Seasonality and Small Spatial-Scale Variation of Chlorophyll a Fluorescence in Bryophyte Syntrichia ruralis [Hedw.] in Semi-Arid Sandy Grassland, Hungary
Abstract
1. Introduction
2. Results
2.1. Effect of Slopes (NE and SW) on Chlorophyll a Fluorescence Parameters
2.2. Effect of Seasons on Chlorophyll a Fluorescence Parameters
2.3. Effect of Slopes and Seasons Interaction on Chlorophyll a Fluorescence Parameters
3. Discussion
3.1. Photochemical Quenching Parameters (Ratio of Variable to Maximum Fluorescence (Fv/Fm), Photochemical Quantum Yield of Photosystem II (ΦPS II), Photochemical Fluorescence Quenching (qP))
3.2. Non-Photochemical Quenching Parameters (Fluorescence Quenching (qN), Non-Photochemical Quenching (NPQ))
4. Materials and Methods
4.1. Sampling of Plant Material
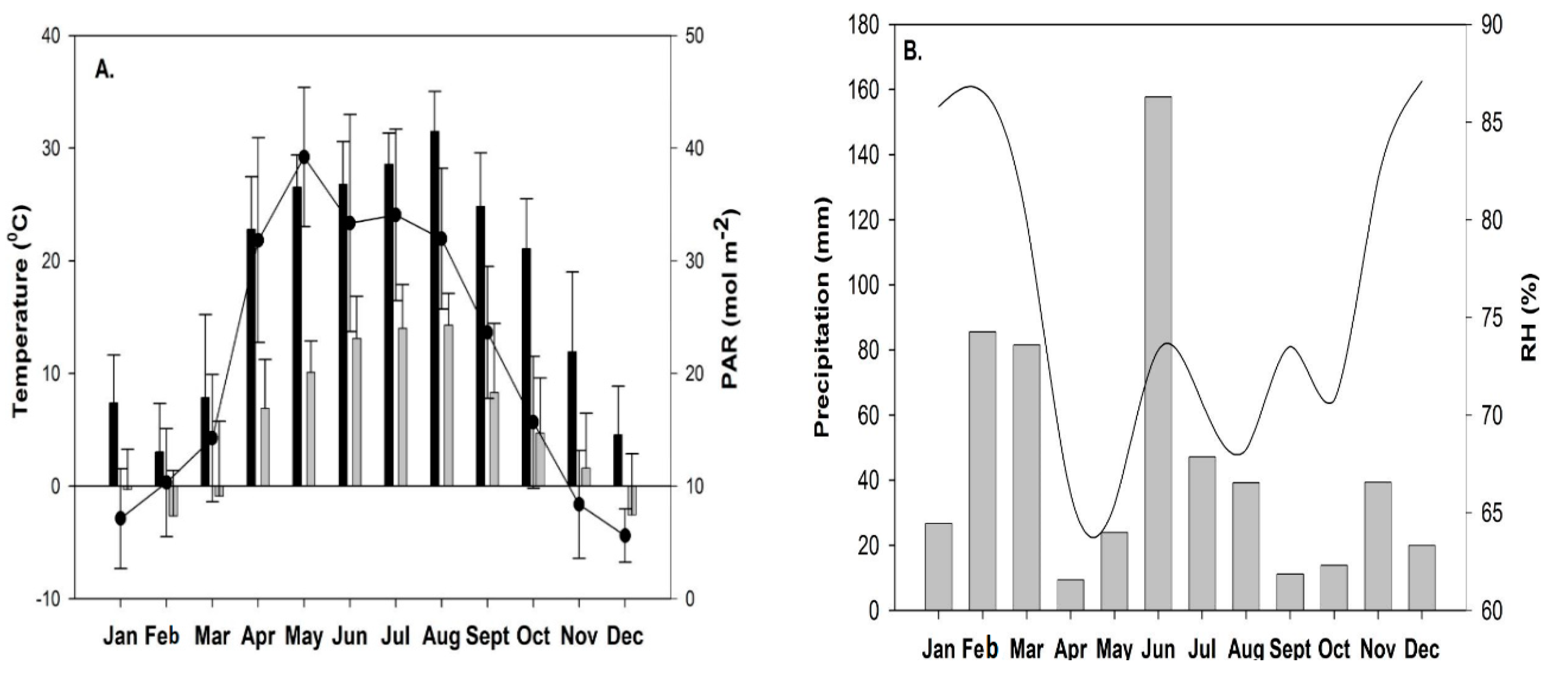
4.2. Climatic Conditions
4.3. Experimental Set-Up
4.4. Chlorophyll a Fluorescence Measurement
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Maestre, F.T.; Bowker, M.A.; Cantón, Y.; Castillo-Monroy, A.P.; Cortina, J.; Escolar, C.; Escudero, A.; Lázaro, R.; Martínez, I. Ecology and functional roles of biological soil crusts in semi-arid ecosystems of Spain. J. Arid Environ. 2011, 75, 1282–1291. [Google Scholar] [CrossRef]
- Proctor, M.C.F.; Pence, V.C. Vegetative tissues: Bryophytes, vascular resurrection plants, and vegetative propagules. In Desiccation and Survival in Plants: Drying without Dying; Black, M., Pritchard, H.W., Eds.; CABI Publishing: Wallingford, CT, USA, 2002. [Google Scholar]
- Tuba, Z.; Proctor, M.C.F.; Csintalan, Z. Ecophysiological responses of homoiochlorophyllous and poikilochlorophyllous desiccation tolerant plants: A comparison and an ecological perspective. Plant Growth Regul. 1998, 24, 211–217. [Google Scholar] [CrossRef]
- Proctor, M.C.F.; Oliver, M.J.; Wood, A.J.; Stark, L. Desiccation tolerance in bryophytes: A review. Bryologist 2007, 110, 595–621. [Google Scholar] [CrossRef]
- Oliver, M.J.; Velten, J.; Mishler, B.D. Desiccation tolerance in bryophytes: A reflection of the primitive strategy for plant survival in dehydrating habitats? Integr. Comp. Biol. 2005, 45, 788–799. [Google Scholar] [CrossRef] [PubMed]
- Csintalan, Z.; Takács, Z.; Proctor, M.C.F.; Nagy, Z.; Tuba, Z. Early morning photosynthesis of the moss Tortula ruralis following summer dewfall in a Hungarian temperate dry sandy grassland. Plant Ecol. 2000, 151, 51–54. [Google Scholar] [CrossRef]
- Tuba, Z.; Csintalan, Z.; Proctor, M.C.F. Photosynthetic responses of a moss Tortula ruralis ssp. ruralis, and the lichens Cladonia convoluta and C. furcata to water deficit and short periods of desiccation and their ecophysiological significance: A baseline study at present-day concentration. New Phytol. 1996, 133, 353–361. [Google Scholar] [CrossRef]
- Peli, E.; Nie, L.; Pócs, T.; Pormbski, S.; Laufer, Z. Chlorophyll fluorescence and CO2 assimilation responses of desiccation-tolerant cyanobacterial crusts of tropical inselberg rocks to rehydration following one and five-years air-dried stage. Cereal Res. Commun. 2005, 3, 437–441. [Google Scholar]
- Margóczi, K.; Szanyi, J.; Aradi, E.; Busa-Fekete, B. Hydrological background of the dune slack vegetation in the Kiskunság. Ann. Warsaw Univ. Life Sci.—SGGW 2007, 38, 105–113. [Google Scholar] [CrossRef]
- Yizhaq, H.; Ashkenazy, Y.; Tsoar, H. Sand dune dynamics and climate change: A modeling approach. J. Geophys. Res. 2009, 114. [Google Scholar] [CrossRef]
- Bartholy, J.; Gelybó, G.; Pongrácz, R. Regional climate change expected in Hungary for 2071–2100. Appl. Ecol. Environ. Res. 2007, 5, 1–17. [Google Scholar] [CrossRef]
- Blanka, V.; Mezősi, G.; Meyer, B. Projected changes in the drought hazard in Hungary due to climate change. Időjárás 2013, 117, 219–237. [Google Scholar]
- Tuba, Z.; Csintalan, Z.; Szente, K.; Nagy, Z.; Grace, J. Carbon gains by desiccation-tolerant plants at elevated CO2. Funct. Ecol. 1998, 12, 39–44. [Google Scholar] [CrossRef]
- Kalopos, T.; Mázsa, K. Juniper shade enables terricolous lichens and mosses to maintain high photochemical efficiency in a semi-arid temperate sand grassland. Photosynthetica 2001, 39, 263–268. [Google Scholar] [CrossRef]
- Genty, B.; Briantais, J.M.; Baker, N.R. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta 1989, 990, 87–92. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence- a practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Proctor, M.C.F.; Bates, J.W. Chlorophyll-fluorescence measurements in bryophytes: Evidence for three main types of light-curve response. J. Bryol. 2018, 40, 1–11. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Adams, W.W. Photoprotection in an ecological context: The remarkable complexity of thermal energy dissipation. New Phytol. 2006, 172, 11–21. [Google Scholar] [CrossRef]
- Marschall, M. Ecophysiology of bryophytes in a changing environment. Acta Biol. Plant. Agriensis 2017, 5, 61–70. [Google Scholar]
- Marschall, M.; Proctor, M.C.F. Are Bryophytes Shade Plants? Photosynthetic Light Responses and Proportions of Chlorophyll a, Chlorophyll b and Total Carotenoids. Ann. Bot. 2004, 94, 593–603. [Google Scholar] [CrossRef]
- Marschall, M.; Beckett, R.P. Photosynthetic responses in the inducible mechanisms of desiccation tolerance of liverwort and moss. Acta Biol. Szeged. 2005, 49, 155–156. [Google Scholar]
- Roháček, K. Chlorophyll fluorescence parameters: The definitions, photosynthetic meaning, and mutual relationships. Photosynthetica 2002, 40, 13–29. [Google Scholar] [CrossRef]
- Proctor, M.C.F. Experiments on the effect of different intensities of desiccation on bryophytes survival, using chlorophyll fluorescence as an index of recovery. J. Bryol. 2003, 25, 201–210. [Google Scholar] [CrossRef]

| Slopes | Fv/Fm | ΦPS II | qP | qN | NPQ |
|---|---|---|---|---|---|
| North-east (NE) | 0.750 ± 0.006 | 0.260 ± 0.008 | 0.582 ± 0.012 | 0.905 ± 0.005 | 4.504 ± 0.217 |
| South-west (SW) | 0.752 ± 0.006 | 0.305 ± 0.009 | 0.659 ± 0.013 | 0.879 ± 0.008 | 3.724 ± 0.229 |
| Season | Fv/Fm | ΦPS II | qP | qN | NPQ |
|---|---|---|---|---|---|
| Spring | 0.714 ± 0.006 a | 0.280 ± 0.013 a | 0.670 ± 0.021 b | 0.904 ± 0.005 bc | 4.086 ± 0.022 a |
| Summer | 0.780 ± 0.002 c | 0.273 ± 0.013 a | 0.613 ± 0.021 ab | 0.920 ± 0.003 c | 5.212 ± 0.187 b |
| Autumn | 0.754 ± 0.005 b | 0.285 ± 0.011 a | 0.590 ± 0.018 a | 0.860 ± 0.010 a | 3.164 ± 0.243 a |
| Winter | 0.756 ± 0.005 b | 0.290 ± 0.016 a | 0.609 ± 0.017 ab | 0.884 ± 0.012 ab | 3.994 ± 0.370 a |
| Chlorophyll a Fluorescence Parameter | Variables | Sum of Squares | Df | F-Value | p-Value |
|---|---|---|---|---|---|
| Fv/Fm | Slopes | 6.07 × 10−5 | 1 | 0.1806 | 0.6731 |
| Seasons | 0.273314 | 3 | 27.0883 | 9.898 × 10−10 *** | |
| Slopes: Seasons | 8.028 × 10−4 | 3 | 0.7956 | 0.5036 | |
| Slopes | 0.25025 | 1 | 14.7795 | 4.235 × 10−4 *** | |
| ΦPS II | Seasons | 1.830 × 10−3 | 3 | 0.3603 | 0.7819465 |
| Slopes: Seasons | 8.346 × 10−3 | 3 | 1.643 | 0.1947031 | |
| Slopes | 0.69312 | 1 | 20.8218 | 4.709 × 10−5 *** | |
| qP | Seasons | 0.042304 | 3 | 4.2362 | 0.01084 |
| Slopes: Seasons | 5.400 × 10−3 | 3 | 0.5408 | 0.65714 | |
| Slopes | 7.8541 × 10−3 | 1 | 12.8124 | 9.211 × 10−4 *** | |
| qN | Seasons | 0.0240497 | 3 | 13.0774 | 4.283 × 10−6 *** |
| Slopes: Seasons | 7.0756 × 10−3 | 3 | 3.8475 | 0.0164519 * | |
| Slopes | 7.3039 | 1 | 11.9309 | 1.32 × 10−3 ** | |
| NPQ | Seasons | 25.4975 | 3 | 13.8834 | 2.379 × 10−6 *** |
| Slopes: Seasons | 5.2303 | 3 | 2.8479 | 0.04949 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruchika; Csintalan, Z.; Péli, E.R. Seasonality and Small Spatial-Scale Variation of Chlorophyll a Fluorescence in Bryophyte Syntrichia ruralis [Hedw.] in Semi-Arid Sandy Grassland, Hungary. Plants 2020, 9, 92. https://doi.org/10.3390/plants9010092
Ruchika, Csintalan Z, Péli ER. Seasonality and Small Spatial-Scale Variation of Chlorophyll a Fluorescence in Bryophyte Syntrichia ruralis [Hedw.] in Semi-Arid Sandy Grassland, Hungary. Plants. 2020; 9(1):92. https://doi.org/10.3390/plants9010092
Chicago/Turabian StyleRuchika, Zsolt Csintalan, and Evelin Ramóna Péli. 2020. "Seasonality and Small Spatial-Scale Variation of Chlorophyll a Fluorescence in Bryophyte Syntrichia ruralis [Hedw.] in Semi-Arid Sandy Grassland, Hungary" Plants 9, no. 1: 92. https://doi.org/10.3390/plants9010092
APA StyleRuchika, Csintalan, Z., & Péli, E. R. (2020). Seasonality and Small Spatial-Scale Variation of Chlorophyll a Fluorescence in Bryophyte Syntrichia ruralis [Hedw.] in Semi-Arid Sandy Grassland, Hungary. Plants, 9(1), 92. https://doi.org/10.3390/plants9010092




