Canavanine Increases the Content of Phenolic Compounds in Tomato (Solanum lycopersicum L.) Roots
Abstract
1. Introduction
2. Results
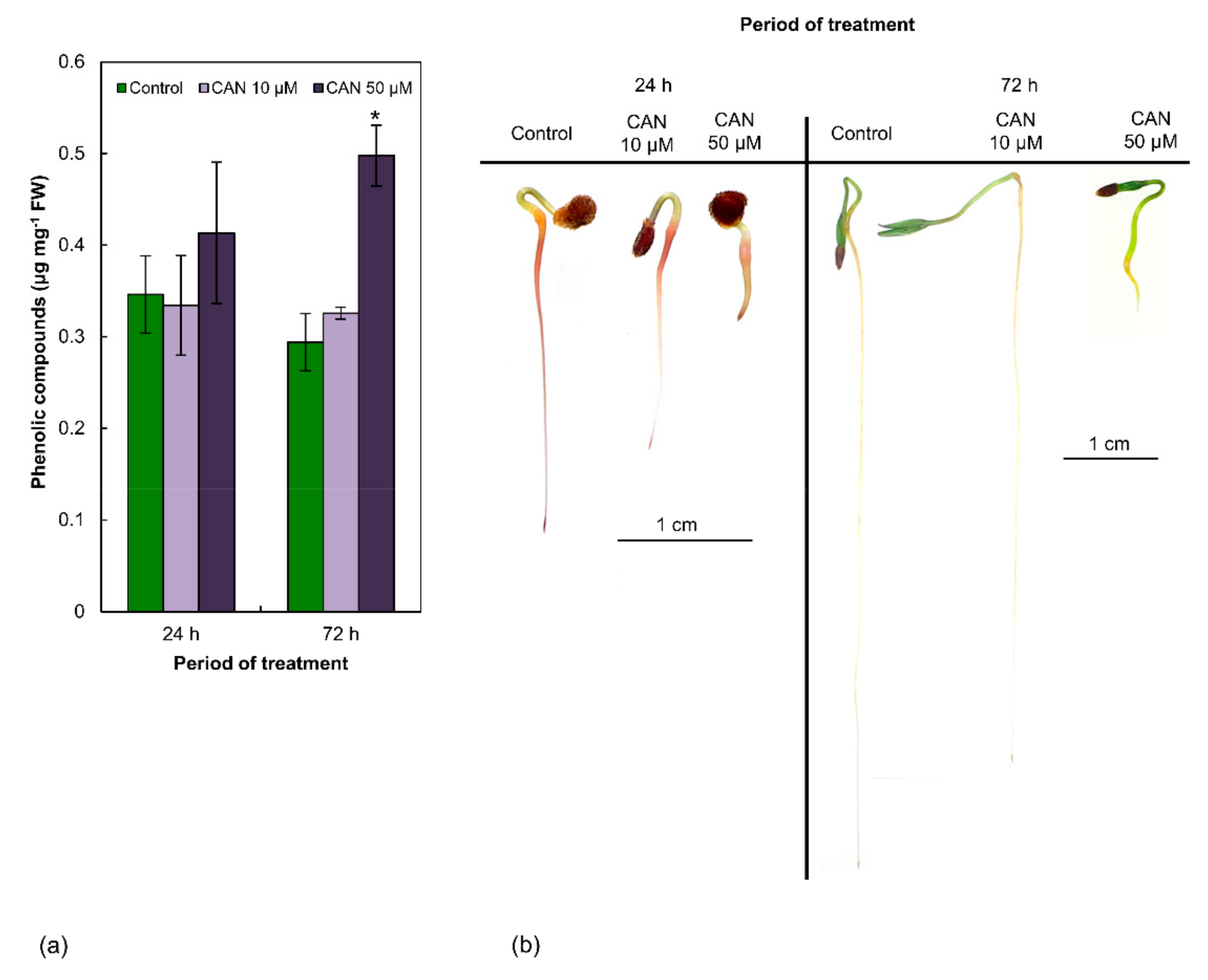
2.1. Concentration of Phenolic Compounds in Tomato Roots Was Increased by Application of 50 µM CAN
2.2. CAN at Higher Concentration (50 µM) Alters Localization of Phenolic Compounds in Roots of Tomato Seedlings
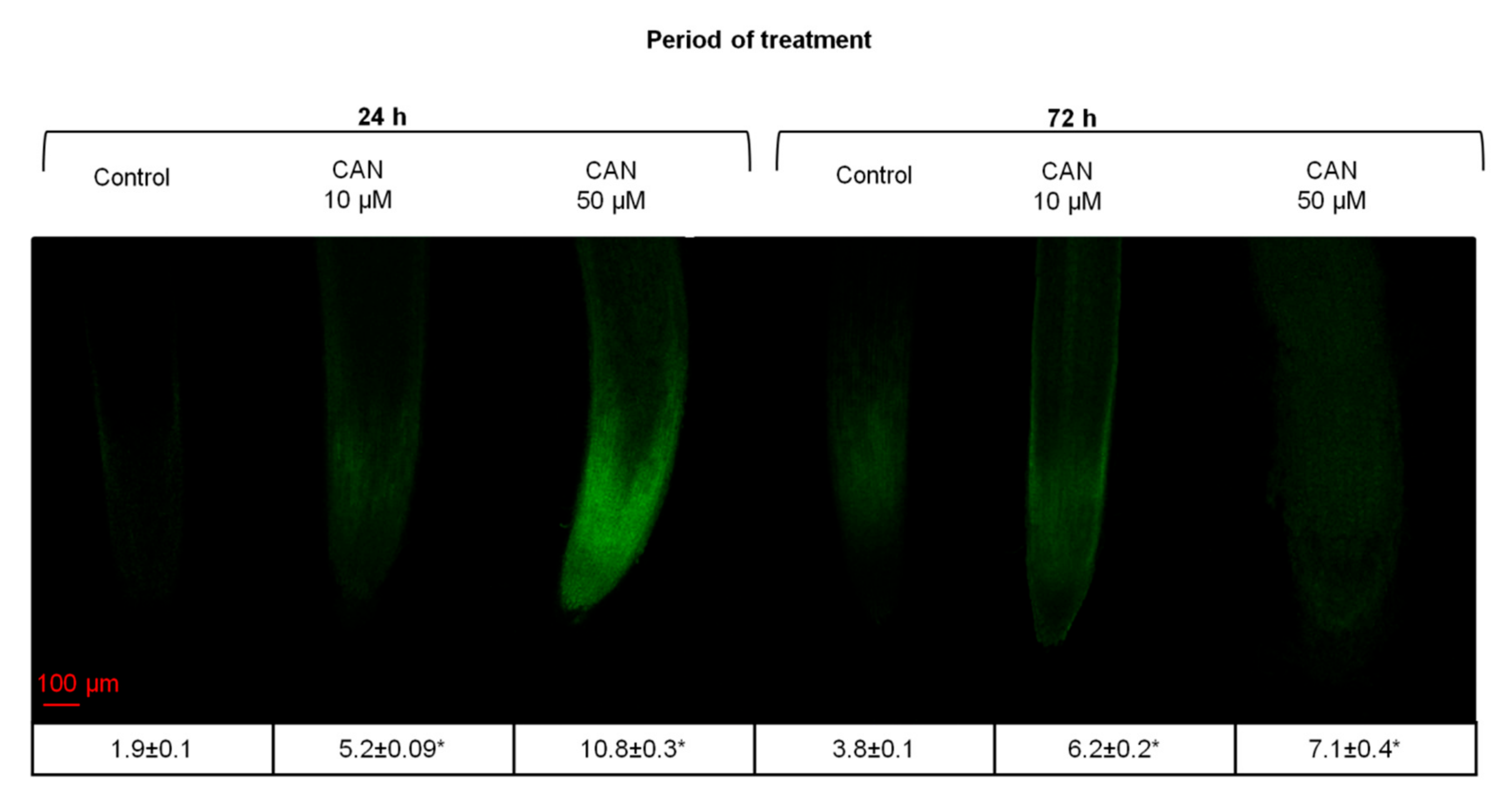
2.3. CAN Increased Flavonoid Accumulation in Tomato Root Tips and Altered Flavonoid Localization in Root Tips
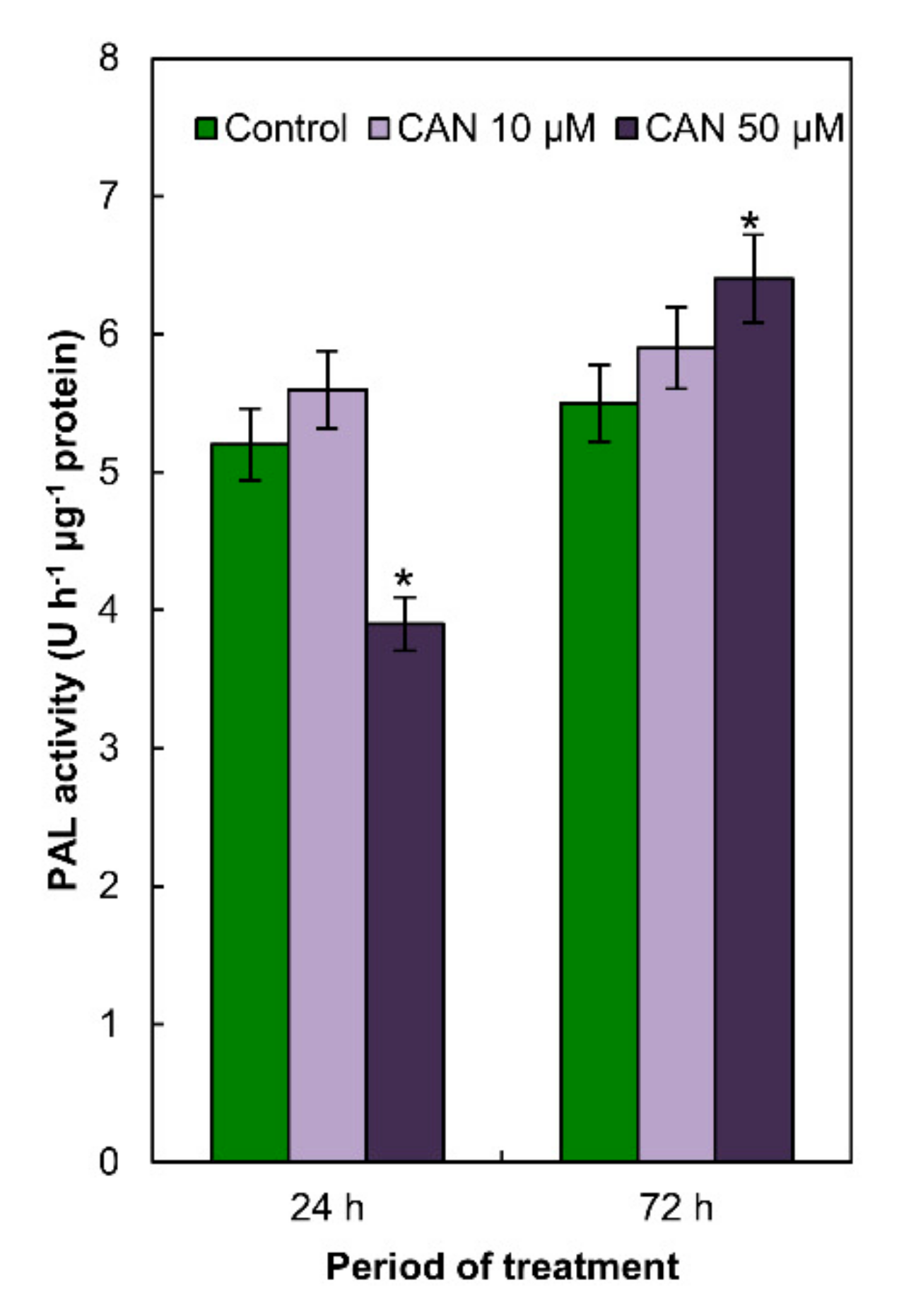
2.4. Activity of PAL in Tomato Roots Was Modified Only by 50 µM CAN
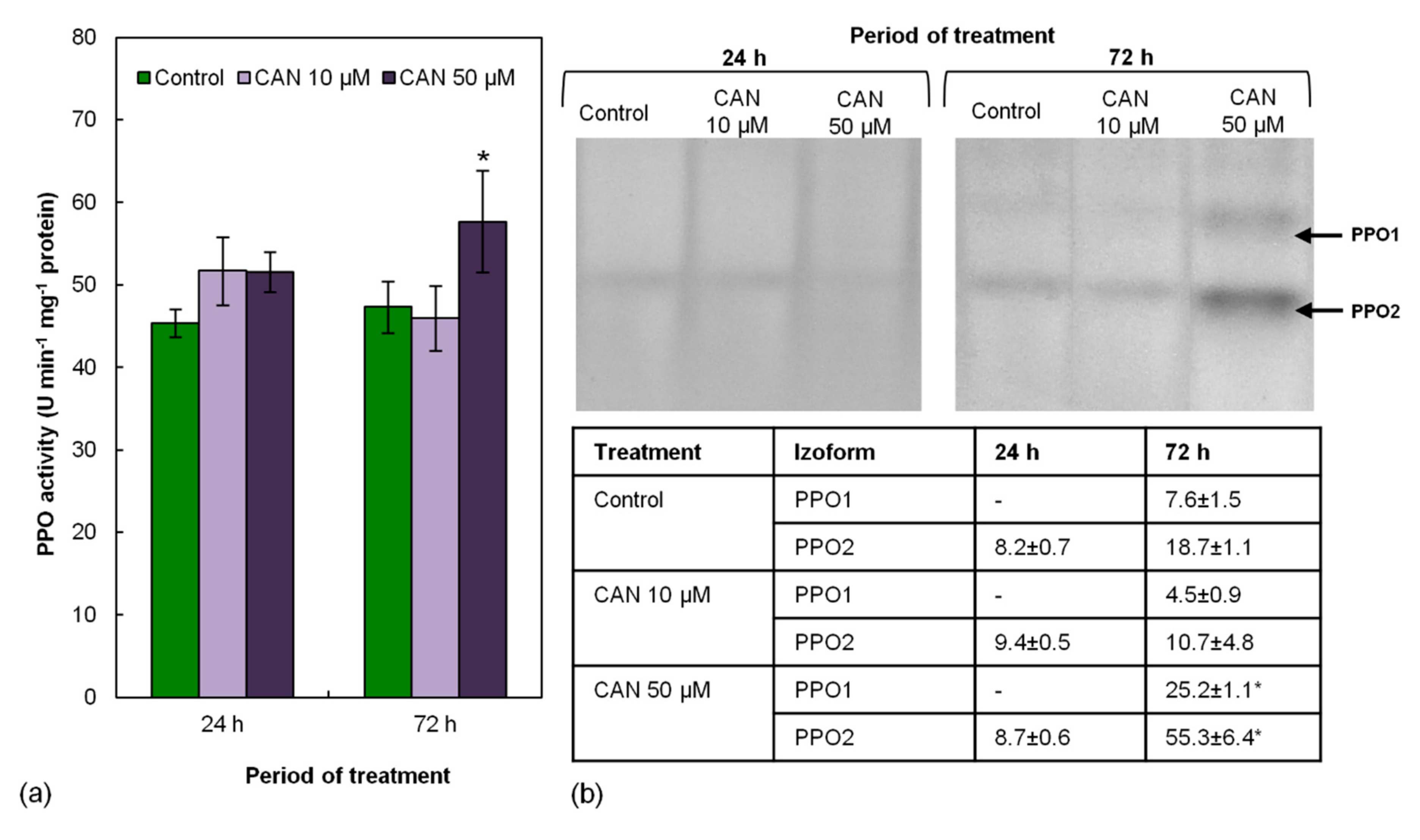
2.5. Activity of PPO in Tomato Roots Increased Only after Prolonged Feeding with 50 µM CAN
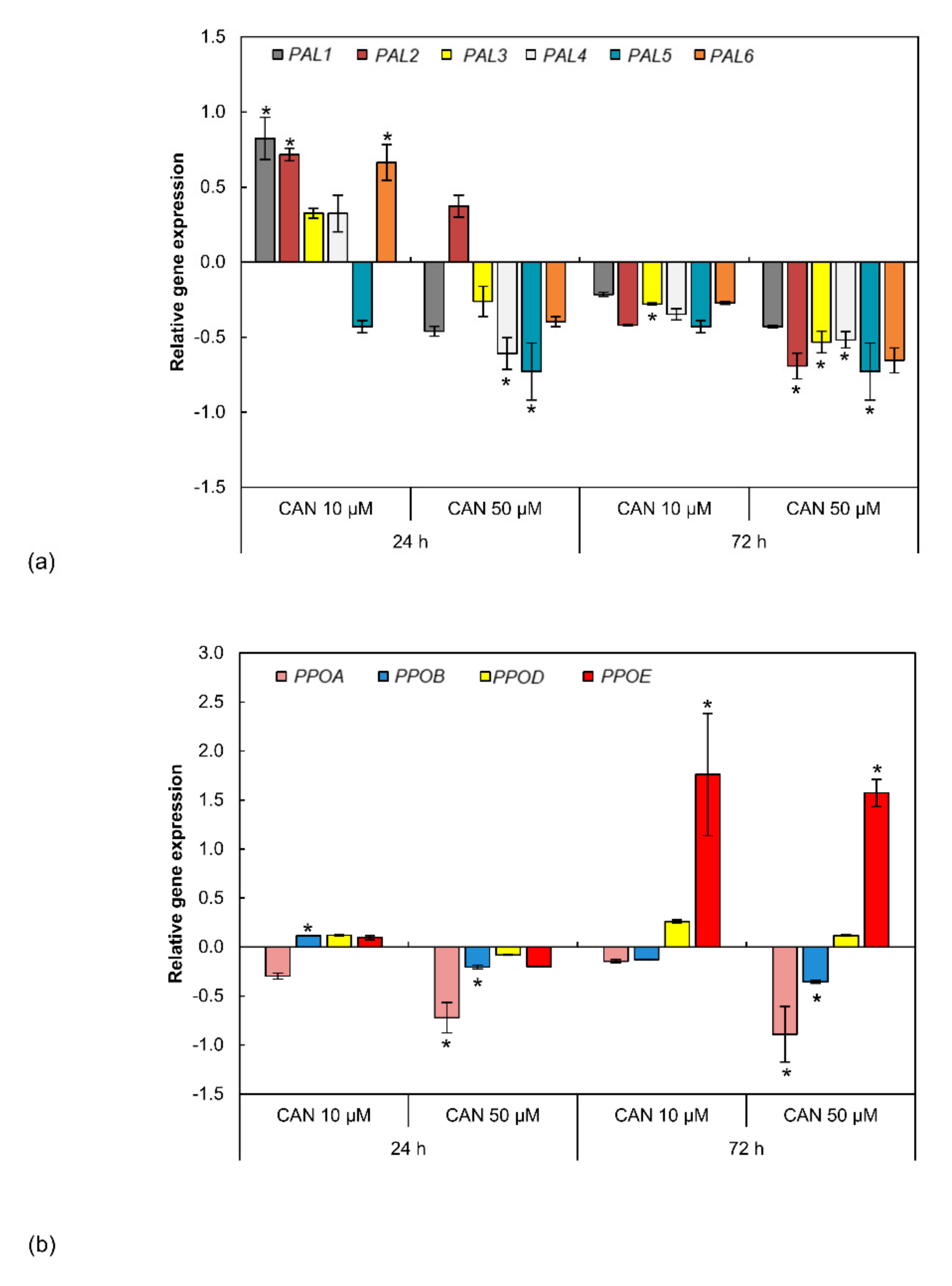
2.6. CAN Modified Expression of Genes Coding PAL and PPO
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Total Phenolic Compounds (PCs) Measurement
4.3. Histochemical Localization of PCs
4.4. Flavonoids Localization
4.5. Measurement of PAL Activity
4.6. Preparation of the Protein Extract for PPO Activity Measurement
4.7. Measurement of PPO Activity
4.7.1. Measurement of PPO Activity in the Root Protein Extract
4.7.2. Measurement of PPO Activity in the Polyacrylamide Gel under Nondenaturing Conditions
4.8. Protein Content Quantification
4.9. PPO and PAL Gene Expression Analysis
4.10. Densitometry Analysis
4.11. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Janků, M.; Luhová, L.; Petřivalský, M. On the origin and fate of reactive oxygen species in plant cell compartments. Antioxidants 2019, 8, 105. [Google Scholar] [CrossRef]
- Laxa, M.; Liebthal, M.; Telman, W.; Chibani, K.; Dietz, K.J. The role of the plant antioxidant system in drought tolerance. Antioxidants 2019, 8, 94. [Google Scholar] [CrossRef] [PubMed]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef] [PubMed]
- Babenko, L.M.; Smirnov, O.E.; Romanenko, K.O.; Trunova, O.K.; Kosakivska, I.V. Phenolic compounds in plants: Biogenesis and functions. Ukr. Biochem. J. 2019, 91, 5–18. [Google Scholar] [CrossRef]
- Pereira, D.M.; Valentão, P.; Pereira, J.A.; Andrade, P.B. Phenolics: From chemistry to biology. Molecules 2009, 14, 2202–2211. [Google Scholar] [CrossRef]
- Jun, S.Y.; Sattler, S.A.; Cortez, G.S.; Vermerris, W.; Sattler, S.E.; Kang, C.H. Biochemical and structural analysis of substrate specificity of a phenylalanine ammonia-lyase. Plant Physiol. 2018, 176, 1452–1468. [Google Scholar] [CrossRef] [PubMed]
- Robards, K.; Antolovich, M. Analytical chemistry of fruit bioflavonoids. A review. Analyst 1997, 122, 11R–34R. [Google Scholar] [CrossRef]
- Croft, K.D. The chemistry and biological effects of flavonoids and phenolic acids. Ann. N. Y. Acad. Sci. 1998, 854, 435–442. [Google Scholar] [CrossRef]
- Mira, L.; Fernandez, M.T.; Santos, M.; Rocha, R.; Florêncio, M.H.; Jennings, K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002, 36, 1199–1208. [Google Scholar] [CrossRef]
- Soares, C.; Carvalho, M.E.A.; Azevedo, R.A.; Fidalgo, F. Plants facing oxidative challenges—A little help from the antioxidant networks. Environ. Exp. Bot. 2019, 161, 4–25. [Google Scholar] [CrossRef]
- Kulbat, K. The role of phenolic compounds in plant resistance. Biotechnol. Food Sci. 2016, 80, 97–108. [Google Scholar]
- Thipyapong, P.; Joel, D.M.; Steffens, J.C. Differential expression and turnover of the tomato polyphenol oxidase gene family during vegetative and reproductive development. Plant Physiol. 1997, 113, 707–718. [Google Scholar] [CrossRef] [PubMed]
- Taranto, F.; Pasqualone, A.; Mangini, G.; Tripodi, P.; Miazzi, M.; Pavan, S.; Montemurro, C. Polyphenol oxidases in crops: Biochemical, physiological and genetic aspects. Int. J. Mol. Sci. 2017, 18, 377. [Google Scholar] [CrossRef]
- Boeckx, T.; Webster, R.; Winters, A.L.; Webb, K.J.; Gay, A.; Kingston-Smith, A.H. Polyphenol oxidase-mediated protection against oxidative stress is not associated with enhanced photosynthetic efficiency. Ann. Bot. 2015, 116, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Newman, S.M.; Eannetta, N.T.; Yu, H.; Prince, J.P.; Carmen de Vicente, M.; Tanksley, S.D.; Steffens, J.C. Organisation of the tomato polyphenol oxidase gene family. Plant Mol. Biol. 1993, 21, 1035–1051. [Google Scholar] [CrossRef]
- Constabel, C.P.; Barbehenn, R. Defensive Roles of Polyphenol Oxidase in Plants. In Induced Plant Resistance to Herbivory; Schaller, A., Ed.; Springer: Dordrecht, The Netherlands, 2008; pp. 253–270. ISBN 9781402081828. [Google Scholar]
- Santisree, P.; Sanivarapu, H.; Gundavarapu, S.; Sharma, K.K.; Bhatnagar-Mathur, P. Nitric Oxide as a Signal in Inducing Secondary Metabolites During Plant Stress. In Co-Evolution of Secondary Metabolites. Reference Series in Phytochemistry; Merillon, J.M., Ramawat, K.G., Eds.; Springer: Cham, Switzerland, 2019; pp. 1–29. [Google Scholar]
- Gupta, K.J.; Kolbert, Z.; Durner, J.; Lindermayr, C.; Corpas, F.J.; Brouquisse, R.; Barroso, J.B.; Umbreen, S.; Palma, J.M.; Hancock, J.T.; et al. Regulating the regulator: Nitric oxide control of post-translational modifications. New Phytol. 2020, 227, 1319–1325. [Google Scholar] [CrossRef]
- Durner, J.; Wendehenne, D.; Klessig, D.F. Defense gene induction in tobacco by nitric oxide, cyclic GMP, and cyclic ADP-ribose. Proc. Natl. Acad. Sci. USA 1998, 95, 10328–10333. [Google Scholar] [CrossRef]
- Staszek, P.; Weston, L.A.; Ciacka, K.; Krasuska, U.; Gniazdowska, A. L-Canavanine: How does a simple non-protein amino acid inhibit cellular function in a diverse living system? Phytochem. Rev. 2017, 16, 1269–1282. [Google Scholar] [CrossRef]
- Nunn, P.B.; Bell, E.A.; Watson, A.A.; Nash, R.J. Toxicity of non-protein amino acids to humans and domestic animals. Nat. Prod. Commun. 2010, 5, 485–504. [Google Scholar] [CrossRef]
- Kim, H.S.; Cha, S.Y.; Jo, C.H.; Han, A.; Hwang, K.Y. The crystal structure of arginyl-tRNA synthetase from Homo sapiens. FEBS Lett. 2014, 588, 2328–2334. [Google Scholar] [CrossRef]
- Krasuska, U.; Ciacka, K.; Orzechowski, S.; Fettke, J.; Bogatek, R.; Gniazdowska, A. Modification of the endogenous NO level influences apple embryos dormancy by alterations of nitrated and biotinylated protein patterns. Planta 2016, 244, 877–891. [Google Scholar] [CrossRef] [PubMed]
- Staszek, P.; Krasuska, U.; Otulak-Kozieł, K.; Fettke, J.; Gniazdowska, A. Canavanine induced decrease in NO synthesis alters activity of antioxidant system but does not impact GSNO catabolism in tomato roots. Front. Plant Sci. 2019, 10, 1077. [Google Scholar] [CrossRef] [PubMed]
- Staszek, P.; Gniazdowska, A. Peroxynitrite induced signaling pathways in plant response to non-proteinogenic amino acids. Planta 2020, 252, 5. [Google Scholar] [CrossRef] [PubMed]
- Astier, J.; Gross, I.; Durner, J. Nitric oxide production in plants: An update. J. Exp. Bot. 2018, 69, 3401–3411. [Google Scholar] [CrossRef] [PubMed]
- Corpas, F.J.; Barroso, J.B. Nitric oxide synthase-like activity in higher plants. Nitric Oxide 2017, 68, 5–6. [Google Scholar] [CrossRef] [PubMed]
- Krasuska, U.; Andrzejczak, O.; Staszek, P.; Bogatek, R.; Gniazdowska, A. Canavanine alters ROS/RNS level and leads to post-translational modification of proteins in roots of tomato seedlings. Front. Plant Sci. 2016, 7, 840. [Google Scholar] [CrossRef] [PubMed]
- Krasuska, U.; Andrzejczak, O.; Staszek, P.; Borucki, W.; Gniazdowska, A. Toxicity of canavanine in tomato (Solanum lycopersicum L.) roots is due to alterations in RNS, ROS and auxin levels. Plant Physiol. Biochem. 2016, 103, 84–95. [Google Scholar] [CrossRef]
- Buer, C.S.; Muday, G.K. The transparent testa4 mutation prevents flavonoid synthesis and alters auxin transport and the response of arabidopsis roots to gravity and light. Plant Cell 2004, 16, 1191–1205. [Google Scholar] [CrossRef]
- Donaldson, L.; Williams, N. Imaging and spectroscopy of natural fluorophores in pine needles. Plants 2018, 7, 10. [Google Scholar] [CrossRef]
- Rodrigues-Corrêa, K.C.d.S.; Fett-Neto, A.G. Abiotic stresses and non-protein amino acids in plants. CRC Crit. Rev. Plant Sci. 2019, 38, 411–430. [Google Scholar] [CrossRef]
- Andrzejczak, O.; Krasuska, U.; Olechowicz, J.; Staszek, P.; Ciacka, K.; Bogatek, R.; Hebelstrup, K.; Gniazdowska, A. Destabilization of ROS metabolism in tomato roots as a phytotoxic effect of meta-tyrosine. Plant Physiol. Biochem. 2018, 123, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Krasuska, U.; Andrzejczak, O.; Staszek, P.; Borucki, W.; Gniazdowska, A. meta-Tyrosine induces modification of reactive nitrogen species level, protein nitration and nitrosoglutathione reductase in tomato roots. Nitric Oxide 2017, 68, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Bertin, C.; Weston, L.A.; Huang, T.; Jander, G.; Owens, T.; Meinwald, J.; Schroeder, F.C. Grass roots chemistry: meta-Tyrosine, an herbicidal nonprotein amino acid. Proc. Natl. Acad. Sci. USA 2007, 104, 16964–16969. [Google Scholar] [CrossRef] [PubMed]
- Silva-Beltrán, N.P.; Ruiz-Cruz, S.; Cira-Chávez, L.A.; Estrada-Alvarado, M.I.; Ornelas-Paz, J.D.J.; López-Mata, M.A.; Del-Toro-Sánchez, C.L.; Ayala-Zavala, J.F.; Márquez-Ríos, E. Total phenolic, flavonoid, tomatine, and tomatidine contents and antioxidant and antimicrobial activities of extracts of tomato plant. Int. J. Anal. Chem. 2015, 2015, 284071. [Google Scholar] [CrossRef]
- Deng, S.P.; Yang, Y.L.; Cheng, X.X.; Li, W.R.; Cai, J.Y. Synthesis, spectroscopic study and radical scavenging activity of kaempferol derivatives: Enhanced water solubility and antioxidant activity. Int. J. Mol. Sci. 2019, 20, 975. [Google Scholar] [CrossRef]
- Banjarnahor, S.D.; Artanti, N. Antioxidant properties of flavonoids. Med. J. Indones. 2015, 23, 239–244. [Google Scholar] [CrossRef]
- Hernández, I.; Alegre, L.; Van Breusegem, F.; Munné-Bosch, S. How relevant are flavonoids as antioxidants in plants? Trends Plant Sci. 2009, 14, 125–132. [Google Scholar] [CrossRef]
- Valifard, M.; Mohsenzadeh, S.; Kholdebarin, B.; Rowshan, V. Effects of salt stress on volatile compounds, total phenolic content and antioxidant activities of Salvia mirzayanii. S. Afr. J. Bot. 2014, 93, 92–97. [Google Scholar] [CrossRef]
- Ksouri, R.; Megdiche, W.; Debez, A.; Falleh, H.; Grignon, C.; Abdelly, C. Salinity effects on polyphenol content and antioxidant activities in leaves of the halophyte Cakile maritima. Plant Physiol. Biochem. 2007, 45, 244–249. [Google Scholar] [CrossRef]
- Sarker, U.; Oba, S. Drought stress enhances nutritional and bioactive compounds, phenolic acids and antioxidant capacity of Amaranthus leafy vegetable. BMC Plant Biol. 2018, 18, 258. [Google Scholar] [CrossRef]
- Naikoo, M.I.; Dar, M.I.; Raghib, F.; Jaleel, H.; Ahmad, B.; Raina, A.; Khan, F.A.; Naushin, F. Role and Regulation of Plants Phenolics in Abiotic Stress Tolerance. In Plant Signaling Molecules; Iqbal, K.M., Antonio, F., Reddy Sudhakar, P., Khan, N.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 157–168. ISBN 9780128164518. [Google Scholar]
- Mahdavikia, F.; Saharkhiz, M.J. Secondary metabolites of peppermint change the morphophysiological and biochemical characteristics of tomato. Biocatal. Agric. Biotechnol. 2016, 7, 127–133. [Google Scholar] [CrossRef]
- Peer, W.A.; Blakeslee, J.J.; Yang, H.; Murphy, A.S. Seven things we think we know about auxin transport. Mol. Plant 2011, 4, 487–504. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.E.; Rashotte, A.M.; Murphy, A.S.; Normanly, J.; Tague, B.W.; Peer, W.A.; Taiz, L.; Muday, G.K. Flavonoids act as negative regulators of auxin transport in vivo in arabidopsis. Plant Physiol. 2001, 126, 524–535. [Google Scholar] [CrossRef] [PubMed]
- De Klerk, G.J.; Guan, H.; Huisman, P.; Marinova, S. Effects of phenolic compounds on adventitious root formation and oxidative decarboxylation of applied indoleacetic acid in Malus ‘Jork 9’. Plant Growth Regul. 2011, 63, 175–185. [Google Scholar] [CrossRef]
- Franco, D.M.; Silva, E.M.; Saldanha, L.L.; Adachi, S.A.; Schley, T.R.; Rodrigues, T.M.; Dokkedal, A.L.; Nogueira, F.T.S.; Rolim de Almeida, L.F. Flavonoids modify root growth and modulate expression of SHORT-ROOT and HD-ZIP III. J. Plant Physiol. 2015, 188, 89–95. [Google Scholar] [CrossRef]
- MacDonald, M.J.; D’Cunha, G.B. A modern view of phenylalanine ammonia lyase. Biochem. Cell Biol. 2007, 85, 273–282. [Google Scholar] [CrossRef]
- Dos Santos, W.D.; Ferrarese, M.D.L.L.; Finger, A.; Teixeira, A.C.N.; Ferrarese-Filho, O. Lignification and related enzymes in Glycine max root growth-inhibition by ferulic acid. J. Chem. Ecol. 2004, 30, 1203–1212. [Google Scholar] [CrossRef]
- Omezzine, F.; Ladhari, A.; Haouala, R. Physiological and biochemical mechanisms of allelochemicals in aqueous extracts of diploid and mixoploid Trigonella foenum-graecum L. S. Afr. J. Bot. 2014, 93, 167–178. [Google Scholar] [CrossRef]
- Rama Devi, S.; Prasad, M.N.V. Ferulic acid mediated changes in oxidative enzymes of maize seedlings: Implications in growth. Biol. Plant. 1996, 38, 387–395. [Google Scholar] [CrossRef]
- Politycka, B. Phenolics and the activities of phenylalanine ammonia-lyase, phenol-β-glucosyltransferase and β-glucosidase in cucumber roots as affected by phenolic allelochemicals. Acta Physiol. Plant. 1998, 20, 405–410. [Google Scholar] [CrossRef]
- Herrig, V.; Ferrarese, M.D.L.L.; Suzuki, L.S.; Rodrigues, J.D.; Ferrarese-Filho, O. Peroxidase and phenylalanine ammonia-lyase activities, phenolic acid contents, and allelochemicals-inhibited root growth of soybean. Biol. Res. 2002, 35, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Kováčik, J.; Klejdus, B.; Bačkor, M. Nitric oxide signals ROS scavenger-mediated enhancement of PAL activity in nitrogen-deficient Matricaria chamomilla roots: Side effects of scavengers. Free Radic. Biol. Med. 2009, 46, 1686–1693. [Google Scholar] [CrossRef] [PubMed]
- De Pinto, M.C.; Tommasi, F.; De Gara, L. Changes in the antioxidant systems as part of the signaling pathway responsible for the programmed cell death activated by nitric oxide and reactive oxygen species in tobacco bright-yellow 2 cells. Plant Physiol. 2002, 130, 698–708. [Google Scholar] [CrossRef] [PubMed]
- Lavanya, S.N.; Udayashankar, A.C.; Raj, S.N.; Mohan, C.D.; Gupta, V.K.; Tarasatyavati, C.; Srivastava, R.; Nayaka, S.C. Lipopolysaccharide-induced priming enhances NO-mediated activation of defense responses in pearl millet challenged with Sclerospora graminicola. 3 Biotech 2018, 8, 475. [Google Scholar] [CrossRef]
- Guo, P.; Cao, Y.; Li, Z.; Zhao, B. Role of an endogenous nitric oxide burst in the resistance of wheat to stripe rust. Plant Cell Environ. 2004, 27, 473–477. [Google Scholar] [CrossRef]
- Manjunatha, G.; Raj, S.N.; Shetty, N.P.; Shetty, H.S. Nitric oxide donor seed priming enhances defense responses and induces resistance against pearl millet downy mildew disease. Pestic. Biochem. Physiol. 2008, 91, 1–11. [Google Scholar] [CrossRef]
- Delledonne, M.; Xia, Y.; Dixon, R.A.; Lamb, C. Nitric oxide functions as a signal in plant disease resistance. Nature 1998, 394, 585–588. [Google Scholar] [CrossRef]
- Arasimowicz, M.; Floryszak-Wieczorek, J. Nitric oxide as a bioactive signalling molecule in plant stress responses. Plant Sci. 2007, 172, 876–887. [Google Scholar] [CrossRef]
- Neill, S.J. Hydrogen peroxide and nitric oxide as signalling molecules in plants. J. Exp. Bot. 2002, 53, 1237–1247. [Google Scholar] [CrossRef]
- Murimwa, J.C.; Rugare, J.T.; Mabasa, S.; Mandumbu, R. Allelopathic effects of aqueous extracts of Sorghum (Sorghum bicolor L. Moench) on the early seedling growth of Sesame (Sesamum indicum L.) varieties and selected weeds. Int. J. Agron. 2019, 2019, 5494756. [Google Scholar] [CrossRef]
- Thipyapong, P.; Stout, M.; Attajarusit, J. Functional analysis of polyphenol oxidases by antisense/sense technology. Molecules 2007, 12, 1569–1595. [Google Scholar] [CrossRef] [PubMed]
- Thipyapong, P.; Hunt, M.D.; Steffens, J.C. Antisense downregulation of polyphenol oxidase results in enhanced disease susceptibility. Planta 2004, 220, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Zhawar, V.K. Hydrogen peroxide and nitric oxide regulation of phenolic metabolism under water stress and ABA in wheat. Acta Biol. Hung. 2017, 68, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Arasimowicz-Jelonek, M.; Floryszak-Wieczorek, J. Nitric oxide: An effective weapon of the plant or the pathogen? Mol. Plant Pathol. 2014, 15, 406–416. [Google Scholar] [CrossRef]
- Arasimowicz-Jelonek, M.; Floryszak-Wieczorek, J. Nitric oxide in the offensive strategy of fungal and oomycete plant pathogens. Front. Plant Sci. 2016, 7, 252. [Google Scholar] [CrossRef]
- Papaiah, S.; Narasimha, G. Peroxidase and polyphenol oxidase activities in healthy and viral infected sunflower (Helianthus annuus L.) leaves. Biotechnol. An Indian J. 2014, 9, 1–5. [Google Scholar]
- Kuvalekar, A.; Redkar, A.; Gandhe, K.; Harsulkar, A. Peroxidase and polyphenol oxidase activities in compatible host-pathogen interaction in Jasminum officinale and Uromyces hobsoni: Insights into susceptibility of host. N. Z. J. Bot. 2011, 49, 351–359. [Google Scholar] [CrossRef]
- Shivalingaiah; Umesha, S.; Sateesh, M.K. Cocculus hirsutus extract inhibits the Xanthomonas oryzae pv. oryzae, the bacterial leaf blight pathogen in rice. Arch. Phytopathol. Plant Prot. 2013, 46, 1885–1894. [Google Scholar]
- Akladious, S.A.; Isaac, G.S.; Abu-Tahon, M.A. Induction and resistance against Fusarium wilt disease of tomato by using sweet basil (Ocimum basilicum L) extract. Can. J. Plant Sci. 2015, 95, 689–701. [Google Scholar] [CrossRef]
- Kampatsikas, I.; Bijelic, A.; Rompel, A. Biochemical and structural characterization of tomato polyphenol oxidases provide novel insights into their substrate specificity. Sci. Rep. 2019, 9, 4022. [Google Scholar] [CrossRef]
- Chi, M.; Bhagwat, B.; Lane, W.; Tang, G.; Su, Y.; Sun, R.; Oomah, B.; Wiersma, P.A.; Xiang, Y. Reduced polyphenol oxidase gene expression and enzymatic browning in potato (Solanum tuberosum L.) with artificial microRNAs. BMC Plant Biol. 2014, 14, 62. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Li, D.; Li, J.; Shao, F.; Lu, S. Characterization of the polyphenol oxidase gene family reveals a novel microRNA involved in posttranscriptional regulation of PPOs in Salvia miltiorrhiza. Sci. Rep. 2017, 7, 44622. [Google Scholar] [CrossRef] [PubMed]
- Serradell, M.D.L.A.; Rozenfeld, P.A.; Martinez, G.A.; Civello, P.M.; Chaves, A.R.; Anon, M.C. Polyphenoloxidase activity from strawberry fruit (Fragariaananassa, Duch., cv Selva): Characterisation and partial purification. J. Sci. Food Agric. 2000, 80, 1421–1427. [Google Scholar] [CrossRef]
- Conforti, F.; Menichini, F. Phenolic compounds from plants as nitric oxide production inhibitors. Curr. Med. Chem. 2011, 18, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Reeve, R.M. Histological and histochemical changes in developing and ripening peaches. II. Cell walls and pectins. Am. J. Bot. 1959, 46, 241–248. [Google Scholar] [CrossRef]
- Sanz, L.; Fernández-Marcos, M.; Modrego, A.; Lewis, D.R.; Muday, G.K.; Pollmann, S.; Dueñas, M.; Santos-Buelga, C.; Lorenzo, O. Nitric oxide plays a role in stem cell niche homeostasis through its interaction with auxin. Plant Physiol. 2014, 166, 1972–1984. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, K.; Hoson, T.; Kamisaka, S. Abscisic acid suppresses the increases in cell wall-bound ferulic and diferulic acid levels in dark-grown wheat (Triticum aestivum L.) coleoptiles. Plant Cell Physiol. 1997, 38, 811–817. [Google Scholar] [CrossRef][Green Version]
- Rescigno, A.; Sanjust, E.; Pedulli, G.F.; Valgimigli, L. Spectrophotometric method for the determination of polyphenol oxidase activity by coupling of 4-tert-butyl-o-benzoquinone and 4-amino-N,N- diethylaniline. Anal. Lett. 1999, 32, 2007–2017. [Google Scholar] [CrossRef]
- Rescigno, A.; Sollai, F.; Rinaldi, A.C.; Soddu, G.; Sanjust, E. Polyphenol oxidase activity staining in polyacrylamide electrophoresis gels. J. Biochem. Biophys. Methods 1997, 34, 155–159. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Staszek, P.; Krasuska, U.; Bederska-Błaszczyk, M.; Gniazdowska, A. Canavanine Increases the Content of Phenolic Compounds in Tomato (Solanum lycopersicum L.) Roots. Plants 2020, 9, 1595. https://doi.org/10.3390/plants9111595
Staszek P, Krasuska U, Bederska-Błaszczyk M, Gniazdowska A. Canavanine Increases the Content of Phenolic Compounds in Tomato (Solanum lycopersicum L.) Roots. Plants. 2020; 9(11):1595. https://doi.org/10.3390/plants9111595
Chicago/Turabian StyleStaszek, Pawel, Urszula Krasuska, Magdalena Bederska-Błaszczyk, and Agnieszka Gniazdowska. 2020. "Canavanine Increases the Content of Phenolic Compounds in Tomato (Solanum lycopersicum L.) Roots" Plants 9, no. 11: 1595. https://doi.org/10.3390/plants9111595
APA StyleStaszek, P., Krasuska, U., Bederska-Błaszczyk, M., & Gniazdowska, A. (2020). Canavanine Increases the Content of Phenolic Compounds in Tomato (Solanum lycopersicum L.) Roots. Plants, 9(11), 1595. https://doi.org/10.3390/plants9111595






