Extracts and individual compounds present anti-inflammatory, antiradical, antiproliferative, and antibacterial activities.
6.2.1. Anti-Inflammatory Activity
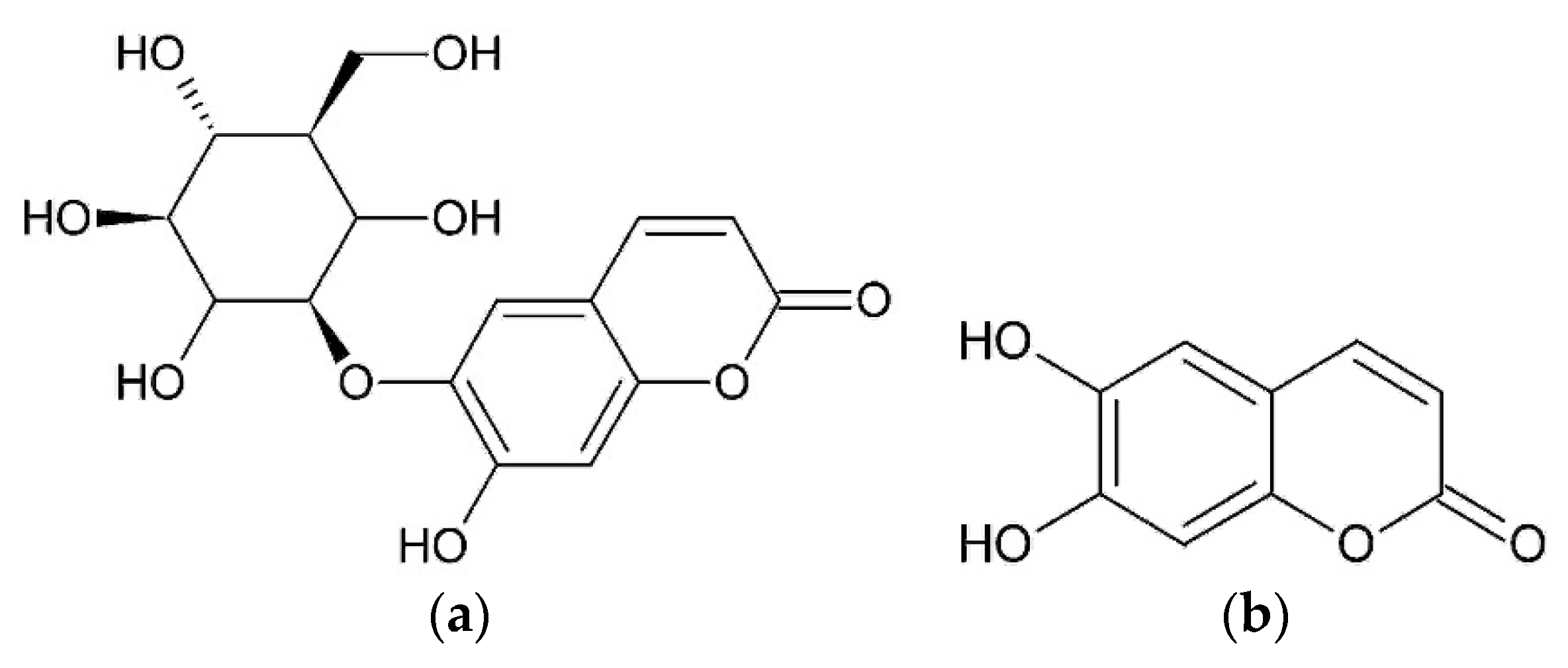
The anti-inflammatory action of hexane and methanol extracts from achenes described below should be connected with the presence of arctiin and isochlorogenic acid [
29]. Hexane and methanol extracts were identified. Paraffins were identified in hexane extract. Methanol extract was found to contain arctiin (arctigenin-4’-O-D-glucoside), arctigenin, and isochlorogenic acid by gas chromatography mass-spectrometry (GC-MS), high-performance liquid chromatography-mass spectrometry (HPLC-MS), high-performance liquid chromatography-ultraviolet (HPLC-UV) and proton nuclear magnetic resonance (
1H-NMR) methods. Anti-inflammatory activity was detected on the immortalized human umbilical vein endothelial cells (HUVECtert). These cells were stimulated by lipopolysacharids and tumor necrosis factor-alpha (TNF-α). Decreased secretion of E-selectin by HUVECtert was measured by real-time quantitative reverse transcription polymerase chain reaction (RT-PCR) and decreased production of interleukin-8 (IL-8) was measured by enzyme-linked immunosorbent assay (ELISA). Furthermore, inhibitory activity of methanol extract on the secretion of E-selectin was detected, as was inhibitory activity of methanol and hexane extracts on the expression of E-selectin. No anti-inflammatory activity was detected by extracts when inflammation was stimulated by TNF-α. Methanol extract at a dose of 15–40 mg/mL or arctiin at a dose of 20–75 μM demonstrated inhibitory activity on E-selectin. The positive control was (E)-3-(4-Methylphenylsulfonyl)-2-propenenitrile (BAY 11-7082, an inhibitor of cytokine-induced IκB-α phosphorylation) at a dose of 5 μM.
Lajter [
24] investigated the anti-inflammatory activity of lignans, flavonoids, and sesquiterpene lactones from the aerial part and roots of
O. acanthium on different targets such as activity on the expression of cyclooxygenase-2 (COX-2) and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB1), and its inhibitory effect on the production of NO, 5-lipoxygenase (5-LOX,) COX-1, and COX-2. The positive controls were quercetin (25 μM) and dexamethasone (2.5 nM) in the analysis of expression of NF-κB1 and COX-2; N
G-monomethyl-L-arginine (L-NMMA) as inhibitor of NO-syntase (100 μM); zileuton as inhibitor of 5-LOX (5 μM); indomethacin and NS-398 (N-[2-(Cyclohexyloxy)-4-nitrophenyl]methanesulfonamide) as inhibitors of COX-1 and COX-2 (1.25 μM and 5 μM, respectively). Also, hexane, chloroform, and aqueous-methanol extracts from aerial parts and roots were investigated at a dose of 10 μg/mL (inhibition of COX-2 and NF-κB1 expression and NO-syntase) and 50 μg/mL (inhibition of 5-LOX, COX-1, and COX-2). Individual compounds were investigated at a dose of 20 μM dissolved in dimethyl suldoxide (DMSO). The results are presented as inhibition % ± SD (
Table 6).
The compounds of roots 4β,15-Dihydro-3-dehydrozaluzanin C and zaluzanin C were also tested for inhibitory activity of the expression of COX-2 and NF-κB1. The most active on the expression of NF-κB1 was found to be 4β,15-Dihydro-3-dehydrozaluzanin C (inhibition of 96.0% for a dose of 10 μM), which was also found to be the most active inhibitor of COX-2 (inhibition of 91.2% on dose 10 μM). Zaluzanin C was demonstrated to have an inhibition of 83.7% for the expression of NF-κB1 and an inhibition of 87.9% for the expression of COX-2. The positive controls were found to have an inhibition of 47.6% for the expression of COX-2 (dexamethasone at a dose of 2.5 nM) and an inhibition of 46.0% for the expression of NF-κB1 (quercetin at a dose of 25 μM). In XTT viability assay (colorimetric assay by using 2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2H-tetrazolium-5-carboxanilide as a substrate which is reduced to purple formazan in living cells) at different concentrations at four, 24, 48 and 72 h), these compounds were demonstrated to have low cytotoxicity [
24].
Onopordopicrin decreases intestinal inflammation [
33]. Zaluzanin C and estafiatone showed an inhibitory effect on the lipopolysaccharide (LPS)/interferon-γ (INF-γ)-induced nitric oxide (NO) and prostaglandin E2 (PGE
2) production on the RAW 264.7 macrophages with IC
50 of 6.61 μM and 3.80 μM, respectively. Additionally, suppression of expression inducible NO syntaze (iNOS) and COX-2 was detected. Zaluzanin C inhibits electron and proton transport and inhibits the synthesis of ATP [
33].
6.2.2. Antitumor Activity
Abuharfeil [
57] investigated aqueous extract of leaves and stems of
O. acanthium against YAC cells (virus-induced murine T-cell lymphoma). The extract increased natural killer cell (NK) activity of splenic lymphoid cells, which exhibit a cytotoxic effect on YAC cells. There were 13 aqueous extracts. Extract from
O. acanthium showed medium stimulation activity. The activity of aqueous extract from fresh material was 40.6% (ratio of effector:target 200:1), and that of aqueous extract from air-dried material was 30.4%. The positive control was interferon α at a dose of 500 U/mL, which increased the activity of NK cells by 78.5%.
Csupor-Löffler [
58] investigated the antitumor activity of aqueous and organic extracts of 26 plants belonging to tribes Cynareae and Lactuceae (Asteraceae) on cell cultures: HeLa (cervix epithelial adenocarcinoma), A431 (skin epidermoid carcinoma), and MCF7 (breast epithelial adenocarcinoma). Living cells were detected in colorimetric MTT assay using [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide. There were water, n-hexane, chloroform, and aqueous methanol extracts from mixture of flowers and fruits, leaves, and roots of
O. acanthium. The most active was chloroform extract from leaves at a dose of 30 μg/mL (HeLa: IC
50 6.53 μg/mL, MCF7: IC
50 6.39 μg/mL, А431: IC
50 4.54 μg/mL) and roots (HeLa: IC
50 6.11 μg/mL, MCF7: IC
50 4.39 μg/mL, А431: IC
50 10.32 μg/mL). The positive controls were doxorubicin (half maximal effective concentration (EC
50) HeLa: 0.081 μg/mL; MCF7: 0.152 μg/mL; А431: 0.081 μg/mL), cisplatin (IC
50 HeLa: 3.7 μg/mL; MCF7: 2.8 μg/mL; А431: 0.85 μg/mL). The most active compounds from
O. acanthium roots, 4β,15-Dihydro-3-dehydrozaluzanin C and zaluzanin C [
24], were measured for their cytotoxic activity by XTT viability assay. The positive control was vinblastine (0.1 μg/mL). In the XTT viability assay THP-1 cells (human monocytic cell line) were used. These compounds had no or low effect on cell line.
Methanol extracts from leaves of
O. acanthium and flowers of
Spartium junceum L. (Fabaceae) were compared to each other on the human glioblastoma U-373 cell line. Methanol extract from
O. acanthium was measured to have inhibitor activity of IC
50 309 μg/mL, while methanol extract from
S. junceum was found to have inhibitor activity of IC
50 1602 μg/mL. This activity was measured by the trypan blue exclusion test. The mechanism of cytotoxicity is connected with inducing caspase-3, a cell enzyme of apoptosis. Thus, methanol extract of
O. acanthium leaves showed proapoptotic activity [
59].
Molnar [
60] connected the antitumor activity of extracts of
O. acanthium with effect on apoptotic enzymes. Sesquiterpene lactones from herb of
Artemisia asiatica and roots of
O. acanthium were investigated on the HL-60 cells (human leukemia cell). The compound from roots of
O. acanthium 4β,15-dihydro-3-dehydrozaluzanin C demonstrated inhibitor activity of IC
50 3.6 μM. Cytotoxicity was measured by colorimetric MTT assay, and effects on the cell cycle were investigated by flow cytometric analysis. Cell changes were detected by staining cell DNA with the fluorescent marker propidium iodide and Hoechst 33258 (2′-(4-hydroxyphenyl)-5-(4-methyl-1-piperazinyl)-2,5′-bi-1H-benzimidazole trihydrochloride hydrate). The mechanism of cytotoxicity was inducing mitochondrial pathway of apoptosis by activation of caspase-3 and caspase-9.
The activity of caspase-3 was measured by fluorimetric assay using Ac-DEVD-AMC (N-Acetyl-Aspartyl-Glutamyl-Valyl-Aspartyl-7-amino-4-methylcoumarin) as a substrate. The activity of caspase-9 was measured using Ac-LEHD-pNA (N-Acetyl-Leucyl-Glutamyl-Histidyl-Aspartyl-p-Nitroanilide) as a substrate.
The lactones increased the amount of cells in G1 and G2/M populations, and decreased the amount of cells in the S population. After 48 h of incubation, an increased number of the hypodiploid (subG1) population was also detected for 5 and 10 μM doses. However, 4β,15-dihydro-3-dehydrozaluzanin C showed the lowest activity on this population. Compound 4β,15-dihydro-3-dehydrozaluzanin C demonstrated inducing activity on the caspase-3 and caspase-9.
Natural compounds that have α-methylen-γ-lactone in their structure may be potential antitumor agents.
NK activity was measured for human blood cells against K562 cells (human immortalized myelogenous leukemia cell line). The activity was tested after treatment with plant extract in the presence of IL-2 (1000 U/mL) and in the absence of IL-2. Production of cytokines IFNγ and TNFα was measured by ELISA; the activity of the cell enzymes granzyme A (N-alpha-benzyloxycarbonyl-L-lysine thiobenzyl ester (N-BLT) serine esterase) and N-acetyl-β-D-glucosaminidase (NAG) was measured by spectrophotometric analysis at 405 nm. In the presence of water extract from leaves and stems of
O. acanthium, cytotoxicity at a dose of 0.1 mg/mL (dilution 1:50) of NK cells against K562 cells was 41.3% in the ratio effector:target of 200:1. In the presence of IL-2 this activity was 56.5%. Synthesis of cytokines after treatment with 0.1 mg/mL of water extracts in the presence of IL-2 was increased by 60% for TNFα and 87% for IFNγ. The activity of the cell enzymes granzyme A and NAG was measured only for water extracts from seeds of
Nigella sativum [
61].
Sesquiterpene lactones also demonstrate antitumor activity [
33]. Thus,
onopordopicrin has cytotoxic activity against the KB cell line (HeLa derivative). Onopordopicrin has an antiproliferative effect with an IC
50 of 15 μM on the HL60 (promyelocytic leukemia). Cytotoxic effect was detected against cell lines P388 (murine leukemia), A549 (adenocarcinomic human alveolar basal epithelial cells), and HT29 (human colon cancer cell line). This effect connects with the presence of the α-methylene-γ-lactone group. Cytotoxic activity is increased by the presence of additional α,β-unsaturated ester group.
Arctiopicrin has cytotoxic activity on cell line of the colon cancer cell line in MTT colorimetric experiment. Toxic effects were investigated by brine shrimp lethality assay.
Zaluzanin C showed a cytotoxic effect on cell lines such as A549, SK-OV-3 (ovarian cancer cell line), SK-MEL-2 (human melanoma cell line), XF498 (human central nervous system (CNS) cancer), HCT15 (human colon adenocarcinoma), HL60, P388, HepG2 (human liver cancer cell line), HeLa, and OVCAR-3 (human ovarian carcinoma cell line). The effects of this compound on peptide synthesis inhibit the translocation of peptidyl-tRNA.
Zaluzanin C and estafiatone inhibit the proliferation of T- and B lymphocytes of mice with dose of 1 × 10−5 М or lower.
6.2.3. Antiradical Activity
There are different data about the antioxidant activity of O. acanthium.
Kiselova [
62] tested 23 aqueous plant extracts; for
O. acanthium, air-dried flowers were used. In water extract, amounts of polyphenols were measured as µM quercetin equivalents. Antioxidant activity was measured by ABTS (2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid)) cation radical decolorization assay. In the presence of potassium persulfate, ABTS radical has absorbance at 734 nm that decreased if water extract contains some antioxidants. Flowers of
O. acanthium demonstrated low antioxidant activity: 0.44 ± 0.06 mM Trolox equivalent antioxidant capacity (TEAC). The highest activity was 4.79 ± 0.14 mM TEAC by water extract from herb of
Alchemilla vulgaris L.; the lowest activity was 0.31 ± 0.01 mM TEAC by water extract from flowers of
Calendula officinalis L. The positive controls were the plants
Camellia sinensis (L.) Kuntze,
Ilex paraguariensis A.St.-Hil.,
Aspalathus linearis (Burm.f.) R.Dahlgren, and
Cyclopia intermedia E.Mey.
Sharifi [
20] carried out experiment using 2,2-diphenyl-1-picrylhydrazyl (DPPH). DPPH assay was based on the decreasing absorbance of DPPH solution at 517 nm then DPPH connection with radicals. Methanol extract of achenes of
O. acanthium was demonstrated to have IC
50 2.6 ± 0.04 μg/mL. The positive controls were 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox) and butylated hydroxytoluene (BHT), which had IC
50 3.3 ± 0.06 μ/mL and IC
50 10.3 ± 0.15 μ/mL, respectively.
In [
55], the antioxidant activity of water-methanol extracts from air-dried achenes of
O. acanthium was found to be IC
50 7.0 ± 0.09 μg/mL. The positive controls were BHT (IC
50 10.3 ± 0.15 μ/mL) and Trolox (IC
50 3.2 ± 0.06 μ/mL).
Koc [
21] measured the antioxidant activity of methanol, ethanol, and acetone extracts from the flowers and leaves of
O. acanthium using DPPH. The total phenolic content was investigated using Folin–Ciocalteu’s reagent, which consists of hexavalent phosphomolybdic/phosphotungstic acid complexes. This reagent is connected with hydroxyl groups of phenols and products have absorbance at 765 nm. Furthermore, authors researched the inhibitor effect on the antioxidant enzymes such as catalase (CAT), glutathione S-transferase (GST), and glutathione peroxidase (GPx). Free-radical scavenging activity was measured as IC
50 and was calculated from the dose–response inhibition curve. Acetone extract had IC
50 842 ng/mL, ethanol extract IC
50 1120 ng/mL, and methanol extract IC
50 723 ng/mL. The positive controls were ascorbic acid, with IC
50 5.144 μg/mL, and quercetin, with IC
50 1.685 μg/mL. Extracts from
O. acanthium materials did not show high inhibitory activity of antioxidant enzymes.
Similar research was conducted by Habibatni [
22], using DPPH and butanol extract from leaves of
O. acanthium. Antioxidant activity was measured as IC
50 134.4 mg/mL. The positive control was ascorbic acid (IC
50 21.4 μg/mL) and the negative control was a solution of DDPH in 80% methanol without
O. acanthium extract. Additionally, authors investigated the hypouricemic effect as an inhibitor of xanthine oxidase. This experiment was carried out using the absorbance of products of xanthine and xanthine oxidase (the dose of xanthine oxidase was 0.1 U/mL). The positive control was allopurinol. The activity of butanol extract was 7.0% of inhibition at a dose of 100 μg/mL, and its IC
50 was 572.9 μg/mL, while allopurinol had IC
50 3.9 μg/mL.
6.2.4. Antibacterial Activity
Methanol extract from the aerial part of
O. acanthium at a concentration of 100 µg/mL had no antibacterial effect on several cultures:
Klebsiella pneumonia,
Proteus vulgaris,
Shigella sonnei,
Vibrio cholerae,
Escherihia coli,
Staphylococcus aureus,
Bacillus anthracis, and
Salmonella paratyphi A [
63].
The antibacterial effect of methanol extract from achenes of
O. acanthium was investigated using the gram-negative cultures
Escherichia coli and
Klebsiella pneumonia and the gram-positive cultures
Staphylococcus epidermidis, S. aureus, and
Micrococcus luteus by agar disc diffusion method. The reference control was amikacin at a dose of 30 mg. The highest antibacterial effects were on the culture of
M. luteus, with a diameter of zone inhibitor of 21 ± 1 mm, and on the culture of
S. epidermidis (18.66 ± 1.53 mm). There was no inhibitory activity against
S. aureus,
K. pneumonia, or
E. coli. Minimum inhibitory concentration (MIC) was measured by the broth macro dilution method and resazurin microtiter method. For the second method, kanamycin was used as the positive control. MICs were obtained for
S. epidermidis and
M. luteus (0.612 mg/mL). The total phenolic content was found to be 168.56 ± 4.89 mg of gallic acid/g of extract [
64].
Ethyl acetate extract from fresh leaves of
O. acanthium was investigated for antibacterial activity after dissolution in 10-times crude extract in ethanol. The extract was separated by TLC and offline overpressure layer chromatography (OPLC) direct bioautography. The most active fractions were analyzed by HPLC-MS. The structure of isolated compounds was detected by NMR. The extract was separated by low-pressure flash chromatography. This work used
Allivibrio fischeri,
Bacillus subtilis strain F1276,
Pseudomonas syringae pv.
maculicola,
Xanthomonas euvesicatoria,
Lactobacillus plantarum,
S. aureus, methicillin resistant
S. aureus (MRSA), and
E. coli. The MIC was also measured for extracts between 27.6–275.9 μg/mL and for isolated onopordopicrin between 2.2–172.4 μg/mL by a broth microdilution method. In the results after TLC analysis, inhibitor activity of growth
B. subtilis was found. There were two zones, one with a retardation factor (hR
f) of 37 and one with an hR
f of 82. The zone with hR
f 82 was active against
B. subtilis,
X. euvesicatoria,
A. fischeri,
S. aureus, and MRSA. All of the bacterial cultures were sensitive to component hR
f 37; however
P. maculicola was inhibited by a high amount of this compound. Onopordopicrin inhibited all of the bacterial cultures; however
P. maculicola was resistant to 20 μg onopordopicrin [
35].
Estafiatone has antibacterial activity against
B. subtilis.
Zaluzanin C inhibits root and shoot growth in lettuce, tomatoes, and cress, and has antifungal activity. Additionally,
onopordopicrin demonstrated antibacterial activity against
S. aureus [
33].