Development of Interspecific Hybrids between a Cultivated Eggplant Resistant to Bacterial Wilt (Ralstonia solanacearum) and Eggplant Wild Relatives for the Development of Rootstocks
Abstract
1. Introduction
2. Results and discussion
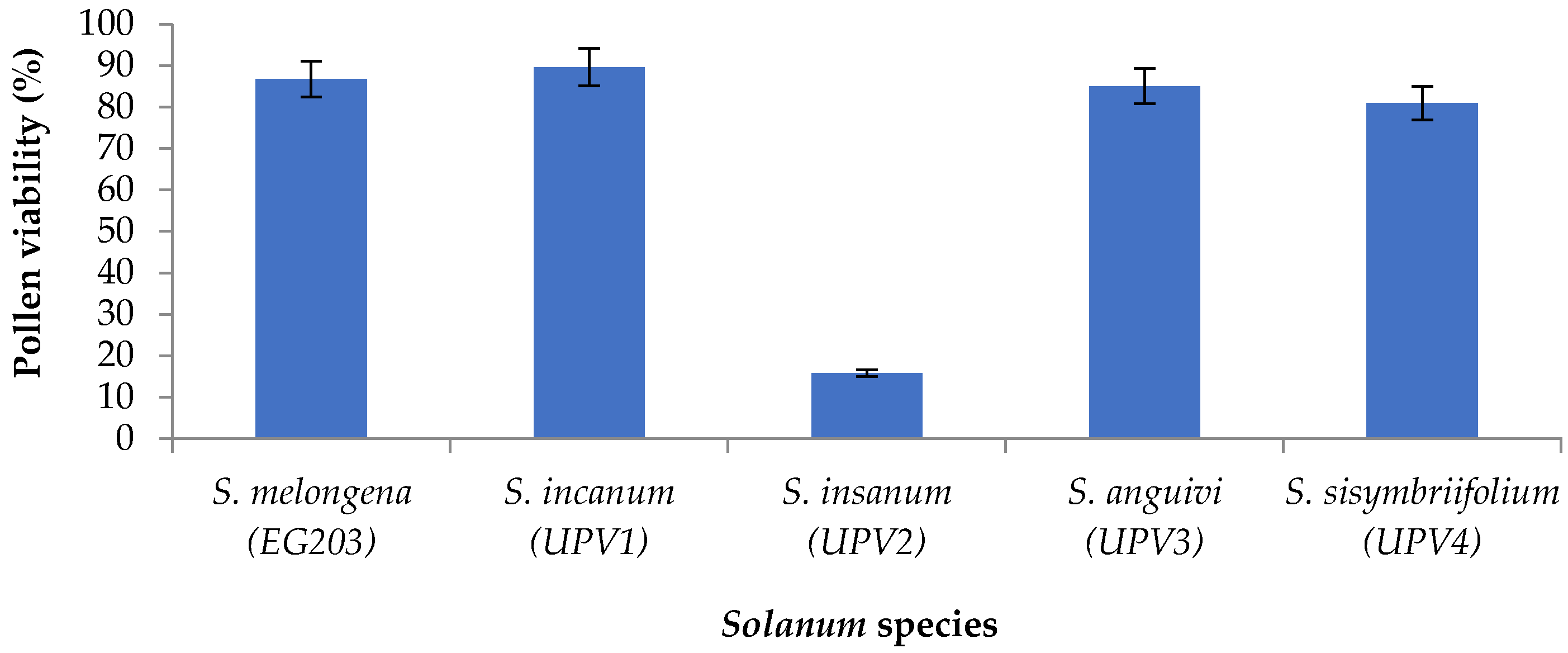
2.1. Production of Interspecific Hybrids
2.2. Hybridity Confirmation
2.3. Assessment of Disease Resistance
3. Material and methods
3.1. Production of Interspecific Hybrids
3.2. Hybridity Confirmation
3.3. Assessment of Disease Resistance
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- FAO. Food and Agriculture Organization of the United Nations. FAO—STAT Production Databases. Available online: http://www.faostat3.fao.org (accessed on 3 August 2020).
- Timberlake, C.F. Anthocyanins in fruit and vegetables. In Recent Advances in the Biochemistry of Fruits and Vegetables; Rhodes, J., Rhodes, M.J.C., Eds.; Academic Press: New York, NY, USA, 1981; pp. 221–247. [Google Scholar]
- Singh, A.P.; Luthria, D.; Wilson, T.; Vorsa, N.; Singh, V.; Banuelos, G.S.; Pasakdee, S. Polyphenols content and antioxidant capacity of eggplant pulp. Food Chem. 2009, 114, 955–961. [Google Scholar] [CrossRef]
- Hayward, A.C. Biology and epidemiology of bacterial wilt caused by Ralstonia solanacearum. Annu. Rev. Phytopathol. 1991, 29, 65–87. [Google Scholar] [CrossRef]
- Schaad, N.W.; Jones, J.B.; Chun, W. Laboratory Guide for the Identification of Plant Pathogenic Bacteria; American Phytopathological Society Press: St. Paul, MN, USA, 2001. [Google Scholar]
- Fegan, M.; Prior, P.; Allen, C.; Hayward, A.C. How complex is the Ralstonia solanacearum species complex. In Bacterial Wilt Disease and the Ralstonia Solanacearum Species Complex; Allen, C., Prior, P., Hayward, A.C., Eds.; American Phytopathological Society Press: St. Paul, MN, USA, 2005; pp. 449–461. [Google Scholar]
- Elphinstone, J.G. The current bacterial wilt situation. A global view. In Bacterial Wilt Disease and the Ralstonia solanacearum Species Complex; Allen, C., Prior, P., Hayward, A.C., Eds.; APS Press: Saint Paul, MN, USA, 2005; pp. 9–28. [Google Scholar]
- Wenneker, M.; Verdel, M.S.W.; Groeneveld, R.M.W.; Kempenaar, C.; Van Beuningen, A.R.; Janse, J.D. Ralstonia (Pseudomonas) solanacearum race3 (biovar2) in surface water and natural weed hosts: First report on stinging nettle (Urtica dioica). Eur. J. Plant. Pathol. 1999, 105, 307–315. [Google Scholar] [CrossRef]
- Huet, G. Breeding for resistances to Ralstonia solanacearum. Front. Plant. Sci. 2014, 5, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Michel, V.V.; Wang, J.F.; Midmore, D.J.; Hartman, G.L. Effects of intercropping and soil amendment with urea and calcium oxide on the incidence of bacterial wilt of tomato and survival of soil-borne Pseudomonas solanacearum in Taiwan. Plant. Pathol. 2003, 46, 600–610. [Google Scholar] [CrossRef]
- Michel, V.V.; Mew, T.W. Effect of a soil amendment on the survival of Ralstonia solanacearum in different soils. Phytopathology 1998, 88, 300–305. [Google Scholar] [CrossRef]
- Pradhanang, P.; Ji, P.; Momol, M.; Olson, S.M.; Mayfield, J.L.; Jones, J. Application of acibenzolar-S-methyl enhances host resistance in tomato against Ralstonia solanacearum. Plant. Dis. 2005, 89, 989–993. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, J.; Genin, S.; Magori, S.; Citovsky, V.; Sriariyanum, M.; Ronald, P.; Dow, M.A.X.; Verdier, V.; Beer, S.V.; Machado, M.A.; et al. Top 10 plant pathogenic bacteria in molecular plant pathology. Mol. Plant. Pathol. 2012, 13, 614–629. [Google Scholar]
- Hartman, G.L.; Hong, W.F.; Hayward, A.C. Potential of biological and chemical control of bacterial wilt. ACIAR Proc. Aust. Cent. Int. Agric. Res. 1993, 45, 322–326. [Google Scholar]
- Lemessa, F.; Zeller, W. Screening rhizobacteria for biological control of Ralstonia solanacearum in Ethiopia. Biol. Control. 2007, 42, 336–344. [Google Scholar] [CrossRef]
- Boshou, L. A broad review and perspective on breeding for resistance to bacterial wilt. In Bacterial Wilt Disease and the Ralstonia solanacearum Species Complex; Allen, C., Prior, P., Hayward, A.C., Eds.; American Phytopathological Society: St. Paul, MN, USA, 2005; pp. 225–238. [Google Scholar]
- Daunay, M.C. Eggplant. In Handbook of Plant Breeding, Vegetables II: Fabaceae, Liliaceae, Umbelliferae, and Solanaceae; Prohens, J., Nuez, F., Eds.; Springer: New York, NY, USA, 2008; pp. 163–220. [Google Scholar]
- Chaudhary, D.R.; Sharma, S.D. Screening of some brinjal cultivars against bacterial wilt and fruit borer. Agric. Sci. Digest 2000, 20, 129–130. [Google Scholar]
- Lebeau, A.; Daunay, M.C.; Frary, A.; Palloix, A.; Wang, J.F.; Dintinger, J.; Chiroleu, F.; Wicker, E.; Prior, P. Bacterial wilt resistance in tomato, pepper, and eggplant: Genetic resources respond to diverse strains in the Ralstonia solanacearum complex. Phytopathology 2011, 101, 154–165. [Google Scholar] [CrossRef]
- Cao, B.-H.; Lei, J.-J.; Wang, Y.; Chen, G.-J. Inheritance and identification of SCAR marker linked to bacterial wilt-resistance in eggplant. Afr. J. Biotech. 2009, 8, 5201–5207. [Google Scholar]
- Xiao, X.; Cao, B.; Li, G.; Lei, J.; Chen, Q.; Jiang, J.; Cheng, Y. Functional characterization of a putative bacterial wilt resistance gene (RE-bw) in eggplant. Plant. Mol. Biol. Rep. 2015, 33, 1058–1073. [Google Scholar]
- Salgon, S.; Raynal, M.; Lebon, S.; Baptiste, J.M.; Daunay, M.C.; Dintinger, J.; Jourda, C. Genotyping by sequencing highlights a polygenic resistance to Ralstonia solanacearum in eggplant (Solanum melongena L.). Intl. J. Mol. Sci. 2018, 19, 357. [Google Scholar] [CrossRef]
- Zamir, D. Improving plant breeding with exotic genetic libraries. Nat. Rev. Genet. 2001, 2, 983–989. [Google Scholar] [CrossRef]
- Taher, D.; Solberg, S.; Prohens, J.; Chou, Y.Y.; Rakha, M.; Wu, T.H. World Vegetable Center Eggplant Collection: Origin, composition, seed dissemination and utilization in breeding. Front. Plant. Sci. 2017, 8, 1484. [Google Scholar] [CrossRef]
- Daunay, M.C.; Salinier, J.; Aubriot, X. Crossability and Diversity of Eggplants and Their Wild Relatives. In The Eggplant Genome; Chapman, M.A., Ed.; Springer: New York, NY, USA, 2019; pp. 135–191. [Google Scholar]
- Devi, C.P.; Munshi, A.D.; Behera, T.K.; Choudhary, H.; Vinod, G.B.; Saha, P. Cross compatibility in interspecific hybridization of eggplant, Solanum melongena, with its wild relatives. Sci. Hort. 2015, 193, 353–358. [Google Scholar] [CrossRef]
- Plazas, M.; Vilanova, S.; Gramazio, P.; Rodríguez-Burruezo, A.; Fita, A.; Herraiz, F.J.; Ranil, R.; Fonseka, R.; Niran, L.; Fonseka, H.; et al. Interspecific hybridization between eggplant and wild relatives from different genepools. J. Am. Soc. Hortic. Sci. 2016, 141, 34–44. [Google Scholar] [CrossRef]
- Behera, T.K.; Singh, N. Inter-specific crosses between eggplant (Solanum melongena L.) with related Solanum species. Sci. Hortic. 2002, 95, 165–172. [Google Scholar] [CrossRef]
- Afful, N.T.; Nyadanu, D.; Akromah, A.H.M.; Annor, C.; Diawouh, R.G. Evaluation of crossability studies between selected eggplant accessions with wild relatives S. torvum, S. anguivi and S. aethopicum (shum group). J. Plant Breed. Crop. Sci. 2018, 10, 1–12. [Google Scholar] [CrossRef]
- Kumchai, J.; Wei, Y.C.; Lee, C.Y.; Chen, F.C.; Chin, S.W. Production of interspecific hybrids between commercial cultivars of the eggplant (Solanum melongena L.) and its wild relative S. torvum. Genet. Mol. Res. 2013, 12, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.N.; Rao, S.K. Chromosome association and pollen fertility in Solanum melongena and S. surattense hybrids. Cytologia 1986, 51, 85–93. [Google Scholar] [CrossRef]
- Nwofia, G.E.; Eneoblong, E.E. Barriers to interspecific hybridization among non-tuberous solanum species. J. Appl. Chem. Agric. Res. 2001, 7, 37–41. [Google Scholar] [CrossRef]
- Vorontsova, M.S.; Stern, S.S.; Bohs, L.; Knapp, S. African spiny Solanum (subgenus Leptostemonum, Solanaceae): A thorny phylogenetic tangle. Bot. J. Linn. Soc. 2013, 173, 176–193. [Google Scholar] [CrossRef]
- García-Fortea, E.; Gramazio, P.; Vilanova, S.; Fita, A.; Mangino, G.; Villanueva, G.; Arrones, A.; Knapp, S.; Prohens, J.; Plazas, M. First successful backcrossing towards eggplant (S. melongena) of a New World species, the silverleaf nightshade (S. elaeagnifolium), and characterization of interspecific hybrids and backcrosses. Sci. Hort. 2019, 246, 253–263. [Google Scholar] [CrossRef]
- Anis, M.; Baksh, S.; Iqbal, M. Cytogenetic studies on F1 hybrid S. incanum X S. melongena var. Am. Wonder. Cytologia 1994, 59, 433–436. [Google Scholar] [CrossRef][Green Version]
- Lester, R.N.; Kang, J.H. Embryo and endosperm function and failure in Solanum species and hybrids. Ann. Bot. 1998, 82, 445–453. [Google Scholar] [CrossRef]
- Narayanaswami, S.; Norstog, K. Plant embryo culture. Bot. Rev. 1964, 30, 587. [Google Scholar] [CrossRef]
- Verba, V.M.; Mamedov, M.I.; Pyshnaya, O.N.; Suprunova, T.N.; Shmykova, N.A. Isolation of eggplant interspecific hybrids by the method of embryo culture. Agric. Biol. 2010, 5, 66–71. [Google Scholar]
- Sharma, D.R.; Ssareen, P.K.; Chowdhary, J.B. Cross-ability and pollination in some non-tuberous Solanum species. Indian J. Agric. Sci. 1984, 59, 514–516. [Google Scholar]
- Nitsch, J.P.; Nitsch, C. Haploid plants from pollen grains. Science 1969, 163, 85–87. [Google Scholar] [CrossRef]
- Sharma, D.R.; Chawdhury, J.B.; Ahuja, U.; Dhankhar, B.S. Interspecific hybridization in genus Solanum. A cross between S. melongena and S. khasianum through embryo culture. Z. Pflanzenzucht. 1980, 85, 248–253. [Google Scholar]
- Blestsos, F.A.; Roupakias, D.G.; Tsaktsira, M.L.; Scaltsojannes, A.B.; Thanassoulopoulos, C.C. Interspecific hybrids between three eggplant (Solanum melongena L.) cultivars and two wild species (Solanum torvum Sw. and Solanum sisymbriifolium Lam.). Plant. Breed. 1998, 117, 159–164. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Ano, G.; Hebert, Y.; Prior, P.; Messiaen, C.M. A new source of resistance to bacterial wilt of eggplants obtained from a cross: Solanum aethiopicum L X Solanum melongena L. Agronomie 1991, 11, 555–560. [Google Scholar] [CrossRef]
- Kaushik, P.; Prohens, J.; Vilanova, S.; Gramazio, P.; Plazas, M. Phenotyping of eggplant wild relatives and interspecific hybrids with conventional descriptors provides insight for their potential utilization in breeding. Front Plant. Sci. 2016, 7, 677. [Google Scholar] [CrossRef] [PubMed]
- Fulton, T.M.; Chunwongse, J.; Tanksley, S.D. Microprep protocol for extraction of DNA from tomato and other herbaceous plants. Plant Mol. Biol. Rep. 1995, 13, 207–209. [Google Scholar] [CrossRef]
- Namisy, A.; Chen, J.R.; Prohens, J.; Metwally, E.; Elmahrouk, M.; Rakha, M. Screening cultivated eggplant and wild relatives for resistance to bacterial wilt (Ralstonia solanacearum). Agriculture 2019, 9, 157. [Google Scholar] [CrossRef]
- Buddenhagen, I.W. Designations of races in Pseudomonas solanacearum. Phytopathology 1962, 52, 726. [Google Scholar]
- He, L.Y.; Sequeira, L.; Kelman, A. Characteristics of strains of Pseudomonas solanacearum from China. Plant. Dis. 1983, 67, 1357–1361. [Google Scholar] [CrossRef]
- Cook, D.; Barlow, E.; Sequeira, L. Genetic diversity of Pseudomonas solanacearum: Detection of restriction fragment polymorphisms with DNA probes that specify virulence and hypersensitive response. Mol. Plant. Microbe Interact. 1989, 2, 113–121. [Google Scholar] [CrossRef]
- Kelman, A. The relationship of pathogenicity in Pseudomonas solanacearum to colony appearance on tetrazolium medium. Phytopathology 1954, 44, 693–695. [Google Scholar]
- Kado, C.I.; Heskett, M. Selective media for isolation of Agrobacterium, Corynebacterium, Erwinia, Pseudomonas, and Xanthomonas. Phytopathology 1970, 60, 969–976. [Google Scholar] [CrossRef]
- Hanson, P.M.; Licardo, O.; Hanudin; Wang, J.F.; Chen, J.T. Diallel analysis of bacterial wilt resistance in tomato derived from different sources. Plant. Dis. 1998, 82, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Winstead, N.N.; Kelman, A. Inoculation techniques for evaluating resistance to Pseudomonas solanacearum. Phytopathology 1952, 42, 623–634. [Google Scholar]
- Aslam, M.N.; Hussain, M.A.; Raheel, M. Assessment of resistance to bacterial wilt incited by Ralstonia solanacearum in tomato germplasm. J. Plant. Dis. Prot. 2017, 124, 585–590. [Google Scholar] [CrossRef]
- Ruiz, J.M.; Belakbir, L.; Ragala, J.M.; Romero, L. Response of plant yield and leaf pigments to saline conditions: Effectiveness of different rootstocks in melon plants (Cucumis melo L.). Soil Sci. Plant. Nutr. 1997, 43, 855–862. [Google Scholar]
- Hassell, R.; Daley, S. Fatty alcohol rootstock treatment: An overview of a new approach to cucurbit grafting. Acta Hort. 2015, 1086, 71–77. [Google Scholar] [CrossRef]
- Rivard, C.L.; O’Connell, S.; Peet, M.M.; Louws, F.J. Grafting tomato with interspecific rootstock to manage diseases caused by Sclerotium rolfsii and southern root-knot nematode. Plant. Dis. 2010, 94, 1015–1021. [Google Scholar] [CrossRef]
- King, S.R.; Davis, A.R.; Zhang, X.; Crosby, K. Genetics, breeding and selection of rootstocks for Solanaceae and Cucurbitaceae. Sci. Hort. 2010, 127, 106–111. [Google Scholar] [CrossRef]
- Kubota, C.; McClure, M.A.; Kokalis-Burelle, N.; Bausher, M.G.; Rosskopf, E.N. Vegetable grafting: History, use, and current technology status in North America. Hort Sci. 2008, 43, 1664–1669. [Google Scholar] [CrossRef]
- Villeneuve, F.; Latour, F.; Théry, T.; Erard, P.; Fournier, C.; Daunay, M.C. Screening of solanaceous wild relatives for graft affinity with eggplant (Solanum melongena L). In Proceedings of the XVIth EUCARPIA Meeting on Capsicum and Eggplant, Budapest, Hungary, 12–14 September 2016; Ertsey-Peregi, K., Füstös, Z., Palotas, G., Csilléry, G., Eds.; Diamond Congress Ltd.: Budapest, Hungary, 2016; pp. 152–160. [Google Scholar]

| Wild Relative Parents | Number of Crosses | Fruit Set (%) | Seeds/Fruit Mean (g) | Germination (%) a |
|---|---|---|---|---|
| S. melongena (male parent) | ||||
| S. incanum UPV1 | 32 | 68.8 | 0.03 | 0 |
| S. insanum UPV2 | 40 | 40.0 | 0.85 | 90 |
| S. anguivi UPV3 | 52 | 46.2 | 0.30 | 88 |
| S. sisymbrifolium UPV4 | 66 | 0.00 | ND | ND |
| S. melongena (female parent) | ||||
| S. incanum UPV1 | 46 | 65.2 | 1.58 | 0 |
| S. insanum UPV2 | 40 | 0.0 | ND | ND |
| S. anguivi UPV3 | 50 | 0.0 | ND | ND |
| S. sisymbrifolium UPV4 | 52 | 0.0 | ND | ND |
| Medium a | Globular Stage | Heart Stage | Torpedo Stage | Abnormal Plants | ||||
|---|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | N | % | |
| S. melongena EG203 × S. incanum UPV1 | ||||||||
| M1 | 30 | 0.0 | 25 | 32.0 | 15 | 86.7 | 0 | 0.0 |
| M2 | 30 | 0.0 | 30 | 43.3 | 35 | 88.6 | 1 | 0.2 |
| M3 | 30 | 0.0 | 30 | 56.7 | 38 | 78.9 | 0 | 0.0 |
| S. incanum UPV1 × S. melongena EG203 | ||||||||
| M1 | 30 | 0.0 | 24 | 8.3 | 39 | 20.5 | 0 | 0.0 |
| M2 | 30 | 0.0 | 24 | 29.2 | 49 | 32.7 | 3 | 13.0 |
| M3 | 30 | 0.0 | 24 | 16.7 | 52 | 30.8 | 0 | 0.0 |
| Genotype | Allele Size | ||
|---|---|---|---|
| smSSR01 | EPSSR04 | EPSSR133 | |
| S. melongena EG203 | 300 | 320 | 220 |
| S. incanum UPV1 | 270 | 320 | 220 |
| S. insanum UPV2 | 270 | 300 | 240 |
| S. anguivi UPV3 | 290 | 300 | 220 |
| S. melongena EG203 × S. incanum UPV1 | 270, 300 | 320 | 220 |
| S. incanum UPV1 × S. melongena EG203 | 270, 300 | 320 | 220 |
| S. melongena EG203 × S. insanum UPV2 | 270, 300 | 300, 320 | 220, 240 |
| S. melongena EG203 × S. anguivi UPV3 | 290, 300 | 300, 320 | 220 |
| Genotype | Strain Pss97 a | Strain Pss2016 a | ||||||
|---|---|---|---|---|---|---|---|---|
| N | W% | DI% | Reaction | N | W% | DI% | Reaction | |
| S. melongena EG048 | 9 | 100.0 | 100.0 | S | 9 | 100.0 | 77.8 | S |
| S. melongena EG203 | 9 | 11.1 | 8.9 | R | 9 | 33.3 | 20.0 | R |
| S. incanum UPV1 | 24 | 100.0 | 100.0 | S | 24 | 66.7 | 23.3 | R |
| S. insanum UPV2 | 24 | 87.5 | 85.0 | S | 24 | 100.0 | 96.7 | S |
| S. anguivi UPV3 | 24 | 100.0 | 100.0 | S | 24 | 95.8 | 45.8 | MR |
| S. melongena EG203 × S. incanum UPV1 | 12 | 83.3 | 56.7 | S | 16 | 94.4 | 56.4 | S |
| S. incanum × S. melongena EG203 | 19 | 90.5 | 67.6 | S | 19 | 95.2 | 58.1 | S |
| S. insanum UPV2 × S. melongena EG203 | 12 | 100.0 | 98.0 | S | 12 | 100.0 | 95.0 | S |
| S. anguivi UPV3 × S. melongena EG203 | 12 | 100.0 | 90.0 | S | 12 | 100.0 | 85.0 | S |
| Species | Accession Code | Country of Origin | Gene Pool |
|---|---|---|---|
| Solanum melongena | EG203 | India | Cultivated |
| S. incanum | UPV1 | Israel | Secondary |
| S. insanum | UPV2 | Sri Lanka | Primary |
| S. anguivi | UPV3 | Ivory Coast | Secondary |
| S. sisymbriifolium | UPV4 | Unknown | Tertiary |
| Marker | Forward Primer | Reverse Primer | Primer Melting Temperature (°C) | Band Size (bp) | Repeat Motif |
|---|---|---|---|---|---|
| smSSR01 | GTGACTACGGTTTCACTGGT | GATGACGACGACGATAATAGA | 50 | 310 | (ATT)21 |
| EPSSR04 | AATGAGTCAGAAACCACGCC | CGTTTAACCTTTGGCTCGAA | 55 | 147 | (CA)10.5 |
| EPSSR133 | AGTGGTAACGTCTGCTTACATTT | AGTTTGAATTCCATGGCTCG | 55 | 230 | (TGT)9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rakha, M.; Namisy, A.; Chen, J.-R.; El-Mahrouk, M.E.; Metwally, E.; Taha, N.; Prohens, J.; Plazas, M.; Taher, D. Development of Interspecific Hybrids between a Cultivated Eggplant Resistant to Bacterial Wilt (Ralstonia solanacearum) and Eggplant Wild Relatives for the Development of Rootstocks. Plants 2020, 9, 1405. https://doi.org/10.3390/plants9101405
Rakha M, Namisy A, Chen J-R, El-Mahrouk ME, Metwally E, Taha N, Prohens J, Plazas M, Taher D. Development of Interspecific Hybrids between a Cultivated Eggplant Resistant to Bacterial Wilt (Ralstonia solanacearum) and Eggplant Wild Relatives for the Development of Rootstocks. Plants. 2020; 9(10):1405. https://doi.org/10.3390/plants9101405
Chicago/Turabian StyleRakha, Mohamed, Ahmed Namisy, Jaw-Rong Chen, Mohammed E. El-Mahrouk, Elmahdy Metwally, Naglaa Taha, Jaime Prohens, Mariola Plazas, and Dalia Taher. 2020. "Development of Interspecific Hybrids between a Cultivated Eggplant Resistant to Bacterial Wilt (Ralstonia solanacearum) and Eggplant Wild Relatives for the Development of Rootstocks" Plants 9, no. 10: 1405. https://doi.org/10.3390/plants9101405
APA StyleRakha, M., Namisy, A., Chen, J.-R., El-Mahrouk, M. E., Metwally, E., Taha, N., Prohens, J., Plazas, M., & Taher, D. (2020). Development of Interspecific Hybrids between a Cultivated Eggplant Resistant to Bacterial Wilt (Ralstonia solanacearum) and Eggplant Wild Relatives for the Development of Rootstocks. Plants, 9(10), 1405. https://doi.org/10.3390/plants9101405







