Achene Morphology and Anatomy of Clematis L. (Ranunculaceae) in Korea and Its Taxonomic Implications
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimens
2.2. Light Microscopy
2.3. Scanning Electron Microscopy
2.4. Morphometry and Data Analysis
3. Results
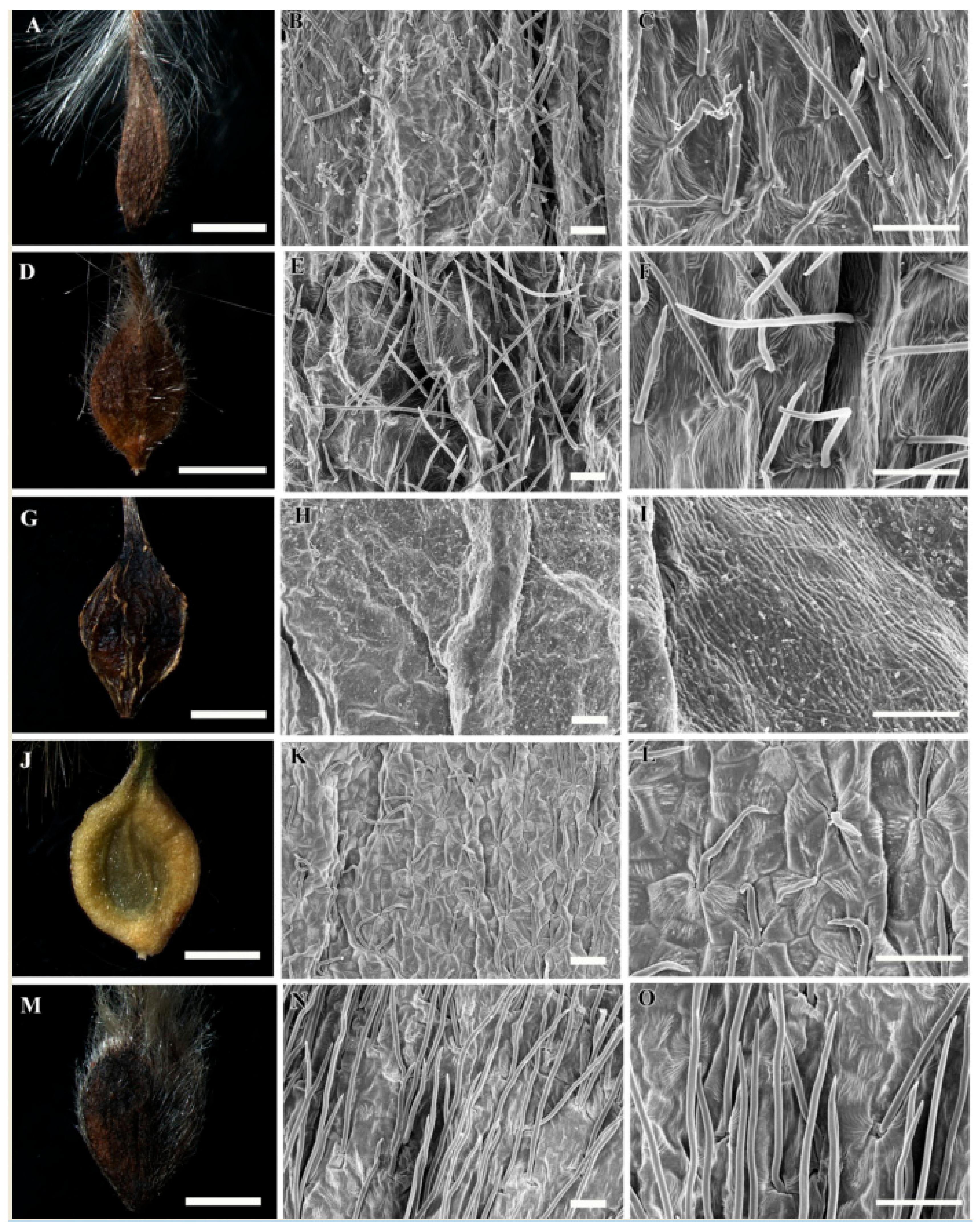
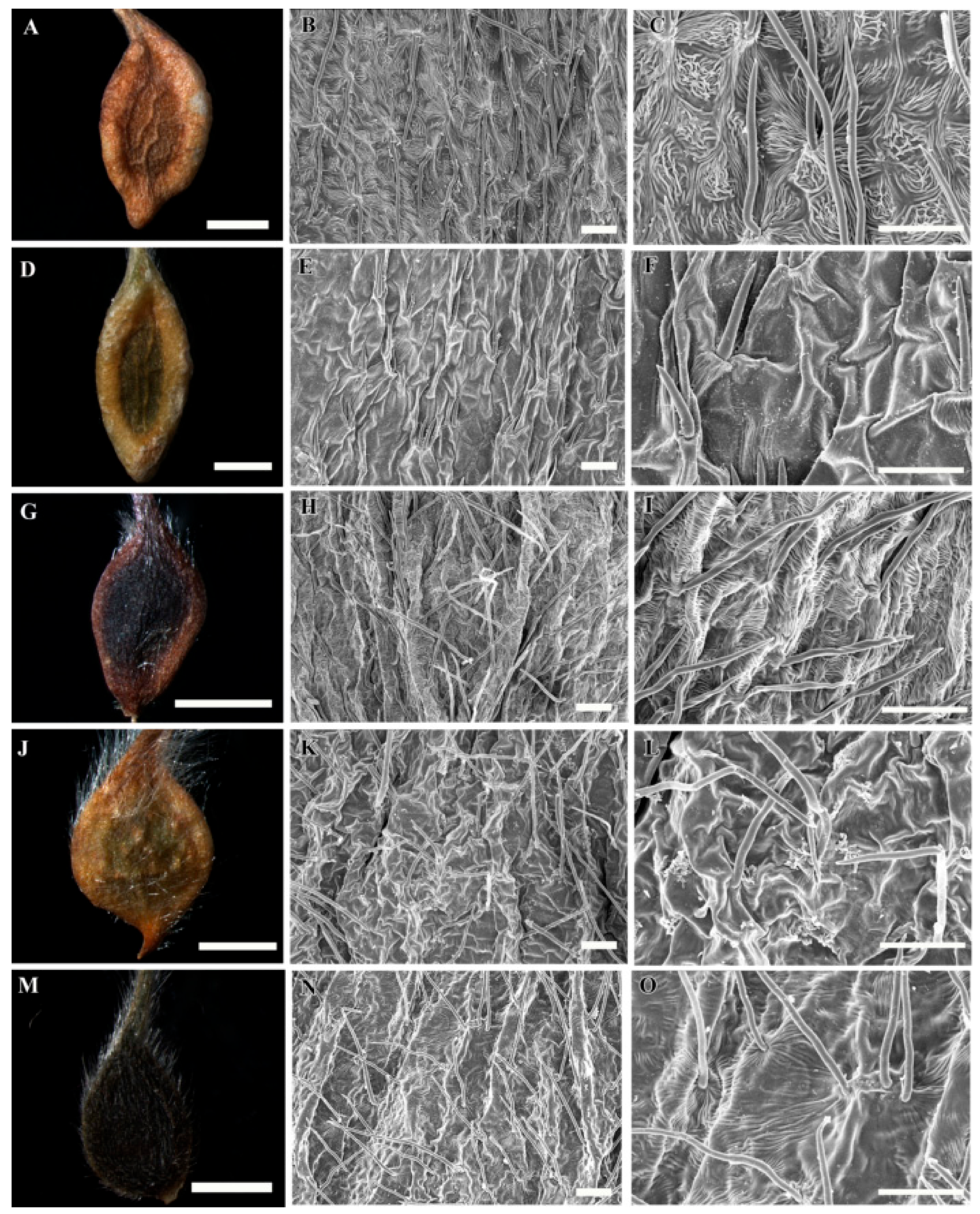
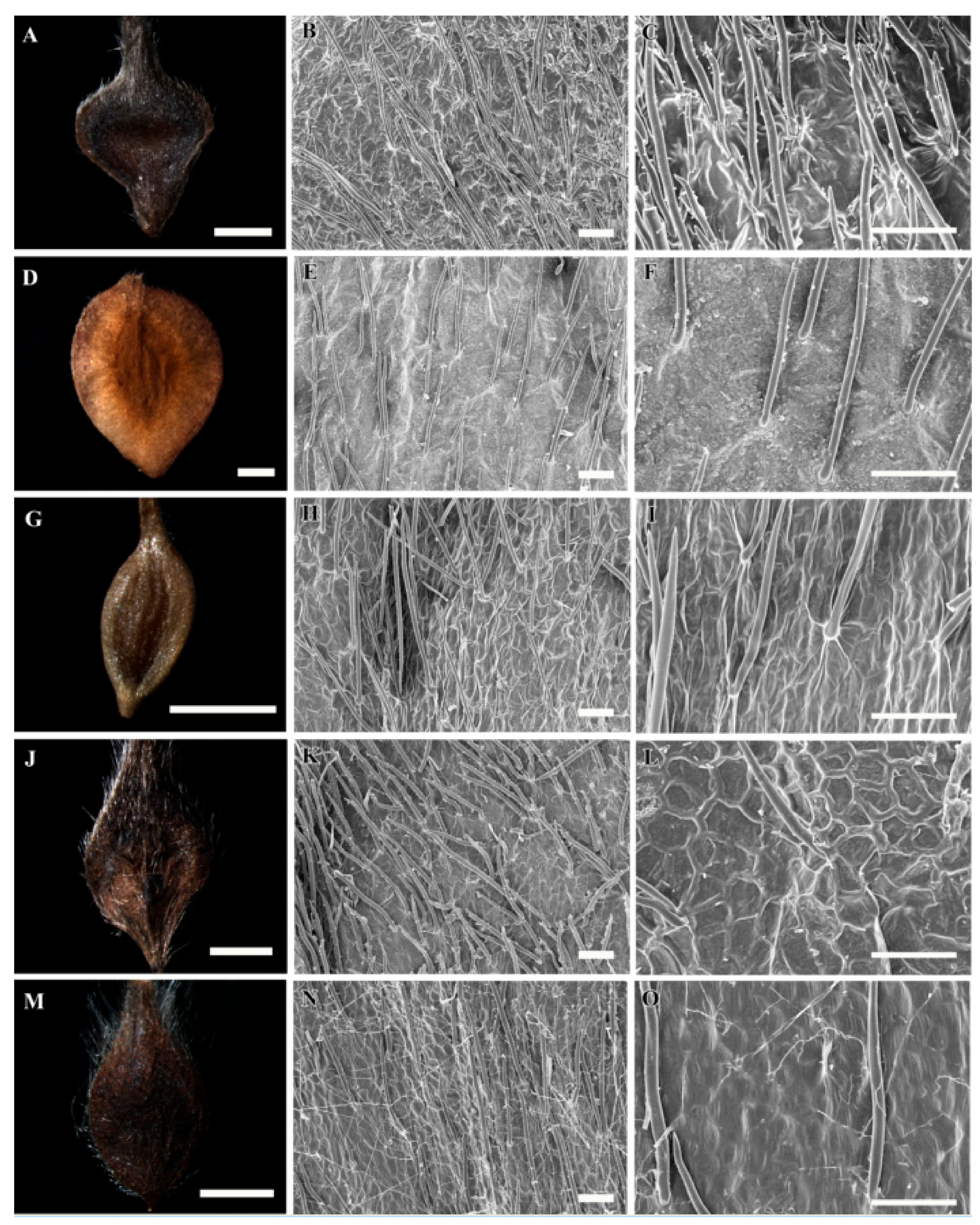
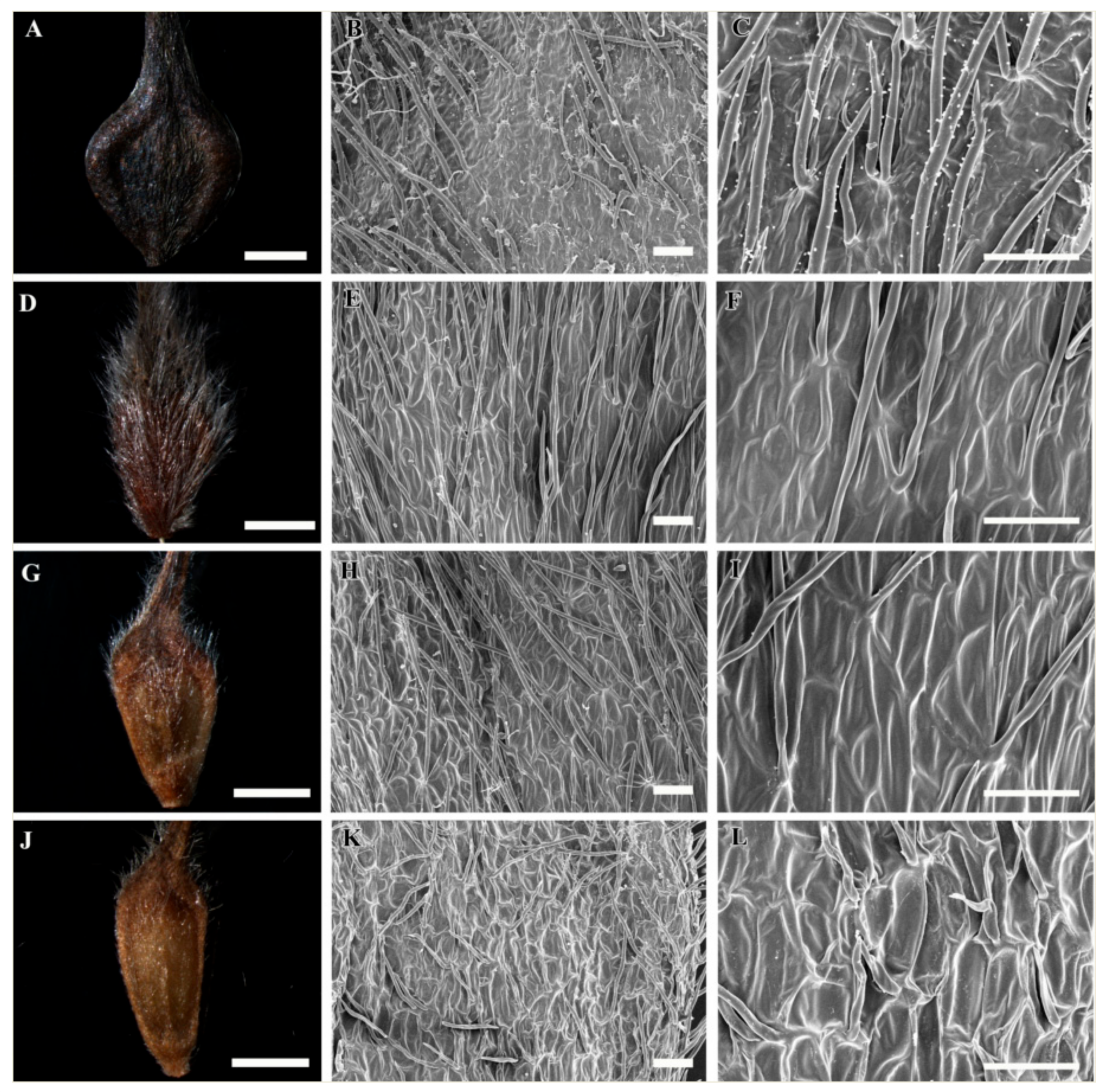
3.1. Gross Achene Morphology
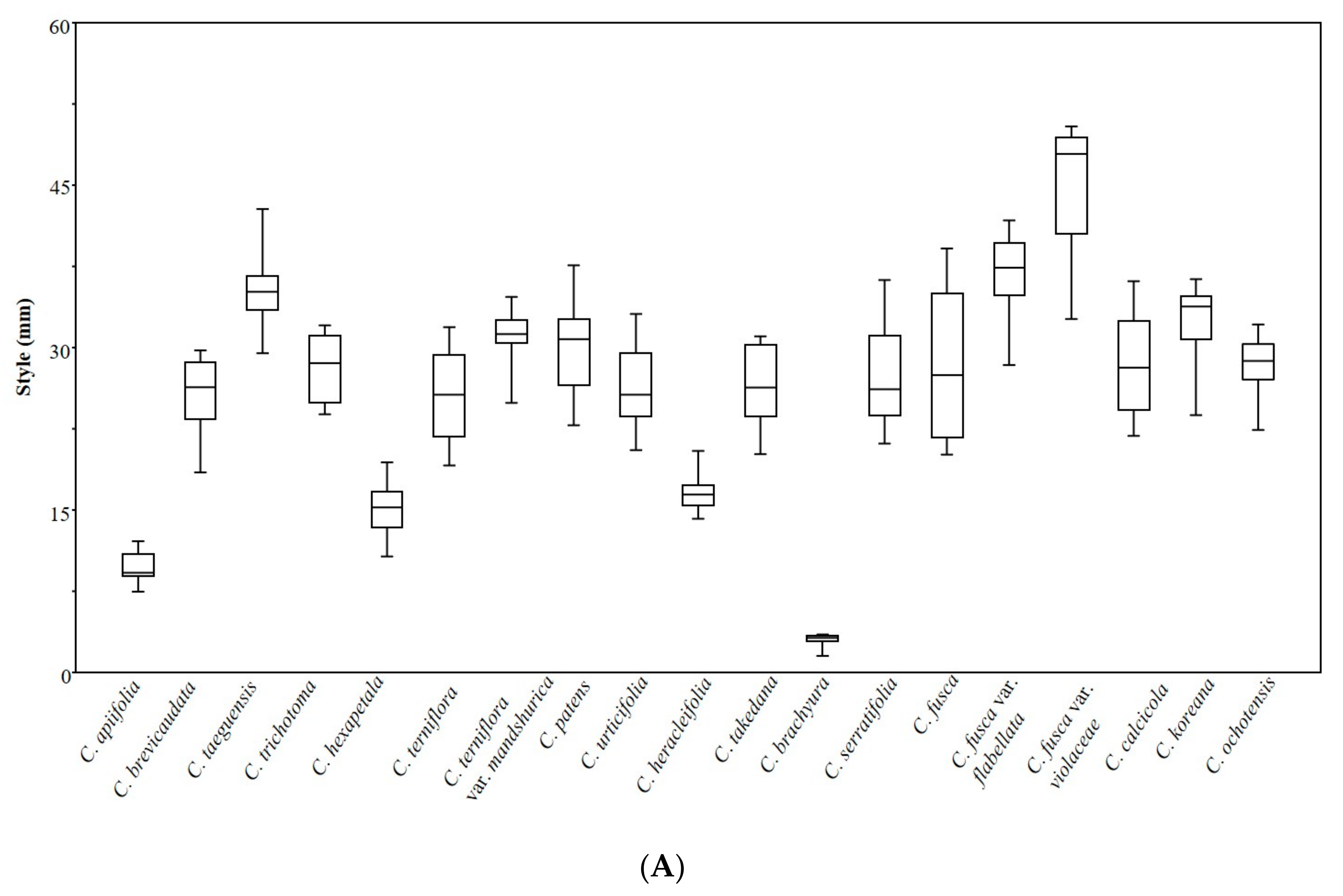
3.2. Achene Indumentum and Style
3.3. Achene Surface Sculpture
3.4. Shape of Epidermal Cells
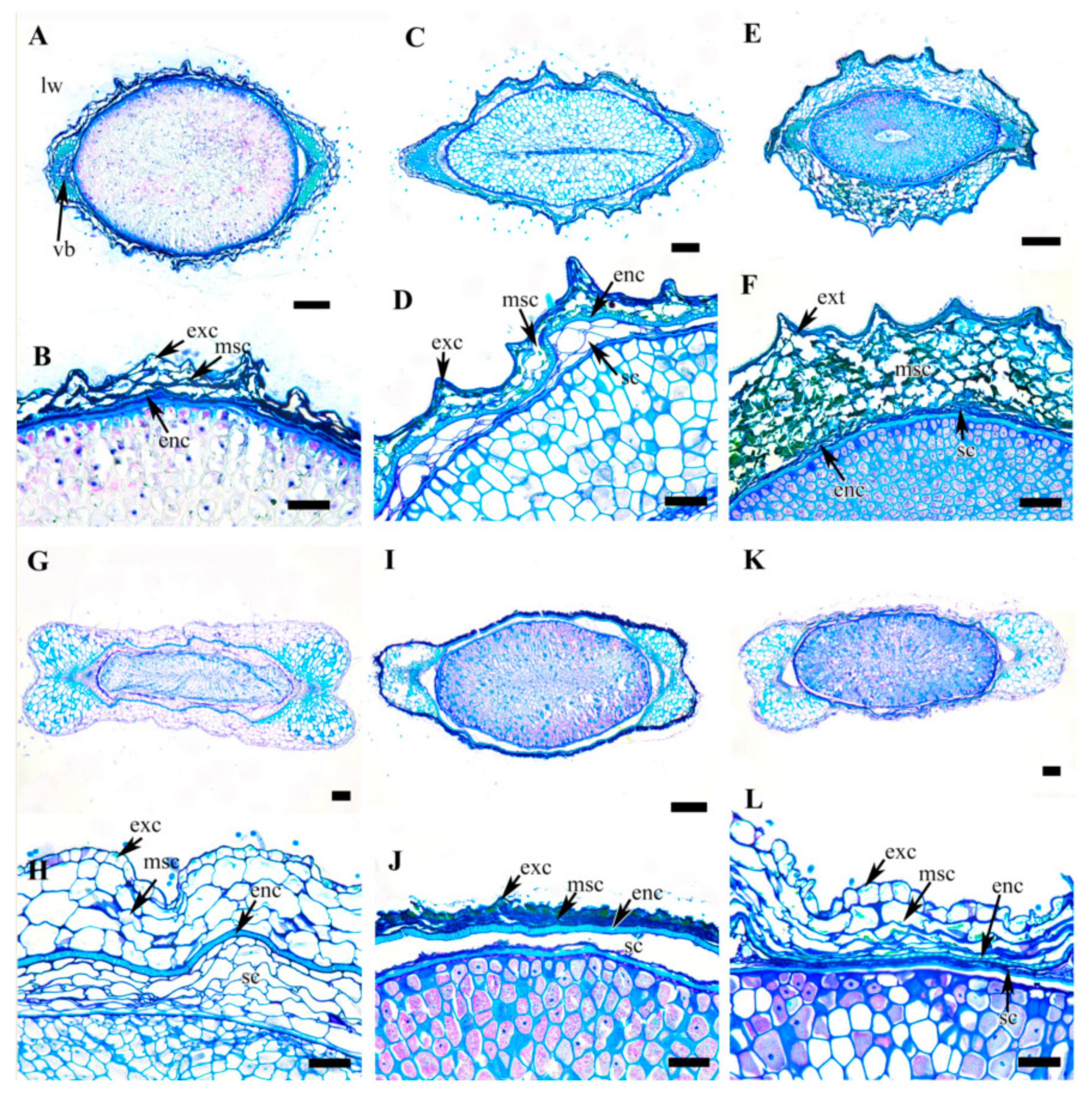
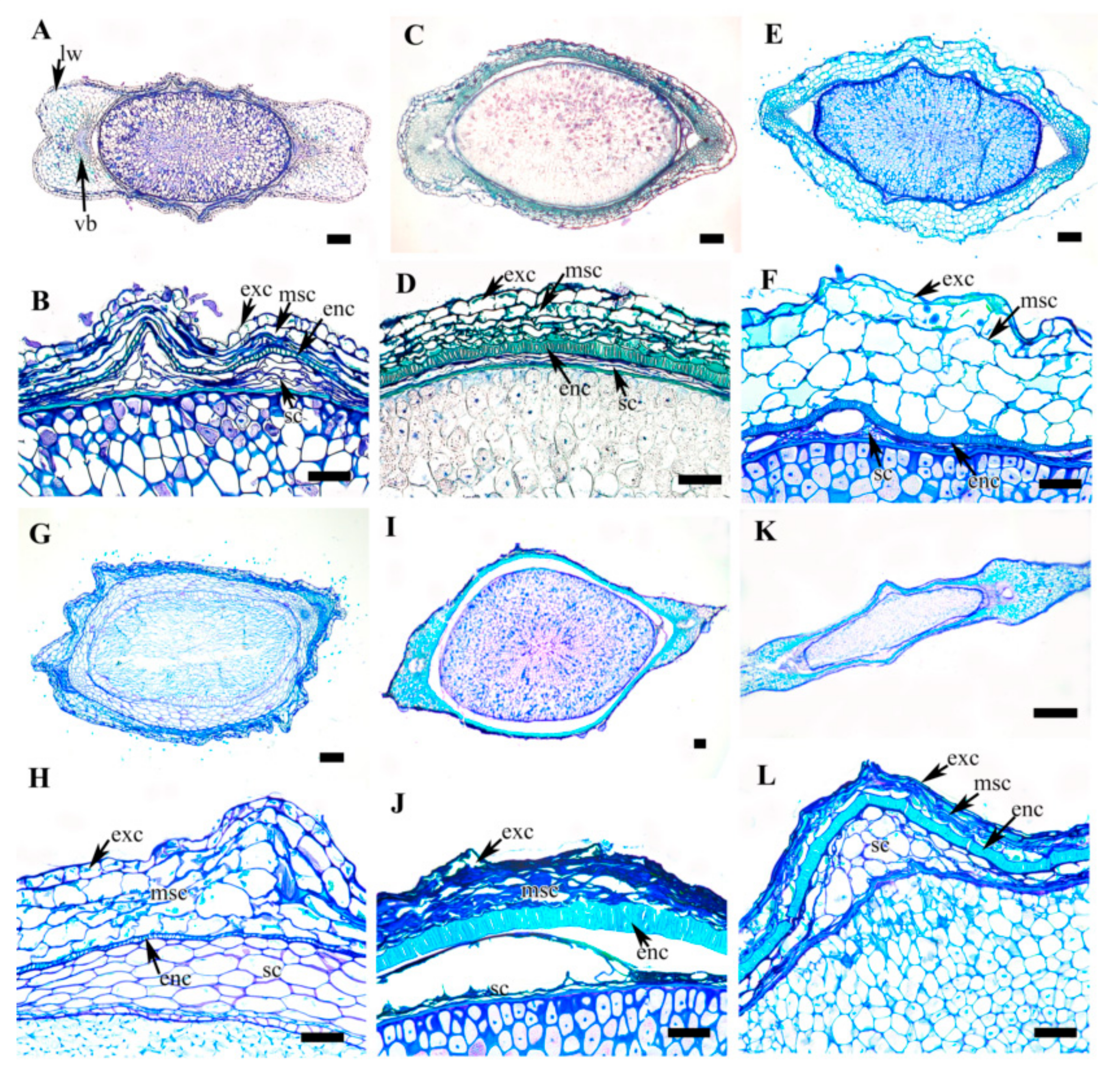
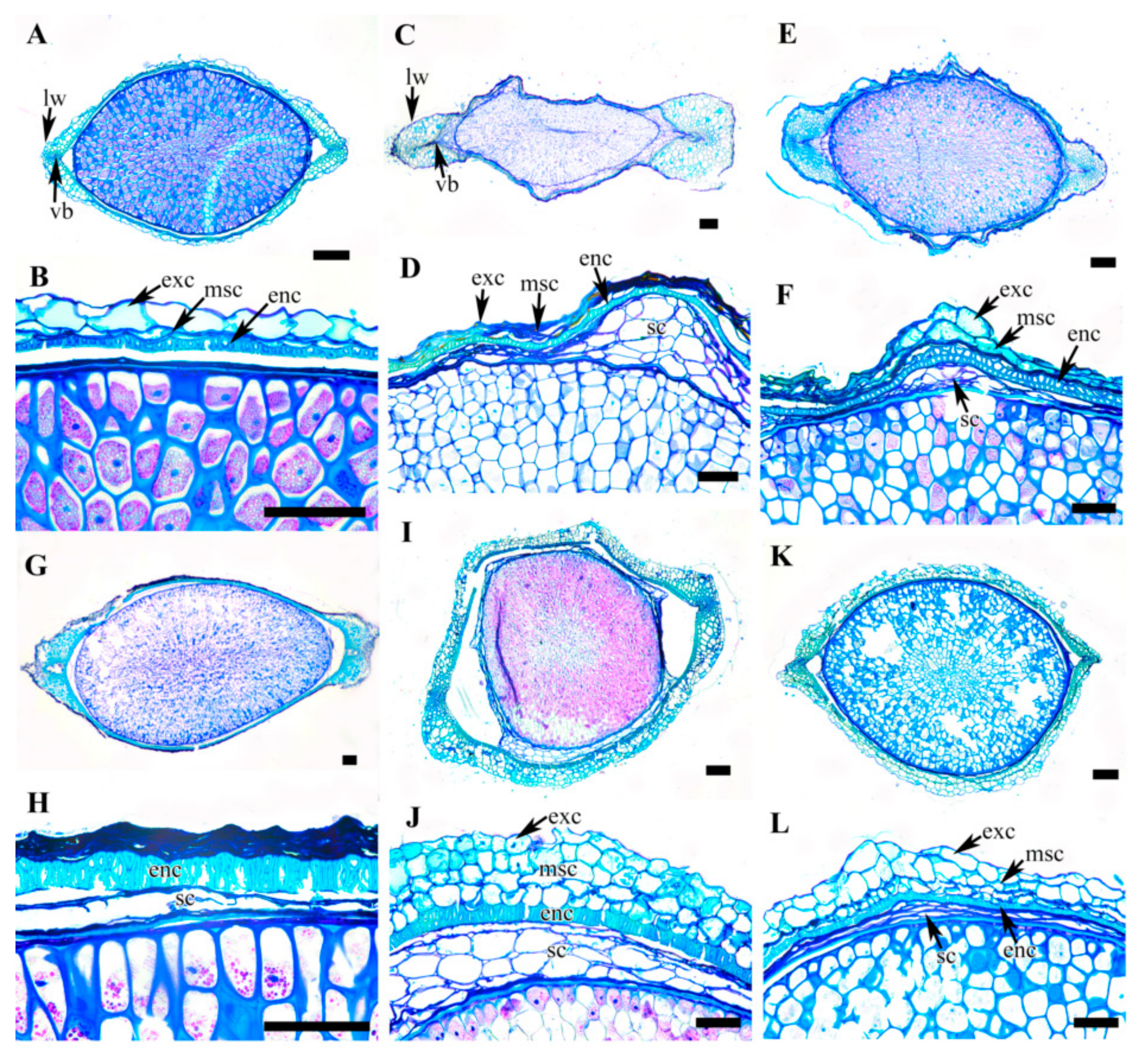
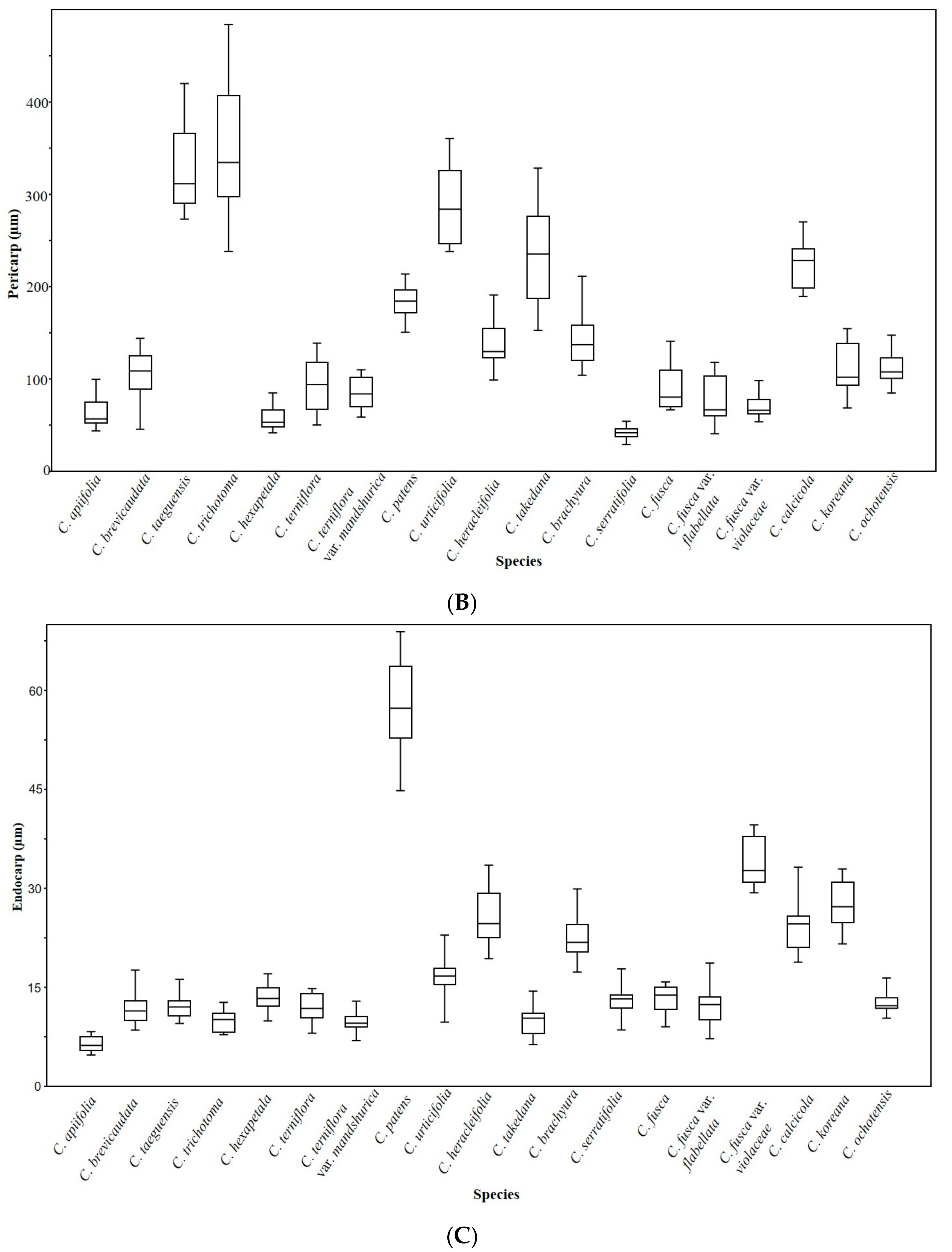
3.5. Achene Anatomy
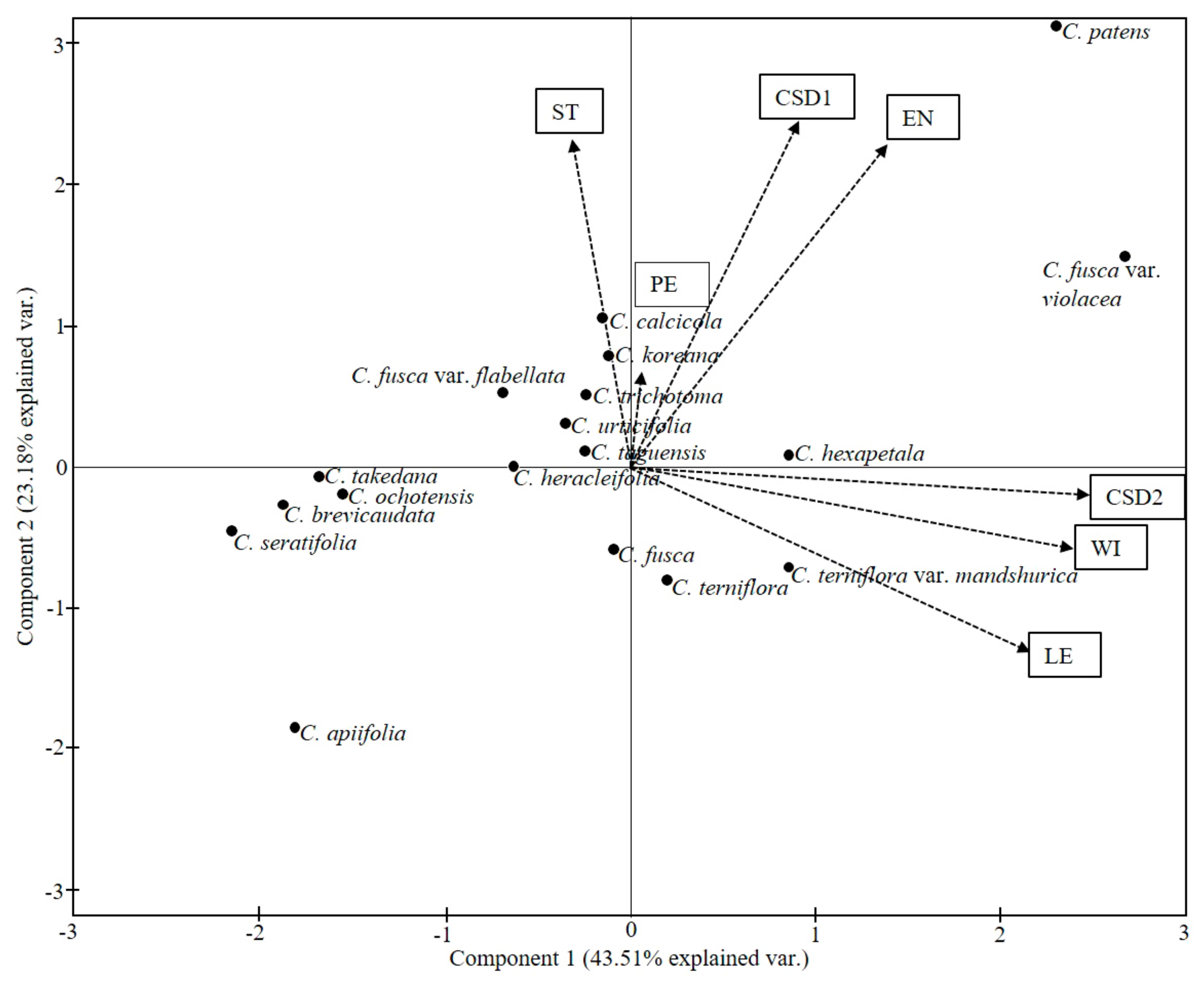
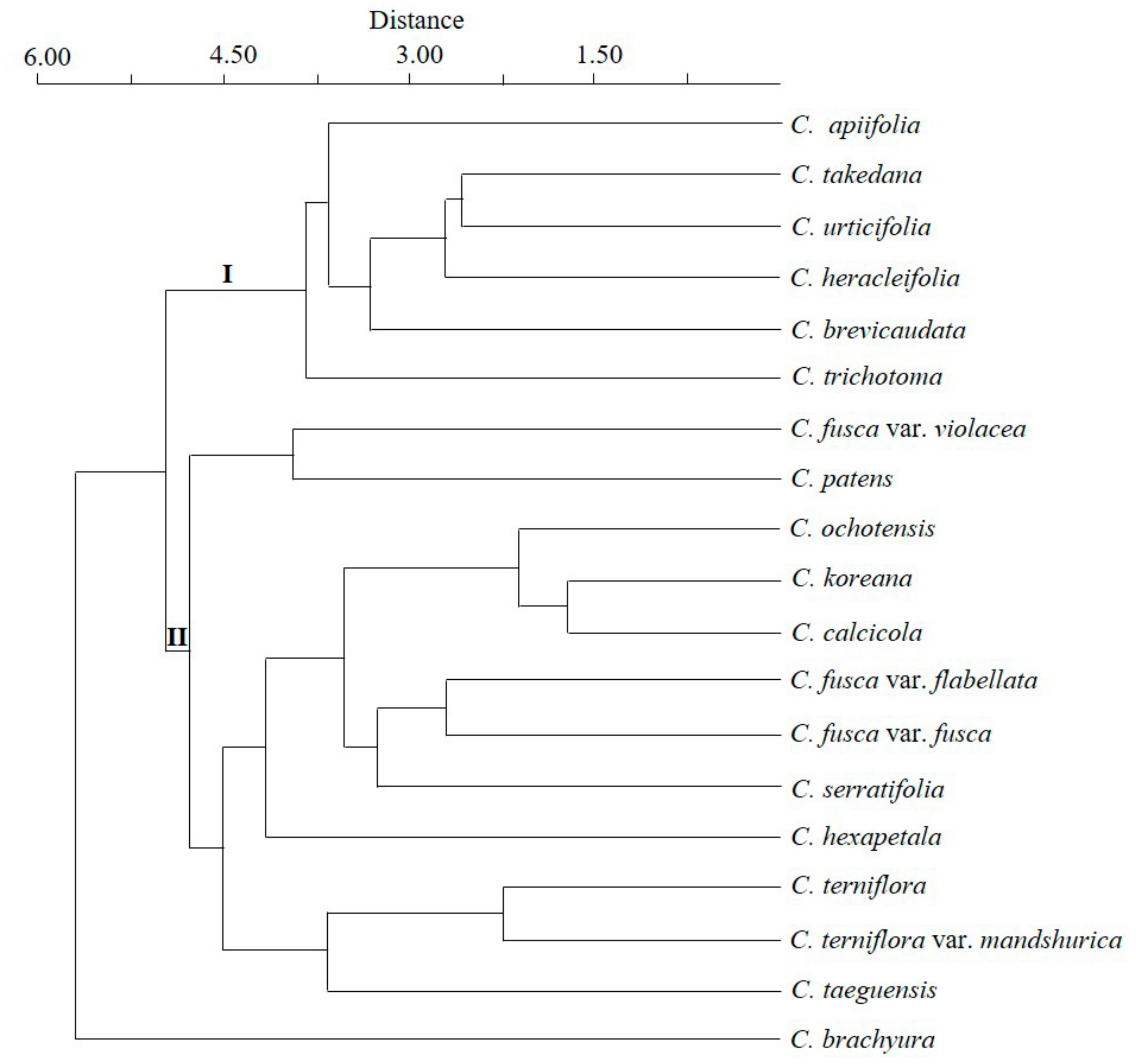
3.6. Statistical Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tamura, M.A. Classification of genus Clematis. Acta Phytotaxon. Geobot. 1987, 38, 33–44. [Google Scholar]
- Tamura, M. Angiospermae: Ordnung Ranunculales. Fam. Ranunculaceae. In Die Natürlichen Pflanzenfamilien; Hiepko, P., Ed.; Duncker and Humblot: Berlin, Germany, 1995; Volume 17, pp. 324–349. [Google Scholar]
- Wang, W.T.; Li, L.Q. A new system of classification of the genus Clematis (Ranunculaceae). Acta Phytotaxon. Sin. 2005, 43, 431–488. [Google Scholar]
- Grey-Wilson, C. Clematis the Genus: A Comprehensive Guide for Gardeners, Horticulturists and Botanists; Timber Press: Portland, OR, USA, 2000. [Google Scholar]
- Johnson, M. The Genus Clematis; Magnus Johnson Plantskola: Stockholm, Sweden, 2001. [Google Scholar]
- Wang, W.T.; Bartholomew, B. Clematis . In Flora of China; Wu, Z.Y., Raven, P., Eds.; Science Press: Beijing, China; Missouri Botanical Garden Press: St. Louis, MO, USA, 2001; Volume 6. [Google Scholar]
- Ellitto, A.C. Systematics of Clematis in Nepal, the Evolution of Tribe Anemoneae DC. (Ranunculaceae) and Phylogeography and Dynamics of Speciation in the Himalaya. Ph.D. Thesis, University of Edinburgh, Edinburgh, UK, 2016. [Google Scholar]
- Prantl, K. Beitrage zur Morphologie und Systematik der Ranunculaceen. Bot. Jahrb. Syst. Pflanzengesch. Pflanzengeogr. 1888, 9, 225–273. [Google Scholar]
- Johnson, M. Slaktet Klematis; Magnus Johnson Plantskola: Stockholm, Sweden, 1997. [Google Scholar]
- Lee, Y.N. New Flora of Korea; Kyo-Hak Publishing Co. Ltd.: Seoul, Korea, 2007; Volume I. [Google Scholar]
- Chang, C.S.; Kim, H.; Chang, K.S. Illustrated Encyclopedia of Fauna and Flora of Korea. Woody Plants; Ministry of Education Science and Technology: Seoul, Korea, 2011; Volume 43, 511p. [Google Scholar]
- Chang, K.S.; Son, D.C.; Lee, D.-H.; Choi, K.; Oh, S.H. Checklist of Vascular Plants in Korea; Korea National Arboretum: Pocheon-Si, Korea, 2017; 1000p. [Google Scholar]
- Kim, J.S. Clematis, L. In Flora of Korea. Magnoliidae; Flora of Korea Editorial Committee and the National Institute of Biological: Gyeongsangbuk-do, Korea, 2017; Volume 2a, pp. 69–76. [Google Scholar]
- Tamura, M. Systema clematidis asiae orientalis. Sci. Rep. 1955, 4, 43–55. [Google Scholar]
- Tamura, M. Ranunculaceae. In The Families and Genera of Vascular Plants; Kubitzki, K., Rohwer, J.G., Bittrich, V., Eds.; Springer: Berlin/Heidelberg, Germany, 1993; Volume 2, pp. 563–583. [Google Scholar]
- Tobe, H. Morphological studies on the genus Clematis Linn. I. Pollen grains. Sci. Rep. Tohoku Univ. Fourth Ser. Biol. 1974, 37, 47–53. [Google Scholar]
- Tobe, H. Morphological studies on the genus Clematis Linn. V. vascular anatomy of the calyx region in four-sepaled flowers. Bot. Mag. Tokyo 1980, 93, 39–54. [Google Scholar] [CrossRef]
- Tobe, H. Morphological studies on the genus Clematis Linn. VI. Vascular anatomy of the androecial and gynoecial regions of the floral receptacle. Bot. Mag. Tokyo. 1980, 93, 125–133. [Google Scholar] [CrossRef]
- Tobe, H. Morphological studies on the genus Clematis Linn. VII. Reinvestigation of Clematis williamsii A. Gray and proposal of its taxonomic transfer to Clematopsis. Bot. Mag. Tokyo. 1980, 93, 135–148. [Google Scholar] [CrossRef]
- Essig, F. Seedling morphology in Clematis (Ranunculaceae) and its taxonomic implications. Sida 1991, 14, 377–390. [Google Scholar]
- Zhang, Y.L. Chromosome studies on 7 species of Clematis in China. J. Wuhan Bot. Res. 1991, 9, 107–113. [Google Scholar]
- Snoeijer, W. A suggested classification for the genus Clematis. Clematis 1992, 7–20. [Google Scholar]
- Yano, Y. Pollen grain morphology in Clematis (Ranunculaceae). Clematis 1993, 42–43. [Google Scholar]
- Yang, T.Y.; Moore, D.M. A revision of the Viorna group of species (section Viorna sensu Prantl) in the genus Clematis (Ranunculaceae). Syst. Geogr. Plant. 1999, 68, 281–303. [Google Scholar] [CrossRef]
- Shi, J.H.; Li, L.Q. Leaf epidermal feature in Clematis (Ranunculaceae) with reference to its systematic significance. Acta Bot. Sin. 2003, 45, 257–268. [Google Scholar]
- Miikeda, O.; Koga, S.; Handa, T.; Yukawa, T. Subgeneric relationships in Clematis (Ranunculaceae) by DNA sequences. In Taxonomy of Cultivated Plants: Third International Symposium; Andrews, S., Leslie, A., Alexander, C., Eds.; Royal Botanic Gardens Kew: London, UK, 1999; pp. 355–358. [Google Scholar]
- Miikeda, O.; Kita, K.; Handa, T.; Yukawa, T. Phylogenetic relationships of Clematis (Ranunculaceae) based on chloroplast and nuclear DNA sequences. Bot. J. Linn. Soc. 2006, 152, 153–168. [Google Scholar] [CrossRef]
- Slomba, J.M.; Garey, J.R.; Essig, F.B. The actin I intron–a phylogenetically informative DNA region in Clematis (Ranunculaceae). Sida 2004, 21, 879–886. [Google Scholar]
- Xie, L.; Wen, J.; Li, L.Q. Phylogenetic analyses of Clematis (Ranunculaceae) based on sequences of nuclear ribosomal ITS and three plastid regions. Syst Bot. 2011, 36, 907–992. [Google Scholar] [CrossRef]
- Jiang, N.; Zhou, Z.; Yang, J.B.; Zhang, S.D.; Guan, K.Y.; Tan, Y.H.; Yu, W.B. Phylogenetic reassessment of tribe Anemoneae (Ranunculaceae): Non-monophyly of Anemone s.l. revealed by plastid datasets. PLoS ONE 2017, 12, e0174792. [Google Scholar] [CrossRef]
- Lehtonen, S.; Christenhusz, M.J.M.; Falck, D. Sensitive phylogenetics of Clematis and its position in Ranunculaceae. Bot. J. Linn. Soc. 2016, 182, 825–867. [Google Scholar] [CrossRef]
- Fukuhara, T. Seed and funicle morphology of Fumariaceae-Fumarioideae: Systematic implications and evolutionary patterns. Int. J. Plant Sci. 1999, 160, 151–180. [Google Scholar] [CrossRef]
- Juan, R.; Pastor, J.; Fernandez, I. SEM and light microscope observations on fruit and seeds in Scrophulariaceae from Southwest Spain and their systematic significance. Ann. Bot. 2000, 86, 323–338. [Google Scholar] [CrossRef]
- Moro, F.V.; Pinto, A.C.R.; Dos Santos, J.M.; Filho, C.F.D. A scanning electron microscopy study of post-seminal development in Angelonia salicarii folia Bonpl. (Scrophulariaceae). Ann. Bot. 2001, 88, 499–506. [Google Scholar] [CrossRef][Green Version]
- Ozcan, T. SEM observations on petals and fruits of some Turkish endemic Bupleurum, L. (Umbelliferae) species. Bot. J. Linn. Soc. 2002, 138, 441–449. [Google Scholar] [CrossRef]
- Xu, F. Sclerotesta morphology and its systematic implications in Magnoliaceous seeds. Bot. J. Linn. Soc. 2003, 142, 407–424. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Yang, D.Z.; Lu, A.M.; Knapp, S. Seed morphology of the tribe Hyoscyameae (Solanaceae). Taxon 2005, 54, 71–83. [Google Scholar] [CrossRef]
- Amini, E.; Zarre, S.; Assadi, M. Seed micro-morphology and its systematic significance in Gypsophila (Caryophyllaceae) and allied genera. Nor. J. Bot. 2011, 29, 660–669. [Google Scholar] [CrossRef]
- Ghimire, B.; Jeong, M.J.; Choi, G.E.; Lee, H.; Suh, G.K.; Heo, K.; Ku, J.J. Seed morphology of the subfamily Helleboroideae (Ranunculaceae) and its systematic implication. Flora 2015, 216, 6–25. [Google Scholar] [CrossRef]
- Ghimire, B.; Jeong, M.J.; Lee, K.M.; Heo, K.; Lee, C.H.; Suh, G.U. Achene morphology of Saussurea species (Asteraceae, Cardueae) in Korea and its systematic implications. Bot. J. Linn. Soc. 2016, 181, 692–710. [Google Scholar] [CrossRef][Green Version]
- Ghimire, B.; Lee, H.; Choi, G.E.; Jeong, M.J.; Lee, C.H.; Suh, G.U.; Heo, K.; Son, S.W. Seed morphology of 12 taxa of the genus Thalictrum, L. (Thalictroideae, Ranunculaceae) and its systematic implication. Phytotaxa 2016, 283, 271–285. [Google Scholar] [CrossRef]
- Cappelletti, E.M.; Poldini, L. Seed morphology in some European aconites (Aconitum, Ranunculaceae). Plant Syst. Evol. 1984, 145, 193–201. [Google Scholar] [CrossRef]
- Chaudhary, R.P.; Trifonova, V.I. Morphology of fruit and comparative anatomy of pericarp and seed coat in the Nepal species of the genus Anemone (Ranunculaceae). Bot. Z. 1988, 73, 803–817. [Google Scholar]
- Constantinidis, T.; Psaras, G.K.; Kamari, G. Seed morphology in relation to infrageneric classification of Consolida (DC.) Gray (Ranunculaceae). Flora 2001, 196, 81–100. [Google Scholar] [CrossRef]
- Dadandi, M.Y.; Kökdil, G.; İlçim, A.; Ozbilgin, B. Seed macro and micro morphology of the selected Nigella (Ranunculaceae) taxa from Turkey and their systematic significance. Biologia 2009, 64, 261–270. [Google Scholar] [CrossRef]
- Heiss, A.G.; Kropf, M.; Sontag, S.; Weber, A. Seed morphology of Nigella s.l. (Ranunculaceae): Identification, diagnostic traits, and their potential phylogenetic relevance. Int. J. Plant Sci. 2011, 172, 267–284. [Google Scholar] [CrossRef][Green Version]
- Ilaraslan, H.; Ilarslan, R.; Blanche, C. Seed morphology of genus Delphinium, L. (Ranunculaceae) in Turkey. Collect. Bot. 1997, 23, 79–95. [Google Scholar] [CrossRef]
- Karcz, J.; Tomzcok, J. Microstructural features of seeds surface in 6 Species of the genus Nigella, L. (Ranunculaceae). Acta Biol. Siles. 1987, 7, 111–125. [Google Scholar]
- Luo, Y.; Zhang, F.M.; Yang, Q.E. Phylogeny of Aconitum subgenus Aconitum (Ranunculaceae) inferred from ITS sequences. Plant Syst. Evol. 2005, 252, 11–25. [Google Scholar] [CrossRef]
- Wang, W.; Lu, A.M.; Ren, Y.; Endress, M.E.; Chen, Z.D. Phylogeny and classification of Ranunculales: Evidence from four molecular loci and morphological data. Perspect. Plant Ecol. Evol. Syst. 2009, 11, 81–110. [Google Scholar] [CrossRef]
- Ziman, S.N.; Bulakh, E.V.; Kadota, Y.; Keener, C.S. Modern view on the taxonomy of the genus Anemone, L. sensu stricto (Ranunculaceae). J. Jap. Bot. 2008, 83, 127–155. [Google Scholar]
- Maciejewska-Rutkowska, I.; Antkowska, W. Taxonomic utility of achene morphology and anatomy in Anemone, L. (Ranunculaceae) species. Acta Biol. Carcov. Bot. 2013, 55, 29–36. [Google Scholar]
- Nakai, T. Synoptical sketch of the Korean Flora. Bull. Natl. Sci. Mus. 1952, 31, 1–152. [Google Scholar]
- Lee, C.B. Illustrated Woody Plants of Korea; Forest Experiment Station: Seoul, Korea, 1967; pp. 1–384. [Google Scholar]
- Moon, A.R.; Han, J.E.; Lee, B.Y.; Park, J.M.; Jang, C. An unrecorded species of genus Clematis (Ranunculaceae) from Korea. J. Asia Pac. Biodivers. 2013, 6, 415–418. [Google Scholar] [CrossRef][Green Version]
- Korea National Arboretum and the Plant Taxonomic Society of Korea. A Synonymic List of Vascular Plants in Korea; Korea National Arboretum: Pocheon, Korea, 2007. (In Korean) [Google Scholar]
- Lee, Y.N. New taxa of Korean flora (4). Korean J. Bot. 1982, 25, 175–180. [Google Scholar]
- World Flora Online (WFO). WFO: Clematis taeguensis Y.N.Lee. 2020. Available online: http://www.worldfloraonline.org/taxon/wfo-0000610734 (accessed on 20 August 2020).
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Makino, T. Observations on the flora of Japan. Bot. Mag. 1907, 21, 86–88. [Google Scholar] [CrossRef][Green Version]
- Ohwi, J. Clematis. In Flora of Japan; Smithsonian Institution: Washington, DC, USA, 1965; pp. 440–442. [Google Scholar]
- Wang, W.T.; Lei, X. A revision of Clematis sect. Tubulosae (Ranunculaceae). Acta Phytotaxon. Sin. 2007, 45, 425–457. [Google Scholar] [CrossRef]
- Shi, J.H. A Preliminary Study of the Hybrid Origin of Clematis pinnata Maxim. (Ranunculaceae). Master’s Dissertation, Beijing Institute of Botany, Chinese Academy of Sciences, Beijing, China, 2003. [Google Scholar]
| Taxon | Locality | Voucher No. |
|---|---|---|
| C. apiifolia DC. | Mt. Sinbul, Icheon-ri, Sangbuk-myeon, Ulju-gun, Ulsan, Korea | Sinbulsan-190911-001 |
| C. brevicaudata DC. | Unchi-ri, Sindong-eup, Jeongseon-gun, Gangwon-do, Korea | Unchiri-191007-001 |
| C. trichotoma Nakai | Mt. Sinbul, Icheon-ri, Sangbuk-myeon, Ulju-gun, Ulsan, Korea | Sinbulsan-190911-001 |
| C. taeguensis Y. Lee | Gyuram-ri, Jeongseon-eup, Jeongseon-gun, Gangwon-do, Korea | Gyuramri-190818-001 |
| C. hexapetala Pall. | Ho-ri, Palbong-myeon, Seosan-si, Chungcheongnam-do, Korea | Hori-190809-001 |
| C. terniflora DC. | Jukpo-ri, Dolsan-eup, Yeosu-si, Jeollanam-do, Korea | Dolsando-191004-002 |
| C. terniflora var. mandshurica (Rupr.) Ohwi | Namhansanseong Fortress, Sanseong-ri, Namhansanseong-myeon, Gwangju-si, Gyeonggi-do, Korea | Namhansanseong-190809-001 |
| C. heracleifolia DC. | Sihwa Lake, Munho-ri, Namyang-eup, Hwaseong-si, Gyeonggi-do, Korea | Sihwaho-190921-016 |
| C.urticifolia Nakai ex Kitag. | Mt. Gariwang, Sugam-ri, Bukpyeong-myeon, Jeongseon-gun, Gangwon-do, Korea | Gariwangsan-191007-001 |
| C. takedana Makino | Sihwa Lake, Munho-ri, Namyang-eup, Hwaseong-si, Gyeonggi-do, Korea | Sihwaho-190921-001 |
| C. patens C.Morren & Dence. | Mt. Johang, Samsong-ri, Cheongcheon-myeon, Goesan-gun, Chungcheongbuk-do, Korea | Johangsan-170831-049 |
| C. brachyura Maxim. | Seondol, Bangjeol-ri, Yeongwol-eup, Yeongwol-gun, Gangwon-do, Korea | Seondol-190719-001 |
| C. serratifolia Rehder | Gasong-ri, Dosan-myeon, Andong-si, Gyeongsangbuk-do, Korea | Gasongri-191007-001 |
| C. fusca var. fusca Turcz. | Mt. Cheongtae, Sapgyo-ri, Dunnae-myeon, Hoengseong-gun, Gangwon-do, Korea | Cheongtaesan-190819-001 |
| C. fusca var. flabellata (Nakai) J. S. Kim | Eundae-bong, Gohan-ri, Gohan-eup, Jeongseon-gun, Gangwon-do, Korea | Eundaebong-190818-001 |
| C. fusca var. violacea Maxim. | Mt. Baekhwa, Mawon-ri, Mungyeong-eup, Mungyeong-si, Gyeongsangbuk-do, Korea | Baekhwasan-150707-007 |
| C. calcicola J. S. Kim | Mt. Deokhang, Daei-ri, Singi-myeon, Samcheok-si, Gangwon-do, Korea | Deokhangsan-190818-001 |
| C. koreana Kom. | Mt. Hambaek, Gohan-ri, Gohan-eup, Jeongseon-gun, Gangwon-do, Korea | Hambaeksan-190818-001 |
| C. ochotensis (Pall.) Poiret | Mt. Gariwang, Sugam-ri, Bukpyeong-myeon, Jeongseon-gun, Gangwon-do, Korea | Gariwangsan-190819-007 |
| Taxon | Shape | Color | Achene Indumentum | Lateral Wings | Style Elongation | Style | Surface Sculpture | Surface Cells Outline | Periclinal Wall | Anticlinal Wall |
|---|---|---|---|---|---|---|---|---|---|---|
| C. apiifolia | Narrow-elliptical | Brown | Completely hairy, hairs short on the achene body and long on the style | Narrow | Elongated | Plumose | Striate-rugose | Not differentiated | Concave with fine folds | Raised, folded |
| C. brevicaudata | Elliptic | Brown | Completely hairy, hairs short on the achene body and long on the style | Narrow | Strongly elongated | Plumose | Striate-rugose | Not differentiated | Concave with fine folds | Raised, folded |
| C. trichotoma | Obovate to fusiform | Black | Glabrous | Narrow | Strongly elongated | Plumose | Striate-rugose | Not differentiated | Concave with fine folds | Slightly raised |
| C. taeguensis | Elliptical to obovate | Light yellow | Sparsely hairy, fine hair on the body and long on the style | Wide | Strongly elongated | Plumose | Striate-colliculate | Elongated, rectangular | Convex with fine folds | Sunken, smooth |
| C. hexapetala | Obovate | Dark brown to black | Completely hairy | Medium | Elongated | Plumose | Striate-rugose | Elongated | Concave with fine folds | Raised, folded |
| C. terniflora | Elliptic to Obovate | Brown | Sparsely hairy, fine hair on the body and long on the style | Wide | Elongated | Plumose | Striate-reticulate | Rectangular, polygonal | Concave with fine folds | Raised, smooth |
| C. terniflora var. mandshurica | Elliptic | Light yellow | Sparsely hairy, fine hair on the body and long on the style | Wide | Elongated | Plumose | Striate-reticulate | Rectangular, polygonal | Concave with fine folds | Sunken, smooth |
| C. heracleifolia | Obovate | Dark brown | Completely hairy | Narrow | Strongly elongated | Plumose | Striate-rugose | Irregular | Concave with fine folds | Raised, folded |
| C. urticifolia | Elliptic to Obovate | Light greenish yellow | Completely hairy | Medium | Strongly elongated | Plumose | Striate-rugose | Irregular | Concave with fine folds | Raised, folded |
| C. takedana | Obovate | Dark brown to black | Completely hairy | Medium | Strongly elongated | Plumose | Striate-rugose | Irregular | Concave with fine folds | Raised, folded |
| C. patens | Obovate | Dark brown to black | Completely hairy, hairs short on the achene body and long on the style | Medium | Strongly elongated | Plumose | Striate-rugose | Irregular | Concave with fine folds | Raised, folded |
| C. brachyura | Obovate | Brown, black | Sparsely hairy | Wide | Not elongated | Glabrous | Rugose, Pustulate | Elongated | Concave pustulate | Raised, pustulate |
| C. serratifolia | Obovate | Medium brown | Sparsely hairy, fine hair on the body and long on the style | Narrow | Strongly elongated | Plumose | Reticulate | Elongated, rectangular | Concave with fine folds | raised, smooth |
| C. fusca var. fusca | Obovate | Dark brown to black | Completely hairy | Wide | Strongly elongated | Plumose | Striate-reticulate | Polygonal | Concave with fine folds | Raised smooth or fine folds |
| C. fusca var. flabellata | Elliptic to Obovate | Brown | Completely hairy | Medium | Strongly elongated | Plumose | Striate-reticulate | Polygonal | Concave with fine folds | Slightly raised, smooth |
| C. fusca var. violacea | Obovate | Black | Completely hairy | Medium | Strongly elongated | Plumose | Striate-reticulate | Polygonal | Concave with fine folds | Raised smooth or fine folds |
| C. calcicola | Narrow-elliptical | Dark brown | Completely hairy | Narrow | Strongly elongated | Plumose | Reticulate | Polygonal | Concave with fine folds | Raised smooth or fine folds |
| C. koreana | Obovate | Medium brown | Completely hairy | Narrow | Strongly elongated | Plumose | Reticulate | Elongated, polygonal | Concave with fine folds | Raised smooth or fine folds |
| C. ochotensis | Obovate | Brown | Sparsely hairy, fine hairs on the body and long on the style | Narrow | Strongly elongated | Plumose | Reticulate | Elongated, polygonal | Concave with fine folds | Raised smooth or fine folds |
| Taxon | Outline in CS | Exocarp | Mesocarp | Endocarp | Seed Coat | |||||
| C. apiifolia | Elliptic | Degenerating cells | Few layers of degenerating cells | Thin, single layer of lignified cells | Degenerated | |||||
| C. brevicaudata | Elliptic | Cutinized single tanniniferous layer | 3–4 layers of parenchyma layer with wavy wall | Thick, single layer (rarely double layer) sclerid cells | Few layers of parenchyma cells | |||||
| C. trichotoma | Elliptic | Cutinized single tanniniferous layer | 8–10 layers of thin walled parenchyma cells with wavy wall | Thin, single layer of lignified cells | 1–2 layers of degenerating cells | |||||
| C. taeguensis | Dumbbell | Cutinized single layer parenchyma cells with thick outer wall | 4–6 layers of thin walled parenchyma cells with wavy wall | Thick, single layer of sclerid cells | 5–7 layers of parenchyma cells | |||||
| C. hexapetala | Fusiform | Cutinized single tanniniferous layer | Few layers of degenerating cells | Thick, single layer of sclerid cells | Few layers of degenerating cells | |||||
| C. terniflora | Dumbbell | Cutinized single layer parenchyma cells with thick outer wall | 2–4 layers of thin walled parenchyma cells with wavy wall | Single layer of sclerid cells | Few layers of degenerating cells | |||||
| C. terniflora var. mandshurica | Dumbbell | Cutinized single layer parenchyma cells with thick outer wall | 4–6 layers of thin walled parenchyma cells with wavy wall | Thick, single layer of sclerid cells | Few layers of parenchyma cells | |||||
| C. heracleifolia | Fusiform | Cutinized single layer parenchyma cells with thick outer wall | 4–6 layers of thin walled parenchyma cells with wavy wall | Thick, single layer of lignified palisade cells | Few layers of degenerating cells | |||||
| C. urticifolia | Elliptic | Cutinized single layer parenchyma cells with thick outer wall | 4–6 layers of thin walled parenchyma cells with wavy wall | Single layer of sclerid cells | Few layers of degenerating cells | |||||
| C. takedana | Elliptic | Cutinized single layer parenchyma cells with thick outer wall | 6–8 layers of thin walled parenchyma cells with wavy wall | Thin, single layer of lignified cells | 5–7 layers of parenchyma cells | |||||
| C. patens | Fusiform | Cutinized single tanniniferous layer | Few layers of degenerating cells | Thick, single layer of lignified palisade cells | Few layers of parenchyma cells | |||||
| C. brachyura | Narrowly fusiform | Cutinized single layer parenchyma cells with thick outer wall | 2–4 layers of thin walled parenchyma cells with wavy wall | Thick, single layer (rarely double layer) sclerid cells | 3–5 layers of parenchyma cells | |||||
| C. serratifolia | Fusiform | Cutinized single layer parenchyma cells with thick outer wall | Few layers of degenerating cells | Thick, single layer of lignified palisade cells | Few layers of degenerating cells | |||||
| C. fusca var. fusca | Fusiform | Cutinized single taniniferous layer | Few layers of degenerating cells | Single layer of sclerid cells | 5–7 layers of parenchyma cells | |||||
| C. fusca var. flabellata | Fusiform | Cutinized single taniniferous layer | Few layers of degenerating cells | Thick, single layer (rarely double layer) sclerid cells | Few layers of degenerating cells | |||||
| C. fusca var. violacea | Fusiform | Degenerated | Few layers of degenerating cells | Thick, single layer of lignified palisade cells | Few layers of degenerating cells | |||||
| C. calcicola | Oval, elliptical | Cutinized single layer parenchyma cells with thick outer wall | 6–8 layers of thin walled parenchyma cells with wavy wall | Thick, single layer (rarely double layer) sclerid cells | Few layers of degenerating cells | |||||
| C. koreana | Oval, elliptical | Cutinized single layer parenchyma cells with thick outer wall | 6–8 layers of thin walled parenchyma cells with wavy wall | Thick, single layer (rarely double layer) sclerid cells | 3–5 layers of parenchyma cells | |||||
| C. ochotensis | Oval, elliptical | Cutinized single layer parenchyma cells with thick outer wall | 2–3 layers of thin walled parenchyma cells with wavy wall | Thin, single layer (rarely double layer) sclerid cells | Few layers of degenerating cells | |||||
| Taxon | Length (mm) | Width (mm) | L/W | Pericarp (µm) | Endocarp (µm) | P/E | Style (mm) | S/B | CSD1 (mm) | CSD2 (mm) | CSD1/CSD2 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| C. apiifolia | 3.95 ± 0.4 | 1.32 ± 0.2 | 3.01 ± 0.21 | 63.51 ± 15.58 | 6.32 ± 1.06 | 10 ± 1.36 | 9.55 ± 1.29 | 2.44 ± 0.38 | 1.66 ± 0.04 | 1.08 ± 0.07 | 1.54 ± 0.08 |
| C. brevicaudata | 2.53 ± 0.19 | 1.62 ± 0.18 | 1.59 ± 0.21 | 104.89 ± 26.35 | 12.03 ± 3 | 8.99 ± 2.56 | 25.74 ± 3.24 | 10.17 ± 1.17 | 2.07 ± 0.05 | 1.15 ± 0.09 | 1.8 ± 0.15 |
| C. trichotoma | 4 ± 0.27 | 2.67 ± 0.24 | 1.5 ± 0.05 | 343.62 ± 68.40 | 9.97 ± 1.52 | 35.94 ± 6.86 | 27.74 ± 3.04 | 6.95 ± 0.83 | 3.06 ± 0.28 | 2.17 ± 0.39 | 1.43 ± 0.16 |
| C. taeguensis | 4.3 ± 0.47 | 3.3 ± 0.48 | 1.34 ± 0.21 | 327.13 ± 46.45 | 12.06 ± 1.64 | 27.19 ± 2.17 | 35.35 ± 3.48 | 8.25 ± 1.2 | 3.28 ± 0.08 | 1.15 ± 0.1 | 2.87 ± 0.24 |
| C. hexapetala | 4.73 ± 0.4 | 2.88 ± 0.2 | 1.64 ± 0.04 | 56.52 ± 11.77 | 13.47 ± 1.93 | 4.17 ± 0.35 | 15.01 ± 2.33 | 3.21 ± 0.63 | 2.93 ± 0.21 | 1.38 ± 0.19 | 2.13 ± 0.14 |
| C. terniflora | 5.44 ± 0.43 | 3.12 ± 0.3 | 1.75 ± 0.06 | 94.85 ± 29.49 | 11.84 ± 2.05 | 7.83 ± 1.24 | 25.97 ± 4.08 | 4.76 ± 0.74 | 3.44 ± 0.3 | 1.33 ± 0.14 | 2.6 ± 0.14 |
| C. terniflora var. mandshurica | 6.75 ± 0.39 | 3.5 ± 0.4 | 1.94 ± 0.13 | 84.89 ± 16.41 | 9.84 ± 1.43 | 8.72 ± 1.65 | 31.66 ± 3.24 | 4.71 ± 0.55 | 3.73 ± 0.21 | 1.47 ± 0.12 | 2.55 ± 0.21 |
| C. heracleifolia | 3.21 ± 0.36 | 2.01 ± 0.25 | 1.6 ± 0.0.08 | 136.06 ± 25.36 | 25.61 ± 4.41 | 5.3 ± 0.17 | 17.07 ± 2.55 | 5.4 ± 1.21 | 3.01 ± 0.48 | 1.42 ± 0.12 | 2.21 ± 0.18 |
| C. urticifolia | 3.83 ± 0.25 | 2.9 ± 0.27 | 1.33 ± 0.09 | 285.8 ± 41.04 | 16.35 ± 3.12 | 17.24 ± 4.21 | 26.55 ± 3.8 | 6.97 ± 1.25 | 2.89 ± 0.37 | 1.49 ± 0.09 | 1.94 ± 0.33 |
| C. takedana | 2.88 ± 0.38 | 1.86 ± 0.22 | 1.56 ± 0.21 | 232.31 ± 52.36 | 9.94 ± 2.27 | 23.92 ± 5.1 | 26.23 ± 3.7 | 9.3 ± 1.64 | 2.08 ± 0.08 | 1.27 ± 0.16 | 1.66 ± 0.26 |
| C. patens | 4.04 ± 0.3 | 3.68 ± 0.51 | 1.12 ± 0.16 | 183.29 ± 18.69 | 57.69 ± 7.41 | 3.33 ± 0.83 | 30.21 ± 5.17 | 7.51 ± 1.41 | 4.95 ± 0.2 | 2.45 ± 0.13 | 2.03 ± 0.18 |
| C. brachyura | 9.12 ± 0.88 | 7.3 ± 0.46 | 1.25 ± 0.10 | 142.19 ± 28.08 | 22.44 ± 3.39 | 6.37 ± 1.04 | 2.99 ± 0.55 | 0.33 ± 0.07 | 7.1 ± 0.14 | 1.08 ± 0.11 | 6.65 ± 0.6 |
| C. serratifolia | 2.86 ± 0.17 | 1.42 ± 0.13 | 2.02 ± 0.11 | 41.44 ± 6.93 | 12.83 ± 2.09 | 3.23 ± 0.14 | 27.16 ± 4.52 | 9.51 ± 1.62 | 1.61 ± 0.04 | 1.04 ± 0.1 | 1.6 ± 0.12 |
| C. fusca var. fusca | 4.64 ± 0.41 | 2.99 ± 0.28 | 1.52 ± 0.07 | 89.63 ± 25.9 | 13.29 ± 1.93 | 6.9 ± 2.26 | 28.61 ± 6.65 | 6.25 ± 1.71 | 3.58 ± 0.17 | 1.21 ± 0.08 | 2.96 ± 0.2 |
| C. fusca var. flabellata | 3.77 ± 0.3 | 2.49 ± 0.23 | 1.14 ± 0.06 | 77.92 ± 25.31 | 12.51 ± 2.71 | 6.3 ± 0.95 | 35.71 ± 4.9 | 9.55 ± 1.72 | 2.86 ± 0.11 | 1.65 ± 0.08 | 1.73 ± 0.02 |
| C. fusca var. violacea | 6.1 ± 0.7 | 5.34 ± 0.51 | 1.18 ± 0.38 | 70.3 ± 12.45 | 34.1 ± 3.54 | 2.05 ± 0.16 | 44.74 ± 5.81 | 7.37 ± 0.9 | 5.16 ± 0.15 | 1.92 ± 0.32 | 2.73 ± 0.34 |
| C. calcicola | 4.44 ± 0.48 | 2.07 ± 0.25 | 2.18 ± 0.38 | 222.95 ± 25.82 | 24.27 ± 4.22 | 9.35 ± 2.25 | 28.48 ± 4.29 | 6.48 ± 1.23 | 2.86 ± 0.25 | 1.85 ± 0.3 | 1.59 ± 0.33 |
| C. koreana | 4.77 ± 0.35 | 2.55 ± 0.36 | 1.88 ± 0.12 | 111.82 ± 26.78 | 27.66 ± 3.51 | 4.11 ± 0.46 | 31.85 ± 4.95 | 6.7 ± 1.14 | 2.36 ± 0.07 | 1.66 ± 0.08 | 1.42 ± 0.04 |
| C. ochotensis | 3.76 ± 0.25 | 1.64 ± 0.26 | 2.35 ± 0.39 | 111.83 ± 16.44 | 12.57 ± 1.5 | 8.96 ± 1.42 | 28.56 ± 2.45 | 7.64 ± 0.89 | 1.81 ± 0.19 | 1.29 ± 0.15 | 1.42 ± 0.3 |
| ANOVA | F = 206.22, p < 0.001 | F = 298.9, p < 0.001 | F = 101.98, p < 0.001 | F = 121.01, p < 0.001 | F = 182.24, p < 0.001 | F = 190.03, p < 0.001 | F = 83.7, p < 0.001 | F = 72.68, p < 0.001 | F = 210.17, p < 0.001 | F = 26.89, p < 0.001 | F = 114.58, p < 0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghimire, B.; Park, B.K.; Son, D.C.; Oh, S.-H. Achene Morphology and Anatomy of Clematis L. (Ranunculaceae) in Korea and Its Taxonomic Implications. Plants 2020, 9, 1279. https://doi.org/10.3390/plants9101279
Ghimire B, Park BK, Son DC, Oh S-H. Achene Morphology and Anatomy of Clematis L. (Ranunculaceae) in Korea and Its Taxonomic Implications. Plants. 2020; 9(10):1279. https://doi.org/10.3390/plants9101279
Chicago/Turabian StyleGhimire, Balkrishna, Beom Kyun Park, Dong Chan Son, and Seung-Hwan Oh. 2020. "Achene Morphology and Anatomy of Clematis L. (Ranunculaceae) in Korea and Its Taxonomic Implications" Plants 9, no. 10: 1279. https://doi.org/10.3390/plants9101279
APA StyleGhimire, B., Park, B. K., Son, D. C., & Oh, S.-H. (2020). Achene Morphology and Anatomy of Clematis L. (Ranunculaceae) in Korea and Its Taxonomic Implications. Plants, 9(10), 1279. https://doi.org/10.3390/plants9101279





