Management of Glyphosate-Resistant Weeds in Mexican Citrus Groves: Chemical Alternatives and Economic Viability
Abstract
1. Introduction
2. Results
2.1. Initial Weed Density
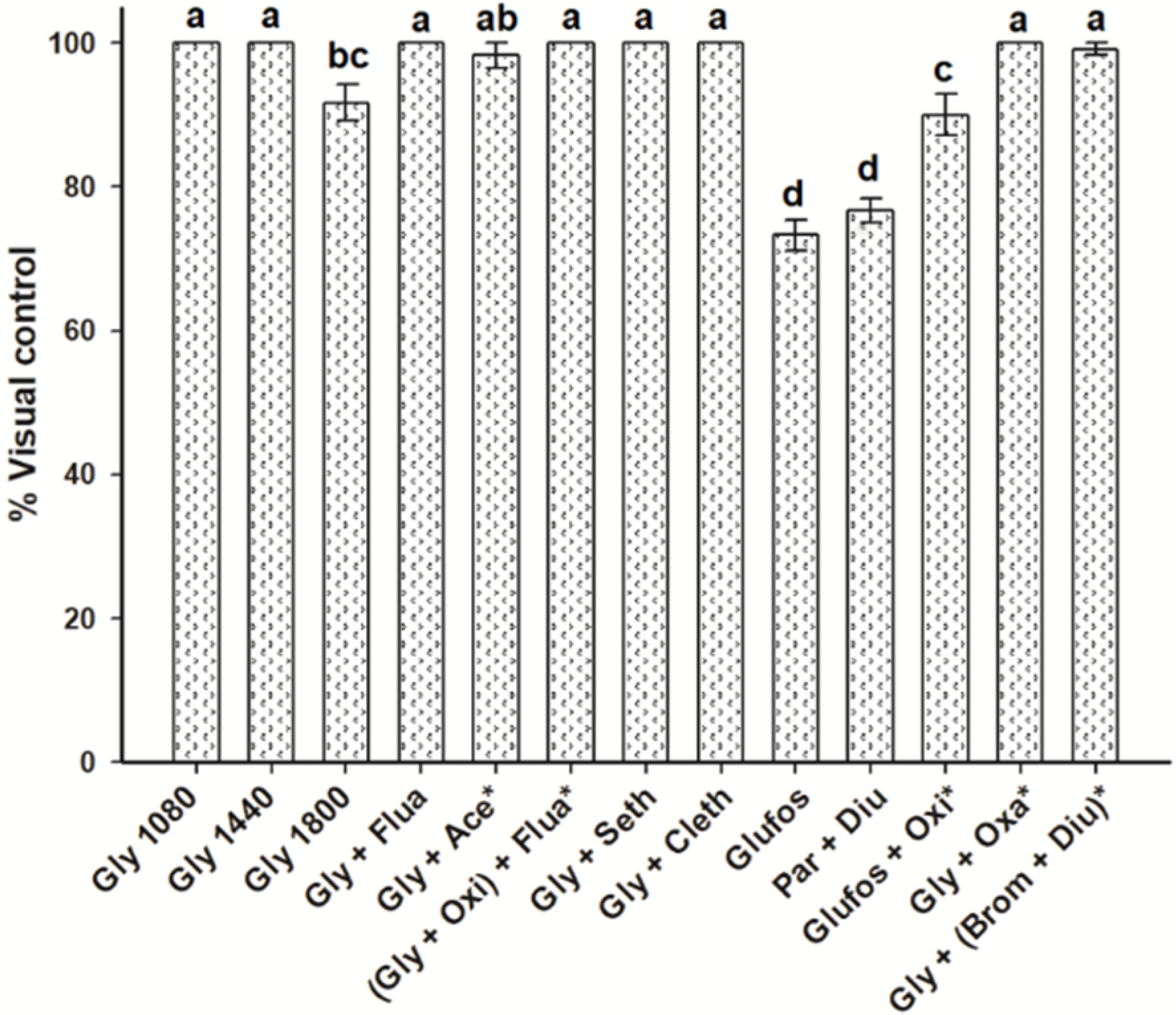
2.2. Total Control of Weeds
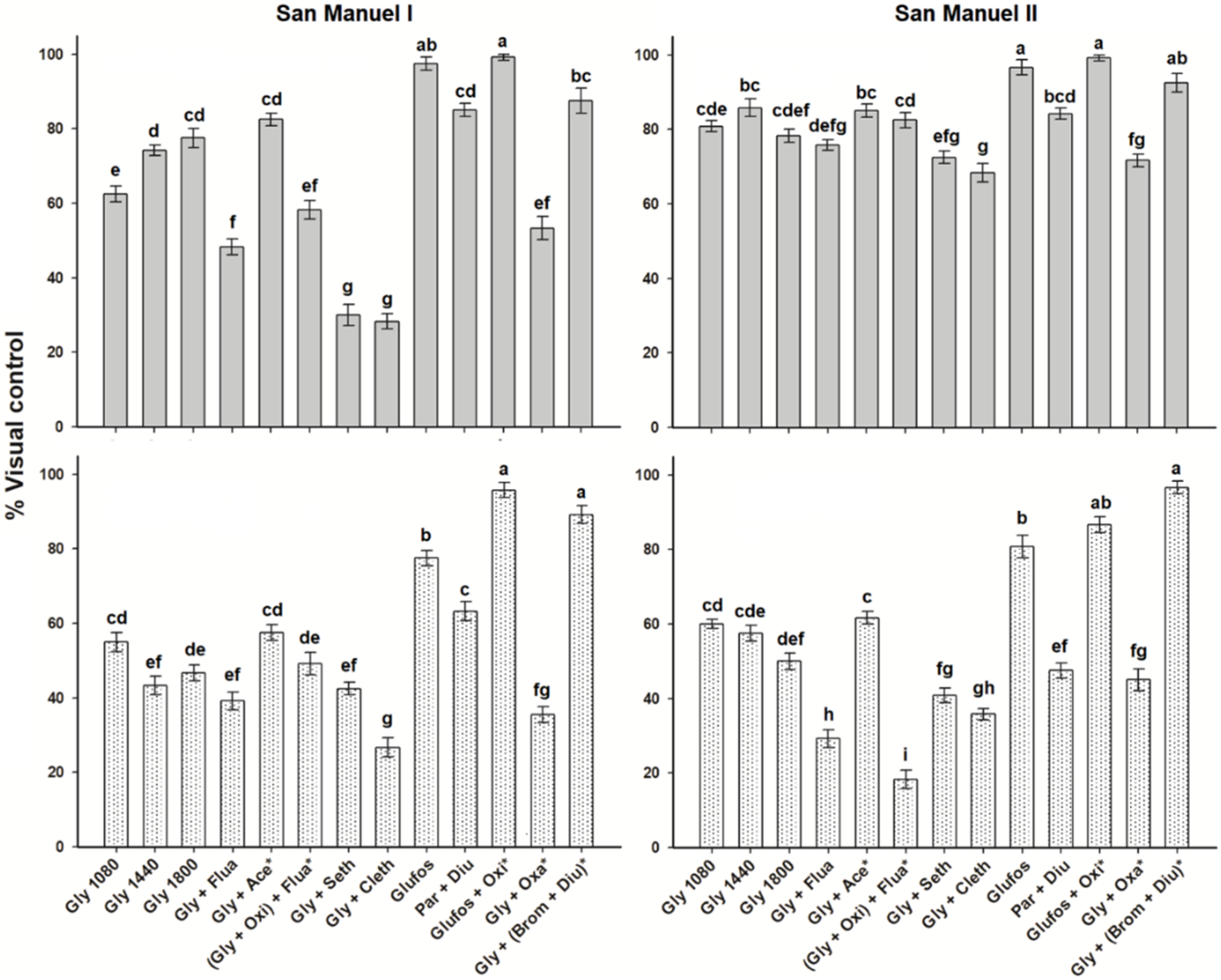
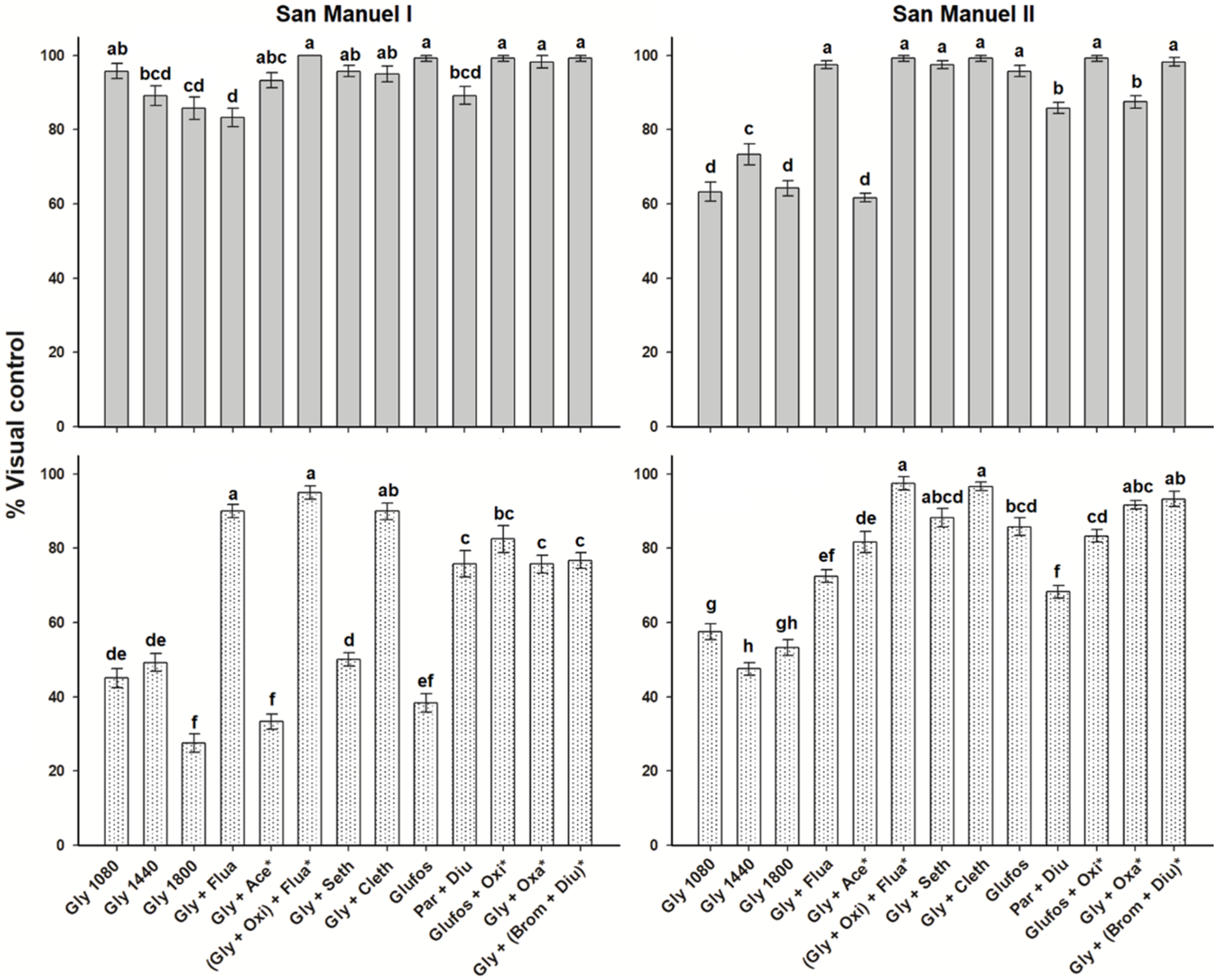
2.3. Control of Bidens Pilosa
2.4. Control of Leptochloa Virgata
2.5. Control of Echinochloa Colona
2.6. Cost of Glyphosate Resistance Management
3. Discussion
4. Materials and Methods
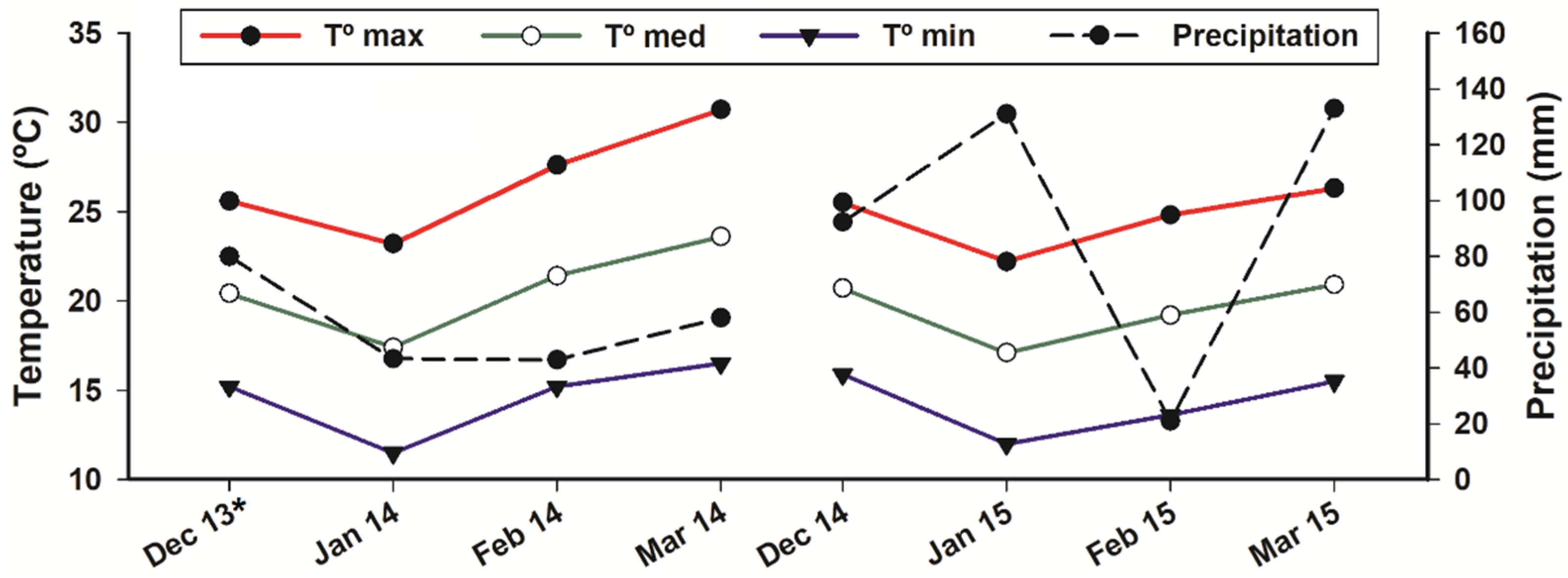
4.1. Local Data and Experimental Design
4.2. Herbicide Application
4.3. Evaluated Variables
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Food and Agriculture Data. 2018. Available online: http://www.fao.org/faostat/en/#rankings/commodities_by_country (accessed on 27 December 2018).
- SIAP-Servicio de Información Agropecuaria y Pesquera. Avance de Siembras y cosechas—Resumen Nacional por Estado. 2019. Available online: http://infosiap.siap.gob.mx:8080/agricola_siap_gobmx/AvanceNacionalCultivo.do (accessed on 1 August 2019).
- USDA-U.S. Department of Agriculture. Citrus: World Markets and Trade. 2019. Available online: http://apps.fas.usda.gov/psdonline/circulars/citrus.pdf (accessed on 1 August 2019).
- Gherekhloo, J.; Fernández-Moreno, P.T.; Alcántara-de la Cruz, R.; Sánchez-González, E.; Cruz-Hipolito, H.E.; Dominguez-Valenzuela, J.A.; De Prado, R. Pro-106-Ser mutation and EPSPS overexpression acting together simultaneously in glyphosate-resistant goosegrass (Eleusine indica). Sci. Rep. 2017, 7, 6702. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.; Mann, R.S.; Rogers, M.E.; Stelinski, L.L. Insecticide resistance in field populations of Asian citrus psyllid in Florida. Pest Manag. Sci. 2011, 67, 1258–1268. [Google Scholar] [CrossRef] [PubMed]
- Torres-Pacheco, I.; López-Arroyo, J.I.; Aguirre-Gómez, J.A.; Guevara-Gonzalez, R.G.; Yáñez-López, R.; Hernández-Zul, M.I.; Quijano-Carraza, J.A. Potential distribution in Mexico of Diaphorina citri (Hemiptera: Psyllidae) vector of huanglongbing pathogen. Fla. Entomol. 2013, 96, 36–47. [Google Scholar] [CrossRef]
- Pérez-López, M.; González-Torralva, F.; Cruz-Hipólito, H.E.; Santos, F.; Domínguez-Valenzuela, J.A.; De Prado, R. Characterization of glyphosate-resistance tropical sprangletop (Leptochloa virgata) and its alternative chemical control in Persian lime. Weed Sci. 2014, 62, 441–450. [Google Scholar] [CrossRef]
- Futch, S.H.; Singh, M. Field evaluation of chemical weed control in Florida citrus. Proc. Fla. State Hortic. Soc. 2000, 113, 68–74. [Google Scholar]
- Bracamonte, E.; Silveira, H.M.; Alcántara-de la Cruz, R.; Domínguez-Valenzuela, J.A.; Cruz-Hipólito, H.E.; De Prado, R. From tolerance to resistance: Mechanisms governing the differential response to glyphosate in Chloris barbata. Pest Manag. Sci. 2018, 74, 1118–1124. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Lambert, G.; Aguilar-Lasserre, A.; Azzaro-Pantel, C.; Miranda-Ackerman, M.A.; Purroy-Vázquez, R.; Pérez-Salazar, M.R. Behavior patterns related to the agricultural practices in the production of Persian lime (Citrus latifolia tanaka) in the seasonal grove. Comput. Electron. Agric. 2017, 116, 162–172. [Google Scholar] [CrossRef]
- Almaguer-Vargas, G.; Ayala-Garay, A.V. Adoption of innovations in ‘Persian’ lemon (Citrus latifolia tan.) in Tlapacoyan, Veracruz. Use of logbook. Rev. Chapingo Ser. Hortic. 2014, 20, 89–100. [Google Scholar] [CrossRef]
- Duke, S.O. The history and current status of glyphosate. Pest Manag. Sci. 2018, 74, 1027–1034. [Google Scholar] [CrossRef]
- Palma-Bautista, C.; Gherekhloo, J.; Domínguez-Martínez, P.A. Characterization of three glyphosate resistant Parthenium hysterophorus populations collected in citrus groves from Mexico. Pestic. Biochem. Physiol. 2019, 155, 1–7. [Google Scholar] [CrossRef]
- Syngenta. Trade Label of Touchdown (Glyphosate 62% m/v). Available online: https://www.syngenta.com.br/sites/g/files/zhg256/f/touchdown.pdf?token=1540408979 (accessed on 26 November 2018).
- Alcántara-de la Cruz, R.; Fernández-Moreno, P.T.; Ozuna, C.V.; Rojano-Delgado, A.M.; Cruz-Hipolito, H.E.; Domínguez-Valenzuela, J.A.; Barro, F.; De Prado, R. Target and non-target site mechanisms developed by glyphosate-resistant Hairy beggarticks (Bidens pilosa L.) populations from Mexico. Front. Plant. Sci. 2016, 7, 1492. [Google Scholar]
- Jhala, A.J.; Ramirez, A.H.M.; Knezevic, S.Z.; Van Damme, P.; Singh, M. Herbicide tank mixtures for broad-spectrum weed control in Florida citrus. Weed Technol. 2013, 27, 129–137. [Google Scholar] [CrossRef]
- Vanhala, P.; Kurstjens, D.; Ascard, J.; Bertram, A.; Cloutier, D.C.; Mead, A.; Raffaelli, M.; Rasmussen, J. Guidelines for physical weed control research: Flame weeding, weed harrowing and intra-row cultivation. In Proceedings of the 6th EWRS—Workshop on Physical and Cultural Weed Control, Lillehammer, Norway, 8–10 March 2004; pp. 208–239. [Google Scholar]
- Alcántara-de la Cruz, R.; Rojano-Delgado, A.M.; Giménez, M.J.; Cruz-Hipolito, H.E.; Domínguez-Valenzuela, J.A.; Barro, F.; De Prado, R. First resistance mechanisms characterization in glyphosate-resistant Leptochloa virgata. Front. Plant. Sci. 2016, 7, 1742. [Google Scholar] [CrossRef] [PubMed]
- Marshall, E.J.P.; Brown, V.K.; Boatman, N.D.; Lutman, P.J.W.; Squire, G.R.; Ward, L.K. The role of weeds in supporting biological diversity within crop fields. Weed Res. 2003, 43, 77–89. [Google Scholar] [CrossRef]
- Pollnac, F.W.; Maxwell, B.D.; Menalled, F.D. Weed community characteristics and crop performance: A neighbourhood approach. Weed Res. 2009, 49, 242–250. [Google Scholar] [CrossRef]
- Alonso-Ayuso, M.; Gabriel, J.L.; García-González, I.; Del Monte, J.P.; Quemada, M. Weed density and diversity in a long-term cover crop experiment background. Crop. Prot. 2018, 112, 103–111. [Google Scholar] [CrossRef]
- Alcántara-de la Cruz, R.; Romano, Y.; Osuna-Ruíz, M.D.; Domínguez-Valenzuela, J.A.; Menendez, J.; De Prado, R. Genetic relationships between tropical sprangletop (Leptochloa virgata) populations from Mexico: Understanding glyphosate resistance spread. Weed Sci. 2016, 64, 579–587. [Google Scholar] [CrossRef]
- Bagavathiannan, M.V.; Davis, A.S. An ecological perspective on managing weeds during the great selection for herbicide resistance. Pest Manag Sci. 2018, 74, 2277–2286. [Google Scholar] [CrossRef] [PubMed]
- Hicks, H.L.; Comont, D.; Coutts, S.R.; Crook, L.; Hull, R.; Norris, K.; Neve, P.; Childs, D.Z.; Freckleton, R.P. The factors driving evolved herbicide resistance at a national scale. Nat. Ecol. Evol. 2018, 2, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Shergill, L.S.; Bish, M.D.; Biggs, M.E.; Bradley, K.W. Monitoring the changes in weed populations in a continuous glyphosate- and dicamba-resistant soybean system: A five-year field-scale investigation. Weed Technol. 2018, 32, 166–173. [Google Scholar] [CrossRef]
- Reddy, K.N.; Singh, M. Germination and emergence of hairy beggarticks (Bidens pilosa). Weed Sci. 1992, 40, 195–199. [Google Scholar] [CrossRef]
- Ganie, Z.A.; Jhala, A.J. Interaction of 2,4-D or dicamba with glufosinate for control of glyphosate-resistant giant ragweed (Ambrosia trifida L.) in glufosinate-resistant maize (Zea mays L.). Front. Plant. Sci. 2017, 8, 1207. [Google Scholar] [CrossRef] [PubMed]
- Bond, J.A.; Eubank, T.W.; Bond, R.C.; Golden, B.R.; Edwards, H.M. Glyphosate-resistant Italian ryegrass (Lolium perenne ssp. multiflorum) control with fall-applied residual herbicides. Weed Technol. 2014, 28, 361–370. [Google Scholar] [CrossRef]
- Ervin, D.E.; Breshears, E.H.; Frisvold, G.B.; Hurley, T.; Dentzman, K.E.; Gunsolus, J.L.; Jussaume, R.A.; Owen, M.D.K.; Norswrothy, J.K.; Al Mamun, M.M.; et al. Farmer attitudes toward cooperative approaches to herbicide resistance management: A common pool ecosystem service challenge. Ecol. Econ. 2019, 157, 237–245. [Google Scholar] [CrossRef]
- Moss, S. Integrated Weed Management (IWM): Why are farmers reluctant to adopt non-chemical alternatives to herbicides? Pest Manag. Sci. 2019, 75, 1205–1211. [Google Scholar] [CrossRef] [PubMed]
- Fried, G.; Cordeau, S.; Metay, A.; Kazakou, E. Relative importance of environmental factors and farming practices in shaping weed communities structure and composition in French vineyards. Agric. Ecosyst. Environ. 2019, 275, 1–13. [Google Scholar] [CrossRef]
- Singh, M.; Malik, M.S.; Ramirez, A.H.M.; Jhala, A.J. Tank mix of saflufenacil with glyphosate and pendimethalin for weed control in Florida citrus. Hortic. Technol. 2011, 21, 606–615. [Google Scholar]
- Lamichhane, J.R.; Devos, Y.; Beckie, H.J.; Owen, M.D.K.; Tillie, P.; Messéan, A.; Kudsk, P. Integrated weed management systems with herbicide-tolerant crops in the European Union: Lessons learnt from home and abroad. Crit. Rev. Biotechnol. 2017, 37, 459–475. [Google Scholar] [CrossRef]
- Livingston, M.; Fernandez-Cornejo, J.; Frisvold, G.B. Economic returns to herbicide resistance management in the short and long run: The role of neighbor effects. Weed Sci. 2016, 64, 595–608. [Google Scholar] [CrossRef]
- Haring, S.C.; Flessner, M.L. Improving soil seed bank management. Pest Manag. Sci. 2018, 74, 2412–2418. [Google Scholar] [CrossRef]
- SMN—Servicio Meteorológico Nacional. Información climatólogica por Estado, Acateno–21143. 2019. Available online: https://smn.conagua.gob.mx/tools/RESOURCES/Mensuales/pue/00021143.TXT (accessed on 20 August 2019).
- Burril, L.C.; Cárdenas, L.; Locatelli, E. Field Manual for Weed Control Research, 1st ed.; Oregon State University Press: Corvallis, OR, USA, 1976; p. 63. [Google Scholar]

| Treatments 1 | MOA 2 | Rate | Liters | Time | USD 3 | |
|---|---|---|---|---|---|---|
| - | Control | - | - | - | - | - |
| 1 | Gly 1080 | EPSPS | 1080 | 3 | POST | 29.0 |
| 2 | Gly 1440 | EPSPS | 1440 | 4 | POST | 38.7 |
| 3 | Gly 1800 | EPSPS | 1800 | 5 | POST | 48.4 |
| 4 | Gly + Flua | EPSPS + ACCase | 1080 + 250 | 3 + 2 | POST | 100.9 |
| 5 | Gly + Ace † | EPSPS + Mitosis | 1080 + 1678 | 3 + 2 | POST+PRE | 57.3 |
| 6 | (Gly + Oxi) + Flua † | (EPSPS+PPO) + ACCase | (1080 + 480) + 250 | (3 + 2) + 2 | POST+PRE | 97.4 |
| 7 | Gly + Seth | EPSPS + ACCase | 1080 + 368 | 3 + 2 | POST | 82.0 |
| 8 | Gly + Cleth | EPSPS + ACCase | 1080 + 236 | 3 + 2 | POST | 70.0 |
| 9 | Glufos | GS | 450 | 2 | POST | 54.1 |
| 10 | Par + Diu | PSI + PSII | 400 + 200 | 2 | POST | 26.8 |
| 11 | Glufos + Oxi † | GS + PPO | 420 + 480 | 2 + 2 | POST + PRE | 134.6 |
| 12 | Gly + Oxa † | GS + PPO | 1080 + 1000 | 4 + 3 | POST + PRE | 112.0 |
| 13 | Gly + (Brom + Diu) † | EPSPS + (PSII + PSII) | 1080 + (1200 + 1200) | 3 + 3 | POST + PRE | 142.4 |
| Species | San Manuel I | San Manuel II | ||
|---|---|---|---|---|
| 2014 | 2015 | 2014 | 2015 | |
| B. pilosa | 114.8 ± 4.6 | 117.3 ± 3.7 | 195.9 ± 8.6 | 182.5 ± 6.3 |
| L. virgata | 98.7 ± 4.7 | 104.8 ± 3.4 | 49.8 ± 3.0 | 58.7 ± 4.6 |
| E. colona | 7.3 ± 3.2 | 11.4 ± 2.08 | 96.7 ± 5.9 | 87.3 ± 4.1 |
| Other weeds | 19.6 ± 2.6 | 15.6 ± 1.7 | 8.0 ± 2.1 | 12.4 ± 3.8 |
| Total | 237.7 | 248.1 | 350.3 | 340.9 |
| Treatment 1 | 15 DAT | 30 DAT | 45 DAT | 60 DAT | 75 DAT |
|---|---|---|---|---|---|
| San Manuel I | |||||
| Control | - | - | - | - | - |
| Gly 1080 | 74.2 ± 2.4 c | 64.2 ± 1.5 e | 68.3 ± 1.7 d | 48.3 ± 2.8 ef | 41.7 ± 2.5 ef |
| Gly 1440 | 72.5 ± 3.1 c | 78.3 ± 1.1 cd | 73.3 ± 2.1 cd | 64.2 ± 3.3 cd | 49.2 ± 2.0 de |
| Gly 1800 | 84.2 ± 2.0 b | 76.7 ± 2.1 d | 78.3 ±1.1 c | 62.5 ± 2.8 cd | 55.0 ± 1.8 cd |
| Gly + Flua | 68.3 ± 1.7 c | 51.7 ± 1.7 f | 53.3 ± 3.1 ef | 41.7 ± 2.5 f | 35.8 ± 2.0 f |
| Gly + Ace † | 69.2 ± 2.4 c | 84.2 ± 2.0 bcd | 71.7 ± 1.7 cd | 59.2 ± 3.0 de | 63.3 ± 2.1 c |
| (Gly + Oxi) + Flua † | 71.7 ± 2.5 c | 64.2 ± 1.5 e | 70.8 ± 1.5 cd | 64.2 ± 2.4 cd | 52.5 ± 1.7 d |
| Gly + Seth | 73.3 ± 2.1 c | 37.5 ± 1.7 g | 58.3 ± 2.1 e | 55.0 ± 2.2 de | 45.0 ± 1.8 def |
| Gly + Cleth | 66.7 ± 1.7 c | 34.2 ± 2.7 g | 46.7 ± 2.5 f | 42.5 ± 2.1 f | 20.0 ± 2.9 g |
| Glufos | 100.0 ± 0 a | 97.5 ± 1.1 a | 88.3 ± 2.1 b | 85.8 ± 1.5 b | 77.5 ± 1.1 b |
| Par + Diu | 86.7 ± 1.1 b | 86.7 ± 2.1 bc | 74.2 ± 0.8 cd | 70.8 ± 0.8 cd | 54.2 ± 2.4 cd |
| Glufos + Oxi † | 100.0 ± 0 a | 99.2 ± 0.8 a | 97.5 ± 1.8 a | 96.3 ± 1.7 a | 91.7 ± 1.7 a |
| Gly + Oxa † | 65.0 ± 1.3 c | 68.3 ± 2.5 de | 59.2 ± 1.5 e | 38.3 ± 2.5 f | 35.8 ± 2.4 f |
| Gly + (Brom + Diu) † | 70.8 ± 2.7 c | 88.3 ± 1.7 b | 91.7 ± 2.5 ab | 92.5 ± 2.5 ab | 89.2 ± 2.7 a |
| San Manuel II | |||||
| Control | - | - | - | - | |
| Gly 1080 | 66.7 ± 2.5 cd | 80.8 ± 4.0 de | 69.2 ± 1.5 bc | 61.7 ± 3.3 cd | 43.3 ± 2.1 ef |
| Gly 1440 | 76.7 ± 2.1 b | 80.8 ± 1.5 de | 69.2 ± 2.0 bc | 70.8 ± 2.0 bc | 51.7 ± 2.5 de |
| Gly 1800 | 75.0 ± 2.6 bc | 85.8 ± 2.0 cde | 76.7 ± 2.1 b | 74.2 ± 3.0 b | 57.5 ± 2.1 cd |
| Gly + Flua | 48.3 ± 1.1 fg | 80.0 ± 2.9 de | 31.7 ± 2.8 f | 48.3 ± 2.5 e | 39.2 ± 2.0 f |
| Gly + Ace † | 75.8 ± 3.0 bc | 85.0 ± 1.8 cde | 88.3 ± 2.5 a | 87.5 ± 2.5 a | 65.8 ± 1.5 c |
| (Gly + Oxi) + Flua † | 53.3 ± 2.1 ef | 86.7 ± 2.5 bcd | 42.5 ± 1.1 de | 36.7 ± 2.1 f | 27.5 ± 2.8 g |
| Gly + Seth | 40.8 ± 2.4 g | 77.5 ± 1.1 de | 45.8 ± 2.7 de | 52.5 ± 1.7 de | 44.4 ± 1.5 ef |
| Gly + Cleth | 51.7 ± 1.1 ef | 75.8 ± 1.5 e | 36.7 ± 3.1 ef | 49.2 ± 2.4 e | 40.8 ± 1.8 f |
| Glufos | 99.2 ± 0.8 a | 96.7 ± 1.7 ab | 93.3 ± 1.7 a | 88.3 ± 1.7 a | 79.2 ± 2.0 b |
| Par + Diu | 98.3 ± 1.1 a | 85.8 ± 1.5 cde | 63.3 ± 2.1 c | 57.5 ± 1.7 de | 48.3 ± 1.7 def |
| Glufos + Oxi † | 100.0 ± 0 a | 99.2 ± 0.8 a | 97.5 ± 1.7 a | 94.2 ± 2.4 a | 90.8 ± 1.5 a |
| Gly + Oxa † | 58.3 ± 2.8 de | 76.7 ± 1.7 d | 50.8 ± 3.0 d | 54.2 ± 2.0 de | 45.0 ± 1.8 ef |
| Gly + (Brom + Diu) † | 74.2 ± 2.0 bc | 94.2 ± 2.0 abc | 95.8 ± 2.4 a | 96.7 ± 2.1 a | 90.8 ± 2.7 a |
| Treatment 1 | Cost | R-$/day | % Inc | San Manuel I | San Manuel II | ||
|---|---|---|---|---|---|---|---|
| DAT > 80% | SC $/day | DAT > 80% | SC $/day | ||||
| Control | - | - | - | - | - | - | - |
| Gly 1080 | 29.0 | 0.38 | RT | - | - | 30 | 0.96 |
| Gly 1440 | 38.7 | 0.51 | 33 | - | - | 30 | 1.29 |
| Gly 1800 | 48.4 | 0.65 | 66 | 15 | 3.22 | 30 | 1.61 |
| Gly + Flua | 100.9 | 1.35 | 247 | - | - | 30 | 3.36 |
| Gly + Ace † | 57.3 | 0.76 | 97 | 30 | 1.91 | 60 | 0.95 |
| (Gly + Oxi) + Flua † | 97.4 | 1.30 | 183 | - | - | 30 | - |
| Gly + Seth | 82.0 | 1.09 | 236 | - | - | - | - |
| Gly + Cleth | 70.0 | 0.93 | 141 | - | - | - | - |
| Glufos | 81.2 | 1.08 | 86 | 60 | 1.35 | 75 | 1.08 |
| Par + Diu | 26.8 | 0.36 | −8 | 30 | 0.89 | 30 | 0.89 |
| Glufos + Oxi † | 134.6 | 1.79 | 364 | 75 | 1.79 | 75 | 1.79 |
| Gly + Oxa † | 112.0 | 1.49 | 286 | - | - | - | - |
| Gly + (Brom + Diu) † | 142.4 | 1.89 | 391 | 75 | 1.89 | 75 | 1.89 |
| Field Activity | Date | |
|---|---|---|
| 2014 | 2015 | |
| Weed mechanical mowing in both experimental plots | 16 December 2013 | 19 December 2014 |
| Initial counting of plants of each weed species | 9 January2014 | 15 January 2015 |
| Application of POST herbicides | 10 January2014 | 16 January 2015 |
| Application of PRE herbicides (treatments 5, 6, 11, 12, and 13), and evaluation at 15 DAT | 25 January 2014 | 31 January 2015 |
| Evaluation at 30 DAT | 9 February 2014 | 15 February 2015 |
| Evaluation at 45 DAT | 23 February 2014 | 1 March 2015 |
| Evaluation at 60 DAT | 9 March 2014 | 15 March 2015 |
| Evaluation at 75 DAT | 24 March 2014 | 29 March 2015 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alcántara-de la Cruz, R.; Domínguez-Martínez, P.A.; da Silveira, H.M.; Cruz-Hipólito, H.E.; Palma-Bautista, C.; Vázquez-García, J.G.; Domínguez-Valenzuela, J.A.; De Prado, R. Management of Glyphosate-Resistant Weeds in Mexican Citrus Groves: Chemical Alternatives and Economic Viability. Plants 2019, 8, 325. https://doi.org/10.3390/plants8090325
Alcántara-de la Cruz R, Domínguez-Martínez PA, da Silveira HM, Cruz-Hipólito HE, Palma-Bautista C, Vázquez-García JG, Domínguez-Valenzuela JA, De Prado R. Management of Glyphosate-Resistant Weeds in Mexican Citrus Groves: Chemical Alternatives and Economic Viability. Plants. 2019; 8(9):325. https://doi.org/10.3390/plants8090325
Chicago/Turabian StyleAlcántara-de la Cruz, Ricardo, Pablo Alfredo Domínguez-Martínez, Hellen Martins da Silveira, Hugo Enrique Cruz-Hipólito, Candelario Palma-Bautista, José Guadalupe Vázquez-García, José Alfredo Domínguez-Valenzuela, and Rafael De Prado. 2019. "Management of Glyphosate-Resistant Weeds in Mexican Citrus Groves: Chemical Alternatives and Economic Viability" Plants 8, no. 9: 325. https://doi.org/10.3390/plants8090325
APA StyleAlcántara-de la Cruz, R., Domínguez-Martínez, P. A., da Silveira, H. M., Cruz-Hipólito, H. E., Palma-Bautista, C., Vázquez-García, J. G., Domínguez-Valenzuela, J. A., & De Prado, R. (2019). Management of Glyphosate-Resistant Weeds in Mexican Citrus Groves: Chemical Alternatives and Economic Viability. Plants, 8(9), 325. https://doi.org/10.3390/plants8090325








