 0.79 ± 0.01 a | 0.78 ± 0.09 a | 0.92 ± 0.05 ab | 1.05 ± 0.03 bc | 1.22 ± 0.06 c |
| Rutin 3 | ND | ND | 0.28 ± 0.06 a | 0.48 ± 0.08 b | 0.60 ± 0.04 b |
| Polyphenolic Compounds | Hexane Fraction | Ethyl Acetate Fraction | Butanol Fraction | Aqueous Fraction |
|---|---|---|---|---|
| Total phenol content 1 | 19.89 ± 2.65 a | 72.59 ± 5.34 c | 87.15 ± 4.66 d | 44.07 ± 2.77 b |
| Total flavonoid content 2 | 6.25 ± 1.25 a | 13.54 ± 0.43 b | 22.25 ± 0.77 c | 5.96 ± 1.11 a |
| Chlorogenic acid 3 | 2.20 ± 0.03 a | 93.12 ± 14.54 b | 912.72 ± 68.04 c | 74.20 ± 2.88 ab |
| Ferulic acid 3 | 0.02 ± 0.004 a | 1.24 ± 0.05 b | 0.11 ± 0.01 a | 0.02 ± 0.001 a |
| Gallic acid 3 | ND | 0.17 ± 0.02 b | 0.04 ± 0.02 a | ND |
| Hydroxybenzoic acid 3 | 0.36 ± 0.02 b | 1.66 ± 0.11 c | 0.13 ± 0.01 a | 0.04 ± 0.003 a |
| Protocatechuic acid 3 | 0.05 ± 0.003 a | 2.20 ± 0.22 b | 2.86 ± 0.01 c | 0.26 ± 0.05 a |
| Rutin 3 | ND | 0.12 ± 0.03 a | 2.59 ± 0.17 b | 0.12 ± 0.02 a |
| Sinapinic acid 3 | ND | 0.13 ± 0.01 b | 0.07 ± 0.01 a | ND |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, S.; Kim, K.C.; Kim, J.-S.; Jang, K.-I.; Hyun, T.K. Anti-Melanoma Activities and Phytochemical Compositions of Sorbus commixta Fruit Extracts. Plants 2020, 9, 1076. https://doi.org/10.3390/plants9091076
Jin S, Kim KC, Kim J-S, Jang K-I, Hyun TK. Anti-Melanoma Activities and Phytochemical Compositions of Sorbus commixta Fruit Extracts. Plants. 2020; 9(9):1076. https://doi.org/10.3390/plants9091076
Chicago/Turabian StyleJin, Sora, Kyeoung Cheol Kim, Ju-Sung Kim, Keum-Il Jang, and Tae Kyung Hyun. 2020. "Anti-Melanoma Activities and Phytochemical Compositions of Sorbus commixta Fruit Extracts" Plants 9, no. 9: 1076. https://doi.org/10.3390/plants9091076





