Elemental Composition and Some Nutritional Parameters of Sweet Pepper from Organic and Conventional Agriculture
Abstract
1. Introduction
2. Results
2.1. Nutritional Characterization of Sweet Peppers
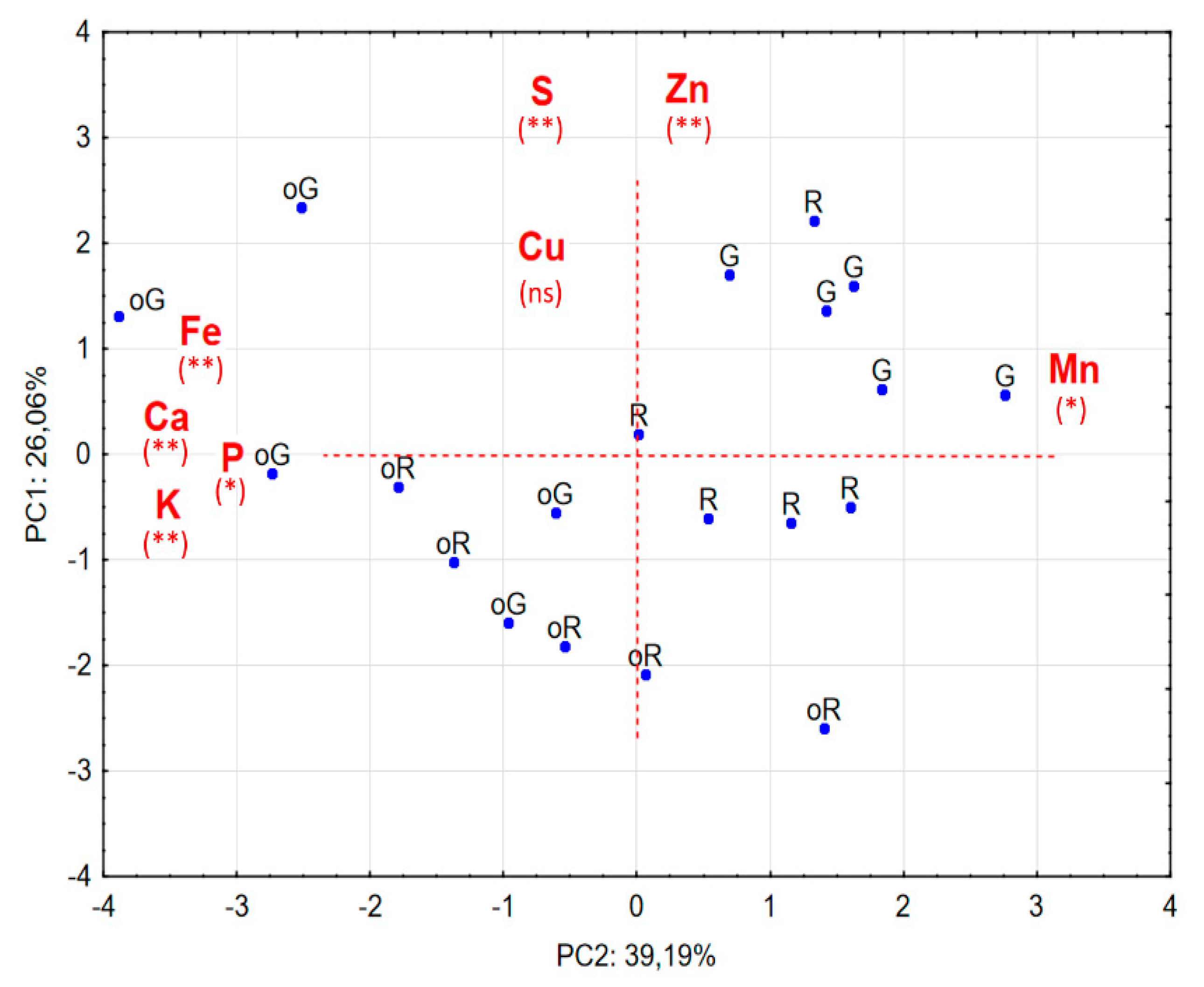
2.2. Principal Component Analysis (PCA)
- 1—The clear separation, with regard to concentrations, between peppers from OA and CA.
- 2—Organic peppers tend to have higher concentrations of Ca, Fe, K and P and less Mn.
- 3—In general, GP from both production methods have higher concentrations of S and Zn.
- 4—In general terms, conventionally grown GP have a higher concentration of Mn and Zn than all the others.
- 5—The Cu shows no significant correlation with any of the axes.
3. Discussion
4. Materials and Methods
4.1. Plant Cultivation
4.2. Sample Collection
4.3. Nutritional Composition
4.4. XRF Preparation and Analysis
4.5. Statistical Analysis and Control Assurance
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smith, L.G.; Kirk, G.J.D.; Jones, P.J.; Williams, A.G. The greenhouse gas impacts of converting food production in England and Wales to organic methods. Nat. Commun. 2019, 10, 4641. [Google Scholar] [CrossRef] [PubMed]
- Willer, H.; Lernoud, J. (Eds.) The World of Organic Agriculture. Statistics and Emerging Trends 2019; Research Institute of Organic Agriculture (FiBL), Frick and IFOAM—Organics International: Bonn, Germany, 2019; Available online: www.organicworld.net/yearbook/yearbook-2019.html (accessed on 6 May 2020).
- Ponisio, L.C.; M’Gonigle, L.K.; Mace, K.C.; Palomino, J.; de Valpine, P.; Kremen, C. Diversification practices reduce organic to conventional yield gap. Proc. R. Soc. Lond. B Biol. Sci. 2015, 282, 20141396. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-H.; Li, S.-X.; Malhi, S. Effects of fertilization and other agronomic measures on nutritional quality of crops. J. Sci. Food Agric. 2008, 88, 7–23. [Google Scholar] [CrossRef]
- Williams, C.M. Nutritional quality of organic food: Shades of grey or shades of green? Proc. Nutr. Soc. 2002, 61, 19–24. [Google Scholar] [CrossRef]
- Worthington, V. Nutritional quality of organic versus conventional fruits, vegetables, and grains. J. Altern. Complement. Med. 2004, 7. [Google Scholar] [CrossRef]
- Benbrook, C. The impacts of yield on nutritional quality: Lessons from organic farming. HortScience 2009, 44, 12–14. [Google Scholar] [CrossRef]
- Reboredo, F.; Pelica, J.; Lidon, F.; Pessoa, M.F.; Ramalho, J.C.; Calvão, T.; Simões, M.; Guerra, M. Heavy metal content of edible plants collected close an area of intense mining activity (Southern Portugal). Environ. Monit. Assess. 2018, 190, 484. [Google Scholar] [CrossRef]
- Reboredo, F.; Simões, M.; Jorge, C.; Martinez, J.; Mancuso, M.; Guerra, M.; Ramalho, J.C.; Pessoa, M.F.; Lidon, F. Metal content in edible crops and agricultural soils due to intensive use of fertilizers and pesticides in Terras da Costa de Caparica (Portugal). Environ. Sci. Pollut. Res. 2019, 26, 2512–2522. [Google Scholar] [CrossRef]
- Reboredo, F.H.; Barbosa, A.; Silva, M.M.; Carvalho, M.L.; Santos, J.P.; Pessoa, M.F.; Lidon, F.; Ramalho, J.C.; Guerra, M. Mineral content of food supplements of plant origin, by energy dispersive x-ray fluorescence. A risk assessment. Expo. Health 2020. [Google Scholar] [CrossRef]
- Reboredo, F.H.S.; Ribeiro, C.A.G. Vertical distribution of Al, Cu, Fe and Zn in the soil salt marshes of the Sado estuary, Portugal. Int. J. Environ. Stud. 1984, 23, 249–253. [Google Scholar] [CrossRef]
- Pelica, J.; Barbosa, S.; Reboredo, F.; Lidon, F.; Pessoa, M.F.; Calvão, T. The paradigm of high concentration of metals of natural or antrophogenic origin in the soils – the case of Neves-Corvo mining area-South of Portugal. J. Geochem. Explor. 2018, 186, 12–23. [Google Scholar] [CrossRef]
- Carrondo, M.; Reboredo, F.; Ganho, R.; Santos Oliveira, J.F. Heavy metal analysis of sediments in Tejo estuary, Portugal, using a rapid flameless atomic absorption procedure. Talanta 1984, 31, 561–564. [Google Scholar] [CrossRef]
- Reboredo, F. Cadmium uptake by Halimione portulacoides. An ecophysiological study. Bull. Environ. Contam. Toxicol. 2001, 67, 926–933. [Google Scholar] [CrossRef] [PubMed]
- Reboredo, F. Zinc compartmentation in Halimione portulacoides (L.) Aellen and some effects on leaf ultrastructure. Environ. Sci Pollut Res. 2012, 19, 2644–2657. [Google Scholar] [CrossRef]
- Matos, A.; Cabo, P.; Ribeiro, M.I.; Fernandes, A. Two decades of organic farming in Portugal. In FONCIMED 2016, Résumés; Instituto Politécnico: Bragança, Portugal, 2016; ISBN 978-972-745-215-6. [Google Scholar]
- Direção Geral de Agricultura e Desenvolvimento Rural (DGADR). A Produção Biológica em Portugal; Direção Geral de Agricultura e Desenvolvimento Rural (DGADR): Lisboa, Portugal, 2019; 77p. (In Portuguese)
- Guilherme, R.; Rodrigues, N.; Marx, I.M.G.; Dias, L.G.; Veloso, A.C.A.; Ramos, A.C.; Peres, A.M.; Pereira, J.A. Sweet peppers discrimination according to agronomic production mode and maturation stage using a chemical-sensory approach and an electronic tongue. Microchem. J. 2020. [Google Scholar] [CrossRef]
- Palma, P.; Alvarenga, P.; Palma, V.L.; Fernandes, R.M.; Soares, A.M.V.M.; Barbosa, I.R. Assessment of anthropogenic sources of water pollution using multivariate statistical techniques: A case study of the Alqueva’s reservoir, Portugal. Environ. Monit. Assess. 2010, 165, 539–552. [Google Scholar] [CrossRef]
- Alvarenga, N.; Taipina, M.; Raposo, N.; Dias, J.; Carvalho, M.J.; Amaral, O.; Lidon, F.C. Development of biscuits with green banana flour irradiated by 60Co: Preservation in modified atmosphere packaging. Emir. J. Food Agric. 2018, 30, 498–502. [Google Scholar]
- Bernardo, A.; Martínez, S.; Álvarez, M.; Fernández, A.; López, M. The composition of two Spanish pepper varieties (Fresno de la Vega and Benavente-los Valles) in different ripening stages. J. Food Qual. 2008, 31, 701–716. [Google Scholar] [CrossRef]
- Rubio, C.; Hardisson, A.; Martín, R.E.; Báez, A.; Martín, M.M.; Álvarez, R. Mineral composition of the red and green pepper (Capsicum annuum) from Tenerife Island. Eur. Food Res. Technol. 2002, 214, 501–504. [Google Scholar] [CrossRef]
- Ribes-Moya, A.M.; Pereira, L.; Guijarro-Real, C.; Raigon, M.D.; Fita, A.M.; Rodriguez-Burruezo, A. Mineral content in Capsicum pepper landraces: Effect of the genotype and the ripening stage. Bull. UASVM Hortic. 2014, 71. [Google Scholar] [CrossRef]
- Esayas, K.; Shimelis, A.; Ashebir, F.; Negussie, R.; Tilahun, B.; Gulelat, D. Proximate composition, mineral content and antinutritional factors of some capsicum (Capsicum annum) varieties grown in Ethiopia. Bull. Chem. Soc. Ethiopia 2011, 25, 451–454. [Google Scholar]
- Kim, E.-H.; Lee, S.-Y.; Baek, D.-Y.; Park, S.-Y.; Lee, S.-G.; Ryu, T.-H.; Lee, S.-K.; Kang, H.-J.; Kwon, O.-H.; Kil, M.; et al. A comparison of the nutrient composition and statistical profile in red pepper fruits (Capsicum annuum L.) based on genetic and environmental factors. Appl. Biol. Chem. 2019, 62. [Google Scholar] [CrossRef]
- Kantar, M.B.; Anderson, J.E.; Lucht, S.A.; Mercer, K.; Bernau, V.; Case, K.A.; Le, N.C.; Frederiksen, M.K.; DeKeyser, H.C.; Wong, Z.-Z.; et al. Vitamin variation in Capsicum Spp. provides opportunities to improve nutritional value of human diets. PLoS ONE 2016, 11, e0161464. [Google Scholar] [CrossRef]
- Howard, L.R.; Talcott, S.T.; Brenes, C.H.; Villalon, B. Changes in phytochemical and antioxidant activity of selected pepper cultivars (Capsicum species) as influenced by maturity. J. Agric. Food Chem. 2000, 48, 1713–1720. [Google Scholar] [CrossRef]
- Ninio, R.; Lewinsohn, E.; Mizrahi, Y.; Sitrit, Y. Changes in sugars, acids, and volatiles during ripening of koubo [Cereus peruvianus (L.) Miller] fruits. J. Agric. Food Chem. 2003, 51, 797–801. [Google Scholar] [CrossRef]
- Tosun, I.; Ustun, N.S.; Tekguler, B. Physical and chemical changes during ripening of blackberry fruits. Sci. agric. (Piracicaba, Braz.) 2008, 65, 87–90. [Google Scholar] [CrossRef]
- Hunter, D.; Foster, M.; McArthur, J.O.; Ojha, R.; Petocz, P.; Samman, S. Evaluation of the micronutrient composition of plant foods produced by organic and conventional agricultural methods. Crit Rev. Food Sci Nutr. 2011, 51, 571–582. [Google Scholar] [CrossRef] [PubMed]
- FAOSTAT, Crops. 2020. Available online: http://www.fao.org/faostat/en/#data/QC/visualize (accessed on 17 June 2020).
- Ogunlade, I.; Alebiosu, A.A.; Osasona, A.I. Proximate, mineral composition, antioxidant activity, and total phenolic content of some pepper varieties (Capsicum species). Int. J. Biol. Chem. Sci. 2012, 6, 2221–2227. [Google Scholar] [CrossRef][Green Version]
- Martínez-Ballesta, M.C.; Dominguez-Perles, R.; Moreno, D.A.; Muries, B.; Alcaraz-López, C.; Bastías, E.; García-Viguera, C.; Carvajal, M. Minerals in plant food: Effect of agricultural practices and role in human health. A review. Agron. Sustain. Dev. 2010, 30, 295–309. [Google Scholar] [CrossRef]
- Kemi, V.E.; Kärkkäinen, M.U.M.; Rita, H.J.; Laaksonen, M.M.L.; Outila, T.A.; Lamberg-Allardt, C.J.E. Low calcium:phosphorus ratio in habitual diets affects serum parathyroid hormone concentration and calcium metabolism in healthy women with adequate calcium intake. Br. J. Nutr. 2010, 103, 561–568. [Google Scholar] [CrossRef]
- O’Dell, B.L. Mineral interactions relevant to nutrient requirements. J. Nutr. 1989, 119, 1832–1838. [Google Scholar] [CrossRef] [PubMed]
- Rossander-Hultén, L.; Brune, M.; Sandström, B.; Lönnerdal, B.; Hallberg, L. Competitive inhibition of iron absorption by manganese and zinc in humans. Am. J. Clin. Nutr. 1991, 54, 152–156. [Google Scholar] [CrossRef]
- Collins, J.F.; Prohaska, J.R.; Knutson, M.D. Metabolic crossroads of iron and copper. Nutr. Rev. 2010, 68, 133–147. [Google Scholar] [CrossRef] [PubMed]
- Linus Pauling Institute. Micronutrient Information Center, Oregon State University, Iron. 2017. Available online: https://lpi.oregonstate.edu/mic/minerals/iron (accessed on 6 May 2020).
- Food and Nutrition Board, Institute of Medicine. Copper. Dietary Reference Intakes for Vitamin A, Vitamin K, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academy Press: Washington, DC, USA, 2001; pp. 224–257. [Google Scholar]
- Osredkar, J.; Sustar, N. Copper and zinc, biological role and significant of copper/zinc imbalance. J. Clin. Toxicol. 2011, S3, 001. [Google Scholar] [CrossRef]
- Kelly, O.J.; Gilman, J.C.; Ilich, J.Z. Utilizing dietary micronutrient ratios in nutritional research may be more informative than focusing on single nutrients. Nutrients 2018, 10, 107. [Google Scholar] [CrossRef] [PubMed]
- Kelly, O.J.; Gilman, J.C.; Ilich, J.Z. Utilizing dietary micronutrient ratios in nutritional research. Expanding the concept of nutrient ratios to macronutrients. Nutrients 2019, 11, 282. [Google Scholar] [CrossRef]
- Powers, K.M.; Smith-Weller, T.; Franklin, G.M.; Longstreth, W.T.; Swanson, P.D.; Checkoway, H. Parkinson’s disease risks associated with dietary iron, manganese, and other nutrient intakes. Neurology 2003, 60, 1761–1766. [Google Scholar] [CrossRef]
- Sandstrom, B. Micronutrient interactions: Effects on absorption and bioavailability. Br. J. Nutr. 2001, 85 (Suppl. 2), S181–S185. [Google Scholar] [CrossRef]
- Pataco, I.M.; Lidon, F.C.; Ramos, I.; Oliveira, K.; Guerra, M.; Pessoa, M.F.; Carvalho, M.L.; Ramalho, J.C.; Leitão, A.E.; Santos, J.P.; et al. Biofortification of durum wheat (Triticum turgidum L. ssp. Durum (Desf.) Husnot) grains with nutrients. J. Plant Interact. 2017, 12, 39–50. [Google Scholar] [CrossRef]
- Lidon, F.C.; Ribeiro, A.; Leitão, A.; Pataco, I.; Ribeiro, M.M.; Reboredo, F.; Almeida, A.; Silva, M.M.; Pais, I.; Pelica, J.; et al. Selenium biofortification of rice grains and implications in the nutritional quality. J. Cereal Sci. 2018, 81, 22–29. [Google Scholar] [CrossRef]
- Mangueze, A.; Pessoa, M.F.; Silva, M.J.; Ndayiragije, A.; Magaia, H.; Cossa, V.; Reboredo, F.; Carvalho, M.L.; Santos, J.P.; Guerra, M.; et al. Simultaneous zinc and selenium biofortification in rice accumulation, localization and implications on the overall mineral content of the flour. J. Cereal Sci. 2018, 82, 34–41. [Google Scholar] [CrossRef]
- Olatunji, T.L.; Afolayan, A.J. The suitability of chili pepper (Capsicum annuum L.) for alleviating human micronutrient dietary deficiencies: A review. Food Sci. Nutr. 2018, 6, 2239–2251. [Google Scholar] [CrossRef]
- AOAC. Association of Official Analytical Chemists (AOAC) International Official Methods of Analysis, 16th ed.; AOAC: Rockville, MD, USA, 1997. [Google Scholar]
- Barroca, M.J.; Guiné, R.P.F.; Amado, A.M.; Ressurreição, S.; Moreira da Silva, A.; Marques, M.P.M.; Batista de Carvalho, L.A.E. The drying process of Sarcocornia perennis: Impact on nutritional and physico-chemical properties. J. Food Sci. Technol. 2020. [Google Scholar] [CrossRef]
- International Pharmacopoeia—Ninth Edition, 2019, method:10.4.2. Measurement of Consistency by Penetrometry. Available online: https://apps.who.int/phint/pdf/b/10.4.2.Measurement-of-consistency-by-penetrometry.pdf (accessed on 21 June 2020).
- Mitcham, B.; Cantwell, M.; Kader, A. Methods for determining quality of fresh commodities. Perish. Handl. Newslett. 1996, 85, 5. [Google Scholar]
- Cardoso, P.; Mateus, T.; Velu, G.; Singh, R.P.; Santos, J.P.; Carvalho, M.L.; Lourenço, V.M.; Lidon, F.; Reboredo, F.; Guerra, M. Localization and distribution of Zn and Fe in grains of biofortified bread wheat lines through micro and triaxial-X-ray spectrometry. Spectrochim. Acta Part B At. Spectrosc. 2018, 141, 70–79. [Google Scholar] [CrossRef]
- Gallardo, H.; Queralt, I.; Tapias, J.; Guerra, M.; Carvalho, M.L.; Marguí, E. Possibilities of low-power X-ray fluorescence spectrometry techniques for rapid multielemental analysis and imaging of vegetal foodstuffs. J. Food Comp. Anal. 2016, 50, 1–9. [Google Scholar] [CrossRef]
| Elements | Green Peppers | Red Peppers | ||
|---|---|---|---|---|
| Organic | Conventional | Organic | Conventional | |
| K | 4.02 ± 0.299 a | 3.55 ± 0.290 ab | 3.65 ± 0.276 ab | 3.27 ± 0.345 b |
| Ca | 1930 ± 144.8 a | 1009 ± 97.10 b | 1314 ± 365.8 b | 1041 ± 203.3 b |
| P | 1974 ± 394.7 a | 1528 ± 208.6 a | 2044 ± 281.38 a | 1704 ± 313.2 a |
| Fe | 84.1 ± 10.4 a | 64.7 ± 5.4 b | 68.0 ± 13.9 ab | 73.6 ± 8.5 ab |
| Zn | 19.2 ± 2.27 b | 25.0 ± 2.43 a | 15.7 ± 1.87 b | 18.6 ± 1.82 b |
| Mn | 7.37 ± 2.07 b | 21.3 ± 4.51 a | 6.49 ± 1.92 b | 22.5 ± 11.5 a |
| S | 1644 ± 329.9 a | 1518 ± 216.3 a | 1232 ± 162.2 a | 1566 ± 204.5 a |
| Cu | 11.8 ± 4.21 a | 11.6 ± 2.21 a | 9.32 ± 0.99 a | 7.94 ± 4.33 a |
| Cl | 3892 ± 630.9 a | 3250 ± 578.5 a | 2862 ± 959.2 a | 2984 ± 437.1 a |
| Ca/P | 0.98 | 0.66 | 0.64 | 0.61 |
| Mn/Fe | 0.087 | 0.329 | 0.095 | 0.306 |
| Fe/Cu | 7.13 | 5.58 | 7.30 | 9.27 |
| Zn/Cu | 1.63 | 2.16 | 1.68 | 2.34 |
| Green Peppers | Red Peppers | |||
|---|---|---|---|---|
| OA | CA | OA | CA | |
| Fruit weight | 159 ± 37.7 a | 127 ± 16.1 ab | 123 ± 13.8 ab | 97.4 ± 18.9 b |
| Moisture * | 93.9 ± 1.30 a | 92.5 ± 0.60 a | 91.8 ± 0.75 a | 90.3 ± 0.43 b |
| Protein * | 10.3 ± 0.62 b | 11.7 ± 0.56 a | 9.17 ± 0.49 b | 11.9 ± 0.42 a |
| Ash * | 6.38 ± 0.58 a | 4.98 ± 0.32 b | 5.87 ± 0.28 a | 5.03 ± 0.27 b |
| Fiber * | 11.4 ± 0.93 a | 11.6 ± 0.74 a | 9.23 ± 1.00 b | 10.3 ± 1.06 ab |
| Firmness ** | 12.1 ± 2.3 a | 7.9 ± 0.9 b | 9.2 ± 1.4 ab | 7.9 ± 1.2 b |
| pH ** | 6.2 ± 0.1 a | 6.0 ± 0.5 a | 5.0 ± 0.1 b | 5.2 ± 0.1 b |
| TSS(°Brix) ** | 3.8 ± 0.3 c | 4.4 ± 0.5 c | 5.8 ± 0.7 b | 7.6 ± 0.5 a |
| TA ** | 63 ± 4.0 b | 67 ± 9.0 b | 162 ± 16.0 a | 171 ± 18.0 a |
| Organic Agriculture | Conventional Agriculture | |||
| Productivity | 28.0 | 30.1 | ||
| Principal Component | Eigenvalue | Total Variance (%) | Cumulative Eigenvalue | Cumulative (%) |
|---|---|---|---|---|
| 1 | 3.13 | 39.19 | 3.13 | 39.19 |
| 2 | 2.08 | 26.06 | 5.22 | 65.24 |
| 3 | 0.89 | 11.09 | 6.11 | 76.33 |
| 4 | 0.63 | 7.93 | 6.74 | 84.26 |
| 5 | 0.44 | 5.51 | 7.18 | 89.76 |
| 6 | 0.33 | 4.17 | 7.52 | 93.94 |
| 7 | 0.25 | 3.17 | 7.77 | 97.11 |
| 8 | 0.23 | 2.89 | 8.00 | 100.00 |
| Components | ||
|---|---|---|
| Attribute | PC1 | PC2 |
| P | −0.68 * | −0.02 |
| Ca | −0.82 ** | 0.09 |
| Cu | −0.37 | 0.55 |
| Fe | −0.75 ** | 0.32 |
| Mn | 0.62 * | 0.57 |
| K | −0.86 ** | −0.19 |
| Zn | 0.26 | 0.83 ** |
| S | −0.29 | 0.79 ** |
| Stage Development | PM | 16/May | 21 | 28 | 04/Jun | 11 | 18 | 25 | 02/Jul | 09 | 16 | 23 | 30 | 06/Aug | 13 | 20 | 27 | 03/Sept | 10 | 17 | 24t |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Transplanting | OA | ||||||||||||||||||||
| CA | |||||||||||||||||||||
| Leaf Development | OA | ≤10% | 10–25% | 25–50% | 50–75% | ≥75% | ≥75% | ||||||||||||||
| CA | ≤10% | 10–25% | 50–75% | 50–75% | ≥75% | ≥75% | |||||||||||||||
| Flowering | OA | ≤10% | 10–25% | 25–50% | 50–75% | ≥75% | 25–50% | 10–25% | |||||||||||||
| CA | ≤10% | 25–50% | 25–50% | 50–75% | ≥75% | 25–50% | 10–25% | ||||||||||||||
| Development of Fruit | OA | ≤10% | 10–25% | 25–50% | 50–75% | ≥75% | ≥75% | 50–75% | 25–50% | ||||||||||||
| CA | ≤10% | 25–50% | 25–50% | 50–75% | ≥75% | ≥75% | 50–75% | 25–50% | |||||||||||||
| Ripening of Fruit | OA | ≤10% | 10–25% | 25–50% | 50–75% | ≥75% | ≥75% | ≥75% | ≥75% | ||||||||||||
| CA | ≤10% | 25–50% | 25–50% | 50–75% | ≥75% | ≥75% | ≥75% | ≥75% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guilherme, R.; Reboredo, F.; Guerra, M.; Ressurreição, S.; Alvarenga, N. Elemental Composition and Some Nutritional Parameters of Sweet Pepper from Organic and Conventional Agriculture. Plants 2020, 9, 863. https://doi.org/10.3390/plants9070863
Guilherme R, Reboredo F, Guerra M, Ressurreição S, Alvarenga N. Elemental Composition and Some Nutritional Parameters of Sweet Pepper from Organic and Conventional Agriculture. Plants. 2020; 9(7):863. https://doi.org/10.3390/plants9070863
Chicago/Turabian StyleGuilherme, Rosa, Fernando Reboredo, Mauro Guerra, Sandrine Ressurreição, and Nuno Alvarenga. 2020. "Elemental Composition and Some Nutritional Parameters of Sweet Pepper from Organic and Conventional Agriculture" Plants 9, no. 7: 863. https://doi.org/10.3390/plants9070863
APA StyleGuilherme, R., Reboredo, F., Guerra, M., Ressurreição, S., & Alvarenga, N. (2020). Elemental Composition and Some Nutritional Parameters of Sweet Pepper from Organic and Conventional Agriculture. Plants, 9(7), 863. https://doi.org/10.3390/plants9070863








