Cunninghamia lanceolata PSK Peptide Hormone Genes Promote Primary Root Growth and Adventitious Root Formation
Abstract
1. Introduction
2. Results
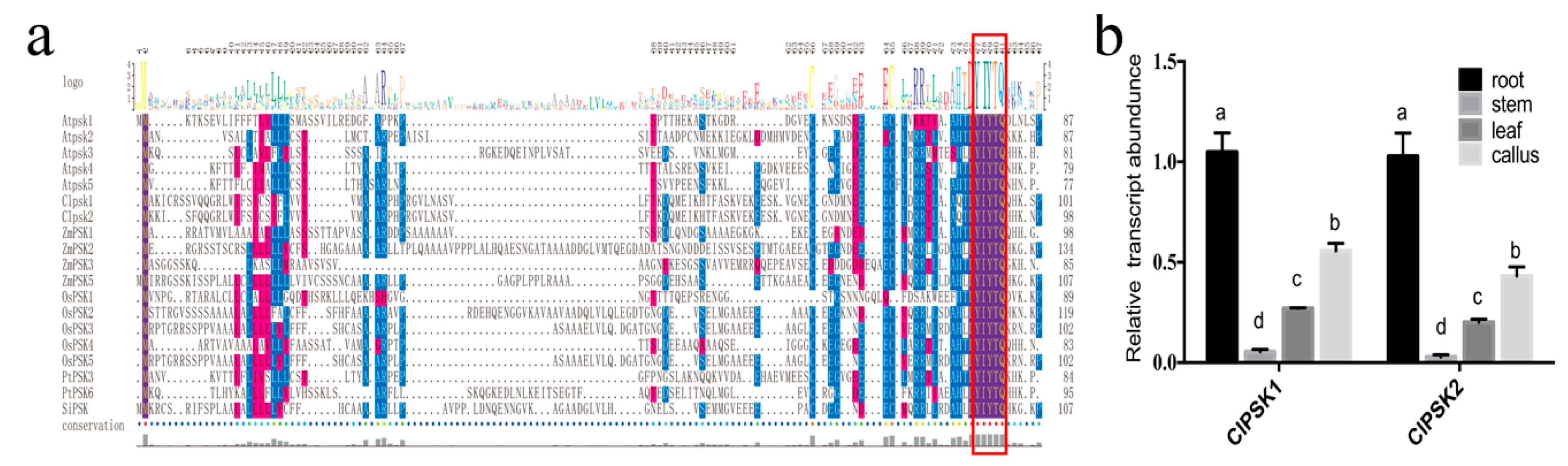
2.1. Molecular Cloning and Characterization of the ClPSK Genes
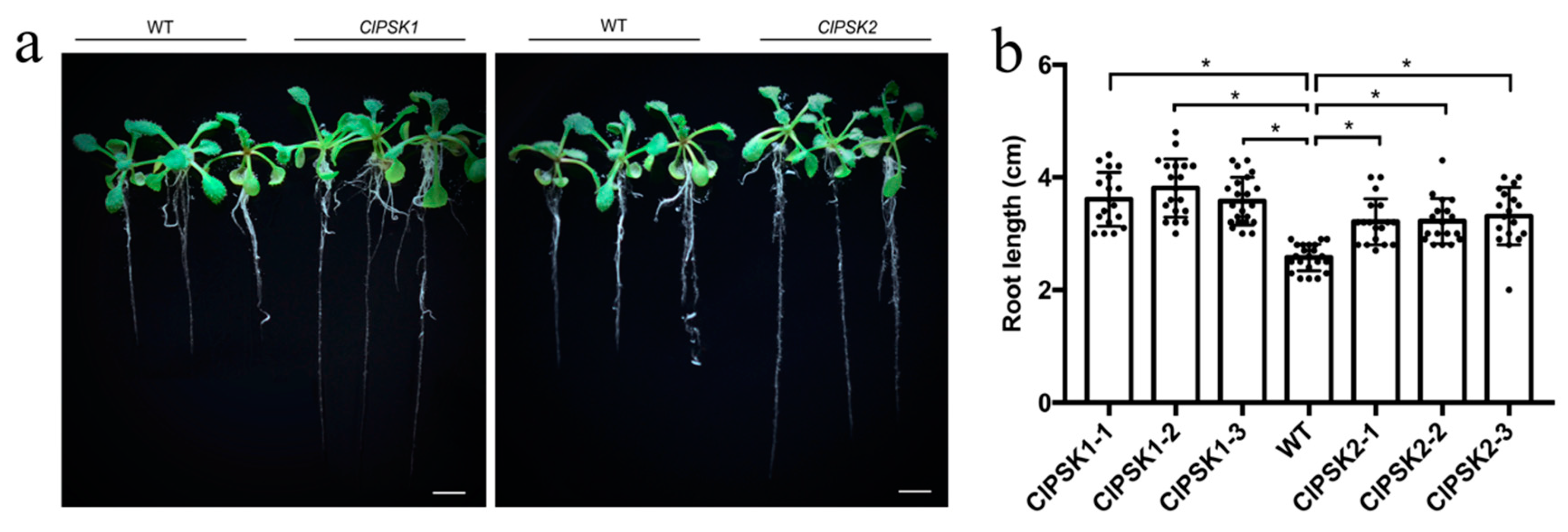
2.2. Overexpression of ClPSK Promotes Root Growth in Arabidopsis Thaliana
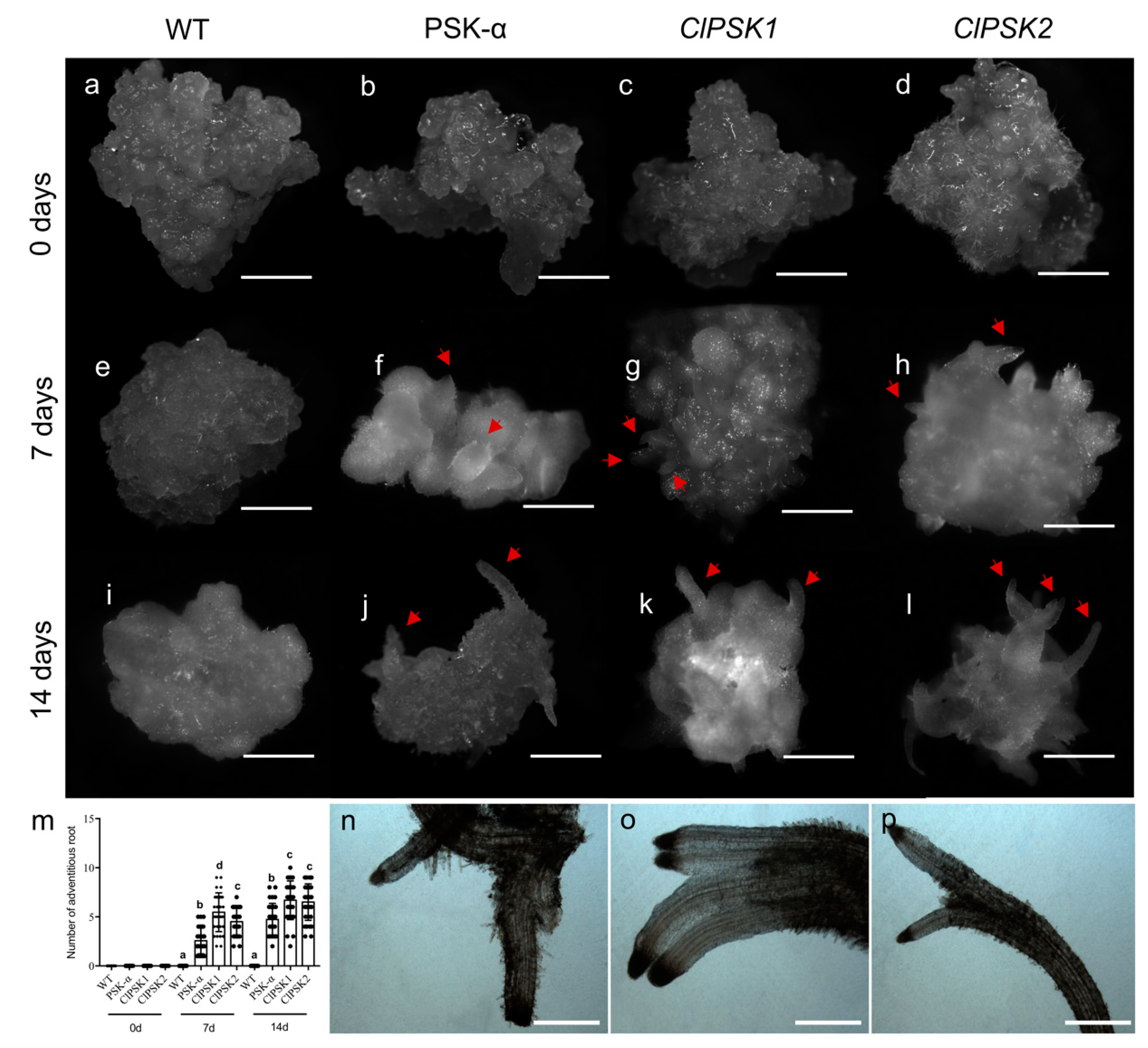
2.3. Overexpression of ClPSK Gene Promotes Organ Differentiation
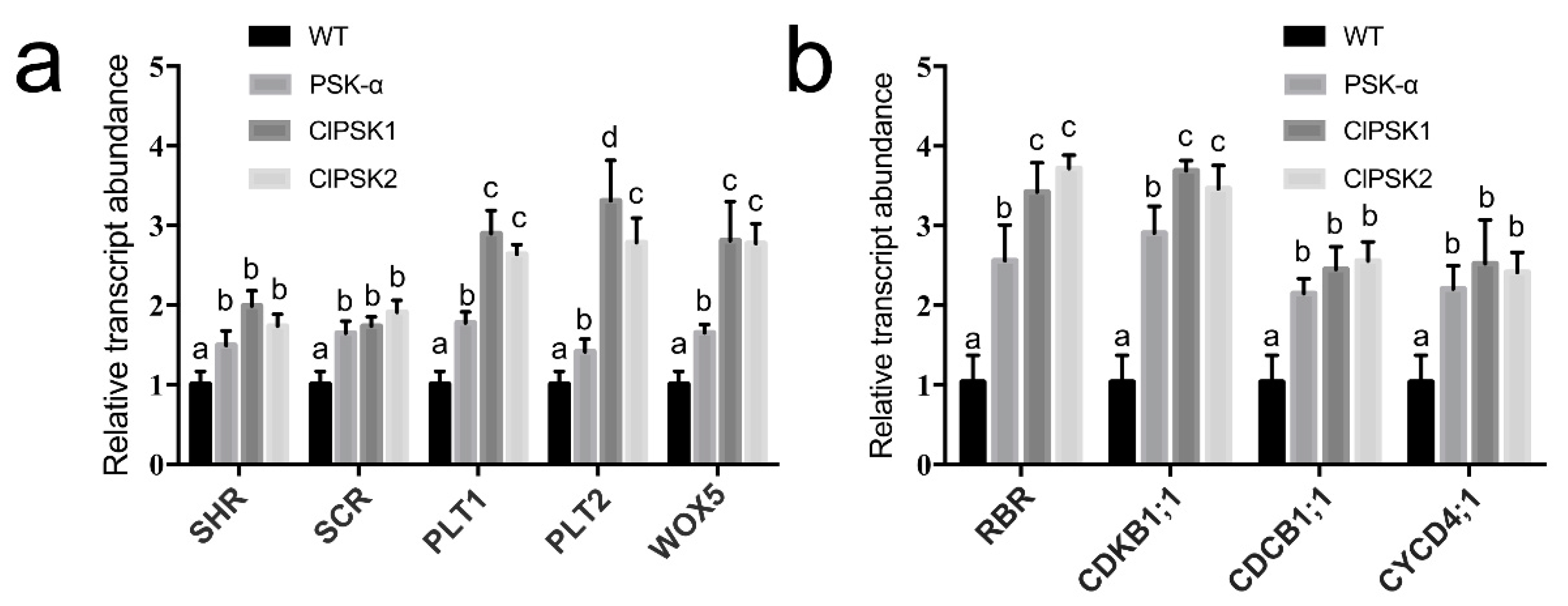
2.4. Overexpression of ClPSK Genes Upregulates the Expression of Genes Related to Root Morphogenesis
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Culture Conditions
4.2. Gene Cloning and Construction of Vectors and ClPSK Overexpression Lines
4.3. Characterization of the ClPSK Genes Analysis
4.4. Quantitative Real-Time PCR
4.5. Morphological Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sauter, M. Phytosulfokine peptide signalling. J. Exp. Bot. 2015, 66, 5161–5169. [Google Scholar] [CrossRef] [PubMed]
- Matsubayashi, Y. Post-Translational Modifications in Secreted Peptide Hormones in Plants. Plant Cell Physiol. 2011, 52, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Matsubayashi, Y.; Nakamura, K.; Sakagami, Y. Diversity of Arabidopsis Genes Encoding Precursors for Phytosulfokine, a Peptide Growth Factor. Plant Physiol. 2001, 127, 842–851. [Google Scholar] [CrossRef] [PubMed]
- Lorbiecke, R.; Sauter, M. Comparative analysis of PSK peptide growth factor precursor homologs. Plant Sci. 2002, 163, 321–332. [Google Scholar] [CrossRef]
- Igasaki, T.; Akashi, N.; Ujinoihara, T.; Matsubayashi, Y.; Sakagami, Y.; Shinohara, K. Phytosulfokine Stimulates Somatic Embryogenesis in Cryptomeria japonica. Plant Cell Physiol. 2003, 44, 1412–1416. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Matsubayashi, Y.; Nakamura, K.; Sakagami, Y. Oryza sativa PSK gene encodes a precursor of phytosulfokine-alpha, a sulfated peptide growth factor found in plants. Proc. Natl. Acad. Sci. USA 1999, 96, 13560–13565. [Google Scholar] [CrossRef]
- Lorbiecke, R.; Steffens, M.; Tomm, J.M.; Scholten, S.; Von Wiegen, P.; Kranz, E.; Wienand, U.; Sauter, M. Phytosulphokine gene regulation during maize (Zea mays L.) reproduction. J. Exp. Bot. 2005, 56, 1805–1819. [Google Scholar] [CrossRef]
- Zhang, H.; Hu, Z.; Lei, C.; Zheng, C.; Wang, J.; Shao, S.; Li, X.; Xia, X.; Cai, X.; Zhou, J. A Plant Phytosulfokine Peptide Initiates Auxin-Dependent Immunity through Cytosolic Ca2+ Signaling in Tomato. Plant Cell 2018, 30, 652–667. [Google Scholar] [CrossRef]
- Kutschmar, A.; Rzewuski, G.; Stuhrwohldt, N.; Beemster, G.T.S.; Inze, D.; Sauter, M. PSK-α promotes root growth in Arabidopsis. New Phytol. 2009, 181, 820–831. [Google Scholar] [CrossRef]
- Matsubayashi, Y.; Ogawa, M.; Kihara, H.; Niwa, M.; Sakagami, Y. Disruption and overexpression of Arabidopsis phytosulfokine receptor gene affects cellular longevity and potential for growth. Plant Physiol. 2006, 142, 45–53. [Google Scholar] [CrossRef]
- Stührwohldt, N.; Dahlke, R.I.; Kutschmar, A.; Peng, X.; Sun, M.X.; Sauter, M. Phytosulfokine peptide signaling controls pollen tube growth and funicular pollen tube guidance in Arabidopsis thaliana. Physiol. Plant. 2015, 153, 643–653. [Google Scholar] [CrossRef] [PubMed]
- Mussig, C.; Shin, G.; Altmann, T. Brassinosteroids Promote Root Growth in Arabidopsis. Plant Physiol. 2003, 133, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, J.; Stührwohldt, N.; Dahlke, R.I.; Sauter, M. Phytosulfokine control of growth occurs in the epidermis, is likely to be non-cell autonomous and is dependent on brassinosteroids. Plant J. 2013, 73, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Heyman, J.; Cools, T.; Vandenbussche, F.; Heyndrickx, K.S.; Van Leene, J.; Vercauteren, I.; Vanderauwera, S.; Vandepoele, K.; De Jaeger, G.; Der Straeten, D.V. ERF115 Controls Root Quiescent Center Cell Division and Stem Cell Replenishment. Science 2013, 342, 860–863. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Li, M.; Cui, W.; Lu, W.; Shen, W. Haem Oxygenase-1 is Involved in Hydrogen Sulfide-induced Cucumber Adventitious Root Formation. J. Plant Growth Regul. 2012, 31, 519–528. [Google Scholar] [CrossRef]
- Nordstrom, A.; Eliasson, L. Levels of endogenous indole-3-acetic acid and indole-3-acetylaspartic acid during adventitious root formation in pea cuttings. Physiol. Plant. 1991, 82, 599–605. [Google Scholar] [CrossRef]
- Sukumar, P.; Maloney, G.S.; Muday, G.K. Localized Induction of the ATP-Binding Cassette B19 Auxin Transporter Enhances Adventitious Root Formation in Arabidopsis. Plant Physiol. 2013, 162, 1392–1405. [Google Scholar] [CrossRef]
- Liu, H.; Wang, S.; Yu, X.; Yu, J.; He, X.; Zhang, S.; Shou, H.; Wu, P. ARL1, a LOB-domain protein required for adventitious root formation in rice. Plant J. 2005, 43, 47–56. [Google Scholar] [CrossRef]
- Maldiney, R.; Pelese, F.; Pilate, G.; Sotta, B.; Sossountzov, L.; Miginiac, E. Endogenous levels of abscisic acid, indole-3-acetic acid, zeatin and zeatin-riboside during the course of adventitious root formation in cuttings of Craigella and Craigella lateral suppressor tomatoes. Physiol. Plant. 1986, 68, 426–430. [Google Scholar] [CrossRef]
- Noiton, D.; Vine, J.H.; Mullins, M.G. Endogenous indole-3-acetic acid and abscisic acid in apple microcuttings in relation to adventitious root formation. Plant Growth Regul. 1992, 11, 63–67. [Google Scholar] [CrossRef]
- Yamakawa, S.; Sakuta, C.; Matsubayashi, Y.; Sakagami, Y.; Kamada, H.; Satoh, S. The promotive effects of a peptidyl plant growth factor, phytosulfokine-α, on the formation of adventitious roots and expression of a gene for a root-specific cystatin in cucumber hypocotyls. J. Plant Res. 1998, 111, 453–458. [Google Scholar] [CrossRef]
- Rigal, A.; Yordanov, Y.S.; Perrone, I.; Karlberg, A.; Tisserant, E.; Bellini, C.; Busov, V.; Martin, F.; Kohler, A.; Bhalerao, R. The AINTEGUMENTA LIKE1 Homeotic Transcription Factor PtAIL1 Controls the Formation of Adventitious Root Primordia in Poplar. Plant Physiol. 2012, 160, 1996–2006. [Google Scholar] [CrossRef] [PubMed]
- Smolka, A.; Welander, M.; Olsson, P.; Holefors, A.; Zhu, L. Involvement of the ARRO-1 gene in adventitious root formation in apple. Plant Sci. 2009, 177, 710–715. [Google Scholar] [CrossRef]
- Wei, K.; Wang, L.; Cheng, H.; Zhang, C.; Ma, C.; Zhang, L.; Gong, W.; Wu, L. Identification of genes involved in indole-3-butyric acid-induced adventitious root formation in nodal cuttings of Camellia sinensis (L.) by suppression subtractive hybridization. Gene 2013, 514, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Kono, A.; Umedahara, C.; Lee, J.; Ito, M.; Uchimiya, H.; Umeda, M. Arabidopsis D-Type Cyclin CYCD4;1 Is a Novel Cyclin Partner of B2-Type Cyclin-Dependent Kinase. Plant Physiol. 2003, 132, 1315–1321. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Wang, H.; Zhuang, D.; Zhu, H.; Du, Y.; Cheng, Z.; Cui, W.; Rogers, H.J.; Zhang, Q.; Jia, C. Roles of MSH2 and MSH6 in cadmium-induced G2/M checkpoint arrest in Arabidopsis roots. Chemosphere 2018, 201, 586–594. [Google Scholar] [CrossRef]
- García-Cruz, K.V.; García-Ponce, B.; Garay-Arroyo, A.; Sanchez, M.D.L.P.; Ugartechea-Chirino, Y.; Desvoyes, B.; Pacheco-Escobedo, M.A.; Tapia-López, R.; Ransom-Rodríguez, I.; Gutierrez, C. The MADS-box XAANTAL1 increases proliferation at the Arabidopsis root stem-cell niche and participates in transition to differentiation by regulating cell-cycle components. Ann. Bot. 2016, 118, 787–796. [Google Scholar] [CrossRef]
- Lee, Y.P.; Fleming, A.; Korner, C.; Meins, F. Differential expression of the CBF pathway and cell cycle-related genes in Arabidopsis accessions in response to chronic low-temperature exposure. Plant Biol. 2009, 11, 273–283. [Google Scholar] [CrossRef]
- De Veylder, L.; Engler, J.D.A.; Burssens, S.; Manevski, A.; Lescure, B.; Van Montagu, M.; Engler, G.; Inze, D. A new D-type cyclin of Arabidopsis thaliana expressed during lateral root primordia formation. Planta 1999, 208, 453–462. [Google Scholar] [CrossRef]
- Wildwater, M.; Campilho, A.; Perezperez, J.M.; Heidstra, R.; Blilou, I.; Korthout, H.; Chatterjee, J.; Mariconti, L.; Gruissem, W.; Scheres, B. The RETINOBLASTOMA-RELATED Gene Regulates Stem Cell Maintenance in Arabidopsis Roots. Cell 2005, 123, 1337–1349. [Google Scholar] [CrossRef]
- Galinha, C.; Hofhuis, H.; Luijten, M.; Willemsen, V.; Blilou, I.; Heidstra, R.; Scheres, B. PLETHORA proteins as dose-dependent master regulators of Arabidopsis root development. Nature 2007, 449, 1053–1057. [Google Scholar] [CrossRef] [PubMed]
- Sozzani, R.; Cui, H.; Morenorisueno, M.A.; Busch, W.; Van Norman, J.M.; Vernoux, T.; Brady, S.M.; Dewitte, W.; Murray, J.A.H.; Benfey, P.N. Spatiotemporal regulation of cell-cycle genes by SHORTROOT links patterning and growth. Nature 2010, 466, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; De Smet, I.; Ding, Z. Shaping a root system: Regulating lateral versus primary root growth. Trends Plant Sci. 2014, 19, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Aichinger, E.; Villar, C.B.R.; Mambro, R.D.; Sabatini, S.; Kohler, C. The CHD3 Chromatin Remodeler PICKLE and Polycomb Group Proteins Antagonistically Regulate Meristem Activity in the Arabidopsis Root. Plant Cell 2011, 23, 1047–1060. [Google Scholar] [CrossRef]
- Aida, M.; Beis, D.; Heidstra, R.; Willemsen, V.; Blilou, I.; Galinha, C.; Nussaume, L.; Noh, Y.; Amasino, R.M.; Scheres, B. The PLETHORA Genes Mediate Patterning of the Arabidopsis Root Stem Cell Niche. Cell 2004, 119, 109–120. [Google Scholar] [CrossRef]
- Helariutta, Y.; Fukaki, H.; Wysockadiller, J.; Nakajima, K.; Jung, J.; Sena, G.; Hauser, M.; Benfey, P.N. The SHORT-ROOT Gene Controls Radial Patterning of the Arabidopsis Root through Radial Signaling. Cell 2000, 101, 555–567. [Google Scholar] [CrossRef]
- Laurenzio, L.D.; Wysockadiller, J.; Malamy, J.E.; Pysh, L.; Helariutta, Y.; Freshour, G.; Hahn, M.G.; Feldmann, K.A.; Benfey, P.N. The SCARECROW Gene Regulates an Asymmetric Cell Division That Is Essential for Generating the Radial Organization of the Arabidopsis Root. Cell 1996, 86, 423–433. [Google Scholar] [CrossRef]
- Nardmann, J.; Reisewitz, P.; Werr, W. Discrete shoot and root stem cell-promoting WUS/WOX5 functions are an evolutionary innovation of angiosperms. Mol. Biol. Evolut. 2009, 26, 1745–1755. [Google Scholar] [CrossRef]
- Sarkar, A.K.; Luijten, M.; Miyashima, S.; Lenhard, M.; Hashimoto, T.; Nakajima, K.; Scheres, B.; Heidstra, R.; Laux, T. Conserved factors regulate signalling in Arabidopsis thaliana shoot and root stem cell organizers. Nature 2007, 446, 811–814. [Google Scholar] [CrossRef]
- Matsubayashi, Y.; Sakagami, Y. PEPTIDE HORMONES IN PLANTS. Annu. Rev. Plant Biol. 2006, 57, 649–674. [Google Scholar] [CrossRef]
- Han, J.; Tan, J.; Tu, L.; Zhang, X. A peptide hormone gene, GhPSK promotes fibre elongation and contributes to longer and finer cotton fibre. Plant Biotechnol. J. 2014, 12, 861–871. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Shen, S.; Kobayashi, T.; Matsubayashi, Y.; Sakagami, Y.; Kamada, H. Stimulatory Effects of a Novel Peptidyl Plant Growth Factor, Phytosulfokine-α, on the Adventitious Bud Formation from Callus of Antirrhinum majus. Plant Biotechnol. 1999, 16, 231–234. [Google Scholar] [CrossRef]
- Porceddu, A.; Stals, H.; Reichheld, J.; Segers, G.; De Veylder, L.; Barroco, R.D.P.; Casteels, P.; Van Montagu, M.; Inze, D.; Mironov, V. A Plant-specific Cyclin-dependent Kinase Is Involved in the Control of G2/M Progression in Plants. J. Biol. Chem. 2001, 276, 36354–36360. [Google Scholar] [CrossRef] [PubMed]
- Segers, G.; Gadisseur, I.; Bergounioux, C.; Engler, J.D.A.; Jacqmard, A.; Van Montagu, M.; Inze, D. The Arabidopsis cyclin-dependent kinase gene cdc2bAt is preferentially expressed during S and G2 phases of the cell cycle. Plant J. 1996, 10, 601–612. [Google Scholar] [CrossRef]
- Andersen, S.U.; Buechel, S.; Zhao, Z.; Ljung, K.; Novak, O.; Busch, W.; Schuster, C.M.; Lohmann, J.U. Requirement of B2-Type Cyclin-Dependent Kinases for Meristem Integrity in Arabidopsis thaliana. Plant Cell 2008, 20, 88–100. [Google Scholar] [CrossRef]
- Borghi, L.; Gutzat, R.; Futterer, J.; Laizet, Y.H.; Hennig, L.; Gruissem, W. Arabidopsis RETINOBLASTOMA-RELATED Is Required for Stem Cell Maintenance, Cell Differentiation, and Lateral Organ Production. Plant Cell 2010, 22, 1792–1811. [Google Scholar] [CrossRef]
- Zhou, X.; Zheng, R.; Liu, G.; Xu, Y.; Zhou, Y.; Laux, T.; Zhen, Y.; Harding, S.A.; Shi, J.; Chen, J. Desiccation treatment and endogenous IAA levels are key factors influencing high frequency somatic embryogenesis in Cunninghamia lanceolata (Lamb.) Hook. Front. Plant Sci. 2017, 8, 2054. [Google Scholar] [CrossRef]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, J.; Liu, W.; Luo, Z.; Wang, P.; Zhang, Y.; Zheng, R.; Shi, J. Transcriptome characteristics and six alternative expressed genes positively correlated with the phase transition of annual cambial activities in Chinese Fir (Cunninghamia lanceolata (Lamb.) Hook). PLoS ONE 2013, 8, e71562. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, H.; Zheng, R.; Hao, Z.; Meng, Y.; Weng, Y.; Zhou, X.; Zhu, L.; Hu, X.; Wang, G.; Shi, J.; et al. Cunninghamia lanceolata PSK Peptide Hormone Genes Promote Primary Root Growth and Adventitious Root Formation. Plants 2019, 8, 520. https://doi.org/10.3390/plants8110520
Wu H, Zheng R, Hao Z, Meng Y, Weng Y, Zhou X, Zhu L, Hu X, Wang G, Shi J, et al. Cunninghamia lanceolata PSK Peptide Hormone Genes Promote Primary Root Growth and Adventitious Root Formation. Plants. 2019; 8(11):520. https://doi.org/10.3390/plants8110520
Chicago/Turabian StyleWu, Hua, Renhua Zheng, Zhaodong Hao, Yan Meng, Yuhao Weng, Xiaohong Zhou, Liming Zhu, Xiangyang Hu, Guibin Wang, Jisen Shi, and et al. 2019. "Cunninghamia lanceolata PSK Peptide Hormone Genes Promote Primary Root Growth and Adventitious Root Formation" Plants 8, no. 11: 520. https://doi.org/10.3390/plants8110520
APA StyleWu, H., Zheng, R., Hao, Z., Meng, Y., Weng, Y., Zhou, X., Zhu, L., Hu, X., Wang, G., Shi, J., & Chen, J. (2019). Cunninghamia lanceolata PSK Peptide Hormone Genes Promote Primary Root Growth and Adventitious Root Formation. Plants, 8(11), 520. https://doi.org/10.3390/plants8110520




