Smoke-Water Enhances Germination and Seedling Growth of Four Horticultural Crops
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Production of Plant-Derived Smoke-Water
2.3. First Experiment
2.3.1. α-Amylase Activity
2.3.2. Extraction and Determination of Abscisic Acid Content
2.4. Second Experiment
2.5. Mineral Nutrition Analysis
2.6. Data Analysis
3. Results
3.1. First Experiment: Effects of Smoke-Water and Light versus Darkness on Germination Parameters, α-Amylase Activity, and Abscisic Acid Content
3.2. Second Experiment: Effects of Smoke-Water on Growth Parameters and Macroelement Contents
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Han, C.; Yang, P. Studies on the molecular mechanisms of seed germination. Proteomics 2015, 15, 1671–1679. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, M.; Light, M.; Van Staden, J. Plant-derived smoke: Old technology with possibilities for economic applications in agriculture and horticulture. S. Afr. J. Bot. 2011, 77, 972–979. [Google Scholar] [CrossRef]
- Mandal, K.; Misra, A.; Hati, K.; Bandyopadhyay, K.; Ghosh, P.; Mohanty, M. Rice residue-management options and effects on soil properties and crop productivity. J. Food Agric. Environ. 2004, 2, 224–231. [Google Scholar]
- De Lange, J.; Boucher, C. Autecological studies on Audouinia capitata (Bruniaceae). I. Plant-derived smoke as a seed germination cue. S. Afr. J. Bot. 1990, 56, 700–703. [Google Scholar] [CrossRef]
- Read, T.; Bellairs, S.; Mulligan, D.; Lamb, D. Smoke and heat effect on soil seed bank germination for the re-establishment of a native forest community in New South Wales. Aust. J. Ecol. 2000, 25, 48–57. [Google Scholar] [CrossRef]
- Van Staden, J.; Jäger, A.; Light, M.; Burger, B. Isolation of the major germination cue from plant-derived smoke. S. Afr. J. Bot. 2004, 70, 654–659. [Google Scholar] [CrossRef]
- Kulkarni, M.; Sparg, S.; Light, M.; Van Staden, J. Stimulation of rice (Oryza sativa L.) seedling vigour by smoke-water and butenolide (3-methyl- 2H-furo [2, 3-c] pyran-2-one). J. Agric. Crop Sci. 2006, 192, 395–398. [Google Scholar] [CrossRef]
- Cox, R.; Chou, Y.; Wester, D. Smoke water and heat influence emergence of short grass prairie species. Fire Ecol. 2017, 13, 138–148. [Google Scholar] [CrossRef]
- Keeley, J.E.; Fotheringham, C.J. Smoke-induced seed germination of California chaparral. Ecology 1998, 79, 2320–2336. [Google Scholar] [CrossRef]
- Dixon, K.; Merritt, D.; Flematti, G.; Ghisalberti, E. Karrikinolide—A phytoreactive compound derived from smoke with applications in horticulture, ecological restoration and agriculture. Acta Hortic. 2009, 813, 155–170. [Google Scholar] [CrossRef]
- Flematti, G.; Ghisalberti, E.; Dixon, K.; Trengove, R. A compound from smoke that promotes seed germination. Science 2004, 305, 977. [Google Scholar] [CrossRef]
- Flematti, G.R.; Merritt, D.J.; Piggott, M.J.; Trengove, R.D.; Smith, S.M.; Dixon, K.W.; Ghisalberti, E.L. Burning vegetation produces cyanohydrins that liberate cyanide and stimulate seed germination. Nat. Commun. 2011, 2, 6150. [Google Scholar] [CrossRef]
- Light, M.; Burger, B.; Staerk, D.; Kohout, L.; Van Staden, J. Butenolides from Plant-Derived Smoke: Natural Plant-Growth Regulators with Antagonistic Actions on Seed Germination. J. Nat. Prod. 2010, 73, 267–269. [Google Scholar] [CrossRef]
- Kamran, M.; Khan, A.L.; Ali, L.; Hussain, J.; Waqas, M.; Al-Harrasi, A.; Imran, Q.M.; Kim, Y.-H.; Kang, S.-M.; Yun, B.; et al. Hydroquinone; A Novel Bioactive Compound from Plant-Derived Smoke Can Cue Seed Germination of Lettuce. Front. Chem. 2017, 5, 30. [Google Scholar] [CrossRef]
- Sparg, S.; Kulkarni, M.; Light, M.; Van Staden, J. Improving seedling vigor of indigenous medicinal plants with smoke. Bioresour Technol. 2005, 96, 1323–1330. [Google Scholar] [CrossRef]
- Stevens, J.; Merritt, D.; Flematti, G.; Ghisalberti, E.; Dixon, K. Seed germination of agricultural weeds is promoted by the butenolide 3-methyl-2 H-furo [2, 3- c] pyran-2-one under laboratory and field conditions. Plant Soil 2007, 298, 113–124. [Google Scholar] [CrossRef]
- Thomas, T.H.; Van Staden, J. Dormancy break of celery (Apium graveolens L.) seeds by plant-derived smoke extract. Plant Growth Regul. 1995, 17, 195–198. [Google Scholar] [CrossRef]
- Drewes, F.E.; Smith, M.T.; Van Staden, J. The effect of plant-derived smoke extract on the germination of light-sensitive lettuce seed. Plant Growth Regul. 1995, 16, 205–209. [Google Scholar] [CrossRef]
- Kulkarni, M.; Ascough, G.; Van Staden, J. Effects of Foliar Applications of Smoke-Water and a Smoke-Isolated Butenolide on Seedling Growth of Okra and Tomato. HortScience 2007, 42, 179–182. [Google Scholar] [CrossRef]
- Graeber, K.; Nakabayashi, K.; Miatton, E.; Leubner-Metzger, G.; Soppe, W. Molecular mechanisms of seed dormancy. Plant Cell Environ. 2012, 35, 1769–1786. [Google Scholar] [CrossRef]
- Miransari, M.; Smith, D.L. Plant hormones and seed germination. Environ. Exp. Bot. 2014, 99, 110–121. [Google Scholar] [CrossRef]
- Meng, Y.; Shuai, H.; Luo, X.; Chen, F.; Zhou, W.; Yang, W.; Shu, K. Karrikins: Regulators Involved in Phytohormone Signaling Networks during Seed Germination and Seedling Development. Front. Plant. Sci. 2017, 7, 2021. [Google Scholar] [CrossRef]
- Yamaguchi, S. Gibberellin metabolism and its regulation. Annu. Rev. Plant Biol. 2008, 59, 225–251. [Google Scholar] [CrossRef]
- Matilla, A.J.; Matilla-Vazquez, M.A. Involvement of ethylene in seed physiology. Plant Sci. 2008, 175, 87–97. [Google Scholar] [CrossRef]
- Cembrowska-Lech, D.; Kępczyński, J. Plant-derived smoke induced activity of amylases, DNA replication and b-tubulin accumulation before radicle protrusion of dormant Avena fatua L. caryopses. Acta Physiol. Plant. 2017, 39, 39. [Google Scholar] [CrossRef]
- Chiwocha, S.D.S.; Dixon, K.W.; Flematti, G.R.; Ghisalberti, E.L.; Merritt, D.J.; Nelson, D.C.; Riseborough, J.A.M.; Smith, S.M.; Stevens, J.C. Karrikins: A new family of plant growth regulators in smoke. Plant Sci. 2009, 177, 252–256. [Google Scholar] [CrossRef]
- Ren, L.; Bai, Y.; Reaney, M. Evidence of different compounds in smoke derived from legumes and grasses acting on seed germination and seedling emergence. Seed Sci. Res. 2017, 27, 154–164. [Google Scholar] [CrossRef]
- Nelson, D.; Flematti, G.; Riseborough, J.; Ghisalberti, E.; Dixon, K.; Smith, S. Karrikins enhance light responses during germination and seedling development in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2010, 107, 7095–7100. [Google Scholar] [CrossRef]
- Moreira, B.; Pausas, J.G. Shedding light through the smoke on the germination of Mediterranean Basin flora. S. Afr. J. Bot. 2018, 115, 244–250. [Google Scholar] [CrossRef]
- Jäger, A.; Light, M.; Van Staden, J. Effects of source of plant material and temperature on the production of smoke extracts that promote germination of light-sensitive lettuce seeds. Environ. Exp. Bot. 1996, 36, 421–429. [Google Scholar] [CrossRef]
- Catav, S.S.; Bekar, I.; Ates, B.S.; Ergan, G.; Oymak, F.; Ulker, E.D.; Tavsanoglu, C. Germination response of five eastern Mediterranean woody species to smoke solution derived from various plant. Turk. J. Bot. 2012, 36, 480–487. [Google Scholar]
- Smith, C.; Perfetti, T.; Garg, R.; Hansch, C. IARC carcinogens reported in cigarette mainstream smoke and their calculated log P values. Food Chem. Toxicol. 2003, 41, 807–817. [Google Scholar] [CrossRef]
- Ren, L.; Bai, Y. Smoke and Ash Effects on Seedling Emergence from Germinable Soil Seed Bank in Fescue Prairie. Rangel. Ecol. Manag. 2016, 69, 499–507. [Google Scholar] [CrossRef]
- Brown, N.; Van Staden, J. Smoke as a germination cue. A review. Plant Growth Regul. 1997, 22, 115–124. [Google Scholar] [CrossRef]
- Waters, M.; Scaffidi, A.; Flematti, G.; Smith, S. The origins and mechanisms of karrikin signalling. Curr. Opin. Plant Biol. 2013, 16, 667–673. [Google Scholar] [CrossRef]
- Daws, M.; Davies, J.; Pritchard, H.; Brown, N.; Van Staden, J. Butenolide from plant-derived smoke enhances germination and seedling growth of arable weed species. Plant Growth Regul. 2007, 51, 73–82. [Google Scholar] [CrossRef]
- Baxter, B.; Van Staden, J.; Granger, J.; Brown, N.A.C. Plant-derived smoke and smoke extracts stimulate seed germination of the fire-climax grass the medatriandra Forssk. Environ. Exp. Bot. 1994, 34, 217–223. [Google Scholar] [CrossRef]
- Coons, J.; Coutant, N.; Lawrence, B.; Finn, D.; Finn, S. An effective system to produce smoke solutions from dried plant tissue for seed germination studies. Appl. Plant Sci. 2014, 2, 1300097. [Google Scholar] [CrossRef]
- Dayamba, S.; Sawadogo, L.; Tigabu, M.; Savadogo, P.; Zida, D.; Tiveau, D.; Oden, P. Effects of aqueous smoke solutions and heat on seed germination of herbaceous species of the Sudanian savanna-woodland in Burkina Faso. Flora 2010, 205, 319–325. [Google Scholar] [CrossRef]
- Raun, S.; Xue, Q.; Thlkowska, K. Effect of seed priming on germination and health of rice (OryzasativaL) seeds. Seed Sci. Technol. 2002, 30, 451–458. [Google Scholar]
- Kamran, M.; Khan, A.; Waqas, M.; Imran, Q.; Hamayun, M.; Kang, S.; Kim, Y.; Kim, M.; Lee, I. Effects of plant-derived smoke on the growth dynamics of Barnyard Grass (Echinochloa crus-galli). Acta Agric. Scand. 2014, 64, 121–128. [Google Scholar] [CrossRef]
- Qi, Q.G.; Rose, P.; Abrams, G.; Taylor, D.; Abrams, S.; Cutler, A. Abscisic acid metabolism, 3-ketoacyl- coenzyme a synthase gene expression and very long-chain monounsaturated fatty acid biosynthesis in Brassica napus embryos. Plant Physiol. 1998, 117, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Xum, Q. Separation and determination of three plant internal hormones by gas chromatography. Se pu = Chin. J. Chromatogr. 2000, 18, 67–69. [Google Scholar]
- Dhindwal, A.; Lather, B.; Singh, J. Efficacy of seed treatment on germination, seedling emergence and vigor of cotton (Gossypium hirsutum) genotypes. Seed Res. 1991, 19, 59–61. [Google Scholar]
- Jackson, M. Soil Chemical Analysis; Prentice Hall: Englewood Cliffs, NJ, USA, 1973. [Google Scholar]
- AOAC. Official method 975.03 Metal in plants. In Official Methods of Analysis of AOAC International, 18th ed.; Association of Official Analytical Chemists: Arlington, VA, USA, 1995. [Google Scholar]
- De Mendiburu, F. agricolae: Statistical Procedures for Agricultural Research. R Package Version 1.2-8. 2017. Available online: https://CRAN.R-project.org/package=agricolae (accessed on 10 April 2019).
- Iqbal, M.; Asif, S.; Ilyas, N.; Raja, N.; Hussain, M.; Shabir, S.; Faz, M.; Rauf, A. Effect of Plant Derived Smoke on Germination and Post Germination Expression of Wheat (Triticumae stivum L). Am. J. Plant Sci. 2016, 7, 806–813. [Google Scholar] [CrossRef]
- Kulkarni, M.; Ascough, G.; Verschaeve, L.; Baeten, K.; Arruda, M.; Van Staden, J. Effect of Smoke Water and a Smoke-Isolated Butenolide on the Growth and Gene Toxicity of Commercial Onion. Sci. Hortic. 2010, 124, 434–439. [Google Scholar] [CrossRef]
- Ghebrehiwot, H.M.; Kulkarni, M.G.; Kirkman, K.P.; Van Staden, J. Smoke-water and a smoke-isolated butenolide improve germination and seedling vigour of Eragrostis tef (Zucc.) Trotter under high temperature and low osmotic potential. J. Agron. Crop Sci. 2008, 194, 270–277. [Google Scholar] [CrossRef]
- Chumpookam, J.; Lin, H.L.; Shiesh, C.C. Effect of smoke-water on seed germination and seedling growth of papaya (Carica papaya cv. Tainung No. 2). HortScience 2012, 47, 741–744. [Google Scholar] [CrossRef]
- Long, R.L.; Stevens, J.C.; Griffiths, E.M.; Adamek, M.; Powles, S.B.; Merritt, D.J. Detecting karrikinolide responses in seeds of the Poaceae. Aust. J. Bot. 2011, 59, 609–619. [Google Scholar] [CrossRef]
- Egerton-Warburton, L. A smoke-induced alteration of the sub-testa cuticle in seeds of the post-fire recruiter, Emmenanthe penduliflora Benth. (Hydrophyllaceae). J. Exp. Bot. 1998, 49, 1317–1327. [Google Scholar] [CrossRef]
- Nelson, D.; Riseborough, J.; Flematti, G.; Stevens, J.; Ghisalberti, E.; Dixon, K.; Smith, S.M. Karrikins discovered in smoke trigger Arabidopsis seed germination by a mechanism requiring gibberellic acid synthesis and light. Plant Physiol. 2009, 149, 863–873. [Google Scholar] [CrossRef]
- Janssen, B.; Snowden, K. Strigolactone and karrikin signal perception: Receptors, enzymes, or both? Front. Plant Sci. 2012, 3, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, R.; Reeves, W.; Ariizumi, T.; Steber, C. Molecular aspects of seed dormancy. Annu. Rev. Plant Biol. 2008, 59, 387–415. [Google Scholar] [CrossRef] [PubMed]
- Shu, K.; Zhang, H.; Wang, S.; Chen, M.; Wu, Y.; Tang, S.; Liu, C.; Feng, Y.; Cao, Y.; Xie, Q. ABI4 regulates primary seed dormancy by regulating the biogenesis of abscisic acid and gibberellins in Arabidopsis. PLoS Genet. 2013, 9, e1003577. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.; Chen, F.; Shuai, H.; Luo, X.; Ding, J.; Tang, S.; Xu, S.; Liu, J.; Du, J.; Liu, J.; et al. Karrikins delay soybean seed germination by mediating abscisic acid and gibberellin biogenesis under shaded conditions. Sci. Rep. 2016, 6, 22073. [Google Scholar] [CrossRef]
- Kucera, B.; Cohn, M.; Leubner-Metzger, G. Plant hormone interactions during seed dormancy release and germination. Seed Sci. Res. 2005, 15, 281–307. [Google Scholar] [CrossRef]
- Fondy, B.; Geiger, D. Diurnal Pattern of Translocation and Carbohydrate Metabolism in Source Leaves of Beta vulgaris L. Plant Physiol. 1982, 70, 671–676. [Google Scholar] [CrossRef]
- Beck, E.; Ziegler, P. Biosynthesis and degradation of starch in higher plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1989, 40, 95–117. [Google Scholar] [CrossRef]
- Sparg, S.; Kulkarni, M.; Van Staden, J. Aerosol smoke and smoke-water stimulation of seedling vigor of a commercial maize cultivar. Crop Sci. 2006, 46, 1336–1340. [Google Scholar] [CrossRef]
- Zhou, J.; Van Staden, J.; Guo, L.; Huang, L. Smoke-water improves shoot growth and indigo accumulation in shoots of Isatis indigotica seedlings. S. Afr. J. Bot. 2011, 77, 787–789. [Google Scholar] [CrossRef]

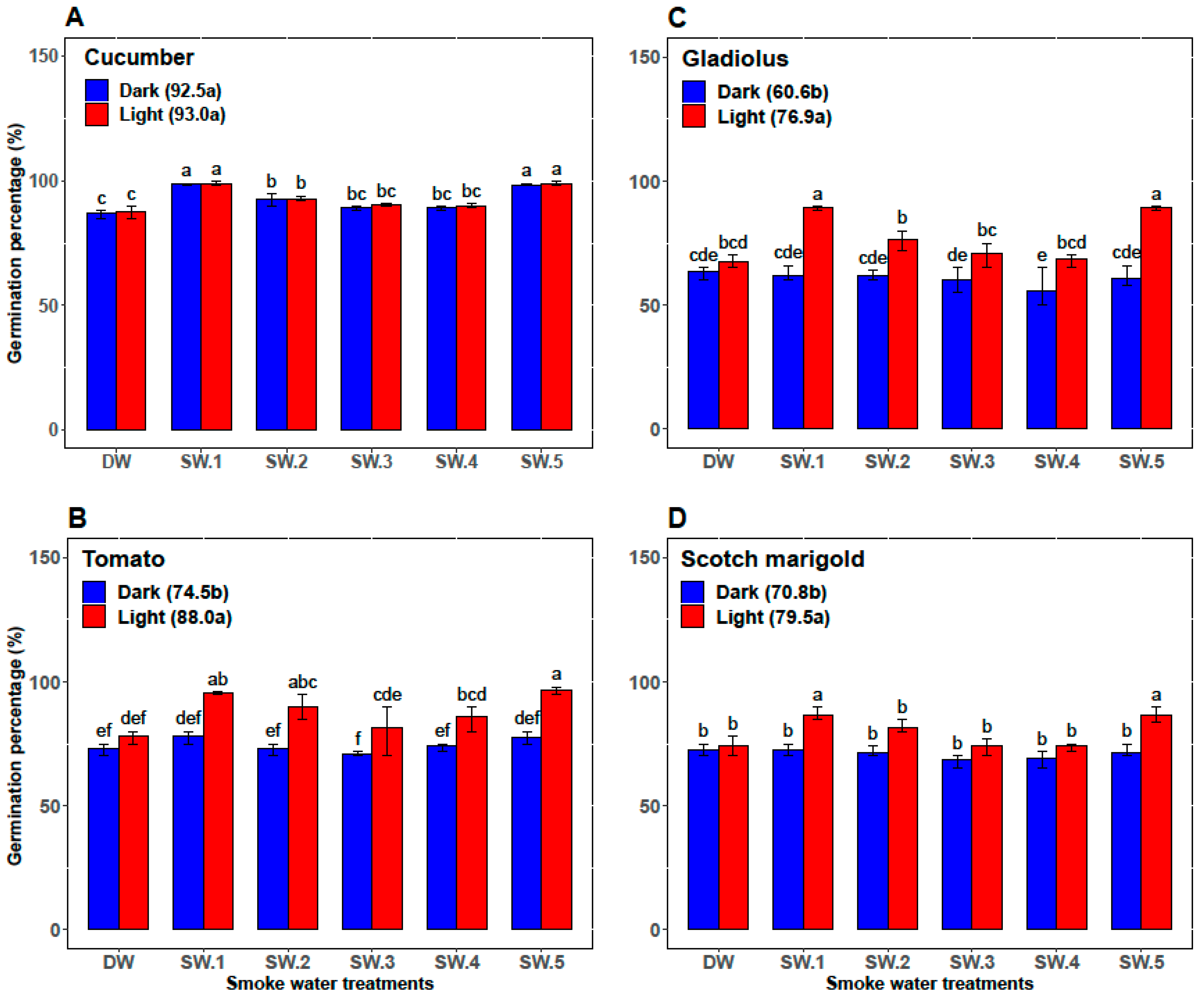
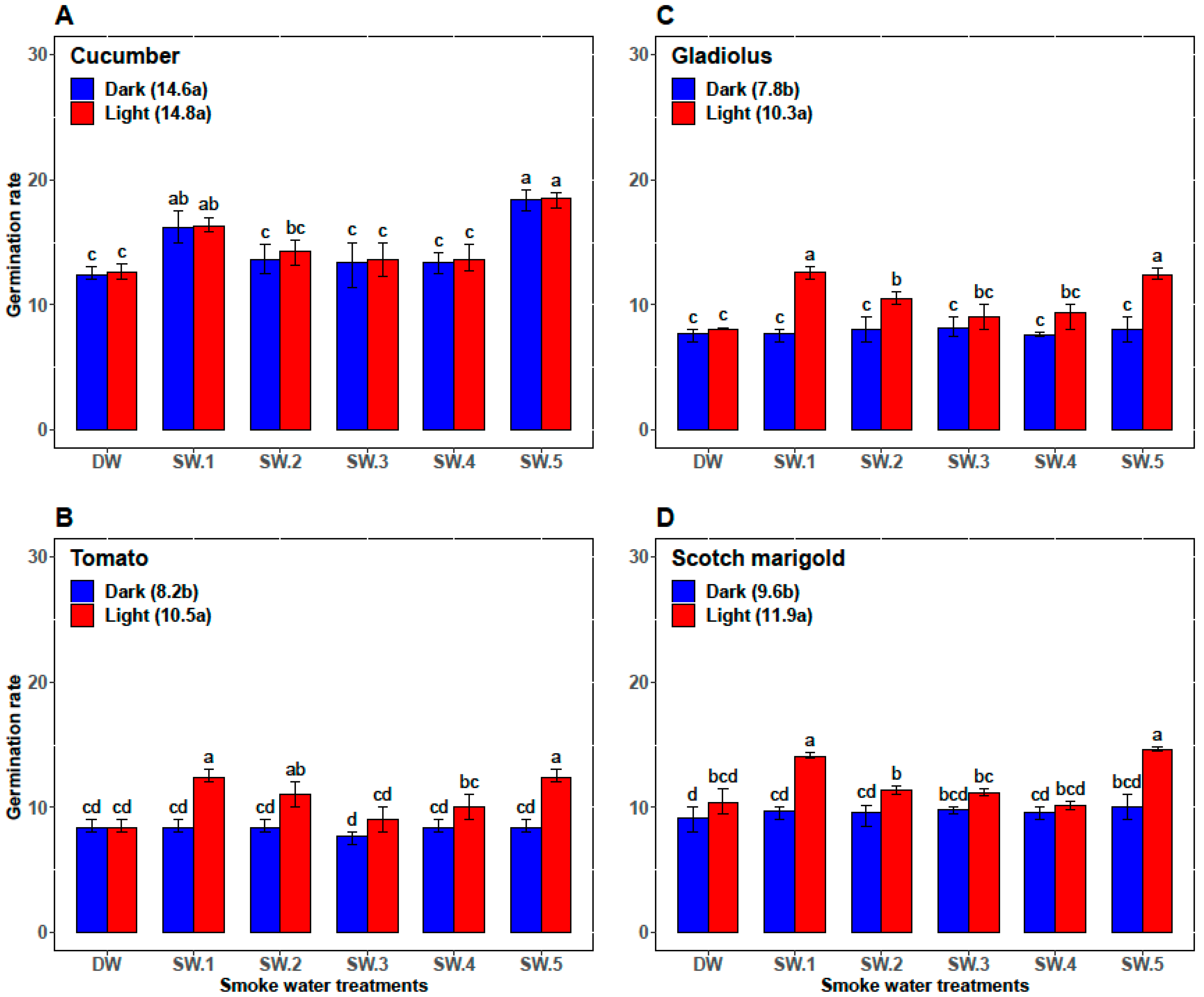
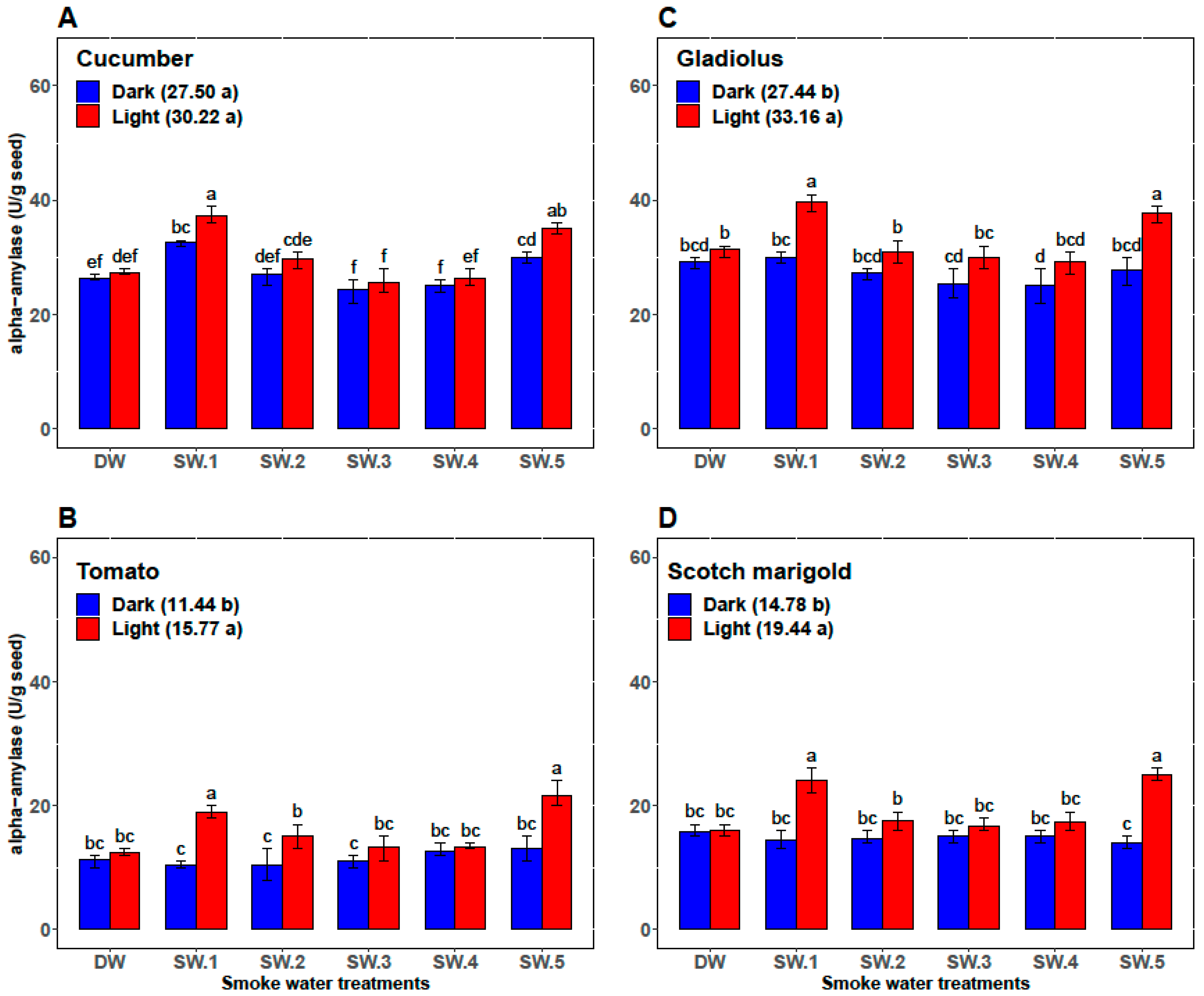
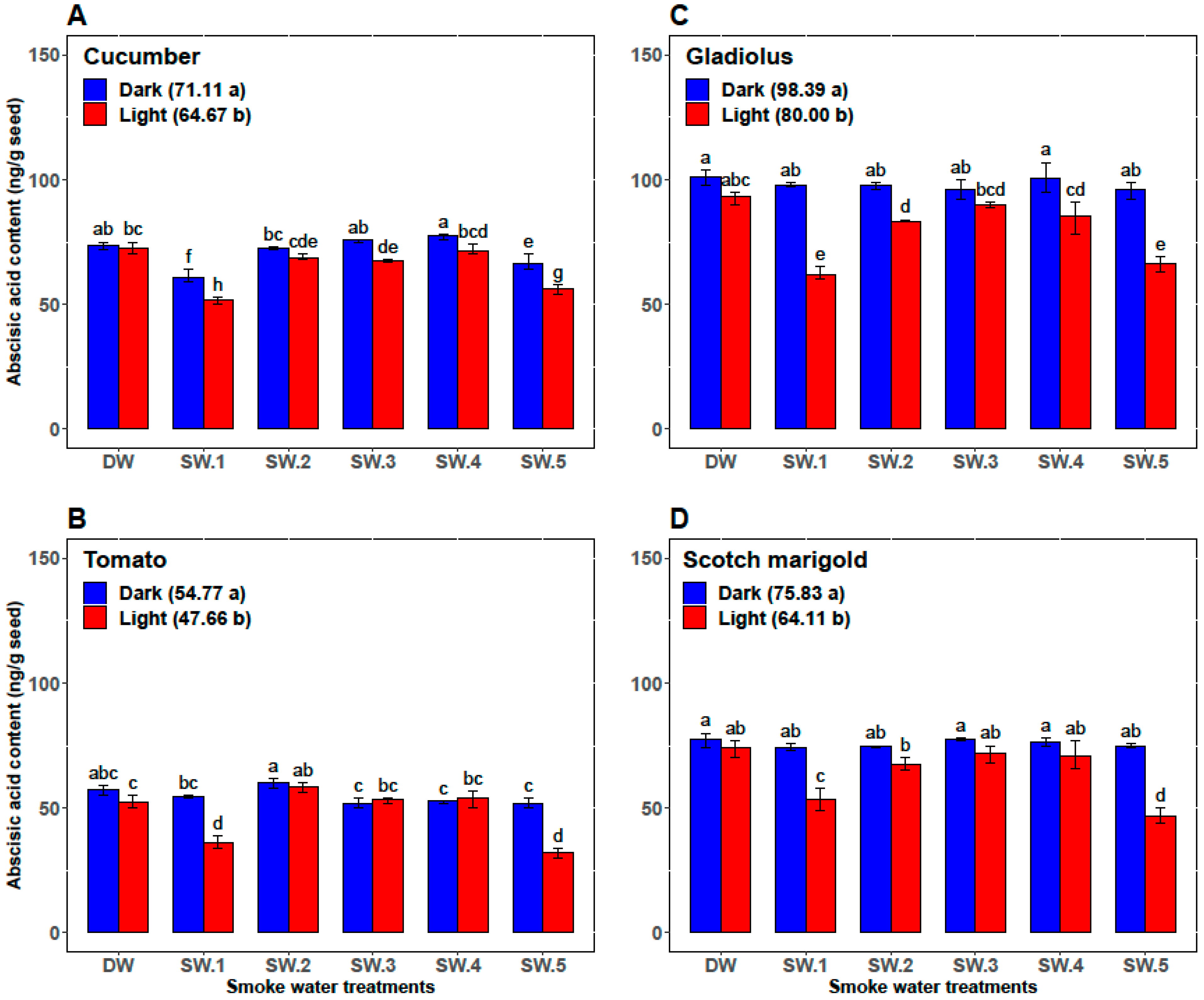

| Plant | Smoke-Water | Germination Percentage (%) | Germination Rate | α-Amylase (U/g Seed) | Abscisic Acid (ng/g Seed) |
|---|---|---|---|---|---|
| Cucumber | DW | 87.33 ± 1.97 c | 12.47 ± 0.57 c | 26.83 ± 0.75 cd | 73.00 ± 2.00 ab |
| SW1 | 98.67 ± 0.82 a | 16.28 ± 0.88 b | 35.00 ± 2.76 a | 56.33 ± 5.47 d | |
| SW2 | 92.67 ± 1.75 b | 13.48 ± 1.42 c | 28.33 ± 2.07 c | 70.67 ± 2.34 b | |
| SW3 | 89.83 ± 0.98 c | 13.97 ± 1.06 c | 25.00 ± 2.00 d | 71.67 ± 4.41 ab | |
| SW4 | 89.67 ± 1.03 c | 13.53 ± 0.87 c | 25.67 ± 1.37 d | 74.50 ± 3.45 a | |
| SW5 | 98.5 ± 0.84 a | 18.46 ± 0.70 a | 32.50 ± 2.88 b | 61.17 ± 6.15 c | |
| Tomato | DW | 75.83 ± 3.76 b | 8.33 ± 0.52 b | 11.83 ± 0.98 b | 54.83 ± 3.43 b |
| SW1 | 86.83 ± 9.50 a | 10.33 ± 2.25 a | 14.67 ± 4.80 ab | 45.33 ± 10.37 c | |
| SW2 | 81.67 ± 9.83 ab | 9.67 ± 1.63 ab | 12.67 ± 2.80 b | 59.17 ± 2.04 a | |
| SW3 | 76.17 ± 8.95 b | 8.33 ± 1.03 b | 12.17 ± 1.94 b | 52.67 ± 1.63 b | |
| SW4 | 80.00 ± 7.46 ab | 9.17 ± 1.17 ab | 13.00 ± 0.89 b | 53.33 ± 2.42 b | |
| SW5 | 87.00 ± 10.75 a | 10.33 ± 2.25 a | 17.33 ± 5.09 a | 42.00 ± 11.10 c | |
| Gladiolus | DW | 65.33 ± 3.27 bc | 7.87 ± 0.43 c | 30.33 ± 1.51 bc | 97.17 ± 5.23 a |
| SW1 | 75.67 ± 15.15 a | 10.12 ± 2.73 a | 34.83 ± 5.42 a | 80.00 ± 19.80 c | |
| SW2 | 69.33 ± 8.55 ab | 9.25 ± 1.54 ab | 29.17 ± 2.71 cd | 90.50 ± 07.92 b | |
| SW3 | 65.33 ± 7.39 bc | 8.58 ± 0.92 bc | 27.67 ± 2.8 cd | 93.17 ± 4.36 ab | |
| SW4 | 62.00 ± 8.83 c | 8.47 ± 1.20 bc | 27.17 ± 3.31 d | 93.00 ± 10.14 ab | |
| SW5 | 75.00 ± 15.99 a | 10.20 ± 2.51 a | 32.67 ± 5.79 ab | 81.33 ± 16.72 c | |
| Scotch marigold | DW | 73.33 ± 3.20 bc | 9.77 ± 1.13 b | 15.83 ± 0.98 c | 75.67 ± 3.50 a |
| SW1 | 79.50 ± 8.22 a | 11.88 ± 2.46 a | 19.17 ± 5.53 ab | 64.00 ± 11.71 b | |
| SW2 | 76.50 ± 6.12 ab | 10.46 ± 1.16 b | 16.17 ± 2.04 bc | 71.17 ± 4.17 a | |
| SW3 | 71.17 ± 4.26 c | 10.49 ± 0.83 b | 15.83 ± 1.33 c | 74.67 ± 3.72 a | |
| SW4 | 71.50 ± 3.73 c | 9.85 ± 0.49 b | 16.17 ± 1.72 bc | 73.50 ± 4.85 b | |
| SW5 | 79.17 ± 8.64 a | 12.32 ± 2.62 a | 19.50 ± 6.09 a | 60.83 ± 15.65 b |
| Plant | Smoke-Water | Shoot Length (cm) | Root Length (cm) | Fresh Weight (gm) | Dry Weight (gm) | Chlorophyll Content | Seedling Vigor Index | N (%) | P (%) | K (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Cucumber | DW | 7.67 ± 0.58 b | 5.33 ± 0.62 b | 2.07 ± 0.41 b | 0.12 ± 0.03 d | 63.00 ± 1.53 a | 1170.00 ± 90.00 c | 1.35 ± 0.15 c | 0.16 ± 0.02 c | 2.07 ± 0.12 a |
| SW1 | 10.00 ± 1.00 a | 7.67 ± 0.58 a | 3.15 ± 0.38 a | 0.33 ± 0.05 a | 71.00 ± 1.50 a | 1702.00 ± 66.36 a | 2.43 ± 0.18 ab | 0.24 ± 0.08 ab | 2.27 ± 0.25 a | |
| SW2 | 9.00 ± 1.00 ab | 7.00 ± 1.00 a | 2.98 ± 0.45 ab | 0.22 ± 0.03 bc | 67.33 ± 1.00 a | 1487.00 ± 146.65 ab | 2.1 0 ± 0.10 b | 0.20 ± 0.02 bc | 2.23 ± 0.32 a | |
| SW3 | 8.00 ± 1.00 ab | 5.67 ± 0.58 b | 2.45 ± 0.23 ab | 0.19 ± 0.01 cd | 64.33 ± 1.53 a | 1252.00 ± 132.18 bc | 1.67 ± 0.15 c | 0.17 ± 0.05 c | 2.17 ± 0.25 a | |
| SW4 | 7.68 ± 0.58 b | 5.35 ± 0.58 b | 2.88 ± 0.38 ab | 0.18 ± 0.03 cd | 63.67 ± 1.00 a | 1196.00 ± 113.25 bc | 1.47 ± 0.12 c | 0.18 ± 0.02 c | 2.10 ± 0.36 a | |
| SW5 | 9.67 ± 0.59 ab | 8.00 ± 1.00 a | 3.20 ± 0.28 a | 0.28 ± 0.03 ab | 72.00 ± 0.58 a | 1684.67 ± 153.58 a | 2.47 ± 0.10 a | 0.27 ± 0.02 a | 2.24 ± 0.12 a | |
| Tomato | DW | 11.00 ± 0.58 b | 7.67 ± 1.00 b | 1.68 ± 0.20 c | 0.16 ± 0.02 b | 60.67 ± 3.12 b | 1512.67 ± 109.12 c | 2.24 ± 0.25 b | 0.23 ± 0.03 b | 3.78 ± 0.29 a |
| SW1 | 14.33 ± 1.15 a | 9.60 ± 0.53 ab | 2.57 ± 0.12 a | 0.23 ± 0.04 a | 75.67 ± 2.35 a | 2194.13 ± 99.68 a | 3.20 ± 0.3 a | 0.35 ± 0.01 a | 4.42 ± 0.14 a | |
| SW2 | 13.00 ± 1.00 ab | 8.43 ± 1.52 b | 2.35 ± 0.13 ab | 0.17 ± 0.01 b | 63.67 ± 3.09 b | 1840.00 ± 83.25 b | 2.77 ± 0.35 ab | 0.33 ± 0.02 a | 4.18 ± 0.50 a | |
| SW3 | 11.33 ± 1.15 b | 8.00 ± 1.85 b | 1.78 ± 0.09 c | 0.16 ± 0.02 b | 61.67 ± 5.29 b | 1517.00 ± 159.12 c | 2.43 ± 0.25 b | 0.28 ± 0.03 ab | 4.00 ± 0.20 a | |
| SW4 | 11.67 ± 1.01 b | 8.83 ± 1.22 b | 1.87 ± 0.19 bc | 0.16 ± 0.04 b | 62.83 ± 7.00 b | 1658.17 ± 92.35 bc | 2.45 ± 0.31 b | 0.25 ± 0.05 b | 4.17 ± 0.26 a | |
| SW5 | 15.00 ± 1.00 a | 11.00 ± 0.76 a | 2.45 ± 0.41 a | 0.26 ± 0.01 a | 74.33 ± 5.13 a | 2423.33 ± 126.23 a | 3.25 ± 0.15 a | 0.34 ± 0.02 a | 4.33 ± 0.42 a | |
| Gladiolus | DW | 13.83 ± 1.00 b | 6.67 ± 1.00 b | 1.13 ± 0.12 b | 0.16 ± 0.02 b | 32.33 ± 5.52 b | 1335.83 ± 155.45 c | 1.86 ± 0.15 b | 0.31 ± 0.03 b | 3.03 ± 0.13 b |
| SW1 | 17.00 ± 1.50 a | 9.00 ± 0.76 a | 2.40 ± 0.15 a | 0.24 ± 0.05 a | 49.33 ± 4.04 a | 2210.00 ± 123.58 a | 3.05 ± 0.24 a | 0.44 ± 0.02 a | 4.21 ± 0.05 a | |
| SW2 | 15.67 ± 1.25 ab | 8.87 ± 1.76 a | 1.40 ± 0.10 b | 0.19 ± 0.01 ab | 37.00 ± 2.65 b | 1689.00 ± 128.36 b | 2.17 ± 0.29 b | 0.34 ± 0.04 b | 3.38 ± 0.33 b | |
| SW3 | 15.00 ± 1.47 ab | 7.97 ± 1.58 ab | 1.30 ± 0.17 b | 0.16 ± 0.01 b | 35.33 ± 1.15 b | 1489.33 ± 155.25 c | 1.94 ± 0.08 b | 0.32 ± 0.03 b | 3.14 ± 0.22 b | |
| SW4 | 14.33 ± 1.22 ab | 8.47 ± 1.47 ab | 1.17 ± 0.15 b | 0.19 ± 0.05 ab | 34.67 ± 5.03 b | 1444.50 ± 106.37 c | 1.92 ± 0.14 b | 0.33 ± 0.02 b | 3.15 ± 0.18 b | |
| SW5 | 16.73 ± 0.47a | 9.17 ± 1.50 a | 2.47 ± 0.11 a | 0.20 ± 0.01 ab | 50.67 ± 5.59 a | 2272.33 ± 136.35 a | 2.90 ± 0.27 a | 0.37 ± 0.02 ab | 3.97 ± 0.16 a | |
| Scotch marigold | DW | 8.83 ± 1.26 c | 7.37 ± 1.58 b | 3.27 ± 1.10 c | 0.28 ± 0.04 b | 42.33 ± 4.16 b | 1160.83 ± 205.31 c | 1.49 ± 0.08 b | 0.25 ± 0.03 b | 2.03 ± 0.16 a |
| SW1 | 13.40 ± 1.22 a | 10.50 ± 1.50 a | 5.93 ± 0.42 a | 0.60 ± 0.09 a | 56.6 ± 3.54 a | 1948.67 ± 169.18 a | 2.03 ± 0.06 a | 0.35 ± 0.09 ab | 2.15 ± 0.20 a | |
| SW2 | 11.23 ± 1.25 abc | 9.00 ± 1.23 ab | 4.66 ± 1.01 b | 0.56 ± 0.06 a | 45.33 ± 4.35 b | 1578.97 ± 119.18 b | 1.70 ± 0.10 b | 0.27 ± 0.06 ab | 2.08 ± 0.17 a | |
| SW3 | 10.5 ± 1.50 bc | 8.77 ± 1.25 b | 3.40 ± 0.61 c | 0.32 ± 0.05 b | 43.33 ± 5.02 b | 1343.33 ± 81.23 bc | 1.55 ± 0.09 b | 0.26 ± 0.05 b | 2.06 ± 0.13 a | |
| SW4 | 10.17 ± 1.76 bc | 7.67 ± 1.10 b | 3.70 ± 0.85 bc | 0.31 ± 0.02 b | 42.67 ± 3.25 b | 1227.33 ± 162.74 c | 1.64 ± 0.06 b | 0.25 ± 0.05 b | 2.03 ± 0.23 a | |
| SW5 | 12.17 ± 1.29 ab | 10.5 ± 1.50 a | 6.20 ± 0.21 a | 0.60 ± 0.02 a | 54.67 ± 2.12 a | 1851.67 ± 89.28 a | 2.06 ± 0.12 a | 0.37 ± 0.03 a | 2.12 ± 0.17 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elsadek, M.A.; Yousef, E.A.A. Smoke-Water Enhances Germination and Seedling Growth of Four Horticultural Crops. Plants 2019, 8, 104. https://doi.org/10.3390/plants8040104
Elsadek MA, Yousef EAA. Smoke-Water Enhances Germination and Seedling Growth of Four Horticultural Crops. Plants. 2019; 8(4):104. https://doi.org/10.3390/plants8040104
Chicago/Turabian StyleElsadek, Mohamed A., and Eltohamy A. A. Yousef. 2019. "Smoke-Water Enhances Germination and Seedling Growth of Four Horticultural Crops" Plants 8, no. 4: 104. https://doi.org/10.3390/plants8040104
APA StyleElsadek, M. A., & Yousef, E. A. A. (2019). Smoke-Water Enhances Germination and Seedling Growth of Four Horticultural Crops. Plants, 8(4), 104. https://doi.org/10.3390/plants8040104






