Screening for Drought Tolerance in Maize (Zea mays L.) Germplasm Using Germination and Seedling Traits under Simulated Drought Conditions
Abstract
1. Introduction
2. Material and Methods
Data Analyses
3. Results
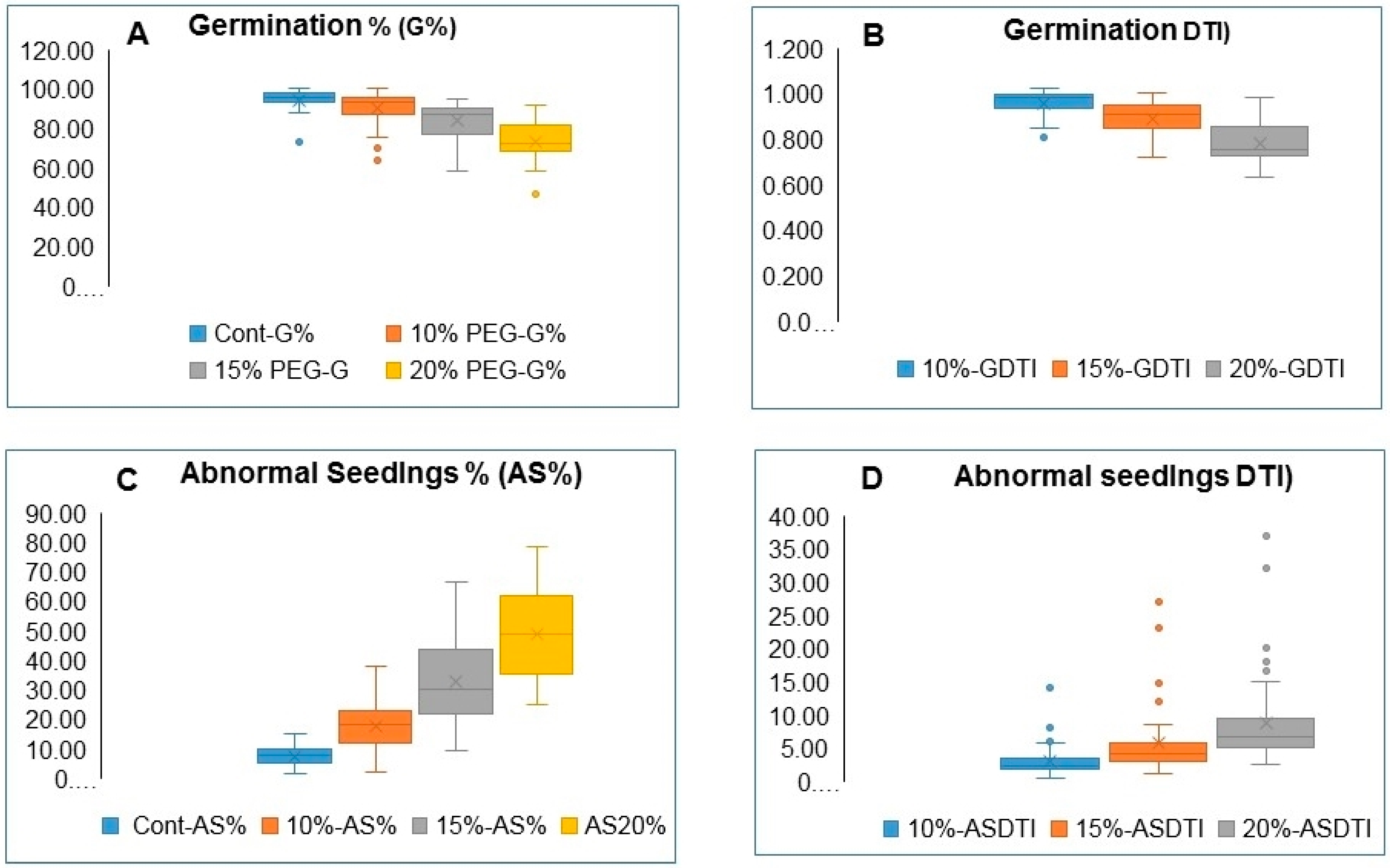
3.1. Variation in Germination and Abnormal Seedlings Percentage
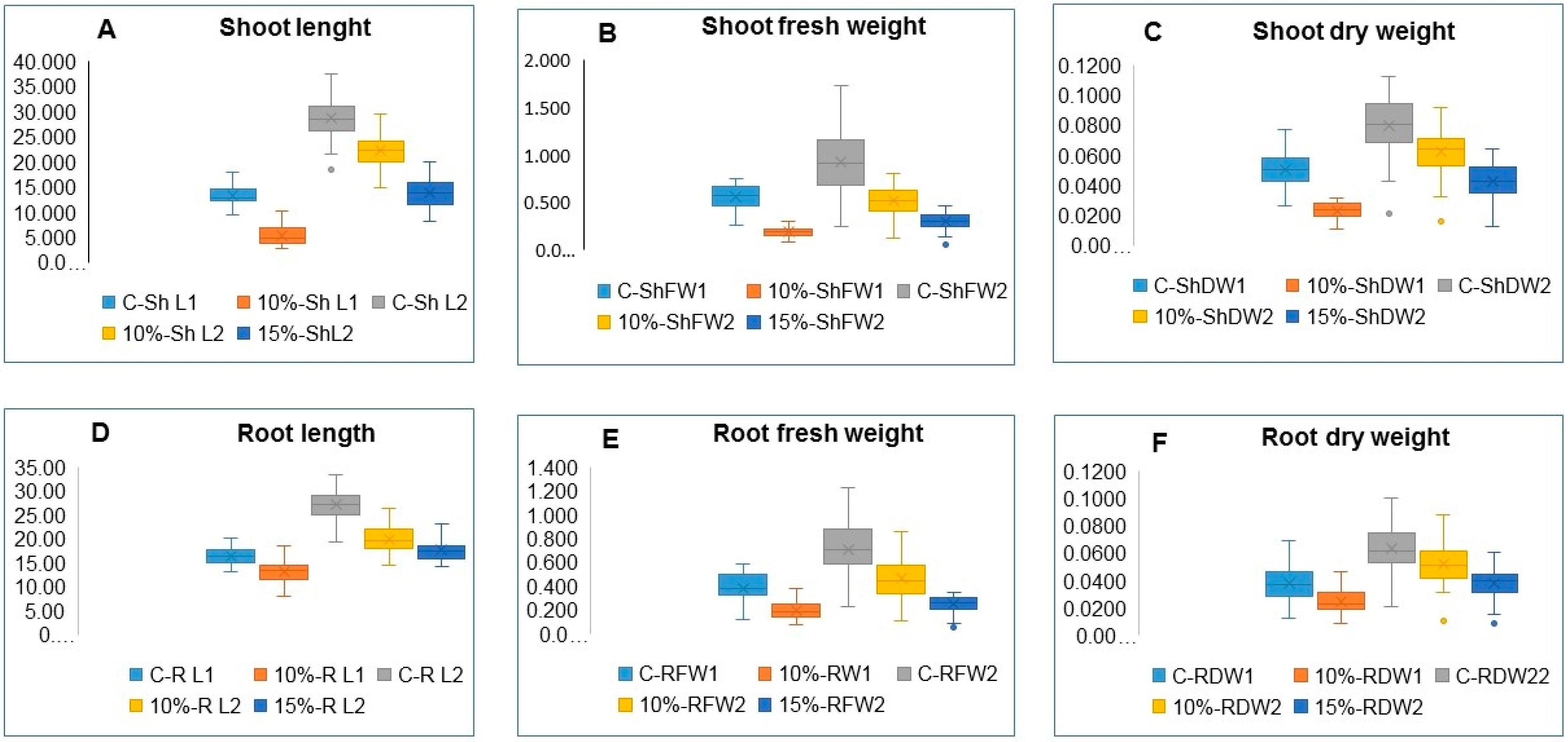
3.2. Variation in Seedling’s Traits
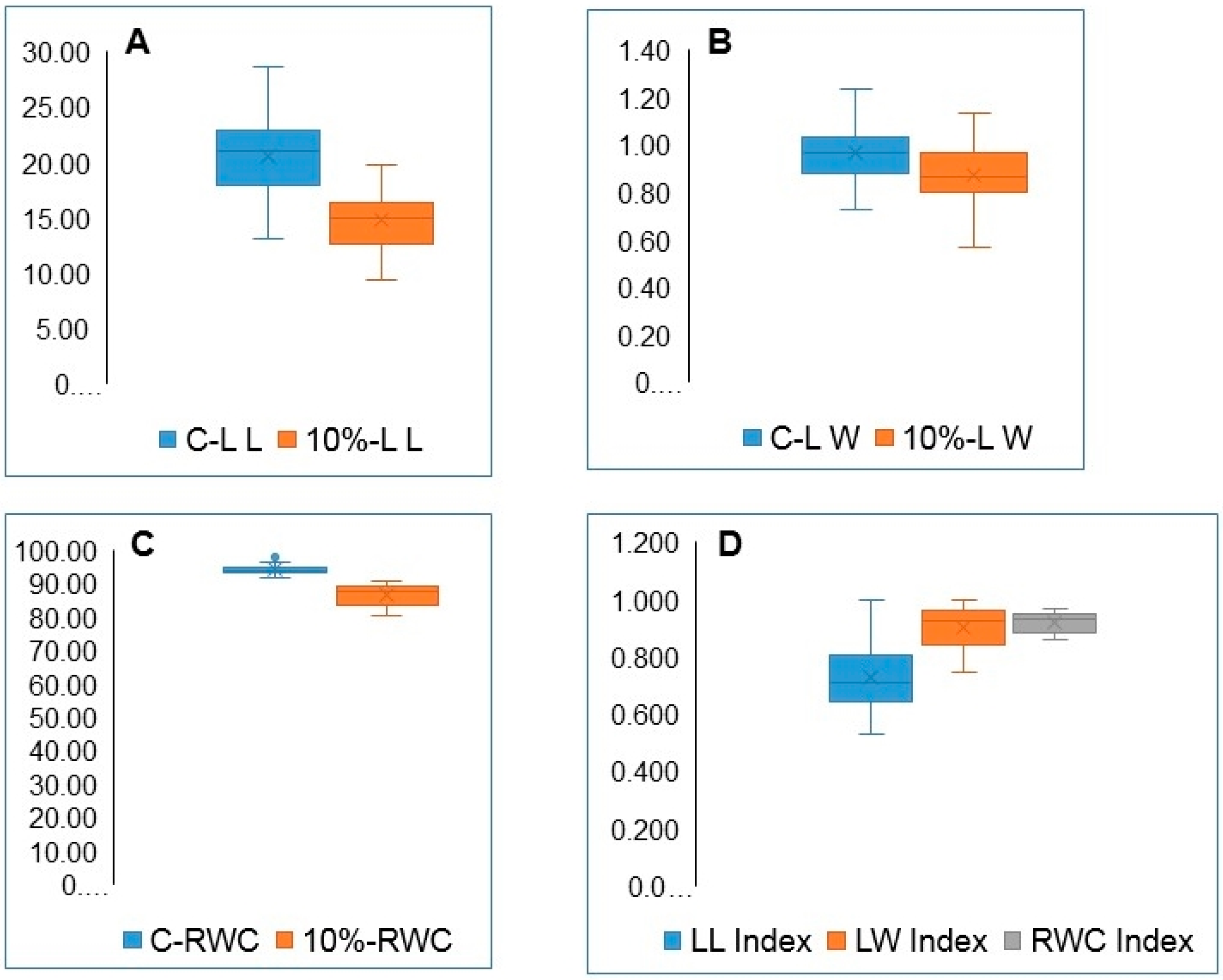
3.3. Variation in Leaf Length, Width, and RWC
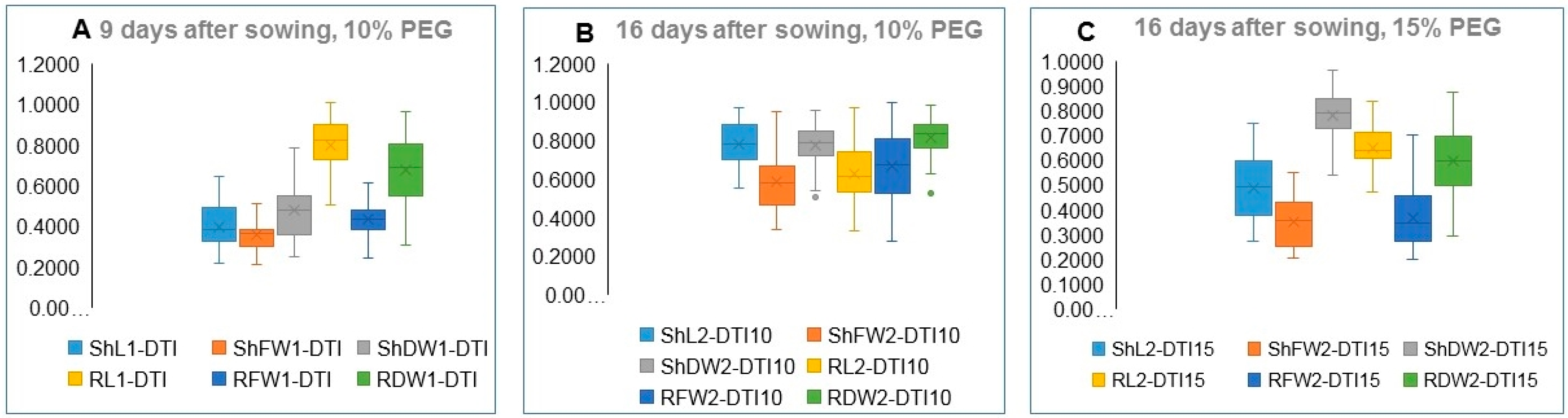
3.4. Heritability of Traits in Control and PEG-Stressed Traits
3.5. Correlations of Traits under Control and PEG Stress
3.6. Screening for Drought-Tolerant Traits and Accessions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Accession | GI 10% | GI 15% | GI 20% | AS DTI10 | AS DTI15 | AS DTI20 | ShL1 DTI | RL1 DTI | ShFW DTI | RFW DTI | ShDW DTI | RDW DTI |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zea 12 | 0.98 | 0.86 | 0.64 | 0.86 | 4.71 | 4.57 | 0.40 | 0.61 | 0.36 | 0.60 | 0.45 | 0.69 |
| Zea242 | 0.96 | 0.97 | 0.92 | 0.50 | 3.50 | 10.5 | 0.42 | 0.82 | 0.50 | 0.60 | 0.56 | 0.81 |
| Zea323 | 0.85 | 0.85 | 0.85 | 1.50 | 8.50 | 18.0 | 0.42 | 0.85 | 0.51 | 0.60 | 0.72 | 0.76 |
| Zea355 | 0.98 | 1.00 | 0.89 | 1.00 | 5.71 | 6.71 | 0.24 | 0.62 | 0.36 | 0.59 | 0.30 | 0.52 |
| Zea382 | 0.93 | 0.93 | 0.88 | 1.00 | 5.00 | 7.50 | 0.49 | 0.79 | 0.36 | 0.46 | 0.51 | 0.79 |
| Zea394 | 0.94 | 0.87 | 0.81 | 0.80 | 3.80 | 7.60 | 0.44 | 0.83 | 0.36 | 0.44 | 0.49 | 0.85 |
| Zea487 | 0.93 | 0.90 | 0.75 | 1.00 | 5.33 | 5.83 | 0.41 | 0.83 | 0.38 | 0.45 | 0.53 | 0.66 |
| Zea630 | 1.00 | 0.93 | 0.85 | 4.00 | 5.50 | 10.0 | 0.54 | 0.93 | 0.38 | 0.41 | 0.53 | 0.31 |
| Zea633 | 0.97 | 0.95 | 0.84 | 3.00 | 4.00 | 5.00 | 0.59 | 0.94 | 0.51 | 0.55 | 0.72 | 0.93 |
| Zea668 | 0.91 | 0.94 | 0.76 | 2.00 | 7.60 | 7.58 | 0.53 | 0.94 | 0.38 | 0.40 | 0.53 | 0.80 |
| Zea677 | 0.93 | 0.91 | 0.64 | 1.78 | 2.89 | 3.22 | 0.35 | 0.56 | 0.34 | 0.61 | 0.42 | 0.37 |
| Zea711 | 1.00 | 0.93 | 0.69 | 8.00 | 27.0 | 37.0 | 0.55 | 1.00 | 0.49 | 0.32 | 0.67 | 0.69 |
| Zea1006 | 0.95 | 0.95 | 0.90 | 1.83 | 2.17 | 5.33 | 0.33 | 0.73 | 0.33 | 0.46 | 0.48 | 0.88 |
| Zea1015 | 1.00 | 0.95 | 0.86 | 3.25 | 3.00 | 6.75 | 0.51 | 0.94 | 0.38 | 0.40 | 0.55 | 0.85 |
| Zea1019 | 1.00 | 0.96 | 0.98 | 2.33 | 2.50 | 5.00 | 0.58 | 0.73 | 0.50 | 0.61 | 0.78 | 0.77 |
| Zea1062 | 0.98 | 0.96 | 0.86 | 14.1 | 23.1 | 32.1 | 0.37 | 0.77 | 0.30 | 0.39 | 0.34 | 0.53 |
| Zea1102 | 0.98 | 0.97 | 0.86 | 6.00 | 12.00 | 20.0 | 0.54 | 0.84 | 0.38 | 0.45 | 0.49 | 0.95 |
| Zea1114 | 0.98 | 0.98 | 0.72 | 3.00 | 8.33 | 9.33 | 0.48 | 0.84 | 0.40 | 0.48 | 0.59 | 0.81 |
| Zea1121 | 0.94 | 0.89 | 0.86 | 3.04 | 6.30 | 7.41 | 0.65 | 1.01 | 0.39 | 0.38 | 0.40 | 0.92 |
| Zea1224 | 1.00 | 0.95 | 0.93 | 0.67 | 1.00 | 5.00 | 0.55 | 1.00 | 0.45 | 0.44 | 0.58 | 0.96 |
| Zea3002 | 0.87 | 0.94 | 0.74 | 2.24 | 3.92 | 3.14 | 0.34 | 0.54 | 0.36 | 0.57 | 0.55 | 0.63 |
| Zea3065 | 0.91 | 0.89 | 0.78 | 5.35 | 4.87 | 12.0 | 0.23 | 0.63 | 0.21 | 0.34 | 0.32 | 0.41 |
| Zea3175 | 0.98 | 0.72 | 0.73 | 2.36 | 2.50 | 4.23 | 0.30 | 0.64 | 0.29 | 0.46 | 0.44 | 0.81 |
| Zea3244 | 0.86 | 0.80 | 0.64 | 2.33 | 2.27 | 2.80 | ||||||
| Zea3257 | 1.00 | 0.95 | 0.74 | 2.33 | 4.33 | 6.67 | 0.33 | 0.50 | 0.25 | 0.51 | 0.35 | 0.40 |
| Zea3280 | 0.98 | 0.93 | 0.75 | 3.00 | 3.00 | 10.0 | 0.33 | 0.75 | 0.29 | 0.38 | 0.38 | 0.58 |
| Zea3282 | 0.97 | 0.78 | 0.76 | 2.17 | 2.00 | 3.83 | 0.38 | 0.85 | 0.39 | 0.45 | 0.49 | 0.58 |
| Zea3301 | 1.00 | 0.72 | 0.69 | 2.29 | 2.57 | 2.57 | 0.28 | 0.90 | 0.30 | 0.33 | 0.43 | 0.80 |
| Zea3324 | 0.80 | 0.78 | 0.73 | 2.75 | 4.10 | 5.10 | 0.27 | 0.63 | 0.22 | 0.35 | 0.32 | 0.38 |
| Zea3325 | 0.96 | 0.91 | 0.85 | 1.00 | 5.00 | 15.0 | 0.37 | 0.82 | 0.33 | 0.40 | 0.48 | 0.55 |
| Zea3346 | 0.98 | 0.75 | 0.73 | 2.20 | 3.80 | 6.60 | 0.38 | 1.00 | 0.31 | 0.30 | 0.25 | 0.67 |
| Zea3392 | 1.00 | 0.93 | 0.73 | 4.00 | 3.40 | 7.00 | 0.24 | 0.63 | 0.29 | 0.31 | 0.30 | 0.55 |
| Zea3400 | 0.96 | 0.91 | 0.72 | 3.49 | 5.66 | 6.17 | 0.22 | 0.84 | 0.23 | 0.27 | 0.33 | 0.76 |
| Zea3424 | 1.02 | 0.88 | 0.70 | 1.83 | 2.44 | 6.72 | 0.32 | 0.78 | 0.37 | 0.48 | 0.52 | 0.74 |
| Zea3425 | 0.99 | 0.81 | 0.64 | 1.19 | 3.50 | 3.85 | 0.40 | 0.75 | 0.34 | 0.46 | 0.47 | 0.60 |
| Zea3576 | 0.98 | 0.85 | 0.65 | 6.00 | 14.0 | 16.0 | 0.39 | 1.00 | 0.40 | 0.40 | 0.57 | 0.73 |
| Zea3582 | 1.00 | 0.81 | 0.75 | 6.33 | 5.33 | 7.33 | 0.37 | 0.75 | 0.34 | 0.45 | 0.70 | 0.52 |
| Zea3602 | 0.98 | 0.89 | 0.74 | 5.67 | 5.67 | 5.50 | 0.32 | 0.78 | 0.25 | 0.32 | 0.36 | 0.55 |
| Zea3712 | 0.98 | 0.81 | 0.81 | 2.75 | 3.75 | 6.00 | 0.40 | 0.85 | 0.35 | 0.41 | 0.47 | 0.79 |
| ZeaIW237 | 0.95 | 0.88 | 0.75 | 3.33 | 6.33 | 7.67 | 0.31 | 0.77 | 0.29 | 0.24 | 0.34 | 0.58 |
| Accession | ShL2 DTI10 | ShFW2 DTI10 | ShDW2 DTI10 | RL2 DTI10 | RFW2 DTI10 | RDW2 DTI10 | ShL2 DTI15 | ShFW2 DTI15 | ShDW2 DTI15 | RL2 DTI15 | RFW2 DTI15 | RDW2 DTI15 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zea 12 | 0.80 | 0.46 | 0.74 | 0.40 | 0.28 | 0.63 | 0.32 | 0.21 | 0.74 | 0.63 | 0.25 | 0.29 |
| Zea242 | 0.93 | 0.63 | 0.87 | 0.69 | 0.67 | 0.90 | 0.64 | 0.47 | 0.87 | 0.62 | 0.33 | 0.59 |
| Zea323 | 0.81 | 0.65 | 0.71 | 0.55 | 0.81 | 0.73 | 0.45 | 0.38 | 0.71 | 0.76 | 0.49 | 0.56 |
| Zea355 | 0.75 | 0.64 | 0.85 | 0.61 | 0.67 | 0.79 | 0.46 | 0.47 | 0.85 | 0.69 | 0.36 | 0.48 |
| Zea382 | 0.88 | 0.85 | 0.78 | 0.69 | 0.84 | 0.70 | 0.60 | 0.48 | 0.78 | 0.64 | 0.46 | 0.65 |
| Zea394 | 0.88 | 0.75 | 0.80 | 0.54 | 0.85 | 0.70 | 0.47 | 0.34 | 0.80 | 0.62 | 0.40 | 0.56 |
| Zea487 | 0.66 | 0.50 | 0.65 | 0.67 | 0.62 | 0.75 | 0.44 | 0.42 | 0.65 | 0.64 | 0.42 | 0.65 |
| Zea630 | 0.88 | 0.65 | 0.85 | 0.63 | 0.69 | 0.85 | 0.55 | 0.55 | 0.85 | 0.64 | 0.34 | 0.73 |
| Zea633 | 0.81 | 0.45 | 0.77 | 0.82 | 0.42 | 0.91 | 0.66 | 0.32 | 0.77 | 0.64 | 0.33 | 0.44 |
| Zea668 | 0.82 | 0.47 | 0.78 | 0.74 | 0.42 | 0.84 | 0.61 | 0.34 | 0.78 | 0.56 | 0.27 | 0.67 |
| Zea677 | 0.89 | 0.66 | 0.77 | 0.84 | 0.63 | 0.78 | 0.75 | 0.51 | 0.77 | 0.71 | 0.70 | 0.65 |
| Zea711 | 0.93 | 0.73 | 0.69 | 0.71 | 0.82 | 0.90 | 0.66 | 0.43 | 0.69 | 0.71 | 0.46 | 0.79 |
| Zea1006 | 0.73 | 0.63 | 0.96 | 0.78 | 0.71 | 0.98 | 0.57 | 0.43 | 0.96 | 0.76 | 0.35 | 0.70 |
| Zea1015 | 0.65 | 0.43 | 0.88 | 0.97 | 0.64 | 0.90 | 0.63 | 0.47 | 0.88 | 0.74 | 0.55 | 0.76 |
| Zea1019 | 0.84 | 0.95 | 0.89 | 0.82 | 0.71 | 0.91 | 0.69 | 0.46 | 0.89 | 0.67 | 0.33 | 0.59 |
| Zea1062 | 0.88 | 0.67 | 0.80 | 0.70 | 0.74 | 0.84 | 0.61 | 0.39 | 0.80 | 0.59 | 0.32 | 0.61 |
| Zea1102 | 0.78 | 0.53 | 0.82 | 0.80 | 0.67 | 0.85 | 0.62 | 0.37 | 0.82 | 0.63 | 0.42 | 0.83 |
| Zea1114 | 0.69 | 0.57 | 0.86 | 0.81 | 0.67 | 0.77 | 0.56 | 0.41 | 0.86 | 0.64 | 0.34 | 0.63 |
| Zea1121 | 0.77 | 0.41 | 0.87 | 0.71 | 0.52 | 0.76 | 0.55 | 0.20 | 0.87 | 0.70 | 0.20 | 0.43 |
| Zea1224 | 0.71 | 0.34 | 0.85 | 0.76 | 0.81 | 0.94 | 0.54 | 0.31 | 0.85 | 0.65 | 0.43 | 0.85 |
| Zea3002 | 0.93 | 0.83 | 0.80 | 0.53 | 0.64 | 0.76 | 0.50 | 0.40 | 0.80 | 0.71 | 0.50 | 0.88 |
| Zea3065 | 0.64 | 0.47 | 0.85 | 0.47 | 0.50 | 0.87 | 0.30 | 0.23 | 0.85 | 0.78 | 0.21 | 0.55 |
| Zea3175 | 0.75 | 0.69 | 0.79 | 0.55 | 0.88 | 0.80 | 0.41 | 0.39 | 0.79 | 0.60 | 0.59 | 0.42 |
| Zea3244 | 0.80 | 0.51 | 0.76 | 0.74 | 0.48 | 0.53 | 0.59 | 0.22 | 0.76 | 0.81 | 0.22 | 0.40 |
| Zea3257 | 0.71 | 0.50 | 0.66 | 0.46 | 0.56 | 0.86 | 0.33 | 0.28 | 0.66 | 0.73 | 0.22 | 0.55 |
| Zea3280 | 0.75 | 0.39 | 0.75 | 0.60 | 0.67 | 0.86 | 0.45 | 0.23 | 0.75 | 0.63 | 0.44 | 0.77 |
| Zea3282 | 0.66 | 0.52 | 0.79 | 0.82 | 0.45 | 0.70 | 0.54 | 0.38 | 0.79 | 0.67 | 0.28 | 0.54 |
| Zea3301 | 0.79 | 0.59 | 0.82 | 0.47 | 0.77 | 0.89 | 0.37 | 0.31 | 0.82 | 0.67 | 0.21 | 0.62 |
| Zea3324 | 0.56 | 0.40 | 0.78 | 0.61 | 0.38 | 0.79 | 0.34 | 0.22 | 0.78 | 0.50 | 0.24 | 0.54 |
| Zea3325 | 0.90 | 0.81 | 0.81 | 0.57 | 0.86 | 0.93 | 0.51 | 0.36 | 0.81 | 0.61 | 0.49 | 0.80 |
| Zea3346 | 0.94 | 0.66 | 0.83 | 0.42 | 0.90 | 0.77 | 0.39 | 0.28 | 0.83 | 0.61 | 0.49 | 0.70 |
| Zea3392 | 0.70 | 0.48 | 0.70 | 0.58 | 0.77 | 0.87 | 0.41 | 0.21 | 0.70 | 0.53 | 0.37 | 0.68 |
| Zea3400 | 0.71 | 0.69 | 0.86 | 0.39 | 1.00 | 0.83 | 0.27 | 0.30 | 0.86 | 0.47 | 0.43 | 0.41 |
| Zea3424 | 0.67 | 0.61 | 0.54 | 0.62 | 0.80 | 0.82 | 0.42 | 0.28 | 0.62 | 0.47 | 0.49 | 0.56 |
| Zea3425 | 0.94 | 0.53 | 0.92 | 0.33 | 0.80 | 0.82 | 0.31 | 0.24 | 0.92 | 0.64 | 0.31 | 0.56 |
| Zea3576 | 0.72 | 0.53 | 0.71 | 0.52 | 0.50 | 0.86 | 0.37 | 0.22 | 0.71 | 0.62 | 0.26 | 0.43 |
| Zea3582 | 0.63 | 0.42 | 0.50 | 0.58 | 0.39 | 0.79 | 0.37 | 0.35 | 0.60 | 0.52 | 0.24 | 0.60 |
| Zea3602 | 0.60 | 0.42 | 0.54 | 0.54 | 0.54 | 0.87 | 0.32 | 0.23 | 0.54 | 0.56 | 0.28 | 0.42 |
| Zea3712 | 0.79 | 0.63 | 0.72 | 0.63 | 0.80 | 0.96 | 0.49 | 0.45 | 0.72 | 0.84 | 0.43 | 0.62 |
| ZeaIW237 | 0.97 | 0.77 | 0.74 | 0.51 | 0.92 | 0.64 | 0.49 | 0.43 | 0.74 | 0.73 | 0.30 | 0.45 |
| Accession | LL Index | LW Index | RWC INDEX |
|---|---|---|---|
| Zea 12 | 0.53 | 0.79 | 0.88 |
| Zea242 | 1.00 | 0.96 | 0.87 |
| Zea323 | 0.78 | 1.00 | 0.90 |
| Zea355 | 0.58 | 0.84 | 0.88 |
| Zea382 | 0.88 | 0.94 | 0.90 |
| Zea394 | 0.77 | 0.94 | 0.88 |
| Zea487 | 0.64 | 0.93 | 0.91 |
| Zea630 | 0.79 | 0.78 | 0.94 |
| Zea633 | 0.66 | 1.01 | 0.95 |
| Zea668 | 0.81 | 0.77 | 0.89 |
| Zea677 | 0.96 | 0.85 | 0.88 |
| Zea711 | 0.81 | 1.00 | 0.93 |
| Zea1006 | 0.74 | 0.93 | 0.95 |
| Zea1015 | 0.68 | 0.92 | 0.97 |
| Zea1019 | 0.96 | 1.00 | 0.95 |
| Zea1062 | 0.80 | 1.00 | 0.94 |
| Zea1102 | 0.57 | 0.77 | 0.96 |
| Zea1114 | 0.72 | 0.81 | 0.89 |
| Zea1121 | 0.73 | 0.89 | 0.94 |
| Zea1224 | 0.65 | 0.93 | 0.95 |
| Zea3002 | 0.83 | 0.90 | 0.89 |
| Zea3065 | 0.59 | 0.97 | 0.96 |
| Zea3175 | 0.60 | 0.89 | 0.93 |
| Zea3244 | 0.71 | 0.88 | 0.86 |
| Zea3257 | 0.65 | 1.00 | 0.87 |
| Zea3280 | 0.75 | 0.97 | 0.94 |
| Zea3282 | 0.69 | 0.77 | 0.93 |
| Zea3301 | 0.65 | 0.97 | 0.88 |
| Zea3324 | 0.55 | 1.00 | 0.94 |
| Zea3325 | 0.92 | 0.98 | 0.95 |
| Zea3346 | 0.70 | 0.96 | 0.95 |
| Zea3392 | 0.56 | 0.76 | 0.89 |
| Zea3400 | 0.63 | 0.94 | 0.87 |
| Zea3424 | 0.67 | 0.93 | 0.92 |
| Zea3425 | 0.84 | 0.88 | 0.90 |
| Zea3576 | 0.56 | 0.93 | 0.94 |
| Zea3582 | 0.70 | 0.96 | 0.95 |
| Zea3602 | 0.69 | 0.86 | 0.95 |
| Zea3712 | 0.90 | 0.96 | 0.96 |
| ZeaIW237 | 0.86 | 0.95 | 0.94 |
References
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef]
- Lobell, D.B.; Roberts, M.J.; Schlenker, W.; Braun, N.L.; Little, B.B.; Rejesus, R.M.; Hammer, G.L. Greater sensitivity to drought accompanies maize yield increase in the U.S-Midwest. Science 2014, 344, 516–519. [Google Scholar] [CrossRef] [PubMed]
- Osborn, D.; Ferguson, J.N. Climate change and abiotic stress mechanisms in plants. Emerg. Top. Life Sci. 2019, 3, 165–181. [Google Scholar]
- Webber, H.; Ewert, F.; Olesen, J.E.; Müller, C.; Fronzek, S.; Ruane, A.C.; Martre, P.; Ababaei, B.; Bindi, M. Diverging importance of drought stress for maize and winter wheat in Europe. Nat. Commun. 2018, 9, 4249. [Google Scholar] [CrossRef]
- Hammer, K. Agrarbiodiversität und pflanzengenetische ressourcen. In Herausforderung und Lösungsansatz. Schriften zu Genetischen Ressourcen; Informationszentrum für Genetische Ressourcen (IGR) u. Zentralstelle für Agrardokumentation und Information (ZADI): Bonn, Germany, 1998; Volume 10, ISSN 0948-8332. [Google Scholar]
- Hammer, K. Checklists and germplasm collecting. FAO/IBPGR. Plant Genet. Resour. Newsl 1991, 85, 15–17. [Google Scholar]
- Abdel-Ghani, A.H.; Neumann, K.; Wabila, C.; Sharma, R.; Dhanagond, S.; Owais, S.J.; Börner, A.; Graner, A.; Kilian, B. Diversity of germination and seedling traits in a spring barley (Hordeum vulgare L.) collection under drought simulated conditions. Genet. Resour. Crop. Evol. 2015, 62, 275–292. [Google Scholar] [CrossRef]
- Thabet, S.G.; Moursi, Y.S.; Karam, M.A.; Graner, A.; Alqudah, A.M. Genetic basis of drought tolerance during seed germination in barley. PLoS ONE 2018, 13, e0206682. [Google Scholar] [CrossRef]
- Tarawneh, R.A.; Szira, F.; Monostori, I.; Behrens, A.; Alqudah, A.M.; Thumm, S.; Lohwasser, U.; Röder, M.S.; Börner, A.; Nagel, M. Genetic analysis of drought response of wheat following either chemical desiccation or the use of a rain-out shelter. J. Appl. Genet. 2019, 60, 137–146. [Google Scholar] [CrossRef]
- Cooper, M.; Gho, C.; Leafgren, R.; Tang, T.; Messina, C. Breeding drought-tolerant maize hybrids for the US corn-belt: Discovery to product. J. Exp. Bot. 2014, 65, 6191–6204. [Google Scholar] [CrossRef]
- Bell, J. Corn Growth Stages and Development; Texas A&M AgriLife Extension and Research Agronomist, Amarillo: Lubbock, TX, USA, 2017. [Google Scholar]
- Edmeades, G.O. A feature in james, drought tolerance in maize: An emerging reality. In Global Status of Commercialized Biotech/GM Crops; Clive, J., Ed.; ISAAA: Ithaca, NY, USA, 2008. [Google Scholar]
- Adewale, S.A.; Akinwale, R.O.; Fakorede, M.A.B.; Badu-Apraku, B. Genetic analysis of drought-adaptive traits at seedling stage in early-maturing maize inbred lines and field performance under stress conditions. Euphytica 2018, 214, 1–18. [Google Scholar] [CrossRef]
- Edmeades, G.O. Progress in Achieving and Delivering Drought Tolerance in Maize an Update; ISAAA: Ithaca, NY, USA, 2013. [Google Scholar]
- Tesfaye, K.; Krusemanb, G.; Cairnsc, J.E.; Zaman-Allahc, M.; Wegarya, D.; Zaidid, P.H.; Bootee, K.J.; Rahutb, D.; Erensteinb, O. Potential benefits of drought and heat tolerance for adapting maize to climate change in tropical environments. Clim. Risk Manag. 2018, 19, 106–119. [Google Scholar] [CrossRef]
- Maiti, R.K.; Maiti, L.E.; Maiti, S.; Maiti, A.M.; Maiti, M.; Maiti, H. Genotypic variability in maize cultivars (Zea mays L.) for resistance to drought and salinity at the seedling stage. J. Plant Physiol. 1996, 148, 741–744. [Google Scholar] [CrossRef]
- Cao, L.Z.X.; Wj, B.X.P. Discuss on evaluating method to drought-resistance of maize in seedling stage. J. Maize Sci. 2004, 12, 73–75. [Google Scholar]
- Fakorede, M.A.B.; Opeke, B.O. Weather factors affecting the response of maize to planting dates in a tropical rainforest location. Exp. Agric. 2008, 21, 31–40. [Google Scholar] [CrossRef]
- Bashir, N.; Mahmood, S.; Zafar, Z.U.; Rasul, S.; Manzoor, H.; Athar, H.-U.-R. Is drought tolerance in maize (Zea mays L.) cultivars at the juvenile stage maintained at the reproductive stage? Pak. J. Bot. 2016, 48, 1385–1392. [Google Scholar]
- Meeks, M.; Murray, S.; Hague, S.; Hays, D. Measuring maize seedling drought response in search of tolerant germplasm. Agronomy 2013, 3, 135–147. [Google Scholar] [CrossRef]
- Liu, M.; Li, M.; Liu, K.; Sui, N. Effects of drought stress on seed germination and seedling growth of different maize varieties. J. Agric. Sci. 2013, 7, 2015. [Google Scholar] [CrossRef]
- Avramova, V.; Nagel, K.A.; Abd, E.H.; Bustos, D.; DuPlessis, M.; Fiorani, F.; Gerrit, T.S.; Beemster, A. Screening for drought tolerance of maize hybrids by multi-scale analysis of root and shoot traits at the seedling stage. J. Exp. Bot. 2016, 67, 2453–2466. [Google Scholar] [CrossRef]
- Akinwale, R.O.; Awosanmi, F.E.; Ogunniyi, O.O.; Fadoju, A.O. Determinants of drought tolerance at seedling stage in early and extra-early maize hybrids. Maydica 2017, 62, 9. [Google Scholar]
- Golbashy, M.; Ebrahimi, M.; Khorasani, S.K.; Choukan, R. Evaluation of drought tolerance of some corn (Zea mays L.) hybrids in Iran. Afr. J. Agric. Res. 2010, 5, 2714–2719. [Google Scholar]
- Grzesiak, M.T.; Marcińska, I.; Janowiak, F.; Rzepka, A.; Hura, T. The relationship between seedling growth and grain yield under drought conditions in maize and triticale genotypes. Acta Physiol. Plant. 2012, 34, 1757–1764. [Google Scholar] [CrossRef]
- Ahmed, H.G.M.; Sajjad, M.; Li, M.; Azmat, M.A.; Rizwan, M.; Maqsood, R.H.; Khan, S.H. Selection criteria for drought-tolerant bread wheat genotypes at seedling stage. Sustainability 2019, 11, 2584. [Google Scholar] [CrossRef]
- Grzesiak, S.; Hordyńska, N.; Szczyrek, P.; Grzesiak, M.T.; Noga, A.; Szechyńska-Hebda, M. Variation among wheat (Triticum easativum L.) genotypes in response to the drought stress: I-selection approaches. J. Plant Interact. 2019, 14, 30–44. [Google Scholar] [CrossRef]
- Arisandy, P.; Suwarno, W.B.; Azrai, M. Evaluation of drought tolerance in maize hybrids using stress tolerance indices. Int. J. Agron. Agric. Res. 2017, 4, 46–54. [Google Scholar]
- Hefny, M.; Ali, A.; Byoumi, T.; Al-Ashry, M.; Okasha, S. Classification of genetic diversity for drought tolerance in maize genotypes through principal component analysis. J. Agric. Sci. Belgrade 2017, 62, 213–227. [Google Scholar] [CrossRef]
- Badr, A.; Brüggmann, W. Comparative analysis of drought stress response of maize genotypes using chlorophyll fluorescence measurements and leaf relative water content. Photosynthetica 2020, 58, 638–645. [Google Scholar] [CrossRef]
- Abdel-Ghani, A.H.; Kumar, B.; Reyes-Matamoros, J.; Gonzalez-Portilla, P.J.; Jansen, C.; San Martin, J.P.; Lee, M.; Lübberstedt, T. Genotypic variation and relationships between seedling and adult plant traits in maize (Zea mays L.) inbred lines grown under contrasting nitrogen levels. Euphytica 2013, 189, 123–133. [Google Scholar] [CrossRef]
- ISTA. International Rules for Seed Testing; International Seed Testing Association: Bassersdorf, Switzerland, 2017. [Google Scholar]
- Hallauer, A.R.; Carena, M.J.; Miranda-Filho, J.B. Quantitative genetics in maize breeding. In Handbook of Plant Breeding; Prohens, J., Nuez, F., Carena, M.J., Eds.; Springer: Berlin/Heidelberg, Germany, 1991; Volume 6. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Kaydan, D.; Yagmur, M. Germination, seedling growth and relative water content of shoot in different seed sizes of triticale under osmotic stress of water and NaCl. Afr. J. Biotechnol. 2008, 16, 2862–2868. [Google Scholar]
- Aktar, S.; Hossain, N.; Azam, M.G.; Billah, M.; Biswas, P.L.; Latif, M.A.; Rohman, M.; Bagum, S.A.; Uddin, M.S. Phenotyping of hybrid maize at seedling stage under drought condition. Am. J. Plant Sci. 2018, 9, 2154–2169. [Google Scholar] [CrossRef][Green Version]
- Kramer, P.J. Cell water relations. In Water Relations of Plants; Kramer, P.J., Ed.; Academic Press: Cambridge, MA, USA, 1983; pp. 23–56. [Google Scholar]
- Shao, H.B.; Chu, L.Y.; Jaleel, C.A.; Zhao, C.X. Water-deficit stress-induced anatomical changes in higher plants. C. R. Biol. 2008, 331, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Anjum, S.A.; Ashraf, U.; Tanveer, M.; Khan, I.; Hussain, S.; Shahzad, B.; Zohaib, A.; Abbas, F.; Saleem, M.F.; Ali, I. Drought induced changes in growth, osmolyte accumulation and antioxidant metabolism of three maize hybrids. Front. Plant Sci. 2017, 8, 69. [Google Scholar] [CrossRef] [PubMed]
- Chloupek, O.; Dostál, V.; Středa, T.; Psota, V.; Dvořáčková, O. Drought tolerance of barley varieties in relation to their root system size. Plant Breed. 2010, 129, 630–636. [Google Scholar] [CrossRef]
- Sayed, M. QTL Analysis for Drought Tolerance Related to Root and Shoot Traits in Barley (Hordeum vulgare L.). Ph.D. Thesis, der Rheinischen Friedrich-Wilhelms-Universitaet, Bonn, Germany, 2011. [Google Scholar]
- Khan, N.H.; Ahsan, M.; Naveed, M.; Sadaqat, H.A.; Javed, I. Genetics of drought tolerance at seedling and maturity stages in Zea mays L. Span. J. Agric. Res. 2016, 14, 13. [Google Scholar] [CrossRef]
- Golabadi, M.; Arzani, A.; Maibody, M. Assessment of drought tolerance in segregating Populations in durum wheat. Afr. J. Agric. Res. 2007, 1, 162–171. [Google Scholar]
- Adee, E.; Roozeboom, K.; Balboa, G.R.; Schlegel, A.; Ciampitti, I.A. Drought-tolerant corn hybrids yield more in drought-stressed environments with no penalty in non-stressed environments. Front. Plant Sci. 2016, 7, 15–34. [Google Scholar] [CrossRef]
- Rachmilevitch, S.; Da Costa, M.; Huang, B. Physiological and biochemical indicators for stress tolerance. In Plant-Environment Interactions, 3rd ed.; Huang, B., Ed.; CRC Press: Boca Raton, FL, USA, 2006; pp. 321–356. [Google Scholar]
- Xu, J.; Yuan, Y.; Xu, Y.; Zhang, G.; Guo, X.; Wu, F.; Wang, Q.; Rong, T.; Pan, G.; Cao, M. Identification of candidate genes for drought tolerance by whole-genome resequencing in maize. BMC Plant. Biol. 2014, 14, 83. [Google Scholar] [CrossRef]
- Korte, A.; Farlow, A. The advantages and limitations of trait analysis with GWAS: A review. Plant Methods 2013, 9, 29. [Google Scholar] [CrossRef]

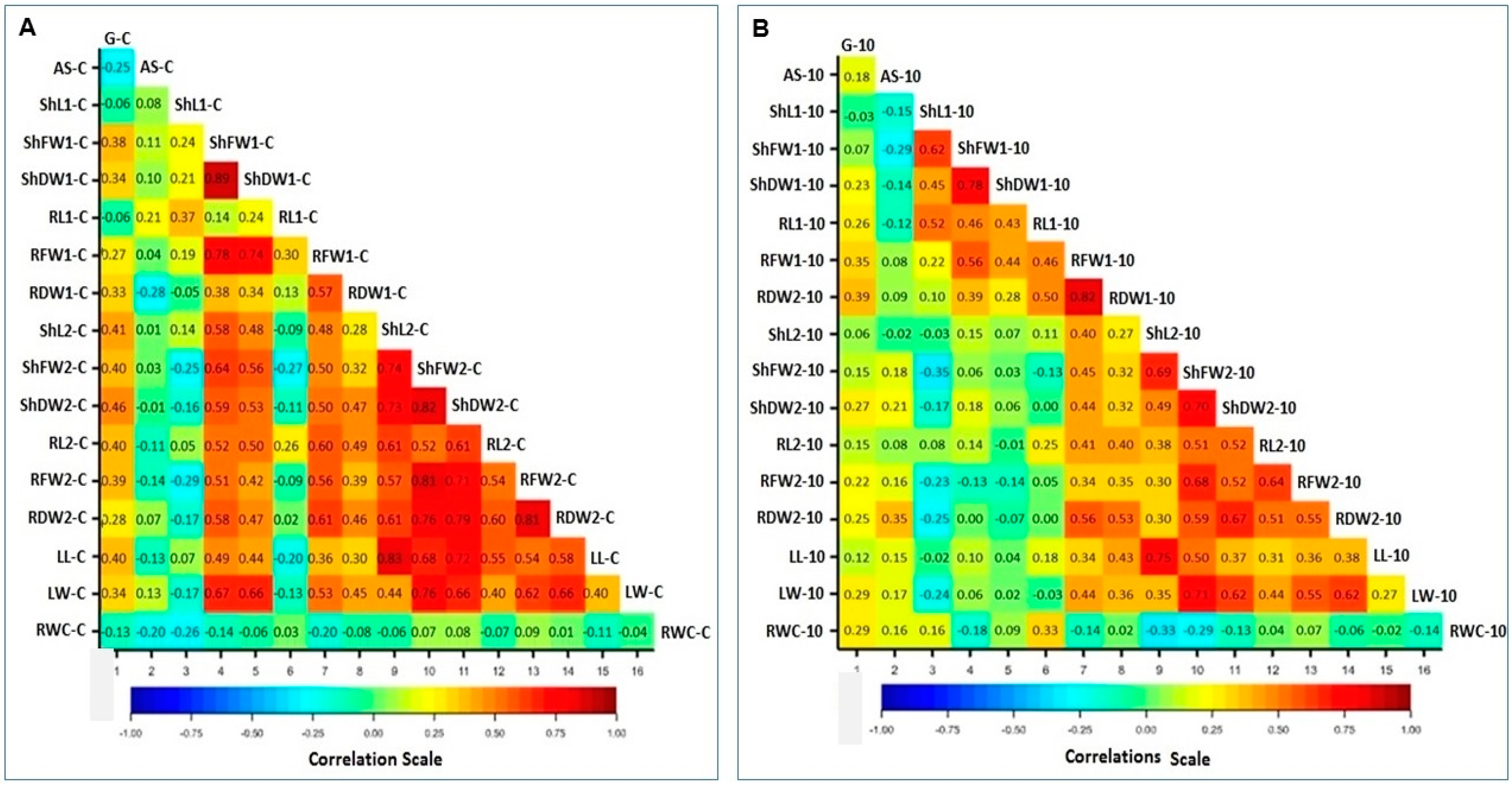
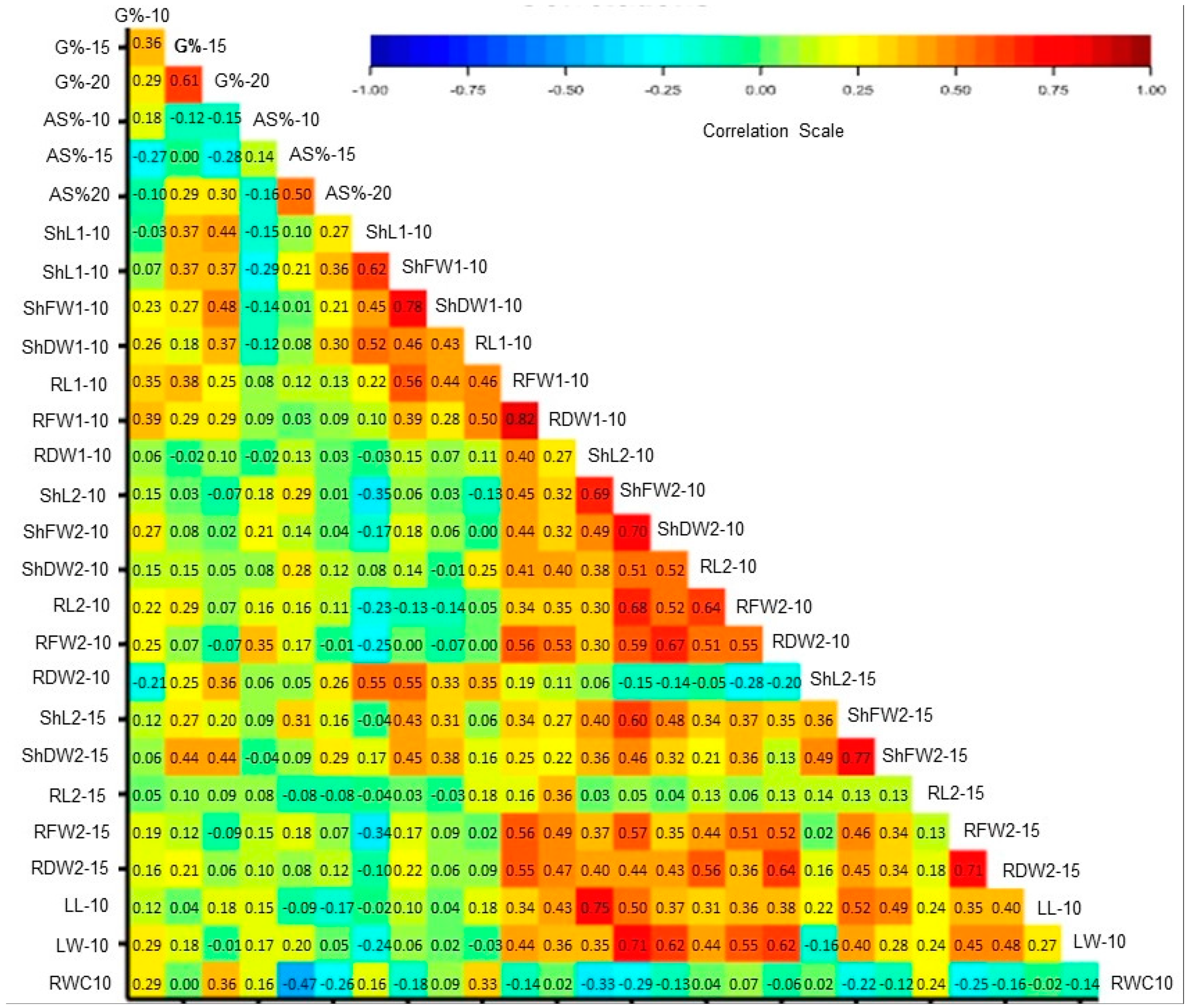
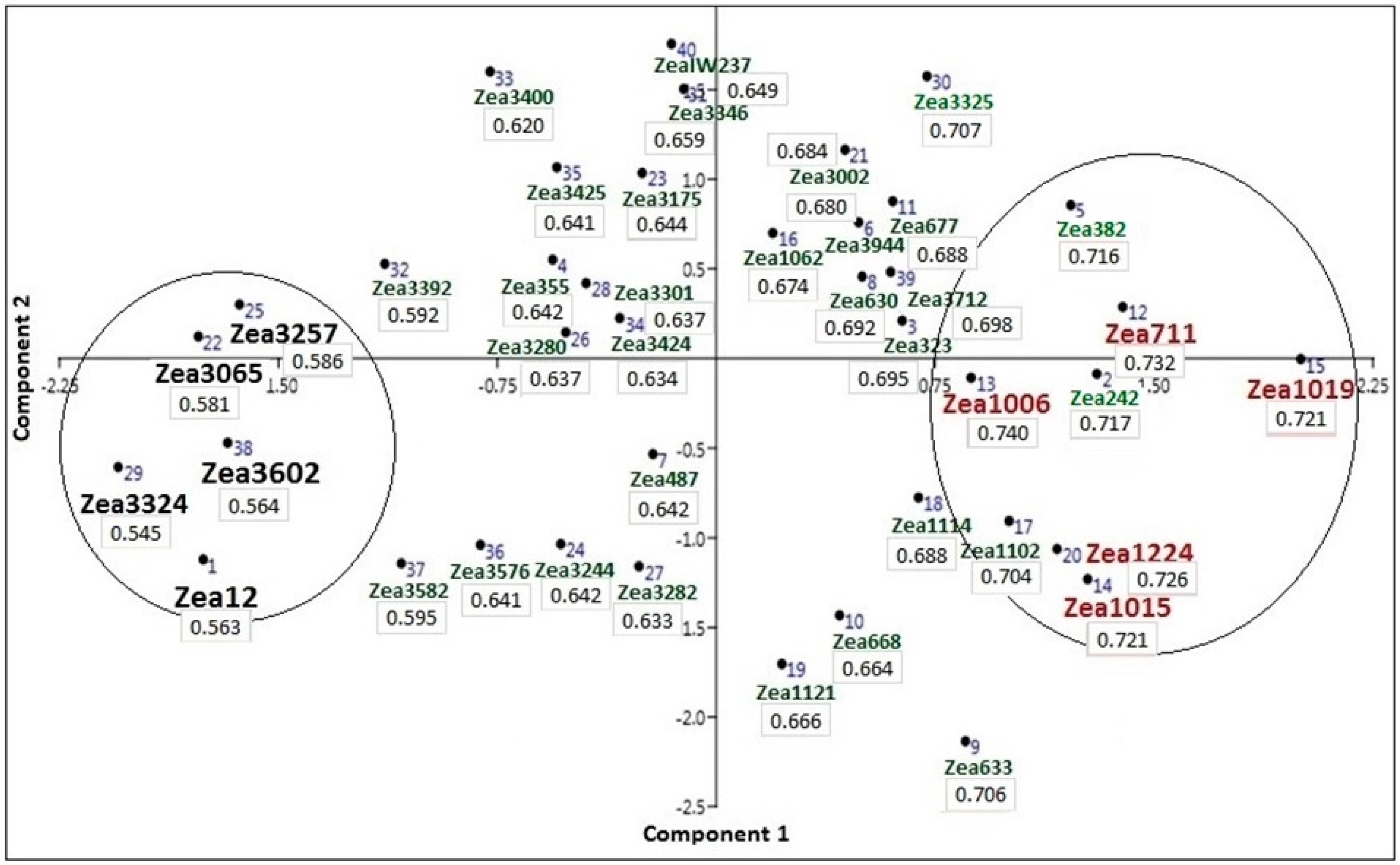
| Serial | Accession ID | Accession Information | Country of Origin |
|---|---|---|---|
| 01 | Zea 12 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Germany |
| 02 | Zea 242 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Germany |
| 03 | Zea 323 | subsp everata (Sturtev) Zhuk. var. oryzoides Körn. | Soviet Union |
| 04 | Zea 355 | subsp saccharata (Körn.) Zhuk var. flavodulcis Körn. | Soviet Union |
| 05 | Zea 382 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Romania |
| 06 | Zea 394 | subsp indentata (Sturtev) Zhuk.Ashoro Zairai. | Japan |
| 07 | Zea 487 | subsp indurata (Sturtev) Zhuk. var. rubropaleata Körn. | Greece |
| 08 | Zea 630 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | China |
| 09 | Zea 633 | subsp everata (Sturtev) Zhuk. var. oryzoides Körn. | China |
| 10 | Zea 668 | subsp indentata (Sturtev) Zhuk. var. xantodon Alef. | Macedonia |
| 11 | Zea 677 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Hungary |
| 12 | Zea 711 | subsp everata (Sturtev) Zhuk. var. oryzoides Körn. | Tschechnia |
| 13 | Zea 1006 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Libya |
| 14 | Zea 1015 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Libya |
| 15 | Zea 1019 | subsp everata (Sturtev) Zhuk. var. oryzoides Körn. | Italy |
| 16 | Zea 1062 | subsp indentata (Sturtev) Zhuk. var. flavorubra Körn. | Korea |
| 17 | Zea 1102 | subsp indurata (Sturtev) Zhuk. | Korea |
| 18 | Zea 1114 | subsp indurata (Sturtev) Zhuk. Var. aurantiaca. | Italy |
| 19 | Zea 1121 | subsp indurata (Sturtev) Zhuk. | Austria |
| 20 | Zea 1224 | subsp everata (Sturtev) Zhuk. var. gracillima Körn. | Rumania |
| 21 | Zea 3002 | subsp indurata (Sturtev) Zhuk. | Georgia |
| 22 | Zea 3065 | subsp indentata (Sturtev) Zhuk. var. leucodon Alef. | Georgia |
| 23 | Zea 3175 | subsp indurata (Sturtev) Zhuk. var. alba Alef. | Georgia |
| 24 | Zea 3244 | subsp everata (Sturtev) Zhuk. var. oxyornis Körn. | Germany |
| 25 | Zea 3257 | subsp indentata (Sturtev) Zhuk. var. xantodon Alef. | Albania |
| 26 | Zea 3280 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Rumania |
| 27 | Zea 3282 | subsp indurata (Sturtev) Zhuk. Oarzan. | Rumania |
| 28 | Zea 3301 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Portugal |
| 29 | Zea 3324 | subsp indentata (Sturtev) Zhuk. | Albania |
| 30 | Zea 3325 | subsp indentata (Sturtev) Zhuk. var. leucodon Alef. | Albania |
| 31 | Zea 3346 | subsp indurata (Sturtev) Zhuk. var. caesia Alef. | USA |
| 32 | Zea 3392 | subsp indurata (Sturtev) Zhuk. var. rubra Bonaf. | Bulgaria |
| 33 | Zea 3400 | subsp semidentata Kuleshov. | Georgia |
| 34 | Zea 3424 | subsp indurata (Sturtev) Zhuk. var. rubropalata Körn. | Italy |
| 35 | Zea 3425 | subsp indurata (Sturtev) Zhuk. var. vulgata Körn. | Germany |
| 36 | Zea 3576 | subsp indurata (Sturtev) Zhuk. var. alba Alef. | Italy |
| 37 | Zea 3582 | subsp semidentata Kuleshov. | Koroatia |
| 38 | Zea 3602 | subsp indentata (Sturtev) Zhuk. | Turkey |
| 39 | Zea 3712 | subsp everata (Sturtev) Zhuk. var. gracillima Körn. | Georgia |
| 40 | IW 237 | Cultivar. imported by the Agriculture Research Center | Egypt |
| Trait | Abbreviation | Description/Methodology |
|---|---|---|
| Germination % | G% | Calculated as G% = n ÷ N × 100, where n is the number of germinated seeds (radicle ≥3 mm) and N is the total number of sown seeds |
| Abnormal Seedling % | AS% | Seedlings that failed to develop into healthy seedlings after two weeks of sowing |
| Shoot length | ShL | Maximum length of shoot measured from the point of attachment to grain (cm) |
| Shoot Fresh weight | ShFW | Weight of fresh shoot detached from the grain and immediately weighed in grams. |
| Shoot Dry weight | ShDW | Weight of dried fresh shoot put in kraft bag and dried in oven at 70 ± 5 °C for 48 h in grams. |
| Root length | RL | Maximum length of fresh roots measured from the point of attachment to the grain (cm) |
| Root Fresh Weight | RFW | Fresh weight of roots of each plant separated from the seed and immediately weighed in grams. |
| Root Dry Weight | RDW | Weight of dried fresh roots put in kraft bag, dried in oven at 70 ± 5 °C for 48 h for complete drying in grams. |
| Leaf Length | LL | Fifth leaf length of 21 days old seedlings in cm |
| Leaf Width | LW | Fifth leaf maximum width of 21 days old seedling in cm |
| Relative Leaf Water Content | RWC | RWC% calculated as: [(FM − DM)/(TM − DM)] × 100, where, FM, TM and DM, are the fresh, turgid and dry masses respectively of leaf disc weighed using Sartorius Cubis MSU balance in grams. |
| Germination Drought Tolerance Index | G-DTI | G% under PEG drought/G% under control × 100 G-DTI10%, G-DTI15%, and G-DTI20% |
| Abnormal Seedlings Drought Tolerance Index | AS-DTI | AS% under PEG drought/AS% under control × 100 AS-DTI10%, AS-DTI15%, and AS-DTI20%, |
| Shoot Length Drought Tolerance Index | ShL-DTI | ShL under PEG drought/ShL under control × 100 ShL-DTI10% and ShL-DTI15% |
| Shoot Fresh Weight Tolerance Index | ShFW-DTI | ShFW under PEG drought/ShFW under control × 100 ShFW-DTI10% and ShFW-DTI15% |
| Shoot Dry Weight Drought Tolerance Index | ShDW-DTI | ShDW under PEG drought/ShDW under control × 100 ShDW-DTI10% and ShDW-DTI15% |
| Root Length Drought Tolerance Index | RL-DTI | RL under PEG drought/RL under control × 100 RL-DTI10% and RL- RL-DTI15% |
| Root Fresh Weight Drought Tolerance Index | RFW-DTI | RFW under PEG drought/RFW under control × 100 RFW-DTI10% and RFW-DTI15% |
| Root Dry Weight Drought Tolerance Index | RDW-DTI | RDW under PEG drought/RDW under control × 100 RDW-DTI10% and RDW-DTI15% |
| Leaf Length Drought Tolerance Index | LL-DTI10% | LL under PEG drought/LL under control × 100 |
| Leaf Width Drought Tolerance Index | LW-DTI10% | LW under drought/LW under control × 100 |
| Relative Water Content Drought Tolerance Index | RWC-DTI10% | RWC under PEG drought/RWC under control × 100 |
| Trait | Control | 10% PEG Treatment | 15% PEG Treatment | 20% PEG Treatment | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Acc. | Herit | Mean | Acc. | Herit | Mean | Acc. | Herit | Mean | Acc. | Herit | |
| G% | 94.28 | *** | 0.72 | 90.39 | *** | 0.71 | 83.86 | *** | 0.58 | 73.41 | *** | 0.58 |
| AS% | 7.41 | *** | 0.76 | 17.68 | *** | 0.73 | 32.44 | *** | 0.77 | 48.68 | *** | 0.84 |
| ShL1 | 13.09 | *** | 0.91 | 5.32 | *** | 0.97 | ||||||
| ShFW1 | 0.550 | *** | 0.86 | 0.190 | *** | 0.86 | ||||||
| ShDW1 | 0.056 | 0.02 | 0.28 | 0.189 | *** | 0.43 | ||||||
| RL1 | 16.34 | *** | 0.60 | 13.00 | *** | 0.85 | ||||||
| RFW1 | 0.382 | *** | 0.84 | 0.189 | *** | 0.89 | ||||||
| RDW1 | 0.056 | 0.51 | 0.37 | 0.070 | 0.44 | 0.46 | ||||||
| ShL2 | 28.68 | *** | 0.85 | 22.12 | *** | 0.81 | 13.86 | *** | 0.78 | |||
| ShFW2 | 0.925 | *** | 0.83 | 0.529 | *** | 0.82 | 0.305 | *** | 0.72 | |||
| ShDW2 | 0.084 | *** | 0.79 | 0.062 | *** | 0.80 | 0.045 | *** | 0.75 | |||
| RL2 | 27.20 | *** | 0.82 | 19.87 | *** | 0.78 | 17.68 | *** | 0.79 | |||
| RFW2 | 0.742 | *** | 0.89 | 0.485 | *** | 0.82 | 0.248 | *** | 0.76 | |||
| RDW2 | 0.063 | *** | 0.85 | 0.052 | *** | 0.82 | 0.038 | *** | 0.76 | |||
| LL | 20.63 | *** | 0.79 | 14.78 | *** | 0.64 | ||||||
| LW | 0.954 | *** | 0.85 | 0.883 | *** | 0.85 | ||||||
| RWC | 94.22 | *** | 0.85 | 86.56 | *** | 0.70 | ||||||
| Trait | Control vs. 10% PEG | Control vs. 15% PEG | Control vs. 20% PEG | |||
|---|---|---|---|---|---|---|
| Significance | LSD | Significance | LSD | Significance | LSD | |
| G% | *** | 7.350 | *** | 8.98 | *** | 10.08 |
| AS% | *** | 7.858 | *** | 9.22 | *** | 10.15 |
| ShL1 | *** | 0.226 | ||||
| ShFW1 | *** | 0.015 | ||||
| ShDW1 | *** | 0.008 | ||||
| RL1 | *** | 0.457 | ||||
| RFW1 | *** | 0.107 | ||||
| RDW1 | *** | 0.572 | ||||
| ShL2 | *** | 0.822 | *** | 0.771 | ||
| ShFW2 | *** | 0.043 | *** | 0.039 | ||
| ShDW2 | *** | 0.010 | *** | 0.009 | ||
| RL2 | *** | 0.835 | *** | 0.853 | ||
| RFW2 | *** | 0.045 | *** | 0.039 | ||
| RDW2 | *** | 0.003 | *** | 0.003 | ||
| LL | *** | 0.618 | ||||
| LW | *** | 0.026 | ||||
| RWC | *** | 0.562 | ||||
| Serial | Accession ID | Number of Best Traits | Total | Number of Least Traits | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | 10% | 15% | 20% | Top 5% | Control | 10% | 15% | 20% | Bottom 5% | ||
| 1 | Zea 12 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 |
| 2 | Zea 242 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 2 |
| 3 | Zea 323 | 1 | 1 | 1 | 1 | 4 | 7 | 4 | 2 | 0 | 13 |
| 4 | Zea 355 | 2 | 1 | 1 | 0 | 4 | 2 | 0 | 1 | 1 | 4 |
| 5 | Zea 382 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 2 |
| 6 | Zea 394 | 1 | 6 | 1 | 0 | 8 | 1 | 0 | 0 | 0 | 1 |
| 7 | Zea 487 | 3 | 0 | 0 | 0 | 3 | 1 | 0 | 1 | 0 | 2 |
| 8 | Zea 630 | 1 | 5 | 3 | 0 | 9 | 0 | 1 | 0 | 0 | 1 |
| 9 | Zea 633 | 0 | 0 | 0 | 1 | 1 | 11 | 9 | 4 | 0 | 24 |
| 10 | Zea 668 | 0 | 0 | 2 | 0 | 2 | 1 | 0 | 1 | 0 | 2 |
| 11 | Zea 677 | 0 | 0 | 1 | 0 | 1 | 9 | 6 | 1 | 1 | 17 |
| 12 | Zea 711 | 2 | 0 | 1 | 0 | 3 | 4 | 0 | 0 | 0 | 4 |
| 13 | Zea 1006 | 2 | 2 | 1 | 0 | 5 | 0 | 0 | 0 | 0 | 0 |
| 14 | Zea 1015 | 1 | 3 | 0 | 0 | 4 | 0 | 2 | 0 | 0 | 2 |
| 15 | Zea 1019 | 1 | 2 | 1 | 1 | 5 | 2 | 3 | 3 | 0 | 8 |
| 16 | Zea 1062 | 5 | 7 | 4 | 0 | 16 | 0 | 0 | 0 | 0 | 0 |
| 17 | Zea 1102 | 0 | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 2 |
| 18 | Zea 1114 | 3 | 3 | 1 | 0 | 7 | 0 | 0 | 0 | 0 | 0 |
| 19 | Zea 1121 | 3 | 5 | 0 | 1 | 9 | 0 | 1 | 1 | 0 | 2 |
| 20 | Zea 1224 | 1 | 2 | 2 | 1 | 6 | 2 | 1 | 0 | 1 | 4 |
| 21 | Zea 3002 | 0 | 0 | 0 | 1 | 1 | 0 | 2 | 0 | 0 | 2 |
| 22 | Zea 3065 | 3 | 1 | 1 | 0 | 5 | 2 | 2 | 0 | 0 | 4 |
| 23 | Zea 3175 | 1 | 0 | 0 | 0 | 1 | 1 | 2 | 1 | 0 | 4 |
| 24 | Zea 3244 | 0 | 0 | 0 | 1 | 1 | 9 | 8 | 5 | 1 | 23 |
| 25 | Zea 3257 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 3 |
| 26 | Zea 3280 | 1 | 2 | 2 | 0 | 5 | 0 | 1 | 0 | 0 | 1 |
| 27 | Zea 3282 | 0 | 0 | 2 | 0 | 2 | 0 | 0 | 3 | 0 | 3 |
| 28 | Zea 3301 | 8 | 2 | 2 | 0 | 12 | 7 | 2 | 1 | 0 | 10 |
| 29 | Zea 3324 | 2 | 0 | 0 | 0 | 2 | 0 | 2 | 0 | 0 | 2 |
| 30 | Zea 3325 | 3 | 3 | 2 | 1 | 9 | 1 | 0 | 0 | 0 | 1 |
| 31 | Zea 3346 | 0 | 0 | 0 | 0 | 0 | 4 | 5 | 1 | 0 | 10 |
| 32 | Zea 3392 | 0 | 1 | 0 | 0 | 1 | 0 | 4 | 0 | 0 | 4 |
| 33 | Zea 3400 | 3 | 6 | 0 | 0 | 9 | 0 | 1 | 0 | 0 | 1 |
| 34 | Zea 3424 | 2 | 5 | 2 | 0 | 9 | 0 | 0 | 1 | 0 | 1 |
| 35 | Zea 3425 | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 3 |
| 36 | Zea 3576 | 2 | 1 | 0 | 0 | 3 | 1 | 0 | 3 | 0 | 4 |
| 37 | Zea 3582 | 0 | 1 | 0 | 0 | 1 | 0 | 2 | 0 | 0 | 2 |
| 38 | Zea 3602 | 11 | 4 | 1 | 0 | 16 | 0 | 1 | 0 | 0 | 1 |
| 39 | Zea 3712 | 1 | 2 | 0 | 0 | 3 | 2 | 2 | 0 | 0 | 4 |
| 40 | Zea IW237 | 0 | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 1 |
| Total | 64 | 67 | 33 | 7 | 171 | 65 | 65 | 32 | 7 | 173 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badr, A.; El-Shazly, H.H.; Tarawneh, R.A.; Börner, A. Screening for Drought Tolerance in Maize (Zea mays L.) Germplasm Using Germination and Seedling Traits under Simulated Drought Conditions. Plants 2020, 9, 565. https://doi.org/10.3390/plants9050565
Badr A, El-Shazly HH, Tarawneh RA, Börner A. Screening for Drought Tolerance in Maize (Zea mays L.) Germplasm Using Germination and Seedling Traits under Simulated Drought Conditions. Plants. 2020; 9(5):565. https://doi.org/10.3390/plants9050565
Chicago/Turabian StyleBadr, Abdelfattah, Hanaa H. El-Shazly, Rasha A. Tarawneh, and Andreas Börner. 2020. "Screening for Drought Tolerance in Maize (Zea mays L.) Germplasm Using Germination and Seedling Traits under Simulated Drought Conditions" Plants 9, no. 5: 565. https://doi.org/10.3390/plants9050565
APA StyleBadr, A., El-Shazly, H. H., Tarawneh, R. A., & Börner, A. (2020). Screening for Drought Tolerance in Maize (Zea mays L.) Germplasm Using Germination and Seedling Traits under Simulated Drought Conditions. Plants, 9(5), 565. https://doi.org/10.3390/plants9050565




