Phosphorus and Nitrogen Modulate Plant Performance in Shrubby Legumes from the Iberian Peninsula
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Culture and Growth Conditions
2.2. Nitrogen and Phosphorus Efficiencies and Allocation Calculations
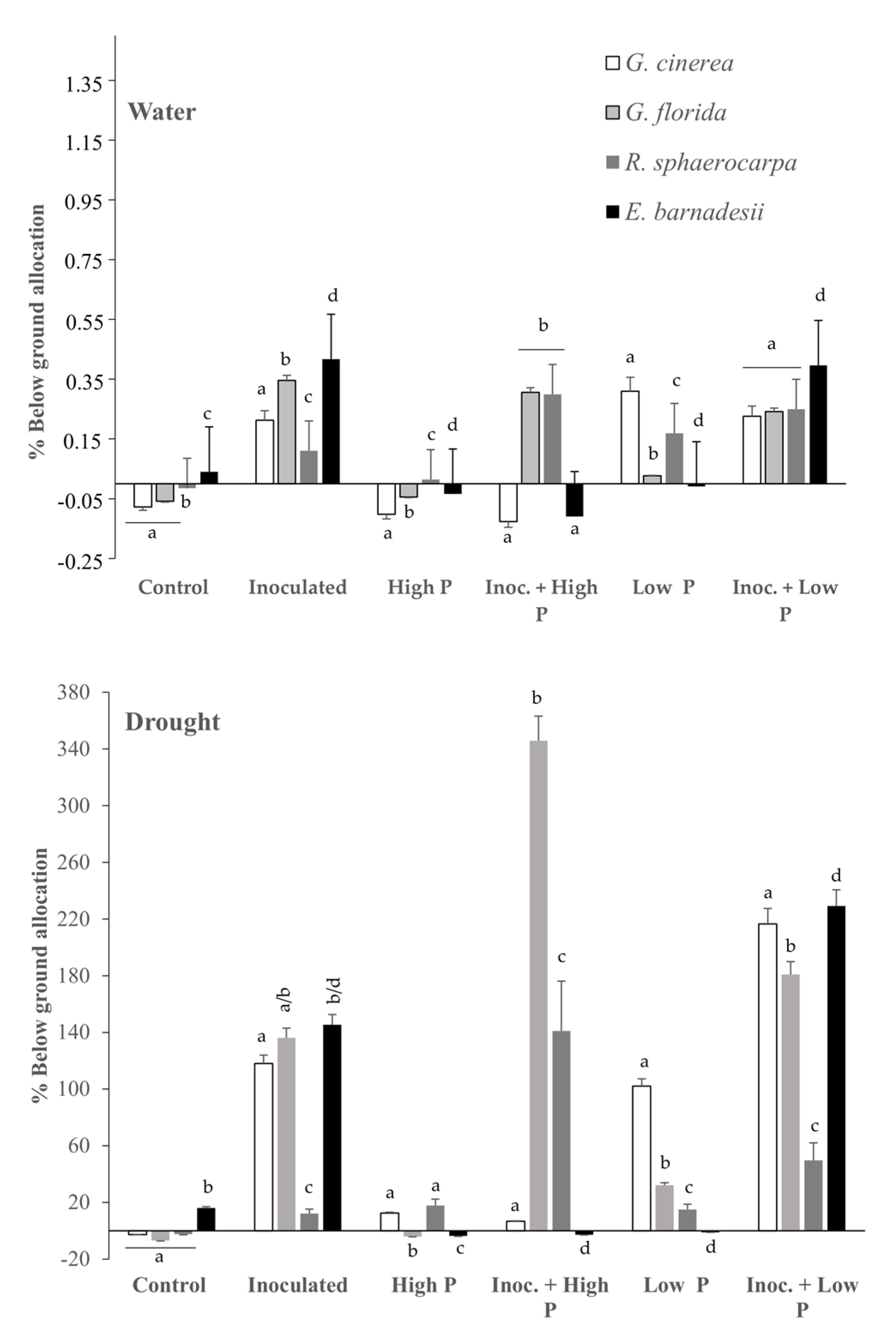
2.3. Below Ground Allocation
2.4. Statistical Design and Analyses
3. Results
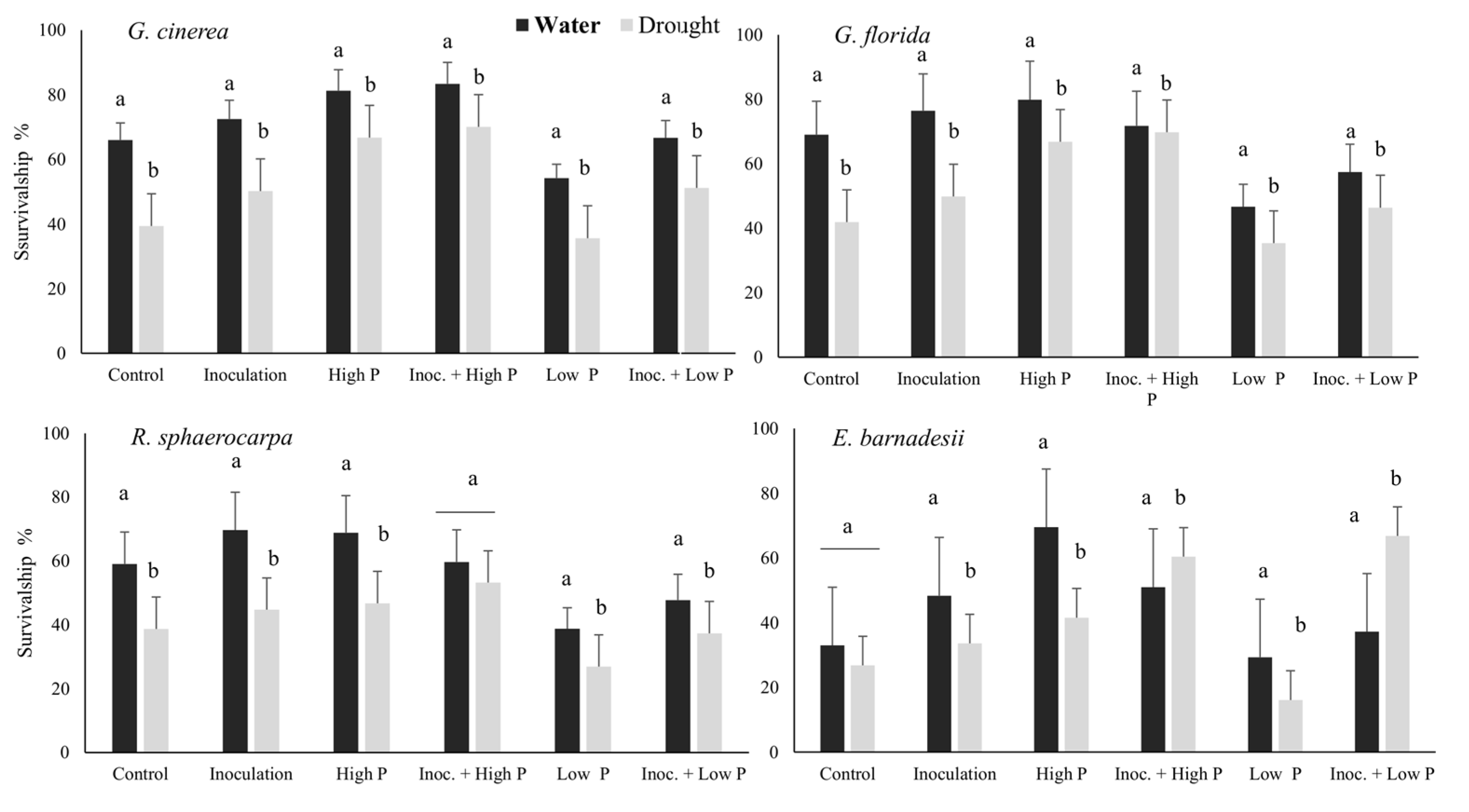
3.1. Plant Survivorship
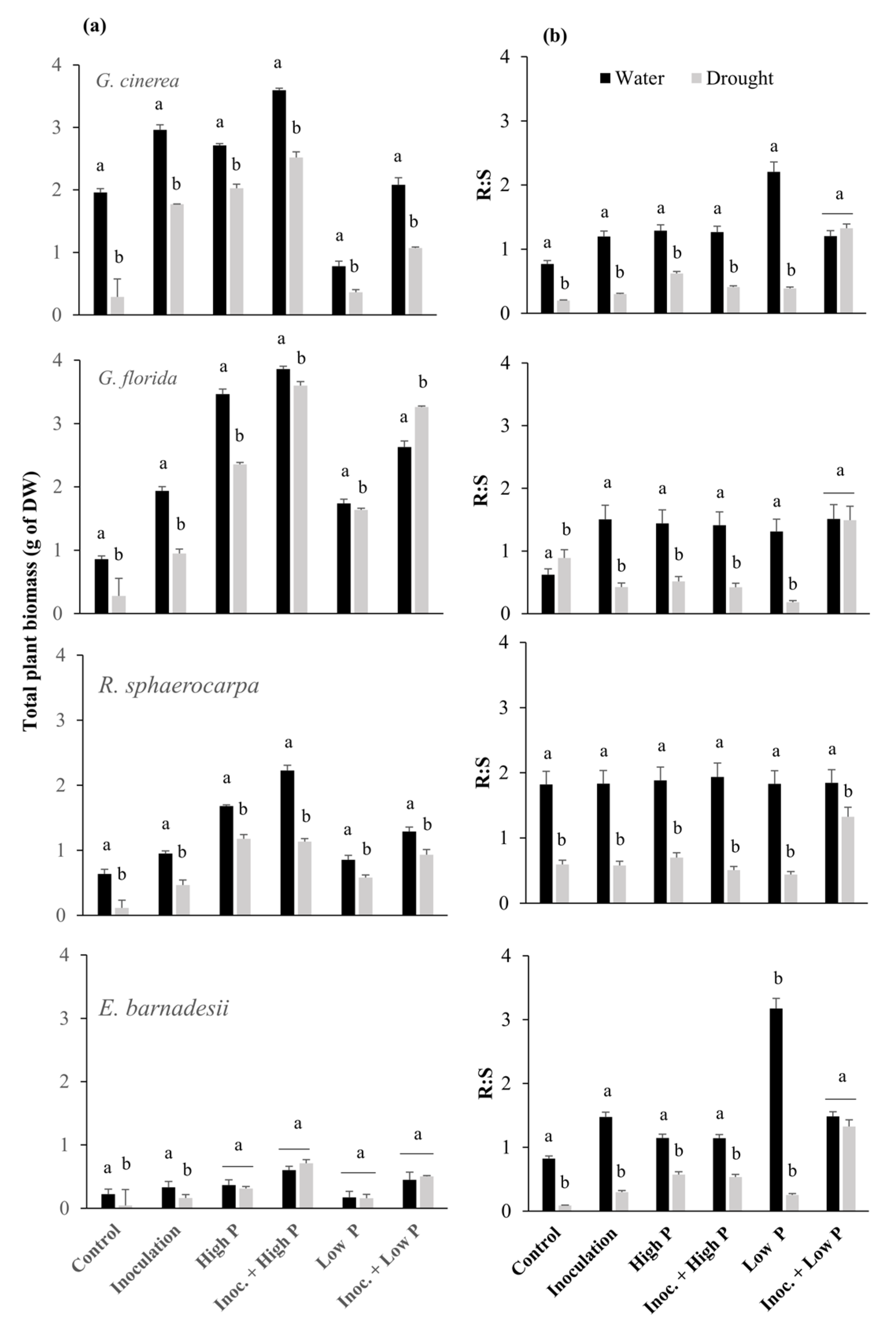
3.2. Biomass Production and Growth Kinetics
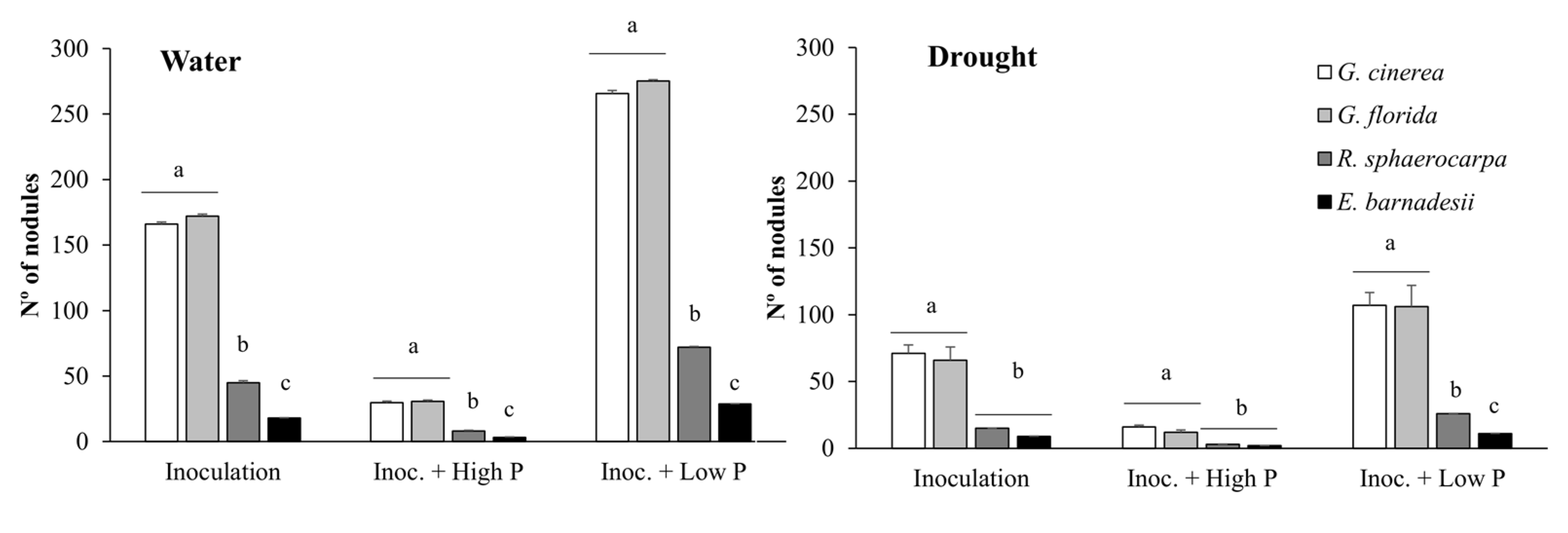
3.3. Nodule Formation and Infection
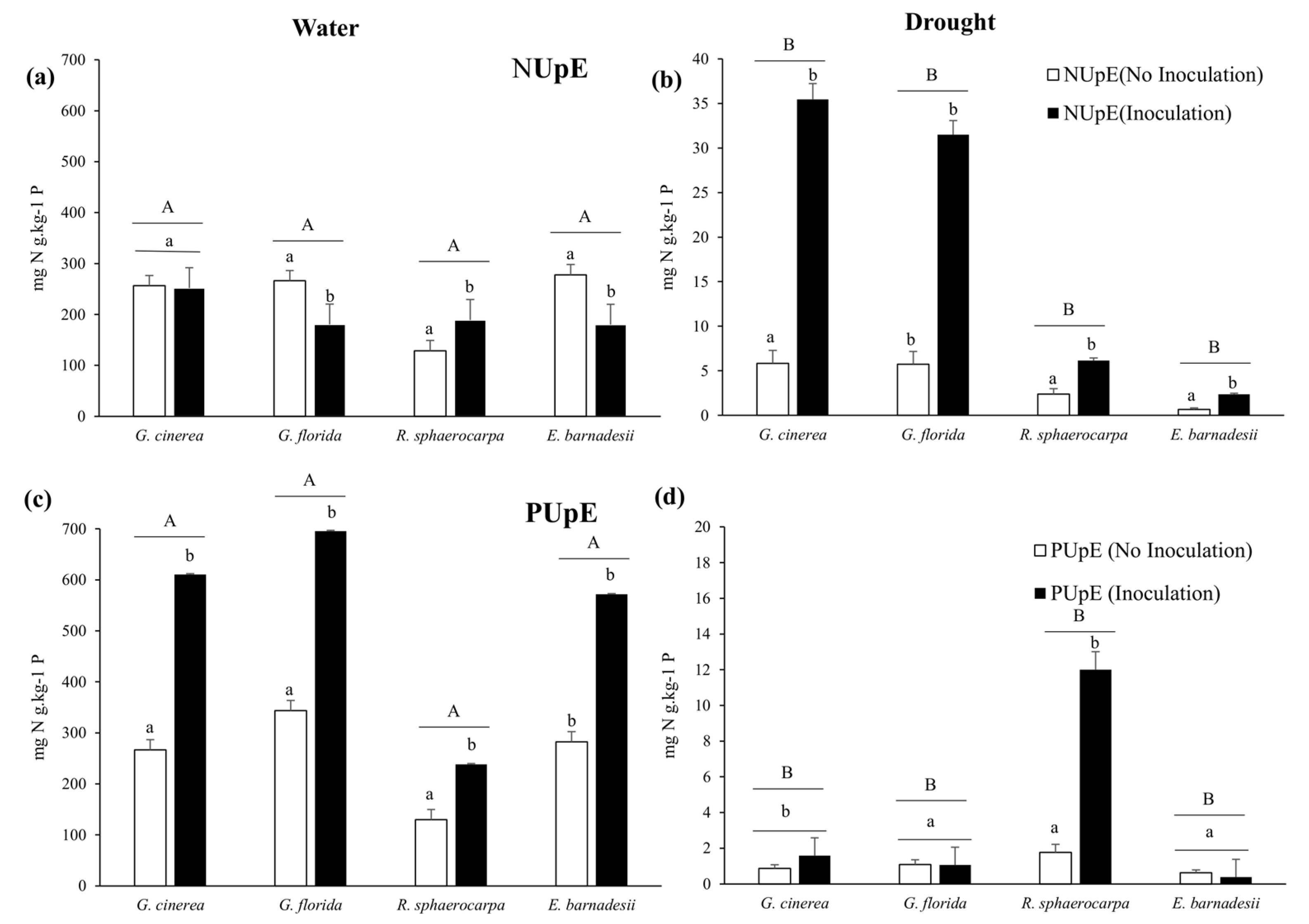
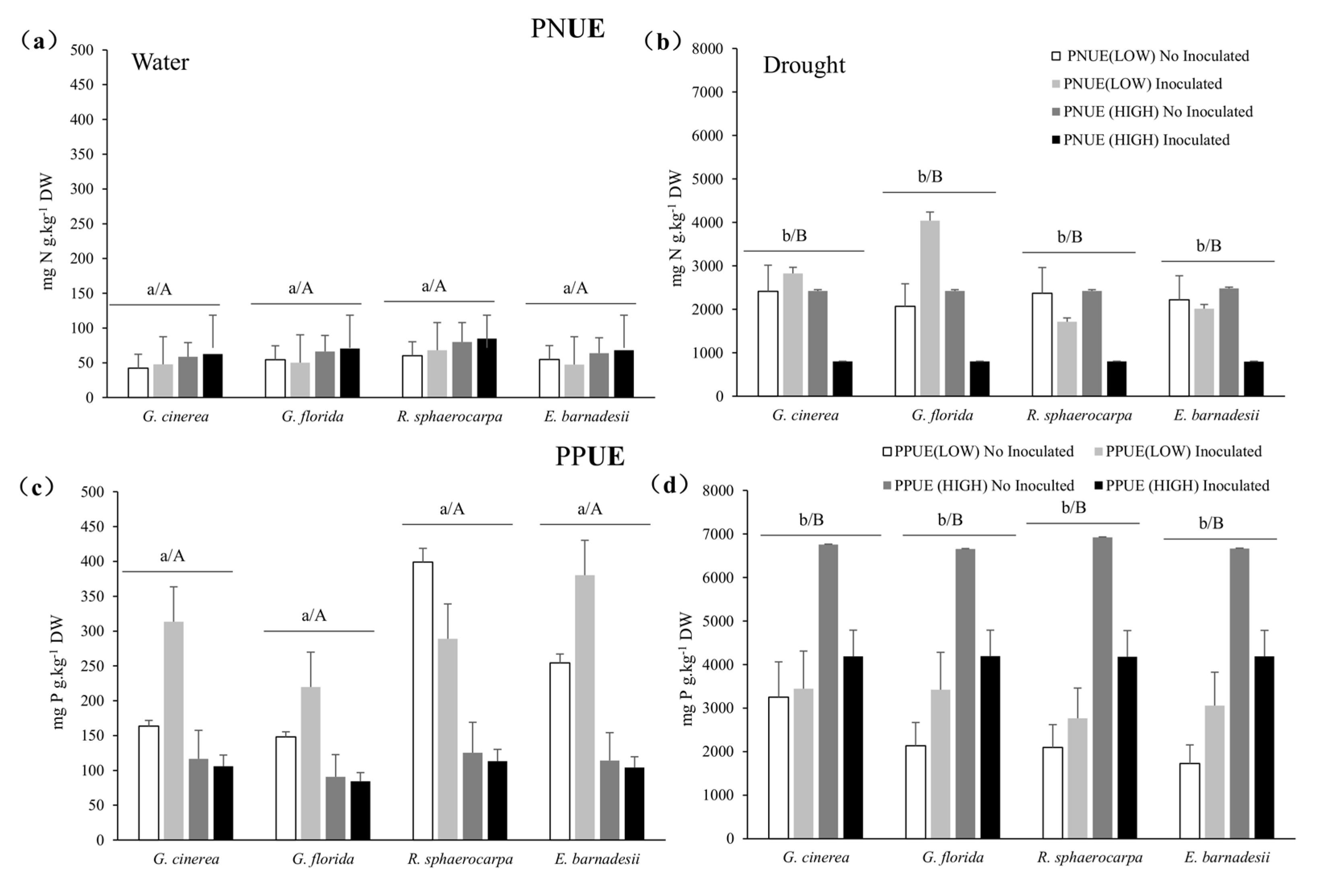
3.4. Nitrogen and Phosphorus Efficiencies and Allocation
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Schimel, D.S. Dry lands in the earth system. Science 2010, 327, 418–419. [Google Scholar] [CrossRef]
- Ruiz-Díez, B.; Fajardo, S.; Puertas-Mejía, M.A.; de Felipe, M.R.; Fernández-Pascual, M. Stress tolerance, genetic analysis and symbiotic properties of root-nodulating bacteria isolated from Mediterranean leguminous shrubs in Central Spain. Arch. Microbiol. 2009, 191, 35–46. [Google Scholar] [CrossRef]
- Fagg, C.W.; Stewart, J.L. The value of Acacia and Prosopis in arid and semi-arid environments. J. Arid. Environ. 1994, 27, 3–25. [Google Scholar] [CrossRef]
- Pérez-Fernández, M.A.; Calvo-Magro, E.; Valentine, A. Benefits of the symbiotic association of shrubby legumes for the rehabilitation of degraded soils under Mediterranean Climatic conditions. Land Degrad. Dev. 2016, 27, 395–405. [Google Scholar] [CrossRef]
- Stevens, G.G.; Pérez-Fernández, M.A.; Morcillo, R.J.L.; Kleinert, A.; Hills, P.; Brand, D.J.; Steenkamp, E.T.; Valentine, A.J. Roots and Nodules Response Differently to P Starvation in the Mediterranean-Type Legume Virgilia divaricata. Front. Plant. Sci. 2019, 10, 73. [Google Scholar] [CrossRef]
- Lal, R. Soil carbon sequestration impacts on global climate change and food security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef]
- Herrera, M.A.; Salamanca, C.P.; Barea, J.M. Mycorrhizal fungi and rhizobia to recover desertified Mediterranean ecosystems. Appl. Environ. Microbiol. 1993, 59, 129–133. [Google Scholar]
- Rodríguez-Echeverría, S.; Pérez-Fernández, M.A. Soil fertility and herb facilitation mediated by Retama sphaerocarpa. J. Veg. Sci. 2003, 14, 807–814. [Google Scholar] [CrossRef]
- Magadlela, A.; Pérez-Fernández, M.A.; Kleinert, A.; Dreyer, L.L.; Valentine, A.J. Source of inorganic N affects the cost of growth in a legume tree species (Virgilia divaricata) from the Mediterrean-type Fynbos ecosystem. J. Plant Ecol. 2016, 9, 752–761. [Google Scholar] [CrossRef]
- Pérez-Fernández, M.A.; Calvo-Magro, E.; Rodríguez-Sánchez, J.; Valentine, A. Differential growth costs and nitrogen fixation in Cytisus multiflorus (L’Hér.) Sweet and Cytisus scoparius (L.) Link are mediated by sources of inorganic N. Plant Biol. 2017, 19, 742–748. [Google Scholar]
- Vadez, V.; Lasso, J.H.; Beck, D.P.; Drevon, J.J. Variability of N2-fixation in common bean (Phaseolus vulgaris L.) under P deficiency is related to P use efficiency. Euphytica 1999, 106, 231–242. [Google Scholar] [CrossRef]
- Hogh-Jensen, H.; Schjoerring, J.K.; Soussana, J.F. The influence of phosphorus deficiency on growth and nitrogen fixation of white clover plants. Annal. Botany 2002, 90, 745–753. [Google Scholar] [CrossRef]
- Olivera, M.; Tejera, N.; Iribarne, C.; Ocana, A.; Lluch, C. Growth, nitrogen fixation and ammonium assimilation in common bean (Phaseolus vulgaris): Effect of phosphorus. Physiol. Plant 2004, 121, 498–505. [Google Scholar] [CrossRef]
- Moro, M.J.; Domingo, F.; Bermudez-de-Castro, F. Acetylene reduction activity (ARA) by the shrub legume Adenocarpus decorticans Boiss. in southern Spain (Almeria). Acta Oecol. 1992, 13, 325–333. [Google Scholar]
- Streeter, J.G. Effects of drought on nitrogen fixation in soybean root nodules. Plant Cell Environ. 2003, 26, 1199–1204. [Google Scholar] [CrossRef]
- Wadisirisuk, P.; Danso, S.K.A.; Hardarson, G.; Bowen, G.D. Influence of Bradyrhizobium japonicum location and movement on nodulation and nitrogen fixation in soybeans. Appl. Environ. Microbiol. 1989, 35, 1711–1716. [Google Scholar]
- Shamseldin, A.; Moawad, H. Inhibition of nitrogenase enzyme and completely suppression of nodulation in common bean (Phaseolus vulgaris L.) at high level of available nitrogen. Am.-Eurasian J. Agric. Environ. Sci. 2010, 7, 75–79. [Google Scholar]
- Venkateswarlu, B.; Rao, A.V.; Lahiri, A.N. Effect of water stress on nodulation and nitrogenase activity of guar (Cyamopsis tetragonoloba (L.) Taub.). Proc. Indian Acad. Sci. Plant. Sci. USA 1983, 92, 297–301. [Google Scholar]
- Abberton, M.T.; MacDuff, J.H.; Vagg, S.A.H.; Marshall, A.H.; Michaelson-Yeates, T.P.T. Nitrogen Fixation in Hybrids of White Clover (Trifolium repens L.) and Caucasian Clover (Trifolium ambiguum M. Bieb). J. Agron. Crop. Sci. 2000, 185, 241–247. [Google Scholar] [CrossRef]
- Sengupta, D.; Kannan, M.; Reddy, A.R. A root proteomics-based insight reveals dynamic regulation of root proteins under progressive drought stress and recovery in Vigna radiata (L.) Wilczek. Planta 2011, 233, 1111–1127. [Google Scholar] [CrossRef]
- López-González, G. Guía de los árboles y arbustos de la Península Ibérica y Baleares (Especies silvestres y las cultivadas más comunes), 3th ed.; Mundi-Prensa Libros, S.A.: Malaga, Spain, 2007. [Google Scholar]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method of Growing Plants Without Soil; California Agricultural Experimental Station: Berkeley, CA, USA, 1950; p. 347. [Google Scholar]
- Ogbaga, C.C.; Stepien, P.; Johnson, G.N. Sorghum (Sorghum bicolor) varieties adopt strongly contrasting strategies in response to drought. Physiol. Plant 2014, 152, 389–401. [Google Scholar] [CrossRef]
- Lachica, M.; Aguilar, A.; Yánez, J. Análisis foliar. Métodos utilizados en la Estación Experimental del Zaidín CSIC (II). An. Edaf. Agrobiol 1973, 32, 1033–1047. [Google Scholar]
- Bouat, A.; Crouzet, C. Notes techniques sur un appareil semiautomatique de clorage de l’azote et de certains composes volatiles. Ann. Agric. 1965, 16, 107–118. [Google Scholar]
- Capitan, F.; Martínez, F. Sobre la determinación espectrofotométrica de fósforo con amidol. An. Edaf. Agribiol 1954, 13, 767–790. [Google Scholar]
- Hammond, J.P.; Broadley, M.R.; White, P.J.; King, G.J.; Bowen, H.C.; Hayden, R.; Meacham, M.C.; Mead, A.; Overs, T.; Spracklen, W.P.; et al. Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits. J. Exp. Bot. 2009, 60, 1953–1968. [Google Scholar] [CrossRef]
- Bazzaz, F.A.; Grace, J. Allocation of Resources in Plants: State of Science and Critical Questions. In Plant Resource Allocation; Bazzaz, F.A., Grace, J., Eds.; Academic Press: San Diego, CA, USA, 1987; pp. 1–37. [Google Scholar]
- Ndzwanana, Z.; Tsvuura, Z.; Valentine, A.J.; Pérez-Fernández, M.A.; Magadlela, A. Differential patterns of nitrogen nutrition and growth cost of the indigenous Vachellia sieberiana and the introduced Chromolaena odorata in the savannah environment. AOB Plants 2019, 11, plz008. [Google Scholar] [CrossRef]
- Míguez-Montero, M.A.; Valentine, A.; Pérez-Fernández, M.A. Regulatory effect of phosphorus and nitrogen on nodulation and plant performance of leguminous shruas. AoB Plants 2019, in press. Available online: https://academic.oup.com/aobpla/advance-article/doi/10.1093/aobpla/plz047/5545401?searchresult=1 (accessed on 2 September 2019).
- Akinrade, E.A.; Iroh, L.; Obigbesan, G.O. Differential expression of alu- miniumtolerance mechanisms in cowpea genotypes under phosphorus limitation. J. Appl. Sci. 2006, 6, 854–859. [Google Scholar]
- Tian, Q.Y.; Chen, F.J.; Liu, J.X.; Zhang, F.S.; Mi, G.H. Inhibition of maize root growth by high nitrate supply is correlated with reduced IAA levels in roots. Plant Physiol. 2008, 165, 942–951. [Google Scholar] [CrossRef]
- López-Pintor, A.; Gómez Sal, A.; Rey Benayas, J.M. Shrubs as a source of spatial heterogeneity—The case of Retama sphaerocarpa in Mediterranean pastures of central Spain. Acta Oecol. 2006, 29, 247–255. [Google Scholar] [CrossRef]
- Pérez-Fernández, M.A.; Calvo-Magro, E.; Ramírez-Rojas, I.; Moreno Gallardo, L.; Valentine, A. Patterns of growth costs and nitrogen acquisition in Cytisus striatus (Hill)Rothm, and Cytisus balansae (Boiss.) Ball are mediated by sources of inorganic N. Plants 2016, 5, 20. [Google Scholar] [CrossRef]
- Kaschuk, G.; Xinyou, Y.; Hungria, M.; Leffelaar, P.; Giller, K.E.; Kuyper, W.T. Photosynthetic adaptation of soy bean due to varying effectiveness of N2 fixation by two distinct Bradyrhizobium japonicum strains. Environ. Exp. Bot. 2012, 76, 1–6. [Google Scholar] [CrossRef]
- Magadlela, A.; Steenkamp, E.T.; Valentine, A.J. Variable P supply affect N metabolism in a legume tree, Virgilia divaricata, from nutrient-poor Mediterranean-type ecosystems. Funct. Plant Biol. 2015, 43, 287–297. [Google Scholar] [CrossRef]
- Bedoussac, L.; Journet, E.P.; Hauggaard-Nielsen, H.; Naudin, C.; Corre-Hellou, G.; Jensen, E.S.; Prieur, L.; Justes, E. Ecological principles underlying the increase of productivity achieved by cereal-grain legume intercrops in organic farming: a review. Agron. Sustain. Dev. 2015, 35, 911–935. [Google Scholar] [CrossRef]
- Valentine, A.J.; Kleinert, A.; Benedito, V.A. Adaptive strategies for nitrogen metabolism in phosphate deficient legume nodules. Plant Sci. 2017, 256, 46–52. [Google Scholar] [CrossRef]
- Parker, M.A.; Wanda, M.; Parker, I.M. Growth of an invasive legume is symbiont limited in newly occupied habitats. Divers. Distrib. 2006, 12, 563–571. [Google Scholar] [CrossRef]
- Pérez-Fernández, M.A.; Lamont, B.B. Competition and facilitation between Australian and Spanish legumes in seven Australian soils. Plant Species Biol. 2016, 31, 256–271. [Google Scholar] [CrossRef]
- Sprent, J.I.; Gehlot, H.S. Nodulated legumes in arid and semi-arid environments: Are they important? Plant Ecol. Divers. 2010, 3, 211–219. [Google Scholar] [CrossRef]
- Lü, X.T.; Reed, S.; Yu, Q.; He, N.P.; Wang, Z.W.; Han, X.G. Convergent responses of nitrogen and phosphorus resorption to nitrogen inputs in a semiarid grassland. Glob. Chang. Biol. 2013, 19, 2775–2784. [Google Scholar] [CrossRef]
- Hardarson, G.; Danso, S.K.A. Methods for measuring biological nitrogen fixation in grain legumes. Plant Soil 1993, 152, 19–23. [Google Scholar] [CrossRef]
- Unkovich, M.; Herridge, D.; Peoples, M.; Cdisch, G.; Boddey, B.; Giller, K.; Alves, B.; Chalk, P. Measuring Plant-Associated Nitrogen Fixation in Agricultural Systems; Australian Centre for International Agricultural Research (ACIAR): Canberra, Australia, 2008; 223p.
- Vardien, W.; Valentine, A.J.; Mesjasz-Przybyłowicz, J.; Przybyłowicz, W.J.; Wang, Y.; Steenkamp, E.T. Nodules from Fynbos legume Virgilia divaricata have high functional plasticity under variable P supply levels. J. Plant Physiol. 2014, 171, 1732–1739. [Google Scholar] [CrossRef]
- Magadlela, A.; Kleinert, A.; Dreyer, L.L.; Valentine, A.J. Low phosphorus conditions affect the nitrogen nutrition and associated carbon costs of two legume tree species from a Mediterranean-type ecosystem. Aust. J. Bot. 2014, 62, 1–9. [Google Scholar] [CrossRef]
- Tsvetkova, G.E.; Georgiev, G.I. Changes in phosphate fractions extracted from different organs of phosphorus starved nitrogen fixing pea plants. J. Plant Nutr. 2007, 30, 2129–2140. [Google Scholar] [CrossRef]
- Maxwell, T.M.R.; Moir, J.L.; Edwards, G.R. Phosphorus response and efficiency of four adventive annual clovers grown in a New Zealand high country soil under glasshouse conditions. New Zeal. J. Agric. Res. 2013, 56, 203–214. [Google Scholar] [CrossRef]
- Almeida, J.P.F.; Hartwig, U.A.; Frehner, M.; Nösberger, J.; Lüscher, A. Evidence that P deficiency induces N feedback regulation of symbiotic N2 fixation in white clover (Trifolium repens). J. Exp. Bot. 2000, 51, 1289–1297. [Google Scholar] [CrossRef]
- Fageria, N.K.; Moreira, A.; Moraes, L.A.C.; Moraes, M.F. Root growth, nutrient uptake, and nutrient-use efficiency by roots of tropical legume cover crops as influenced by phosphorus fertilization. Commun. Soil. Sci. Plant 2014, 45, 555–569. [Google Scholar] [CrossRef][Green Version]
- Pang, J.; Tibbett, M.; Denton, M.D.; Lambers, H.; Siddique, K.H.M.; Bolland, M.D.A.; Revell, C.K.; Ryan, K.H. Variation in seedling growth of 11 perennial legumes in response to phosphorus supply. Plant Soil 2010, 328, 133–143. [Google Scholar] [CrossRef]
- Markham, J.H.; Zekveld, C. Nitrogen fixation makes biomass allocation to roots independent of soil nitrogen supply. Can. J. Bot. 2007, 85, 787–793. [Google Scholar] [CrossRef]
- Power, S.C.; Cramer, M.D.; Verboom, G.A.; Chimphango, S.B.M. Does phosphate acquisition constrain legume persistence in the fynbos of the Cape Floristic Region? Plant Soil 2010, 334, 33–46. [Google Scholar] [CrossRef]
- Maistry, P.M.; Muasya, A.M.; Valentine, A.J.; Chimphango, S.B.M. Increasing nitrogen supply stimulates phosphorus acquisition mechanisms in the fynbos species Aspalathus linearis. Funct. Plant Biol. 2015, 42, 52–62. [Google Scholar] [CrossRef]
- MacAlister, D.; Muthama Muasya, A.; Chimphango, S.M. Linking root traits to superior phosphorus uptake and utilization efficiency in three Fabales in the Core Cape Subregion, South Africa. Funct. Plant Biol. 2018, 45, 760–770. [Google Scholar] [CrossRef]

| Control | Inoculated | High P | Inoc + High P | Low P | Inoc + Low P | |
|---|---|---|---|---|---|---|
| Water | ||||||
| G. cinerea | 51.38 ± 1.202 a,a | 133.74 ± 9.03 b,a | 63.34 ± 4.01 c,a | 55.97 ± 6.04 c,a | 117.85 ± 9.14 a,a | 171.60 ± 8.43 b,a |
| G. florida | 14.68 ± 1.86 a,b | 138.39 ± 16.49 b,b | 16.20 ± 1.66 b,b | 207.86 ± 12.29 c,b | 82.40 ± 4.11 a,b | 155.86 ± 12.77 b,b |
| R. sphaerocarpa | 8.60 ± 0.03 a,b | 56.78 ± 3.21 b,b | 62.99 ± 10.44 a,c | 136.97 ± 14.35 c,c | 60.26 ± 1.12 a,c | 90.23 ± 7.91 b,b |
| E. barnadesii | 76.12 ± 7.87 a,c | 142.71 ± 12.67 b,a | 9.99 ± 0.65 c,d | 39.50 ± 3.08 d,a | 5.66 ± 0.63 a,c | 172.86 ± 6.14 d,b |
| Drought | ||||||
| G. cinerea | 0.41 ± 0.002 a,a | 1.06 ± 0.003 a,a | 0.40 ± 0.018 b,a | 0.74 ± 0.012 b,a | 0.94 ± 0.002 c,a | 1.36 ± 0.041 a,a |
| G. florida | 0.55 ± 0.07 a,b | 1.10 ± 0.003 b,b | 1.29 ± 0.055 c,b | 1.65 ± 0.038 c,b | 0.60 ± 0.017 a,b | 1.24 ± 0.055 b,b |
| R. sphaerocarpa | 0.07 ± 0.004 a,b | 0.45 ± 0.040 b,b | 0.50 ± 0.031 a,a | 1.09 ± 0.029 b,b | 0.48 ± 0.022 a,b | 0.72 ± 0.060 b,b |
| E. barnadesii | 0.60 ± 0.002 a,a | 1.13 ± 0.007 b,a | 0.12 ± 0.003 a,c | 0.15 ± 0.019 b,a | 0.71 ± 0.031 a,a | 1.37 ± 0.146 b,b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez-Fernández, M.; Míguez-Montero, Á.; Valentine, A. Phosphorus and Nitrogen Modulate Plant Performance in Shrubby Legumes from the Iberian Peninsula. Plants 2019, 8, 334. https://doi.org/10.3390/plants8090334
Pérez-Fernández M, Míguez-Montero Á, Valentine A. Phosphorus and Nitrogen Modulate Plant Performance in Shrubby Legumes from the Iberian Peninsula. Plants. 2019; 8(9):334. https://doi.org/10.3390/plants8090334
Chicago/Turabian StylePérez-Fernández, María, Ángel Míguez-Montero, and Alexandre Valentine. 2019. "Phosphorus and Nitrogen Modulate Plant Performance in Shrubby Legumes from the Iberian Peninsula" Plants 8, no. 9: 334. https://doi.org/10.3390/plants8090334
APA StylePérez-Fernández, M., Míguez-Montero, Á., & Valentine, A. (2019). Phosphorus and Nitrogen Modulate Plant Performance in Shrubby Legumes from the Iberian Peninsula. Plants, 8(9), 334. https://doi.org/10.3390/plants8090334





