Cytokinins Are Abundant and Widespread among Insect Species
Abstract
1. Introduction
2. Materials and Methods
2.1. Insect Species
2.2. Cytokinin Analysis by High Performance Liquid Chromatography-Electrospray Ionization Tandem Mass Spectrometry (HPLC-(ESI)-MS/MS)
3. Results and Discussion
3.1. Cytokinin Profiles and Levels in Insects
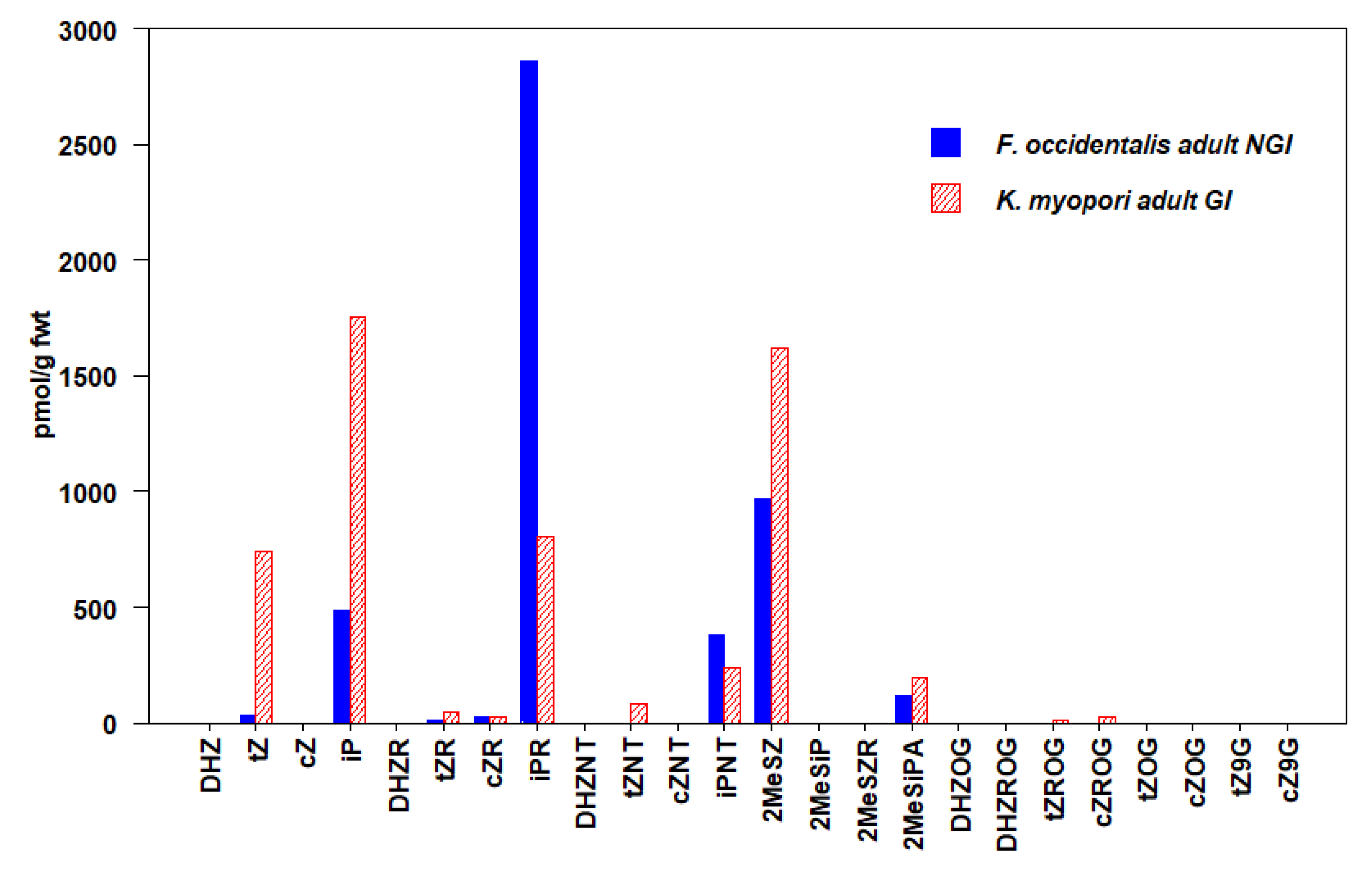
3.2. Thysanoptera
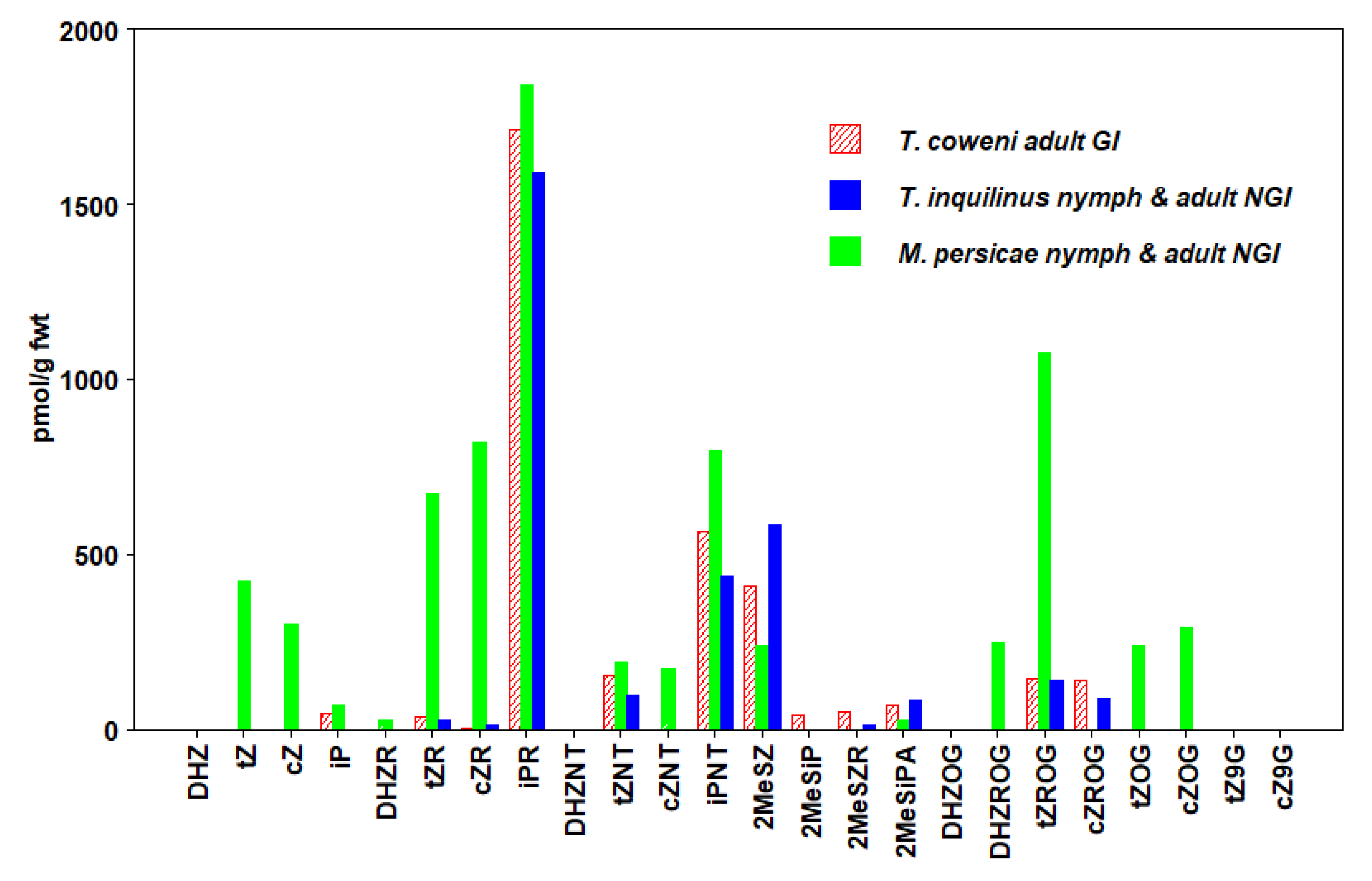
3.3. Hemiptera
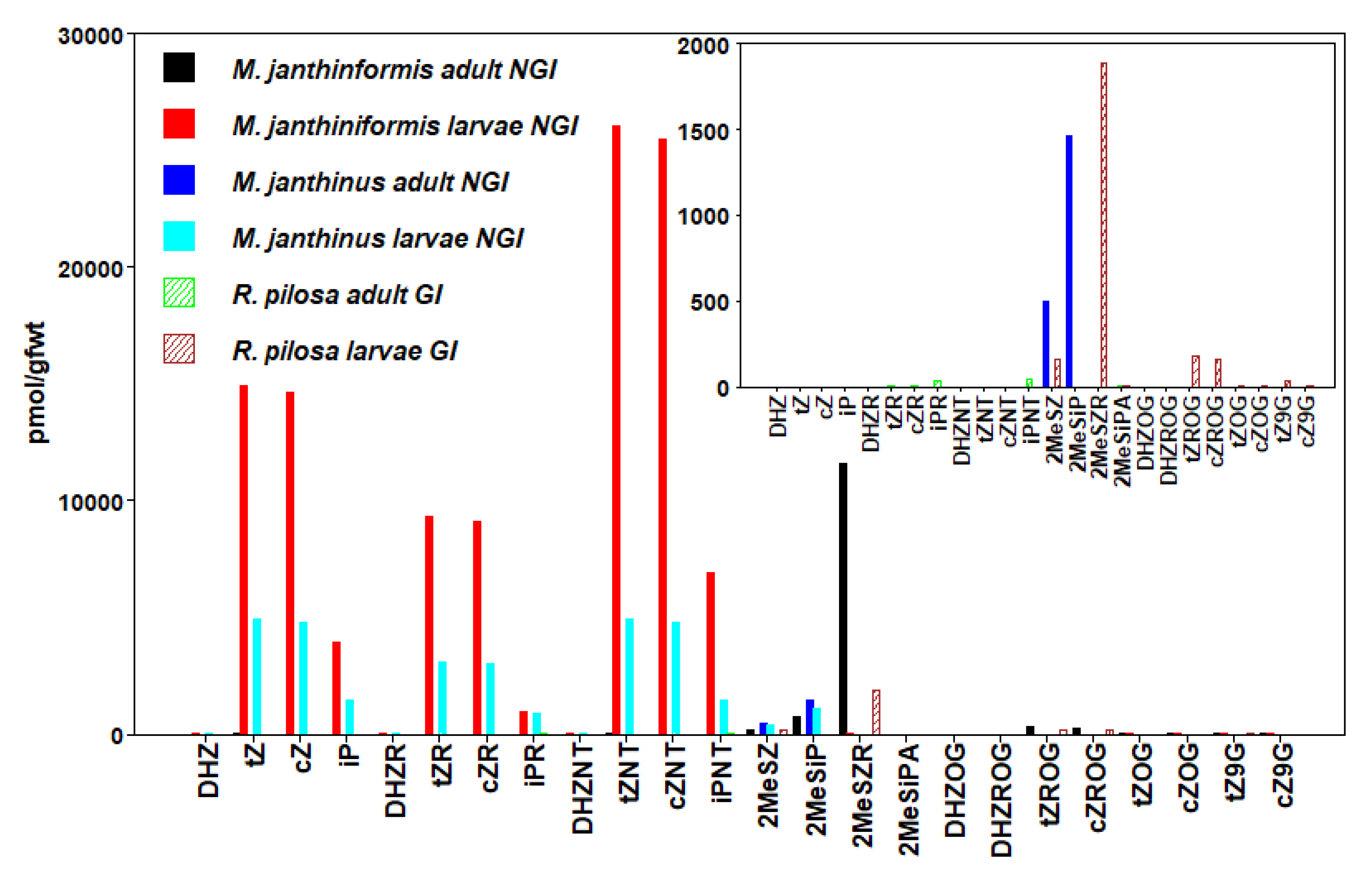
3.4. Coleoptera
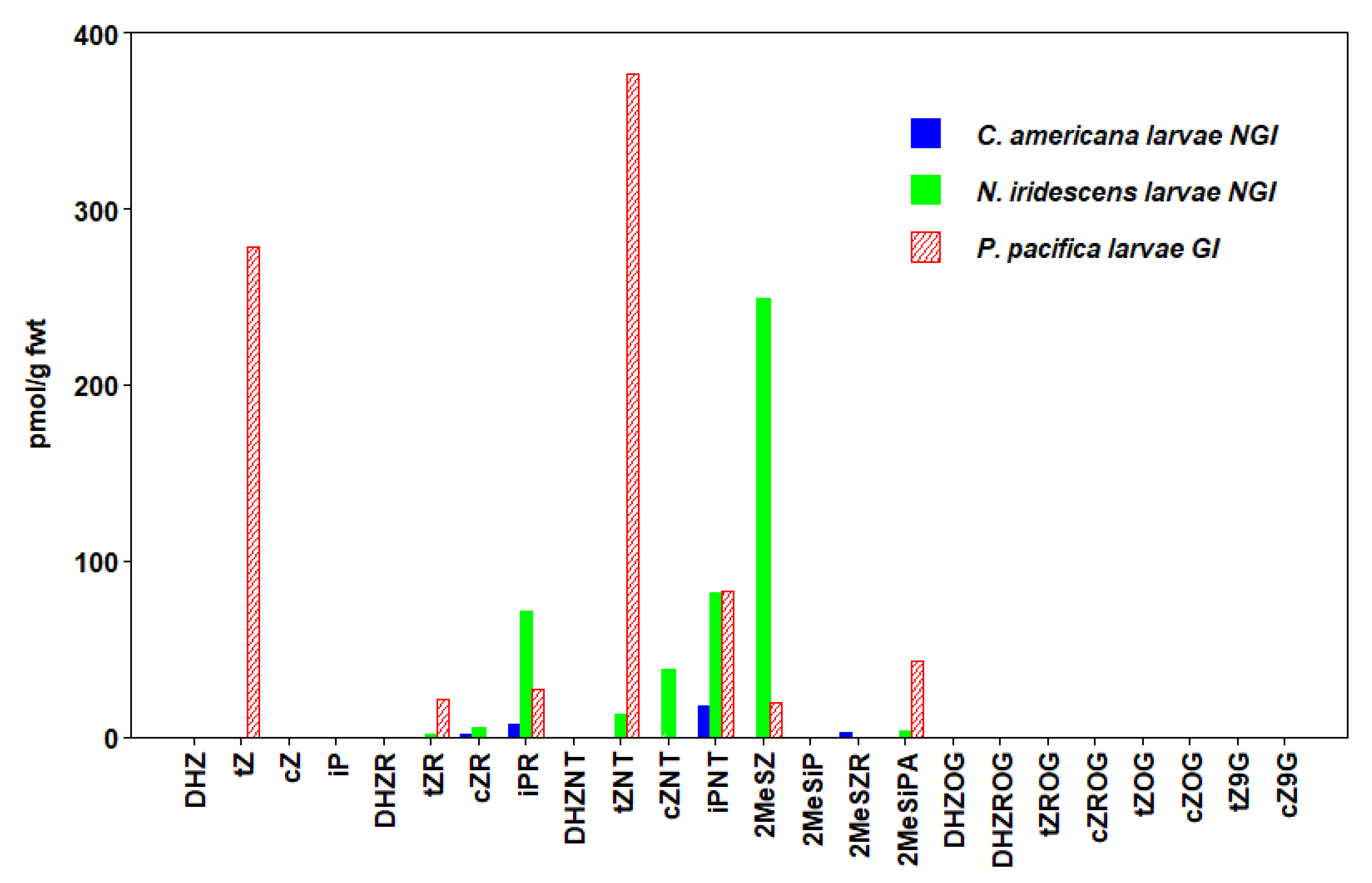
3.5. Hymenoptera
3.6. Lepidoptera
3.7. Diptera
3.8. Cytokinins in Insects
3.9. Evidence for Cytokinin Biosynthesis Pathway in Insects
3.10. Role of Cytokinins in Gall Induction
3.11. Role of Cytokinins in Manipulating Nutrient Allocation and Host-Plant Defenses
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mok, D.W.S.; Mok, M.C. Cytokinin metabolism and action. Annu. Rev. Plant Phys. 2001, 52, 89–118. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, H. Cytokinins: Activity, biosynthesis, and translocation. Annu. Rev. Plant Biol. 2006, 57, 431–449. [Google Scholar] [CrossRef] [PubMed]
- Kieber, J.J.; Schaller, G.E. Cytokinins. Arabidopsis Book 2014, 12, e0168. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Oger, P.M.; Schrammeijer, B.; Hooykaas, P.J.J.; Farrand, S.K.; Winans, S.C. The bases of crown gall tumorigenesis. J. Bacteriol. 2000, 182, 3885–3895. [Google Scholar] [CrossRef]
- Kisiala, A.; Laffont, C.; Emery, R.J.N.; Frugier, F. Bioactive cytokinins are selectively secreted by Sinorhizobium meliloti nodulating and nonnodulating strains. Mol. Plant Microbe Interact. 2013, 26, 1225–1231. [Google Scholar] [CrossRef]
- Samanovic, M.I.; Tu, S.; Novák, O.; Iyer, L.M.; McAllister, F.E.; Aravind, L.; Gygi, S.P.; Hubbard, S.R.; Strnad, M.; Darwin, K.H. Proteasomal control of cytokinin synthesis protects Mycobacterium tuberculosis against nitric oxide. Mol. Cell 2015, 57, 984–994. [Google Scholar] [CrossRef]
- Samanovic, M.I.; Hsu, H.; Jones, M.B.; Jones, V.; McNeil, M.R.; Becker, S.H.; Jordan, A.T.; Strnad, M.; Xu, C.; Jackson, M.; et al. Cytokinin signaling in Mycobacterium tuberculosis. mBio 2018, 9, e00989-18. [Google Scholar] [CrossRef]
- Seo, H.; Kim, S.; Sagong, H.Y.; Son, H.F.; Jin, K.S.; Kim, I.K.; Kim, K.J. Structural basis for cytokinin production by LOG from Corynebacterium glutamicum. Sci. Rep. 2016, 6, 31390. [Google Scholar] [CrossRef]
- Jorge, G.L.; Kisiala, A.; Morrison, E.; Aoki, M.; Nogueira, A.P.O.; Emery, R.J.N. Endosymbiotic Methylbacterium oryzae mitigates the impact of limited water availability in lentil (Lens culinaris Medik.) by increasing plant cytokinin levels. Environ. Exp. Bot. 2019, 162, 525–540. [Google Scholar] [CrossRef]
- Hinsch, J.; Vrabka, J.; Oeser, B.; Novak, O.; Galuszka, P.; Tudzynski, P. De novo biosynthesis of cytokinins in the biotrophic fungus Claviceps purpurea. Environ. Microbiol. 2015, 17, 2935–2951. [Google Scholar] [CrossRef]
- Morrison, E.N.; Knowles, S.; Thorn, R.G.; Saville, B.J.; Emery, R.J.N. Detection of phytohormones in temperate forest fungi predicts consistent abscisic acid production and a common pathway for cytokinin biosynthesis. Mycologia 2015, 107, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Morrison, E.N.; Emery, R.J.N.; Saville, B.J. Phytohormone involvement in the Ustilago maydis-Zea mays pathosystem: Relationships between abscisic acid and cytokinin levels and strain virulence in infected cob tissue. PLoS ONE 2015, 10, e0130945. [Google Scholar] [CrossRef] [PubMed]
- Malinowski, R.; Novák, O.; Borhan, M.H.; Spíchal, L.; Strnad, M.; Rolfe, S.A. The role of cytokinins in clubroot disease. Eur. J. Plant. Pathol. 2016, 145, 543–557. [Google Scholar] [CrossRef]
- Siddique, S.; Radakovic, Z.S.; De La Torre, C.M.; Chronis, D.; Novák, O.; Ramireddy, E.; Holbein, J.; Matera, C.; Hütten, M.; Gutbrod, P.; et al. A parasitic nematode releases cytokinin that controls cell division and orchestrates feeding site formation in host plants. Proc. Natl. Acad. Sci. USA 2015, 112, 12669–12674. [Google Scholar] [CrossRef] [PubMed]
- Mapes, C.C.; Davies, P.J. Cytokinins in the ball gall of Solidago altissima and the gall forming larvae of Eurosta solidaginis. New Phytol. 2001, 151, 203–212. [Google Scholar] [CrossRef]
- Straka, J.R.; Hayward, A.R.; Emery, R.J.N. Gall-inducing Pachypsylla celtidis (Psyllidae) infiltrate hackberry trees with high concentrations of phytohormones. J. Plant Interact. 2010, 5, 197–203. [Google Scholar] [CrossRef]
- Zhang, H.; Guiguet, A.; Dubreuil, G.; Kisiala, A.; Andreas, P.; Emery, R.J.N.; Huguet, E.; Body, M.; Giron, D. Dynamics and origin of cytokinins involved in plant manipulation by a leaf-mining insect. Insect Sci. 2017, 24, 1065–1078. [Google Scholar] [CrossRef]
- Brütting, C.; Crava, C.M.; Schäfer, M.; Schuman, M.C.; Meldau, S.; Adam, N.; Baldwin, I.T. Cytokinin transfer by a free-living mirid to Nicotiana attenuata recapitulates a strategy of endophytic insects. eLife 2018, 7, e36268. [Google Scholar] [CrossRef]
- Body, M.; Heidi, M.; Edger, P.; Schultz, J. A gall-forming insect manipulates hostplant phytohormone synthesis, concentrations, and signaling. bioRxiv 2019, 658823. [Google Scholar] [CrossRef]
- Seegobin, M.; Kisiala, A.; Noble, A.; Kaplan, D.; Brunetti, C.; Emery, R.J.N. Canis familiaris tissues are characterized by different profiles of cytokinins typical of the tRNA degradation pathway. FASEB J. 2018, 32, 6575–6581. [Google Scholar] [CrossRef]
- Aoki, M.; Seegobin, M.; Kisiala, A.; Noble, A.; Brunetti, C.; Emery, R.J.N. Phytohormone metabolism in human cells: Cytokinins are taken up and interconverted in HeLa cell culture. FASEB BioAdv. 2019, 1, 320–331. [Google Scholar] [CrossRef]
- Akiyoshi, D.E.; Klee, H.; Amasino, R.M.; Nester, E.W.; Gordon, M.P. T-DNA of Agrobacterium tumefaciens encodes an enzyme of cytokinin biosynthesis. Proc. Natl. Acad. Sci. USA 1984, 81, 5994–5998. [Google Scholar] [CrossRef] [PubMed]
- Powell, G.K.; Morris, R.O. Nucleotide sequence and expression of a Pseudomonas savastanoi cytokinin biosynthetic gene: Homology with Agrobacterium tumefaciens tmr and tzs loci. Nucleic Acids Res. 1986, 14, 2555–2565. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, H.; Kasahara, H.; Ueda, N.; Kojima, M.; Takei, K.; Hishiyama, S.; Asami, T.; Okada, K.; Kamiya, Y.; Yamaya, T.; et al. Agrobacterium tumefaciens increases cytokinin production in plastids by modifying the biosynthetic pathway in the host plant. Proc. Natl. Acad. Sci. USA 2005, 102, 9972–9977. [Google Scholar] [CrossRef] [PubMed]
- Barash, I.; Manulis-Sasson, S. Virulence mechanisms and host-specificity of gall forming Pantoea agglomerans. Trends Microbiol. 2007, 15, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Pertry, I.; Václavíková, K.; Gemrotová, M.; Spíchal, L.; Galuszka, P.; Depuydt, S.; Temmerman, W.; Stes, E.; De Keyser, A.; Riefler, M.; et al. Rhodococcus fascians impacts plant development through the dynamic Fas-mediated production of a cytokinin mix. Mol. Plant Microbe Interact. 2010, 23, 1164–1174. [Google Scholar] [CrossRef] [PubMed]
- Morrison, E.N.; Emery, R.J.N.; Saville, B.J. Fungal derived cytokinins are necessary for normal Ustilago maydis infection of corn. Plant Pathol. 2016, 66, 726–742. [Google Scholar] [CrossRef]
- Chanclud, E.; Kisiala, A.; Emery, R.J.N.; Chalvon, V.; Ducasse, A.; Romiti-Michel, C.; Gravot, A.; Kroj, T.; Morel, J. Cytokinin production by the rice blast fungus is a pivotal requirement for full virulence. PLoS Pathog. 2016, 12, e1005457. [Google Scholar] [CrossRef]
- Frébort, I.; Kowalska, M.; Hluska, T.; Frébortová, J.; Galuszka, T. Evolution of cytokinin biosynthesis and degradation. J. Exp. Bot. 2011, 62, 2431–2452. [Google Scholar] [CrossRef]
- Spíchal, L. Cytokinins—Recent news and views of evolutionally old molecules. Funct. Plant Biol. 2012, 39, 267–284. [Google Scholar] [CrossRef]
- Nishii, K.; Wright, F.; Chen, Y.Y.; Möller, M. Tangled history of a multigene family: The evolution of ISOPENTENYLTRANSFERASE genes. PLoS ONE 2018, 13, e0201198. [Google Scholar] [CrossRef]
- Konevega, A.L.; Soboleva, N.G.; Makhno, V.I.; Peshekhonov, A.V.; Katunin, V.I. The effect of modification of tRNA nucleotide-37 on the tRNA interaction with the P- and the A-site of the 70s ribosome Escherichia coli. Mol. Biol. 2006, 40, 669–683. [Google Scholar] [CrossRef]
- Chimnaronk, S.; Forouhar, F.; Sakai, J.; Yao, M.; Tron, C.M.; Atta, M.; Fontecave, M.; Hunt, J.F.; Tanaka, I. Snapshots of dynamics in synthesizing N6-isopentenyladenosine at the tRNA anticodon. Biochemistry 2009, 48, 5057–5065. [Google Scholar] [CrossRef]
- Bellés, X.; Martin, D.; Piulachs, M. The mevalonate pathway and the synthesis of juvenile hormone in insects. Annu. Rev. Entomol. 2005, 50, 181–199. [Google Scholar] [CrossRef]
- Tarkowská, D.; Strnad, M. Isoprenoid-derived plant signaling molecules: Biosynthesis and biological importance. Planta 2018, 247, 1051–1066. [Google Scholar] [CrossRef]
- Miyawaki, K.; Tarkowski, P.; Matsumoto-Kitano, M.; Kato, T.; Sato, S.; Tarkowski, D.; Tabala, S.; Sanberg, G.; Kakimoto, T. Roles of Arabidopsis ATP/ADP isopentenyltransferases and tRNA isopentenyltransferases in cytokinin biosynthesis. Proc. Natl. Acad. Sci. USA 2006, 103, 16598–16603. [Google Scholar] [CrossRef]
- Kamada-Nobusada, T.; Sakakibara, H. Molecular basis for cytokinin biosynthesis. Phytochemistry 2009, 70, 444–449. [Google Scholar] [CrossRef]
- Takei, K.; Yamaya, T.; Sakakibara, H. Arabidopsis CYP735A1 and CYP735A2 encode cytokinin hydrolases that catalyze biosynthesis of trans-zeatin. J. Biol. Chem. 2004, 279, 41866–41872. [Google Scholar] [CrossRef]
- MacDonald, E.M.S.; Powell, G.K.; Regier, D.A.; Glass, N.L.; Roberto, F.; Kosuge, T.; Morris, R.O. Secretion of zeatin, ribosylzeatin, and ribosyl-1”-methylzeatin by Pseudomonas savastanoi. Plant Physiol. 1986, 82, 742–747. [Google Scholar] [CrossRef]
- Lichter, A.; Barash, I.; Valinsky, L.; Manulis, S. The genes involved in cytokinin biosynthesis in Erwinia herbicola pv. gypsophilae: Characterization and role in gall formation. J. Bacteriol. 1995, 177, 4457–4465. [Google Scholar] [CrossRef]
- Jameson, P. Cytokinins and auxins in plant pathogen interactions—An overview. Plant Growth Regul. 2000, 32, 369–380. [Google Scholar] [CrossRef]
- Reineke, G.; Heinze, B.; Schiraski, J.; Buettner, H.; Kahmann, R.; Basse, C.W. Indole-3-acetic acid (IAA) biosynthesis in the smut fungus Ustilago maydis and its relevance for increased IAA levels in infected tissue and host tumor formation. Mol. Plant Pathol. 2008, 9, 339–355. [Google Scholar] [CrossRef]
- Bruce, S.A.; Saville, B.J.; Emery, R.J.N. Ustilago maydis produces cytokinins and abscisic acid for potential regulation of tumor formation in maize. J. Plant Growth Regul. 2011, 30, 51–63. [Google Scholar] [CrossRef]
- Dorchin, N.; Hoffman, J.; Stirk, W.; Novak, O.; Strnad, M.; van Staden, J. Sexually dimorphic gall structures correspond to differential phytohormone contents in male and female wasps. Physiol. Entomol. 2009, 34, 359–369. [Google Scholar] [CrossRef]
- Connor, E.F.; Bartlett, L.; O’Toole, S.; Byrd, S.; Biskar, K.; Orozco, J. The mechanism of gall induction makes galls red. Arthropod-Plant Interact. 2012, 6, 489–495. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Tanaka, H.; Hasegawa, M.; Tokuda, M.; Asami, T.; Suzuki, Y. Phytohormones and willow gall induction by a gall-inducing sawfly. New Phytol. 2012, 196, 586–595. [Google Scholar] [CrossRef]
- Tanaka, Y.; Okada, K.; Asami, T.; Suzuki, Y. Phytohormones in Japanese mugwort gall induction by a gall-inducing gall midge. Biosci. Biotech. Biochem. 2013, 9, 1942–1948. [Google Scholar] [CrossRef]
- Bartlett, L.; Connor, E.F. Exogenous phytohormones and the induction of plant galls by insects. Arthropod-Plant Interact. 2014, 8, 339–348. [Google Scholar] [CrossRef]
- Takei, M.; Yoshida, S.; Kawai, T.; Hasegawa, M.; Suzuki, Y. Adaptive significance of gall formation for a gall-inducing aphids on Japanese elm trees. J. Insect Physiol. 2015, 72, 43–51. [Google Scholar] [CrossRef]
- Kai, S.; Kumashiro, S.; Adachi, S.; Suzuki, Y.; Shiomi, Y.; Matsunaga, K.; Gyoutoku, N.; Asami, T.; Tokuda, M. Life history of Stenopsylla nigricornis (Hemiptera: Psylloidea: Triozidae) and phytohormones involved in gall induction. Arthropod-Plant Interact. 2017, 11, 99–108. [Google Scholar] [CrossRef]
- Angra-Sharma, R.; Sharma, D.K. Cytokinins in pathogenesis and disease resistance of Pyrenophora teres-barley and Dreschslera maydis-maize interactions during early stages of infection. Mycopathologia 1999, 148, 87–95. [Google Scholar] [CrossRef]
- Giron, D.; Kaiser, W.; Imbault, N.; Casas, J. Cytokinin-mediated leaf manipulation by a leafminer caterpillar. Biol. Lett. 2007, 3, 340–343. [Google Scholar] [CrossRef]
- Walters, D.R.; McRoberts, N.; Fitt, B.D.L. Are green islands red herrings? Significance of green islands in plant interactions with pathogens and pests. Biol. Rev. 2008, 83, 79–102. [Google Scholar] [CrossRef]
- Kaiser, W.; Huguet, E.; Casas, J.; Commin, C.; Giron, D. Plant green-island phenotype induced by leaf-miners is mediated by bacterial symbionts. Proc. R. Soc. B 2010, 277, 2311–2319. [Google Scholar] [CrossRef]
- Body, M.; Kaiser, W.; Dubreuil, G.; Casas, J.; Giron, D. Leaf-miners co-opt microorganisms to enhance their nutritional environment. J. Chem. Ecol. 2013, 39, 969–977. [Google Scholar] [CrossRef]
- Chanclud, E.; Morel, J.B. Plant hormones: A fungal point of view. Mol. Plant Pathol. 2016, 17, 1289–1297. [Google Scholar] [CrossRef]
- Zhang, H.; Dubreuil, G.; Faivre, N.; Dobrev, P.; Kaiser, W.; Huguet, E.; Vankova, R.; Giron, D. Modulation of plant cytokinin levels in the Wolbachia free leaf-mining species Phyllonorycter mespilella. Entomol. Exp. Appl. 2018, 166, 428–438. [Google Scholar] [CrossRef]
- Larson, K.C.; Whitham, T.G. Manipulation of food resources by a gall-forming aphid. The physiology of sink-source interactions. Oecologia 1991, 88, 15–21. [Google Scholar] [CrossRef]
- Inbar, M.; Eshel, A.; Wool, D. Interspecific competition among phloem-feeding insects mediation by induced host-plant sinks. Ecology 1995, 76, 1505–1515. [Google Scholar] [CrossRef]
- Roitsch, T.; Ehneß, R. Regulation of source/sink relations by cytokinins. Plant Growth Regul. 2000, 32, 359–367. [Google Scholar] [CrossRef]
- Giron, D.; Frago, E.; Glevarec, G.; Pieterse, C.M.; Dicke, M. Cytokinins as key regulators in plant-microbe-insect interactions: Connecting plant growth and defence. Funct. Ecol. 2013, 27, 599–609. [Google Scholar] [CrossRef]
- Schäfer, M.; Meza-Canales, I.D.; Navarro-Quezada, A.; Brütting, C.; Vanková, R.; Baldwin, I.T.; Meldau, S. Cytokinin levels and signaling respond to wounding and the perception of herbivore elicitors in Nicotiana attenuata. J. Integr. Plant Biol. 2015, 57, 198–212. [Google Scholar] [CrossRef] [PubMed]
- Diekman, J.; Hammer, P.E. Induction of anthocyanin accumulation by cytokinins in Arabidopsis thaliana. Plant Physiol. 1995, 108, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Hu, X.; Duan, R. Interactive effects of cytokinins, light, and sucrose on the phenotypes and the syntheses of anthocyanins and lignins in cytokinin overproducing transgenic Arabidopsis. J. Plant Growth Regul. 2005, 24, 93–101. [Google Scholar] [CrossRef]
- Allan, A.C.; Hellens, R.P.; Liang, W.A. MYB transcription factors that colour our fruit. Trends Plant Sci. 2008, 13, 99–102. [Google Scholar] [CrossRef]
- McCalla, D.R.; Genthe, M.K.; Hovanitz, W. The chemical nature of an insect gall-growth factor. Plant Physiol. 1962, 37, 98–103. [Google Scholar] [CrossRef]
- Engelbrecht, L. Cytokinin in den “grünen inseln” des herbstlaubes. Flora 1968, 159, 208–214. [Google Scholar] [CrossRef]
- Engelbrecht, L.; Orban, U.; Hesse, W. Leaf-miner caterpillars and cytokinins in the “green islands” of autumn leaves. Nature 1969, 223, 319–321. [Google Scholar] [CrossRef]
- Ohkawa, M. Isolation of zeatin from larvae of Drycocosmus kuriplilus Yasumatsu. Hortscience 1974, 9, 458–459. [Google Scholar]
- van Staden, J. Cytokinins from larvae in Erythrina latissima galls. Plant Sci. Lett. 1975, 5, 227–230. [Google Scholar] [CrossRef]
- Connor, E.F.; Taverner, M.P. The evolution and adaptive significance of the leaf-mining habit. Oikos 1997, 79, 6–25. [Google Scholar] [CrossRef]
- Quesnelle, P.; Emery, R.J.N. Cis-cytokinins that predominate in Pisum sativa during early embryogenesis will accelerate embryo growth in vitro. Can. J. Bot. 2007, 85, 91–103. [Google Scholar] [CrossRef]
- Farrow, S.C.; Emery, R.J.N. Concurrent profiling of indole-3-acetic acid, abscisic acid, and cytokinins and structurally related purines by high-performance-liquid-chromatography tandem electrospray mass spectrometry. Plant Methods 2012, 8, 42. [Google Scholar] [CrossRef] [PubMed]
- Mound, L.; Morris, D. A new thrips pest of Myoporum cultivars in California, in a new genus of leaf-galling Australian Phlaeothripidae (Thysanoptera). Zootaxa 2007, 1495, 35–45. [Google Scholar] [CrossRef]
- Reitz, S. Biology and ecology of the western flower thrips (Thysanoptera: Thripidae): The making of a pest. Fla. Entomol. 2009, 92, 7–14. [Google Scholar] [CrossRef]
- Miller III, D.G. Life history, ecology and communal gall occupation in the manzanita leaf-gall aphid, Tamalia coweni (Cockerell) (Homoptera: Aphididae). J. Nat. Hist. 1998, 32, 351–366. [Google Scholar] [CrossRef]
- Miller III, D.G. The ecology of inquilinism in communally parasitic Tamalia aphids (Hemiptera: Aphididae). Ann. Entomol. Soc. Am. 2004, 97, 1233–1241. [Google Scholar] [CrossRef]
- Andres, M.R.; Connor, E.F. The community-wide and guild-specific effects of pubescence on the folivorous insects of manzanitas Arctostaphylos spp. Ecol. Entomol. 2003, 28, 383–396. [Google Scholar] [CrossRef]
- Miller III, D.G.; Crespi, B. The evolution of inquilinism, host-plant use and mitochondrial substitution rates in Tamalia gall aphids. J. Evolut. Biol. 2003, 16, 731–743. [Google Scholar] [CrossRef]
- Miller III, D.G.; Lawson, S.P.; Rinker, D.C.; Estby, H.; Abbot, P. The origin and genetic differentiation of the socially parasitic aphid Tamalia inquilinus. Mol. Ecol. 2015, 24, 5751–5766. [Google Scholar] [CrossRef]
- Caldara, R.; Sassi, D.; Toševski, I. Phylogeny of the weevil genus Rhinusa Stephens based on adult morphological characters and host plant information (Coleoptera: Curculionidae). Zootaxa 2010, 2627, 39–56. [Google Scholar] [CrossRef]
- Toševski, I.; Caldara, R.; Jović, J.; Hernández-Vera, G.; Baviera, C.; Gassmann, A.; Emerson, B.C. Morphological, molecular and biological evidence reveal two cryptic species in Mecinus janthinus Germar (Coleoptera, Curculionidae), a successful biological control agent of Dalmatian toadflax, Linaria dalmatica (Lamiales, Plantaginaceae). Syst. Entomol. 2011, 36, 741–753. [Google Scholar] [CrossRef]
- Barnewall, E.C.; De Clerck-Floate, R.A. A preliminary histological investigation of gall induction in an unconventional galling system. Arthropod-Plant Interact. 2012, 6, 449–459. [Google Scholar] [CrossRef]
- Caltagirone, L.E. Notes on the biology, parasites, and inquilines of Pontania pacifica (Hymenoptera: Tenthredinidae), a leaf-gall incitant on Salix lasiolepis. Ann Entomol. Soc. Am. 1964, 57, 279–291. [Google Scholar] [CrossRef]
- Sage, R.D. Wet and dry-weight estimates of insects and spiders based on length. Am. Midl. Nat. 1982, 108, 407–411. [Google Scholar] [CrossRef]
- Bullock, J.A.; Smith, P.H. The relation between dry and fresh weight in some caterpillars. Entomol. Exp. Appl. 1971, 14, 125–131. [Google Scholar] [CrossRef]
- Harris, M.O.; Freeman, T.P.; Rohfritsch, O.; Anderson, K.G.; Payne, S.A.; Moore, J.A. Virulent Hessian fly (Diptera: Cecidomyiidae) larvae induce a nutritive tissue during compatible interactions with wheat. Ann. Entomol. Soc. Am. 2006, 99, 305–316. [Google Scholar] [CrossRef]
- Ehler, L.E. Ecology of Rhopalomyia californica Felt (Diptera: Cecidomyiidae) and its parasites in an urban environment. Hilgardia 1982, 50, 1–31. [Google Scholar] [CrossRef]
- Gajdošová, S.; Spíchal, L.; Kamínek, M.; Hoyerová, K.; Novák, O.; Dobrev, P.I.; Galuszka, P.; Klíma, P.; Gaudinová, A.; Žižková, E.; et al. Distribution, biological activities, metabolism, and conceivable function of cis-zeatin-type cytokinins in plants. J. Exp. Bot. 2011, 62, 2287–2840. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Kanehisa, M.; Sato, Y.; Furumichi, M.; Morishima, K.; Tanabe, M. New approach for understanding genome variations in KEGG. Nucleic Acids Res. 2019, 47, D590–D595. [Google Scholar] [CrossRef]
- Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 2019, 28, 1947–1951. [Google Scholar] [CrossRef]
- Gutzwiller, F.; Dedeine, F.; Kaiser, W.; Giron, D.; Lopez-Vaamonde, C. Correlation between the green-island phenotype and Wolbachia infections during the evolutionary diversification of Gracillariidae leaf-mining moths. Ecol. Evol. 2015, 5, 4049–4062. [Google Scholar] [CrossRef]
- Klámbt, D. The biogenesis of cytokinins in higher plants: Our present knowledge. In Physiology and Biochemistry of Cytokinins in Plants; Kamínek, M., Mok, D.W.S., Zažímalová, E., Eds.; SPB Academic Publishing: The Hague, The Netherlands, 1992; pp. 25–27. ISBN 978-9051030662. [Google Scholar]
- Persson, B.C.; Esberg, B.; Ólafsson, Ó.; Björk, G.R. Synthesis and function of isopentenyl adenosine derivatives in tRNA. Biochimie 1994, 76, 1152–1160. [Google Scholar] [CrossRef]
- Linder, A.C.; Lang, D.; Seifert, M.; Podlešáková, K.; Novák, O.; Strnad, M.; Reski, R.; von Schwartzenberg, K. Isopentenyltransferase-1 (IPT1) knockout in Physcomitrella together with phylogenetic analyses of IPTs provide insights into evolution of plant cytokinin biosynthesis. J. Exp. Bot. 2014, 65, 2533–2543. [Google Scholar] [CrossRef]
- Taller, B.J.; Murai, N.; Skoog, F. Endogenous cytokinins in the ribosomal RNA of higher plants. Plant Physiol. 1987, 83, 755–760. [Google Scholar] [CrossRef]
- Mapes, C.C.; Davies, P.J. Indole-3-acetic acid and ball gall development on Solidago altissima. New Phytol. 2001, 151, 195–202. [Google Scholar] [CrossRef]
- Sedlarević, A.; Morina, F.; Toševski, I.; Gašić, U.; Natić, M.; Jović, J.; Krstić, O.; Veljović-Jovanović, S. Comparative analysis of phenolic profiles of ovipositional fluid of Rhinusa pilosa (Mecinini, Curculionidae) and its host plant Linaria vulgaris (Plantaginaceae). Arthropod-Plant Interact. 2016, 10, 311–322. [Google Scholar] [CrossRef]
- Suzuki, H.; Yokokaura, J.; Ito, T.; Arai, R.; Yokoyama, C.; Toshima, H.; Nagat, S.; Asami, T.; Suzuki, Y. Biosynthetic pathway of the phytohormone auxin in insects and screening of its inhibitors. Insect Biochem. Mol. Biol. 2014, 53, 66–72. [Google Scholar] [CrossRef]
- Zhao, C.; Navarro Escalante, L.; Chen, H.; Benatti, T.R.; Qu, J.; Chellapilla, S.; Waterhouse, R.M.; Wheeler, D.; Andersson, M.N.; Bao, R.; et al. A massive expansion of effector genes underlies gall-formation in the wheat pest Mayetiola destructor. Curr. Biol. 2015, 25, 613–620. [Google Scholar] [CrossRef]
- Aggarwal, R.; Subramanyam, S.; Zhao, C.; Chen, M.S.; Harris, M.O.; Stuart, J.J. Avirulence effector discovery in a plant galling and plant parasitic arthropod, the Hessian fly (Mayetiola destructor). PLoS ONE 2014, 9, e100958. [Google Scholar] [CrossRef]
- Zhao, C.; Shukle, R.; Navarro-Escalante, L.; Chen, M.C.; Richards, S.; Stuart, J.J. Avirulence mapping in the Hessian fly (Mayetiola destructor) reveals a protein phosphatase 2C effector gene family. J. Insect Physiol. 2016, 84, 22–31. [Google Scholar] [CrossRef]
- Wang, Z.; Ge, J.Q.; Chen, H.; Cheng, X.; Yang, Y.; Li, J.; Whitworth, R.J.; Chen, M.S. An insect nucleoside diphosphate kinase (NDK) functions as an effector protein in wheat—Hessian fly interactions. Insect Biochem. Mol. Biol. 2018, 100, 30–38. [Google Scholar] [CrossRef]
- Zhao, C.; Rispe, C.; Nabity, P.D. Secretory RING finger proteins function as effectors in a grapevine galling insect. BMC Genom. 2019, 20, 923. [Google Scholar] [CrossRef]
- Weis, A.E.; Walton, R.; Crego, C.L. Reactive plant tissue sites and the population biology of gall makers. Annu. Rev. Entomol. 1988, 33, 467–486. [Google Scholar] [CrossRef]
- Abrahamson, W.G.; Weis, A.R. Evolutionary Ecology across Three Trophic Levels: Goldenrods, Gallmakers, and Natural Enemies; Princeton University Press: Princeton, NJ, USA, 1997; ISBN 9780691012087. [Google Scholar]
- Redfern, M. Plant Galls; Harper Collins: London, UK, 2011; ISBN 978-0002201438. [Google Scholar]
- Silvia, M.S.; Connor, E.F. Differences in meristems between monocots and dicots and susceptibility to attack by gall-inducing insects. Arthropod-Plant Interact. 2017, 11, 485–494. [Google Scholar] [CrossRef]
- Ikeuchi, M.; Sugimoto, K.; Iwase, A. Plant callus: Mechanisms of induction and repression. Plant Cell 2013, 25, 3159–3173. [Google Scholar] [CrossRef]
- Janocha, D.; Lohmann, J.U. From signals to stem cells and back again. Curr. Opin. Plant Biol. 2018, 45, 136–142. [Google Scholar] [CrossRef]
- Roitsch, T.; Lara, M.E.B.; Hoffman, M.; Proels, R.; Sinha, A.K. Extracellular invertase: Key metabolic enzyme and PR protein. J. Exp. Bot. 2003, 54, 513–524. [Google Scholar] [CrossRef]
- Werner, T.; Holst, K.; Pörs, Y.; Guivarc’h, A.; Mustroph, A.; Chriqui, D.; Schmülling, T. Cytokinin deficiency causes distinct changes of sink and source parameters in tobacco shoots and roots. J. Exp. Bot. 2008, 59, 2659–2672. [Google Scholar] [CrossRef]
- Siemens, J.; Keller, I.; Sarx, J.; Kunz, S.; Schuller, A.; Nagel, W.; Schmülling, T.; Parkiske, M.; Ludwig-Müller, J. Transcriptome analysis of Arabidopsis clubroots indicate a key role for cytokinins in disease development. Mol. Plant-Microbe Interact. 2006, 19, 480–494. [Google Scholar] [CrossRef]
- Kyndt, T.; Denil, S.; Haegeman, A.; Trooskens, G.; Bauters, L.; Van Criekinge, W.; De Meyer, T.; Gheysen, G. Transcriptional reprogramming by root knot and migratory nematode infection in rice. New Phytol. 2012, 196, 887–900. [Google Scholar] [CrossRef]
- Rawat, N.; Neerja, C.N.; Mair, S.; Bentur, J.S. Differential gene expression in gall midge susceptible rice genotypes revealed by suppressive subtraction hybridization (SSH) cDNA libraries and microarray analysis. Rice 2012, 5, 8. [Google Scholar] [CrossRef]
- Nabity, P.D.; Haus, M.J.; Berenbaum, M.R.; DeLucia, E.H. Leaf-galling phylloxera on grapes reprograms host metabolism and morphology. Proc. Natl. Acad. Sci. USA 2013, 110, 16663–16668. [Google Scholar] [CrossRef]
- Zhang, H.; de Bernonville, T.D.; Body, M.; Glevarec, G.; Reichelt, M.; Unsicker, S.; Bruneau, M.; Renou, J.; Huguet, E.; Dubreuil, G.; et al. Leaf-mining by Phyllonorycter blancardella reprograms the host-leaf transcriptome to modulate phytohormones associated with nutrient mobilization and plant defense. J. Insect Physiol. 2016, 84, 114–127. [Google Scholar] [CrossRef]
- Takeda, S.; Yoza, M.; Amano, T.; Ohshima, I.; Hirano, T.; Sata, M.H.; Sakamoto, T.; Kimura, S. Comparative transcriptome analysis of galls from four different host plants suggests the molecular mechanism of gall development. PLoS ONE 2019, 14, e0223686. [Google Scholar] [CrossRef]
- Eitle, M.W.; Greisser, M.; Vankova, R.; Dobrev, P.; Aberer, S. Grape phylloxera (D. vitifoliae) manipulates SA/JA concentrations and signaling pathways in root galls of Vitis spp. Plant Physiol. Biochem. 2019, 144, 85–91. [Google Scholar] [CrossRef]
- Hearn, J. Exploring Population History and Gall Induction in Cynipid Gall Wasps Using Genomics and Transcriptomics. Ph.D. Thesis, University of Edinburgh, Edinburgh, UK, 2013. [Google Scholar]
- Bailey, S.; Percy, D.M.; Hefer, C.A.; Cronk, Q.C.B. The transcriptional landscape of insect galls: Psyllid (Hemiptera) gall formation in Hawaiian Metrosideros polymorpha. BMC Genom. 2015, 16, 943. [Google Scholar] [CrossRef]
- Martinson, E.O.; Hackett, J.D.; Machado, C.A.; Arnold, A.E. Metatranscriptome analysis of fig flowers provides insights into potential mechanisms for mutualism stability and gall induction. PLoS ONE 2015, 10, e0130745. [Google Scholar] [CrossRef]
- Schultz, J.C.; Edger, P.P.; Body, M.J.A.; Appel, H.M. A galling insect activates plant reproductive programs during gall development. Sci. Rep. 2019, 9, 1833. [Google Scholar] [CrossRef]
- Shih, T.H.; Lin, S.H.; Huang, M.Y.; Sun, C.W.; Yang, C.M. Transcriptome profile of cup-shaped galls in Litsea acuminata leaves. PLoS ONE 2018, 13, e0205265. [Google Scholar] [CrossRef]
- Santner, A.; Calderon-Villalobos, L.I.A.; Estelle, M. Plant hormones are versatile chemical regulators of plant growth. Nat. Chem. Biol. 2009, 5, 301–307. [Google Scholar] [CrossRef]
- Tooker, J.F.; De Moraes, C.M. Feeding by a gall-inducing caterpillar species alters levels of indole-3-acetic and abscisic acid in Solidago altissima stems. Arthropod-Plant Interact. 2011, 5, 115–124. [Google Scholar] [CrossRef]
- Tooker, J.F.; De Moraes, C.M. Feeding by Hessian fly (Mayetiola destructor [Say]) larvae on wheat increases levels of fatty acids and indole-3-acetic acid but not hormones involved in plant-defense signaling. J. Plant Growth Regul. 2011, 30, 158–165. [Google Scholar] [CrossRef]


| Order (Family) | Gall Inducer | Non-Gall Inducer | Life Stage | Host Plant | Locality | Site-Collector |
|---|---|---|---|---|---|---|
| Thysanoptera | ||||||
| (Phlaeothripidae) | Klambothrips myopori | Adult | Myoporum laetum | Pacifica, CA | Field-EFC | |
| (Thripidae) | Frankliniella occidentalis | Adult | Rosa sp. | Half Moon Bay, CA | Greenhouse-EFC | |
| Hemiptera | ||||||
| (Aphididae) | Tamalia coweni | Adult | Arctostaphylos viscida | Chico, CA | Field-DGM & EFC | |
| Tamalia inquilinus | Adult + Nymph | A. viscida | Chico, CA | Field-EFC | ||
| Myzus persicae | Adult + Nymph | Salix lasiolepis | San Francisco, CA | Field-EFC | ||
| Coleoptera | ||||||
| (Curculionidae) | Rhinusa pilosa | Adult & Larvae | Linaria vulgaris | Lethbridge, CAN | Lab Colonies-RDF | |
| Mecinus janthinus | Adult & Larvae | L. vulgaris | Lethbridge, CAN | Lab & Field colonies-RDF | ||
| Mecinus janthiniformis | Adult & Larvae | Linaria dalmatica | Fort Macleod, CAN | Field-RDF | ||
| Hymenoptera | ||||||
| (Tenthredinoidea) | Pontania pacifica | Larvae | S. lasiolepis | Pacifica, CA | Field-EFC | |
| Cimbex americana | Larvae | S. lasiolepis | Pacifica, CA | Field-EFC | ||
| Nematus iridescens | Larvae | Populus angustifolia x P. fremontii | Flagstaff, AZ | Field-PWP | ||
| Lepidoptera | ||||||
| (Gelechiidae) | Gnorimoschema gallaesolidaginis | Larvae | Solidago altissima | University Park, PA | Field-JFT | |
| Dichomeris sp. | Larvae | S. altissima | University Park, PA | Field-JFT | ||
| Diptera | ||||||
| (Tephritidae) | Eurosta solidaginis | Adult & Larvae | S. altissima | Northfield, MN | Field-EFC | |
| Bactrocera oleae | Adult & Larvae | Olea europea | Santa Rosa, CA | Field-EFC | ||
| (Cecidomyiidae) | Mayetiola destructor | Larvae | Triticum aestivum | Manhattan, KS | Greenhouse-MSC | |
| Rhopalomyia californica | Larvae | Baccharis pilularis | San Francisco, CA | Field-EFC |
| Isoprenoid Cytokinins | Labeled CK Internal Standard | |
|---|---|---|
| Nucleotides (NTs) | ||
| 1. | Trans-zeatin riboside-5′-monophosphate (tZNT) | 2H5[9RMP]Z |
| 2. | Cis-zeatin riboside-5′-monophosphate (cZNT) | |
| 3. | Dihydrozeatin riboside -5′-monophosphate (DHZNT) | 2H3[9RMP]DHZ |
| 4. | Isopentyladenosine-5′monophosphate (iPNT) | 2H6[9RMP]iP |
| Ribosides (RBs) | ||
| 5. | Trans-zeatin riboside (tZR) | 2H5[9R]Z |
| 6. | Cis-zeatin riboside (cZR) | |
| 7. | Dihydrozeatin riboside (DHZR) | 2H3[9R]DHZ |
| 8. | Isopentyladenosine (iPR) | 2H6[9R]iP |
| Free bases (FBs) | ||
| 9. | Trans-zeatin (tZ) | 2H3DHZ |
| 10. | Cis-zeatin (cZ) | |
| 11. | Dihydrozeatin (DHZ) | |
| 12. | Isopentyladenine (iP) | 2H6iP |
| Glucosides (GLUCs) | ||
| 13. | Trans-zeatin-O-glucoside (tZOG) | 2H5ZOG |
| 14. | Cis-zeatin-O-glucoside (cZOG) | |
| 15. | Dihydrozeatin-O-glucoside (DHZOG) | 2H7DHZOG |
| 16. | Trans-zeatin-O-glucoside riboside (tZROG) | 2H5ZROG |
| 17. | Cis-zeatin-O-glucoside riboside cZROG | |
| 18. | Dihydrozeatin-O-glucoside riboside (DHZROG) | 2H7DHZROG |
| 19. | Trans-zeatin-9-glucoside (tZ9G) | 2H5Z9G |
| 20. | Cis-zeatin-9-glucoside (cZ9G) | |
| 21. | Dihydrozeatin-9-glucoside (DHZ9G) | 2H3DHZ9G |
| 22. | Isopentenyladenine-7-glucoside (iP7G) | 2H5iP7G |
| 23. | Isopentenyladenine-9-glucoside (iP9G) | |
| Methylthiols (METs) | ||
| 24. | 2-Methylthio-trans-zeatin (2MeSZ) | 2H5MeSZ |
| 25. | 2-Methylthio-trans-zeatin riboside (2MeSZR) | 2H5MeSZR |
| 26. | 2-Methylthio-isopentyladenine (2MeSiP) | 2H6MeSiP |
| 27. | 2-Methylthio-isopentyladenosine (2MeSiPA) | 2H6MeSiPR |
| Aromatic Cytokinins | ||
| 28. | Benzylaminopurine (BA) | 2H7BA |
| 29. | Benzylaminopurine riboside (BAR) | 2H7BAR |
| Insect Order & Species | tZ +tZR + tZNT Mean ± SE | cZ + cZR + cZNT Mean ± SE | iP + iPR + iPNT Mean ± SE | Total CKs (all forms) Mean ± SE |
|---|---|---|---|---|
| Thysanoptera | ||||
| F. occidentalis (NGI) Adult | 43 ± 10 | 23 ± 7 | 3730 ± 281 | 4876 ± 376 |
| K.myopori (GI) Adult | 869 ± 377 | 28 ± 6 | 2799 ± 265 | 5548 ± 945 |
| Hemiptera | ||||
| T. coweni (GI) Adult | 191 ± 68 | 4 ± 3 | 2323 ± 196 | 3369 ± 220 |
| T. inquilinus (NGI) Adult + Nymph | 120 ± 35 | 11 ± 4 | 2030 ± 96 | 3068 ± 169 |
| M. persicae (NGI) Adult + Nymph | 1290 ± 64 | 1295 ± 33 | 2705 ± 109 | 7436 ± 177 |
| Coleoptera | ||||
| R. pilosa (GI) Larvae | 2439 ± 1172 | |||
| M. janthinus (NGI) Larvae | 12,910 ± 1584 | 12,550 ± 2464 | 3865 ± 3283 | 30,928 ± 499 |
| M. janthiniformis (NGI) Larvae | 50,287 ± 10,134 | 49,268 ± 9383 | 11,765 ± 4023 | 111,630 ± 21,383 |
| R. pilosa (GI) Adult | 2 ± 0.7 | 2 ± 0.7 | 76 ± 8 | 83 ± 8 |
| M. janthinus (NGI) Adult | 1961 ± 352 | |||
| M. janthiniformis (NGI) Adult | 16 ± 16 | 6 ± 6 | 13,184 ± 6687 | |
| Hymenoptera | ||||
| P. pacifica (GI) Larvae | 676 ± 314 | 109 ± 31 | 848 ± 330 | |
| C. americana (NGI) Larvae | 2 ± 0.4 | 25 ± 6 | 29 ± 6 | |
| N. iridescens (NGI) Larvae | 14 ± 2 | 44 ± 2 | 154 ± 35 | 463 ± 74 |
| Lepidoptera | ||||
| G. gallaesolidaginis (GI) Larvae | 96 ± 53 | 104 ± 67 | 252 ± 78 | 526 ± 167 |
| Dichomeris sp. (NGI) Larvae | 2 ± 1.5 | 54 ± 17 | 849 ± 130 | 1424 ± 302 |
| Diptera | ||||
| E. solidaginis (GI) Larvae | 154,251 ± 40,883 | 54,646 ± 4191 | 318,405 ± 47,781 | |
| B. oleae (NGI) Larvae | 71 ± 11 | 138 ± 11 | ||
| E. solidaginis (GI) Adult | 12 ± 1 | 695 ± 174 | 769 ± 191 | |
| B. oleae (NGI) Adult | 475 ± 354 | 989 ± 473 | ||
| M. destructor (GI) Larvae D3 | 181 ± 28 | 754 ± 57 | 151 ± 20 | 2600 ± 72 |
| R. californica (GI) Larvae | 486 ± 163 | 631 ± 252 | 1118 ± 401 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andreas, P.; Kisiala, A.; Emery, R.J.N.; De Clerck-Floate, R.; Tooker, J.F.; Price, P.W.; Miller III, D.G.; Chen, M.-S.; Connor, E.F. Cytokinins Are Abundant and Widespread among Insect Species. Plants 2020, 9, 208. https://doi.org/10.3390/plants9020208
Andreas P, Kisiala A, Emery RJN, De Clerck-Floate R, Tooker JF, Price PW, Miller III DG, Chen M-S, Connor EF. Cytokinins Are Abundant and Widespread among Insect Species. Plants. 2020; 9(2):208. https://doi.org/10.3390/plants9020208
Chicago/Turabian StyleAndreas, Peter, Anna Kisiala, R. J. Neil Emery, Rosemarie De Clerck-Floate, John F. Tooker, Peter W. Price, Donald G. Miller III, Ming-Shun Chen, and Edward F. Connor. 2020. "Cytokinins Are Abundant and Widespread among Insect Species" Plants 9, no. 2: 208. https://doi.org/10.3390/plants9020208
APA StyleAndreas, P., Kisiala, A., Emery, R. J. N., De Clerck-Floate, R., Tooker, J. F., Price, P. W., Miller III, D. G., Chen, M.-S., & Connor, E. F. (2020). Cytokinins Are Abundant and Widespread among Insect Species. Plants, 9(2), 208. https://doi.org/10.3390/plants9020208








