Comprehensive Evaluation of Five Pecan (Carya illinoinensis) Cultivars at Seedling Stage Based on Principal Component Analysis
Abstract
1. Introduction
2. Results
2.1. Comparison of Growth at Different Stages of Pecan Seedlings
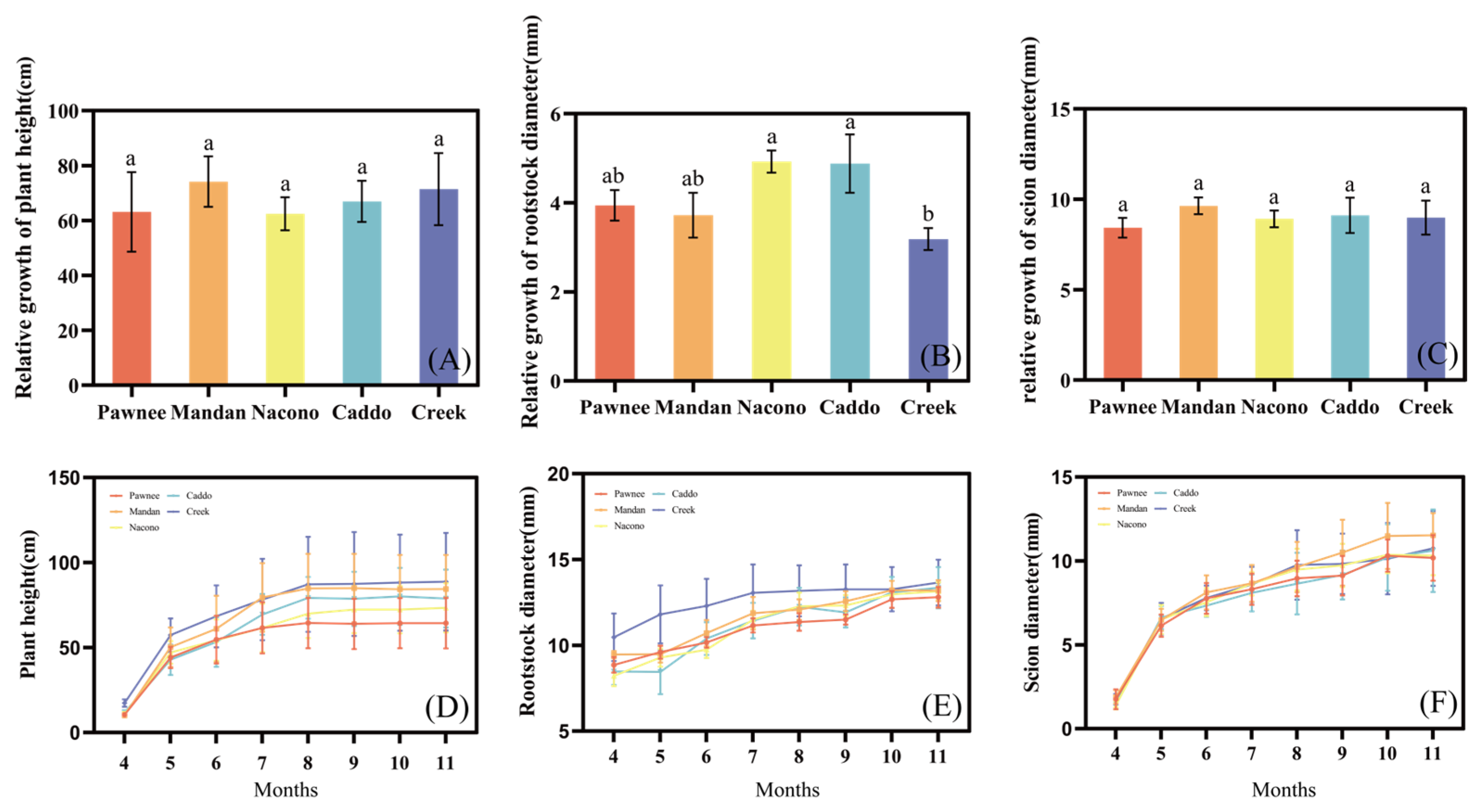
2.1.1. Plant Height, Rootstock Diameter, and Scion Diameter
2.1.2. Leaf Length, Leaf Width, and Leaf Area
2.1.3. Biomass and Quality Index of Seedlings
2.2. Comparison of the Anatomical Structures of Different Pecans at the Seedling Stage
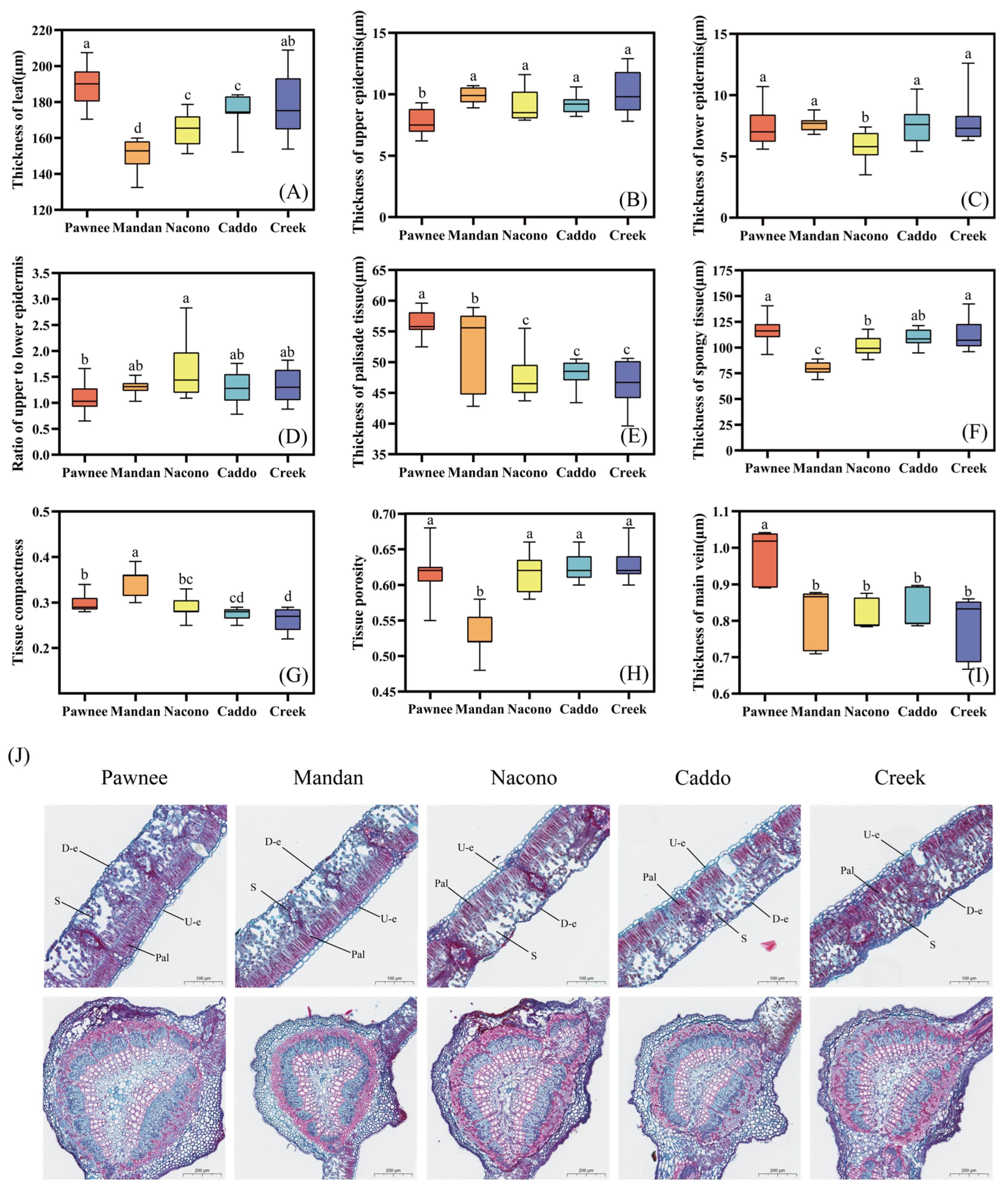
2.2.1. Leaf Anatomical Structure
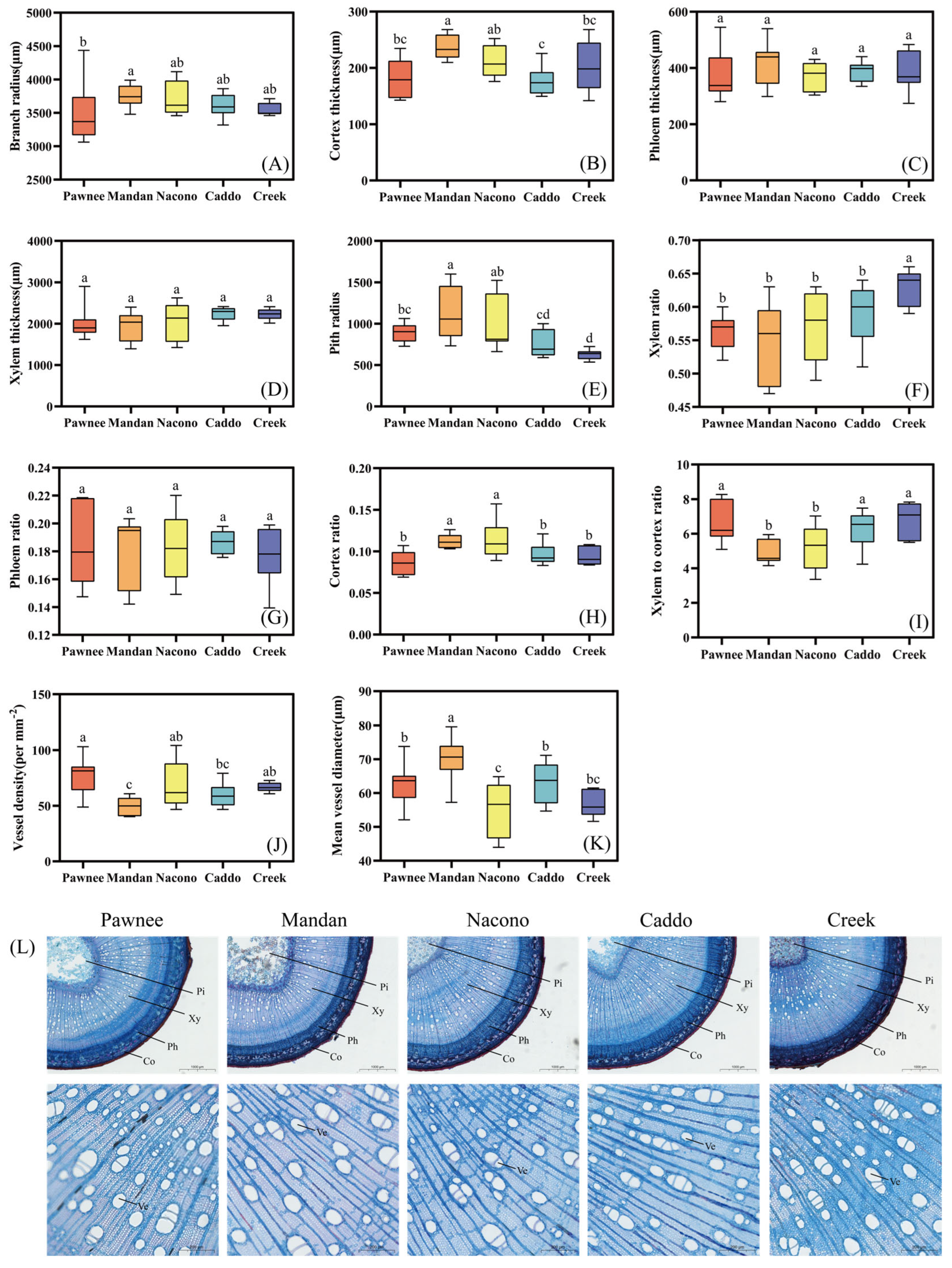
2.2.2. Stem Anatomical Structure
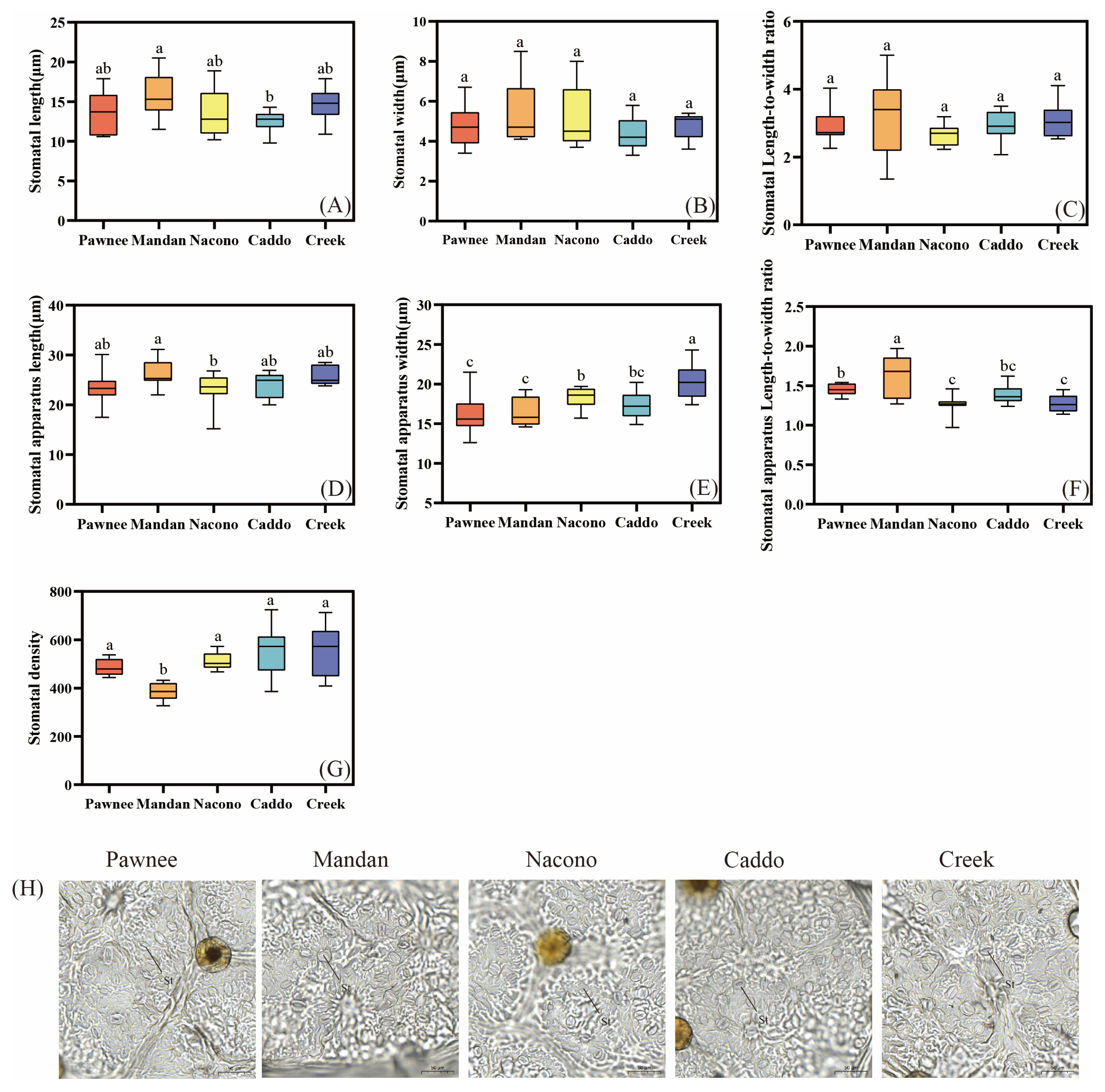
2.2.3. Leaf Epidermal Micromorphology
2.3. Comparison of Photosynthetic Physiology of Different Cultivars of Pecan at the Seedling Stage
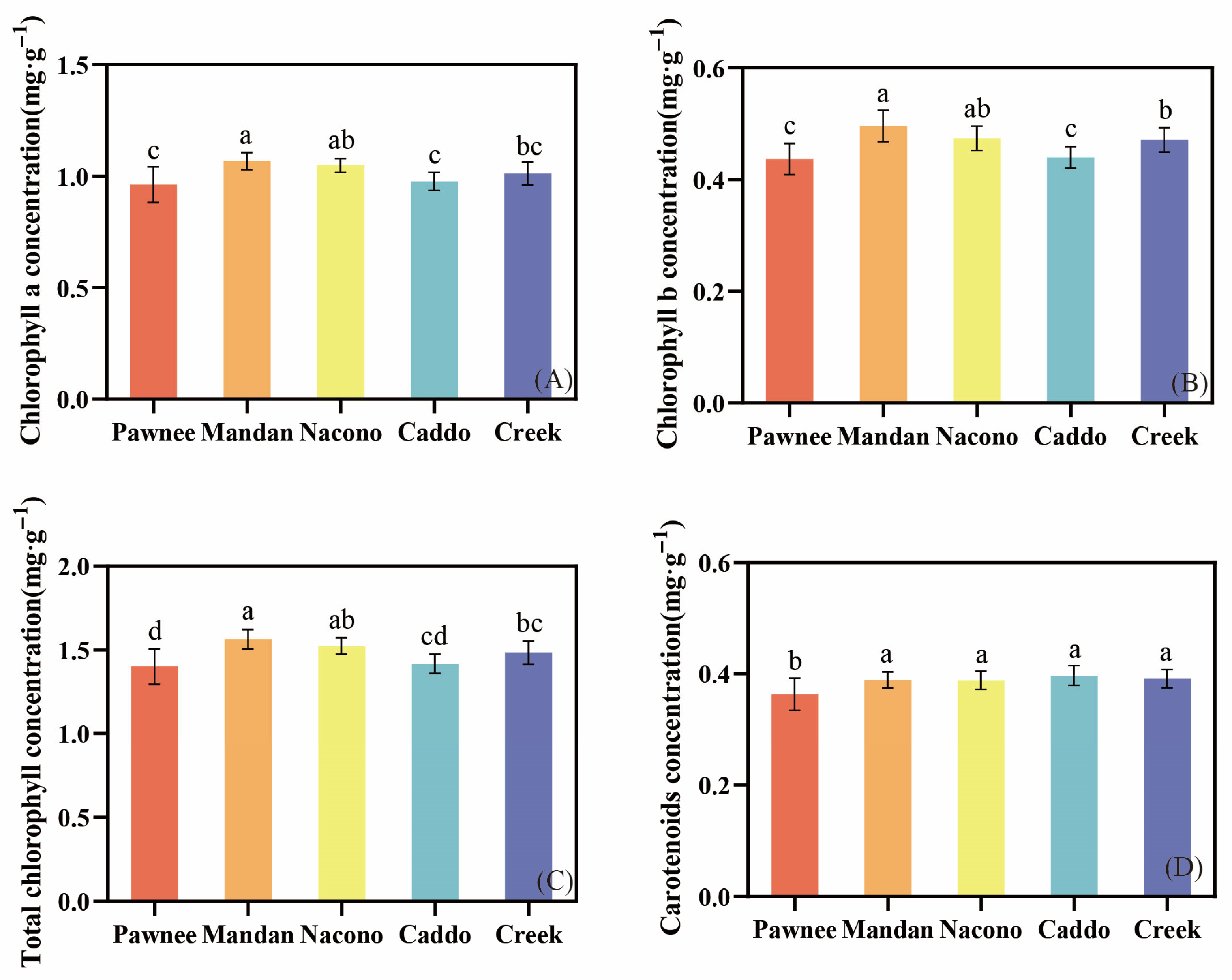
2.3.1. Photosynthetic Pigment Content
2.3.2. Photosynthetic Parameters
2.3.3. Chlorophyll Fluorescence Parameters
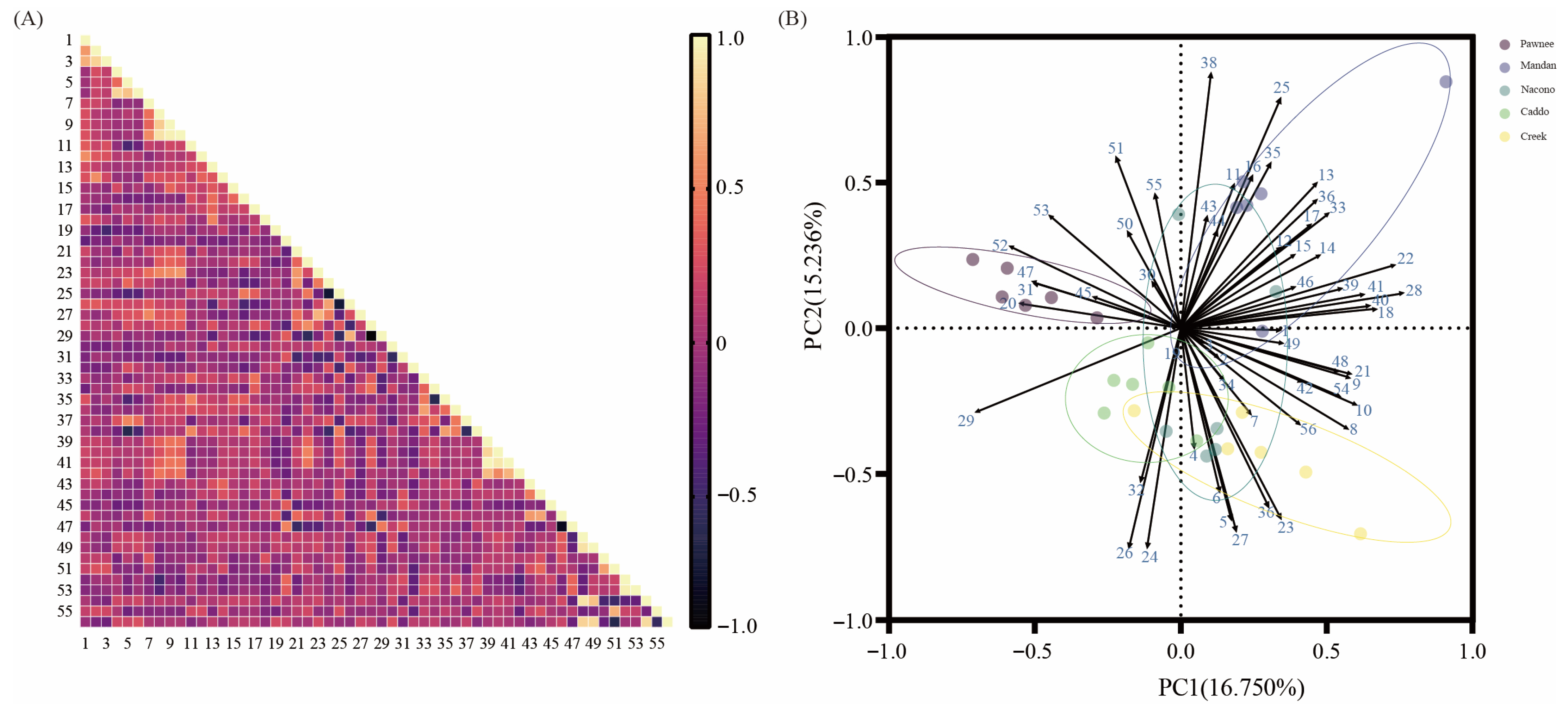
2.4. Comprehensive Evaluation of Different Cultivars of Pecan
3. Discussion
3.1. Differences in the Vegetative Growth of Grafted Pecan Seedlings of Different Cultivars During the Seedling Stage
3.2. Differences in the Anatomical Structure of Grafted Seedlings of Different Cultivars of Pecan at the Seedling Stage
3.3. Differences in Photosynthetic Capacity of Grafted Seedlings of Different Cultivars of Pecan at the Seedling Stage
3.4. Principal Component Analysis of Seedlings Among Different Pecan Cultivars and Limitations
4. Materials and Methods
4.1. Plant Materials
4.2. Experimental Site
4.3. Experimental Design
4.4. Measurement
4.4.1. Measurement of Growth Indicators
4.4.2. Observation of Anatomical Structure
4.4.3. Measurement of Photosynthetic Indicators
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhang, R.; Peng, F.; Li, Y. Pecan Production in China. Sci. Hortic. 2015, 197, 719–727. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Sabharanjak, S.M.; Zengin, G.; Mollica, A.; Szostak, A.; Simirgiotis, M.; Huminiecki, Ł.; Horbanczuk, O.K.; Nabavi, S.M.; Mocan, A. Pecan Nuts: A Review of Reported Bioactivities and Health Effects. Trends Food Sci. Technol. 2018, 71, 246–257. [Google Scholar] [CrossRef]
- Villarreal-Lozoya, J.E.; Lombardini, L.; Cisneros-Zevallos, L. Phytochemical Constituents and Antioxidant Capacity of Different Pecan [Carya Illinoinensis (Wangenh.) K. Koch] Cultivars. Food Chem. 2007, 102, 1241–1249. [Google Scholar] [CrossRef]
- Hilbig, J.; Policarpi, P.D.B.; Grinevicius, V.M.A.D.S.; Mota, N.S.R.S.; Toaldo, I.M.; Luiz, M.T.B.; Pedrosa, R.C.; Block, J.M. Aqueous Extract from Pecan Nut [Carya Illinoinensis (Wangenh) C. Koch] Shell Show Activity against Breast Cancer Cell Line MCF-7 and Ehrlich Ascites Tumor in Balb-C Mice. J. Ethnopharmacol. 2018, 211, 256–266. [Google Scholar] [CrossRef]
- Han, M.; Peng, F.; Marshall, P. Pecan Phenology in Southeastern China. Ann. Appl. Biol. 2018, 172, 160–169. [Google Scholar] [CrossRef]
- Luo, X.; Li, Z.; Sun, Z.; Wan, X. Analysis of Pecan Cultivars Mahan and Western in East China. Genet. Mol. Res. 2016, 15, 12. [Google Scholar] [CrossRef]
- Sparks, D. Adaptability of Pecan as a Species. HortScience 2005, 40, 1175–1189. [Google Scholar] [CrossRef]
- USDA-FAS. Global Pecan Production Estimates 2024/2025. USDA Foreign Agricultural Service, Office of Global Analysis. 2025. Available online: https://www.fas.usda.gov/data/oilseeds-world-markets-and-trade (accessed on 20 July 2025).
- Reid, W.; Hunt, K.L. Pecan Production in the Northern United States. Horttechnology 2000, 10, 298–301. [Google Scholar] [CrossRef]
- Zheng, J.; Hänninen, H.; Lin, J.; Shen, S.; Zhang, R. Extending the Cultivation Area of Pecan (Carya Illinoinensis) Toward the South in Southeastern Subtropical China May Cause Increased Cold Damage. Front. Plant Sci. 2021, 12, 768963. [Google Scholar] [CrossRef]
- Mo, Z.; Feng, G.; Su, W.; Liu, Z.; Peng, F. Transcriptomic Analysis Provides Insights into Grafting Union Development in Pecan (Carya Illinoinensis). Genes 2018, 9, 71. [Google Scholar] [CrossRef]
- Wang, X.; Stein, L.; Black, M.; Kubenka, K.; Randall, J.; Ding, C. Phenotypic Diversity and Population Structure of Pecan (Carya Illinoinensis) Collections Reveals Geographic Patterns. Sci. Rep. 2024, 14, 18592. [Google Scholar] [CrossRef]
- Ojeda-Barrios, D.; Abadía, J.; Lombardini, L.; Abadía, A.; Vázquez, S. Zinc Deficiency in Field-grown Pecan Trees: Changes in Leaf Nutrient Concentrations and Structure. J. Sci. Food Agric. 2012, 92, 1672–1678. [Google Scholar] [CrossRef]
- Dobbertin, M. Tree Growth as Indicator of Tree Vitality and of Tree Reaction to Environmental Stress: A Review. Eur. J. For. Res. 2006, 125, 89. [Google Scholar] [CrossRef]
- Trugman, A.T.; Anderegg, L.D.L.; Sperry, J.S.; Wang, Y.; Venturas, M.; Anderegg, W.R.L. Leveraging Plant Hydraulics to Yield Predictive and Dynamic Plant Leaf Allocation in Vegetation Models with Climate Change. Glob. Change Biol. 2019, 25, 4008–4021. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.-P.; Zhao, X.-D.; Han, L.-J.; Li, H.-R. Leaf Dry Mass per Unit Area and Leaf Pigments Underlying the Higher Stomatal Conductance of Deciduous Species Relative to Evergreen Species in Dendrobium. Bot. Stud. 2025, 66, 11. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, J.O.; Park, B.B. The Leaf Trichome, Venation, and Mesophyll Structural Traits Play Important Roles in the Physiological Responses of Oak Seedlings to Water-Deficit Stress. Int. J. Mol. Sci. 2022, 23, 8640. [Google Scholar] [CrossRef]
- Jahan, E.; Sharwood, R.E.; Tissue, D.T. Effects of Leaf Age during Drought and Recovery on Photosynthesis, Mesophyll Conductance and Leaf Anatomy in Wheat Leaves. Front. Plant Sci. 2023, 14, 1091418. [Google Scholar] [CrossRef]
- García-Cervigón, A.I.; García-López, M.A.; Pistón, N.; Pugnaire, F.I.; Olano, J.M. Co-Ordination between Xylem Anatomy, Plant Architecture and Leaf Functional Traits in Response to Abiotic and Biotic Drivers in a Nurse Cushion Plant. Ann. Bot. 2021, 127, 919–929. [Google Scholar] [CrossRef]
- Taratima, W.; Ritmaha, T.; Jongrungklang, N.; Maneerattanarungroj, P.; Kunpratum, N. Effect of Stress on the Leaf Anatomy of Sugarcane Cultivars with Different Drought Tolerance (Saccharum Officinarum, Poaceae). Rev. De-Biol. Trop. 2020, 68, 1159–1170. [Google Scholar] [CrossRef]
- Bao, X.-G.; Chong, P.-F.; He, C.; Lu, X.-M.; Wang, X.-Y.; Zhang, F.; Tan, B.-B.; Yang, J.-L.; Gao, L.-L. Enterobacter-Inoculation Altered the C, N Contents and Regulated Biomass Allocation in Reaumuria Soongorica to Promote Plant Growth and Improve Salt Stress Tolerance. Front. Plant Sci. 2024, 15, 1502659. [Google Scholar] [CrossRef]
- Wu, Y.; Zhao, Y.; Sang, X.; Yang, X. Selecting Superior Variety of Atractylodes Lancea through Photosynthetic Characters and Chlorophyll Fluorescence Parameters. Planta Medica 2011, 77, PD14. [Google Scholar] [CrossRef]
- Bussotti, F.; Gerosa, G.; Digrado, A.; Pollastrini, M. Selection of Chlorophyll Fluorescence Parameters as Indicators of Photosynthetic Efficiency in Large Scale Plant Ecological Studies. Ecol. Indic. 2020, 108, 105686. [Google Scholar] [CrossRef]
- Feghhi, J.; Teimouri, S.; Makhdoum, M.F.; Erfanifard, Y.; Abbaszadeh Tehrani, N. The Assessment of Degradation to Sustainability in an Urban Forest Ecosystem by GIS. Urban For. Urban Green. 2017, 27, 383–389. [Google Scholar] [CrossRef]
- Deshmukh, R.; Sonah, H.; Belanger, R.R. New Evidence Defining the Evolutionary Path of Aquaporins Regulating Silicon Uptake in Land Plants. J. Exp. Bot. 2020, 71, 6775–6788. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, P.; Brilhante, M.Â.; Messerschmid, T.F.E.; Serrano, H.C.; Kadereit, G.; Branquinho, C.; De Vos, J.M. Plant Growth Forms Dictate Adaptations to the Local Climate. Front. Plant Sci. 2022, 13, 1023595. [Google Scholar] [CrossRef] [PubMed]
- Picchio, R.; Tavankar, F.; Nikooy, M.; Pignatti, G.; Venanzi, R.; Lo Monaco, A. Morphology, Growth and Architecture Response of Beech (Fagus Orientalis Lipsky) and Maple Tree (Acer Velutinum Boiss.) Seedlings to Soil Compaction Stress Caused by Mechanized Logging Operations. Forests 2019, 10, 771. [Google Scholar] [CrossRef]
- Branch, H.A.; Moxley, D.R.; Angert, A.L. Regional Differences in Leaf Evolution Facilitate Photosynthesis Following Severe Drought. New Phytol. 2024, 243, 2457–2469. [Google Scholar] [CrossRef]
- Vicente, E.; Didion-Gency, M.; Morcillo, L.; Morin, X.; Vilagrosa, A.; Grossiord, C. Aridity and Cold Temperatures Drive Divergent Adjustments of European Beech Xylem Anatomy, Hydraulics and Leaf Physiological Traits. Tree Physiol. 2022, 42, 1720–1735. [Google Scholar] [CrossRef]
- Shi, F.; Pan, Z.; Dai, P.; Shen, Y.; Lu, Y.; Han, B. Effect of Waterlogging Stress on Leaf Anatomical Structure and Ultrastructure of Phoebe Sheareri Seedlings. Forests 2023, 14, 1294. [Google Scholar] [CrossRef]
- Zhang, F.; Lu, K.; Gu, Y.; Zhang, L.; Li, W.; Li, Z. Effects of Low-Temperature Stress and Brassinolide Application on the Photosynthesis and Leaf Structure of Tung Tree Seedlings. Front. Plant Sci. 2020, 10, 1767. [Google Scholar] [CrossRef]
- Henn, J.J.; Buzzard, V.; Enquist, B.J.; Halbritter, A.H.; Klanderud, K.; Maitner, B.S.; Michaletz, S.T.; Pötsch, C.; Seltzer, L.; Telford, R.J.; et al. Intraspecific Trait Variation and Phenotypic Plasticity Mediate Alpine Plant Species Response to Climate Change. Front. Plant Sci. 2018, 9, 1548. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Huang, X.; Chen, J.; Li, L.; Cheng, J.; Wang, S.; Liu, J. Plasticity of Leaf Traits of Juglans Regia L. Luodianense Liu et Xu Seedlings Under Different Light Conditions in Karst Habitats. Forests 2021, 12, 81. [Google Scholar] [CrossRef]
- Aaltonen, H.; Lindén, A.; Heinonsalo, J.; Biasi, C.; Pumpanen, J. Effects of Prolonged Drought Stress on Scots Pine Seedling Carbon Allocation. Tree Physiol. 2016, 37, 418–427. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhao, D.; Zhang, P.; Liu, F.; Jia, M.; Liang, J. Seedling Evaluation of Six Walnut Rootstock Species Originated in China Based on Principal Component Analysis and Cluster Analysis. Sci. Hortic. 2020, 265, 109212. [Google Scholar] [CrossRef]
- De La Riva, E.G.; Tosto, A.; Pérez-Ramos, I.M.; Navarro-Fernández, C.M.; Olmo, M.; Anten, N.P.R.; Marañón, T.; Villar, R. A Plant Economics Spectrum in Mediterranean Forests along Environmental Gradients: Is There Coordination among Leaf, Stem and Root Traits? J. Veg. Sci. 2016, 27, 187–199. [Google Scholar] [CrossRef]
- Ristova, D.; Busch, W. Natural Variation of Root Traits: From Development to Nutrient Uptake. Plant Physiol. 2014, 166, 518–527. [Google Scholar] [CrossRef]
- Contador, M.L.; Comas, L.H.; Metcalf, S.G.; Stewart, W.L.; Porris Gomez, I.; Negron, C.; Lampinen, B.D. Root Growth Dynamics Linked to Above-Ground Growth in Walnut (Juglans Regia). Ann. Bot. 2015, 116, 49–60. [Google Scholar] [CrossRef]
- Soheili, F.; Heydari, M.; Woodward, S.; Naji, H.R. Adaptive Mechanism in Quercus Brantii Lindl. Leaves under Climatic Differentiation: Morphological and Anatomical Traits. Sci. Rep. 2023, 13, 3580. [Google Scholar] [CrossRef]
- Yu-Bing, L.; Xin-Rong, L.; Meng-Meng, L.; Dan, L.; Wen-Li, Z. Leaf (or Assimilation Branch) Epidermal Micromorphology of Desert Plant in Arid and Semiarid Areas of China. Chin. J. Plant Ecol. 2016, 40, 1189–1207. [Google Scholar] [CrossRef]
- Li, S.; Tan, T.; Fan, Y.; Raza, M.A.; Wang, Z.; Wang, B.; Zhang, J.; Tan, X.; Chen, P.; Shafiq, I.; et al. Responses of Leaf Stomatal and Mesophyll Conductance to Abiotic Stress Factors. J. Integr. Agric. 2022, 21, 2787–2804. [Google Scholar] [CrossRef]
- Singh, M.; Mohan, R.; Mishra, S.; Goyal, N.; Shanker, K.; Gupta, N.; Kumar, B. Ultra Performance Liquid Chromatography Coupled with Principal Component and Cluster Analysis of Swertia Chirayita for Adulteration Check. J. Pharm. Biomed. Anal. 2019, 164, 302–308. [Google Scholar] [CrossRef]
- Momayyezi, M.; Rippner, D.A.; Duong, F.V.; Raja, P.V.; Brown, P.J.; Kluepfel, D.A.; Earles, J.M.; Forrestel, E.J.; Gilbert, M.E.; McElrone, A.J. Structural and Functional Leaf Diversity Lead to Variability in Photosynthetic Capacity across a Range of Juglans Regia Genotypes. Plant Cell Environ. 2022, 45, 2351–2365. [Google Scholar] [CrossRef] [PubMed]
- Karabourniotis, G.; Liakopoulos, G.; Bresta, P.; Nikolopoulos, D. The Optical Properties of Leaf Structural Elements and Their Contribution to Photosynthetic Performance and Photoprotection. Plants 2021, 10, 1455. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Shen, F.; Yang, L.; Xing, W.; Clothier, B. Limited Acclimation in Leaf Morphology and Anatomy to Experimental Drought in Temperate Forest Species. Biology 2022, 11, 1186. [Google Scholar] [CrossRef]
- Tan, F.; Cao, W.; Li, X.; Li, Q. Characteristics, Relationships, and Anatomical Basis of Leaf Hydraulic Traits and Economic Traits in Temperate Desert Shrub Species. Life 2024, 14, 834. [Google Scholar] [CrossRef]
- Arab, M.M.; Askari, H.; Aliniaeifard, S.; Mokhtassi-Bidgoli, A.; Estaji, A.; Sadat-Hosseini, M.; Sohrabi, S.S.; Mesgaran, M.B.; Leslie, C.A.; Brown, P.J.; et al. Natural Variation in Photosynthesis and Water Use Efficiency of Locally Adapted Persian Walnut Populations under Drought Stress and Recovery. Plant Physiol. Biochem. 2023, 201, 107859. [Google Scholar] [CrossRef]
- Tang, J.; Ji, X.; Li, A.; Zheng, X.; Zhang, Y.; Zhang, J. Effect of Persistent Salt Stress on the Physiology and Anatomy of Hybrid Walnut (Juglans Major × Juglans Regia) Seedlings. Plants 2024, 13, 1840. [Google Scholar] [CrossRef]
- Xiao, Y.; Tholen, D.; Zhu, X.-G. The Influence of Leaf Anatomy on the Internal Light Environment and Photosynthetic Electron Transport Rate: Exploration with a New Leaf Ray Tracing Model. J. Exp. Bot. 2016, 67, 6021–6035. [Google Scholar] [CrossRef]
- Jiang, C.-D.; Wang, X.; Gao, H.-Y.; Shi, L.; Chow, W.S. Systemic Regulation of Leaf Anatomical Structure, Photosynthetic Performance, and High-Light Tolerance in Sorghum. Plant Physiol. 2011, 155, 1416–1424. [Google Scholar] [CrossRef]
- Li, S.; Wang, J.; Lu, S.; Salmon, Y.; Liu, P.; Guo, J. Trade-Off between Hydraulic Safety and Efficiency in Plant Xylem and Its Influencing Factors. Forests 2023, 14, 1817. [Google Scholar] [CrossRef]
- Pratt, R.B.; Jacobsen, A.L.; Percolla, M.I.; De Guzman, M.E.; Traugh, C.A.; Tobin, M.F. Trade-Offs among Transport, Support, and Storage in Xylem from Shrubs in a Semiarid Chaparral Environment Tested with Structural Equation Modeling. Proc. Natl. Acad. Sci. USA 2021, 118, e2104336118. [Google Scholar] [CrossRef]
- Pan, S.; Wang, X.; Yan, Z.; Wu, J.; Guo, L.; Peng, Z.; Wu, Y.; Li, J.; Wang, B.; Su, Y.; et al. Leaf Stomatal Configuration and Photosynthetic Traits Jointly Affect Leaf Water Use Efficiency in Forests along Climate Gradients. New Phytol. 2024, 244, 1250–1262. [Google Scholar] [CrossRef]
- Li, Y.; Li, H.; Li, Y.; Zhang, S. Improving Water-Use Efficiency by Decreasing Stomatal Conductance and Transpiration Rate to Maintain Higher Ear Photosynthetic Rate in Drought-Resistant Wheat. Crop J. 2017, 5, 231–239. [Google Scholar] [CrossRef]
- Li, S.; Lu, S.; Wang, J.; Chen, Z.; Zhang, Y.; Duan, J.; Liu, P.; Wang, X.; Guo, J. Responses of Physiological, Morphological and Anatomical Traits to Abiotic Stress in Woody Plants. Forests 2023, 14, 1784. [Google Scholar] [CrossRef]
- Dai, X.; Zhai, R.; Lin, J.; Wang, Z.; Meng, D.; Li, M.; Mao, Y.; Gao, B.; Ma, H.; Zhang, B.; et al. Cell-Type-Specific PtrWOX4a and PtrVCS2 Form a Regulatory Nexus with a Histone Modification System for Stem Cambium Development in Populus Trichocarpa. Nat. Plants 2023, 9, 96–111. [Google Scholar] [CrossRef]
- Lombardini, L.; Restrepo-Diaz, H.; Volder, A. Photosynthetic Light Response and Epidermal Characteristics of Sun and Shade Pecan Leaves. J. Am. Soc. Hortic. Sci. 2009, 134, 372–378. [Google Scholar] [CrossRef]
- Li, Z.; Jiang, E.; Liu, M.; Sun, Q.; Gao, Z.; Du, Y. Effects of Coverlys TF150® on the Photosynthetic Characteristics of Grape. Int. J. Mol. Sci. 2023, 24, 16659. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, Y.; Yan, H.; Cai, K.; Li, H.; Wu, Z.; Wu, J.; Yang, X.; Jiang, H.; Wang, Q.; et al. Integrated Metabolomic and Transcriptomic Analyses Reveal Different Metabolite Biosynthesis Profiles of Juglans Mandshurica in Shade. Front. Plant Sci. 2022, 13, 991874. [Google Scholar] [CrossRef] [PubMed]
- Al-Gaadi, K.A.; Tola, E.; Madugundu, R.; Zeyada, A.M.; Alameen, A.A.; Edrris, M.K.; Edrees, H.F.; Mahjoop, O. Response of Leaf Photosynthesis, Chlorophyll Content and Yield of Hydroponic Tomatoes to Different Water Salinity Levels. PLoS ONE 2024, 19, e0293098. [Google Scholar] [CrossRef]
- Yu, L.; Luo, X.; Croft, H.; Rogers, C.A.; Chen, J.M. Seasonal Variation in the Relationship between Leaf Chlorophyll Content and Photosynthetic Capacity. Plant Cell Environ. 2024, 47, 3953–3965. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.R. Chlorophyll Fluorescence: A Probe of Photosynthesis In Vivo. Annu. Rev. Plant Biol. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [PubMed]
- Raihan, M.R.H.; Antala, M.; Stróżecki, M.; Haque, M.I.; Hasanuzzaman, M.; Juszczak, R.; Rastogi, A. Silicon-Induced Photosynthetic Adaptations in Common Buckwheat under Salt Stress Revealed by Prompt Chlorophyll a Fluorescence Analysis. Sci. Rep. 2025, 15, 19343. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Zhang, Y.; Zhang, A.; Lu, C. Non-Photochemical Quenching: From Light Perception to Photoprotective Gene Expression. Int. J. Mol. Sci. 2022, 23, 687. [Google Scholar] [CrossRef] [PubMed]
- Bertamini, M.; Muthuchelian, K.; Rubinigg, M.; Zorer, R.; Velasco, R.; Nedunchezhian, N. Low-Night Temperature Increased the Photoinhibition of Photosynthesis in Grapevine (Vitis Vinifera L. Cv. Riesling) Leaves. Environ. Exp. Bot. 2006, 57, 25–31. [Google Scholar] [CrossRef]
- Ren, Y.-F.; Chen, G.-P.; Pu, T.; Chen, C.; Zeng, J.-X.; Peng, X.; Ma, Y.-W.; Yang, W.-Y.; Wang, X.-C. Responses of Photosynthetic Characteristics to Low Light Stress in Ear Leaves of High Photosynthetic Efficiency Maize at Narrow Row of Maize-Soybean Strip in-Tercropping System. Acta Agron. Sin. 2019, 45, 728. [Google Scholar] [CrossRef]
- Grauke, L.J.; Mendoza-Herrera, M.A.; Stelly, D.M.; Klein, P.E. ‘Jones Hybrid’ Hickory: A Case Study in Carya Curation. SpringerPlus 2016, 5, 1860. [Google Scholar] [CrossRef]
- Grauke, L.J.; Wood, B.W.; Harris, M.K. Crop Vulnerability: Carya. HortScience 2016, 51, 653–663. [Google Scholar] [CrossRef]
- Chen, H.; Wu, J.; Liu, J.; Tan, P.; Zhu, K.; Peng, F. Effects of Truffle Inoculation on Root Physiology and Mycorrhizosphere Microbial Communities of Carya Illinoinensis Seedlings. Forests 2023, 14, 2078. [Google Scholar] [CrossRef]
- Patel, I.; Gorim, L.Y.; Tanino, K.; Vandenberg, A. Diversity in Surface Microstructures of Trichomes, Epidermal Cells, and Stomata in Lentil Germplasm. Front. Front. Plant Sci. 2021, 12, 697692. [Google Scholar] [CrossRef]
- Chen, M.; Zhu, K.; Tan, P.; Liu, J.; Xie, J.; Yao, X.; Chu, G.; Peng, F. Ammonia–Nitrate Mixture Dominated by NH4+–N Promoted Growth, Photosynthesis and Nutrient Accumulation in Pecan (Carya Illinoinensis). Forests 2021, 12, 1808. [Google Scholar] [CrossRef]
| Cultivars | Leaf Length (cm) | Leaf Width (cm) | Leaf Area (mm2) | Length/Width Ratio |
|---|---|---|---|---|
| Pawnee | 71.39 ± 14.33 b | 37.94 ± 5.09 c | 2057.02 ± 543.61 b | 1.90 ± 0.40 a |
| Mandan | 76.04 ± 15.67 ab | 37.59 ± 7.50 c | 2258.92 ± 734.31 ab | 2.03 ± 0.31 a |
| Nacono | 70.70 ± 23.35 b | 49.09 ± 4.80 a | 2702.37 ± 991.47 a | 1.43 ± 0.39 b |
| Caddo | 67.29 ± 12.86 b | 41.51 ± 6.38 bc | 2136.19 ± 602.86 b | 1.63 ± 0.26 b |
| Creek | 87.67 ± 17.95 a | 44.36 ± 7.44 b | 2776.31 ± 692.52 a | 1.99 ± 0.36 a |
| Cultivars | Leaf Biomass (g) | Stem Biomass (g) | Root Biomass (g) | Total Biomass (g) | Shoot/Root Ratio | Graftling Quality Index |
|---|---|---|---|---|---|---|
| Pawnee | 24.85 ± 4.08 a | 26.66 ± 7.44 b | 99.98 ± 22.71 b | 151.50 ± 31.54 b | 1.92 ± 0.13 a | 27.10 ± 4.61 b |
| Mandan | 28.90 ± 13.23 a | 41.84 ± 12.88 ab | 142.98 ± 32.45 a | 213.72 ± 50.58 a | 2.13 ± 0.37 a | 30.41 ± 5.36 b |
| Nacono | 31.27 ± 6.62 a | 32.47 ± 9.89 ab | 109.17 ± 25.74 ab | 172.90 ± 34.78 ab | 1.80 ± 0.20 a | 27.36 ± 3.66 b |
| Caddo | 30.59 ± 8.30 a | 36.21 ± 9.54 ab | 111.20 ± 23.20 ab | 178.00 ± 39.14 ab | 1.66 ± 0.08 a | 27.06 ± 5.52 b |
| Creek | 27.47 ± 8.65 a | 48.41 ± 19.57 a | 139.55 ± 38.38 a | 215.43 ± 60.21 a | 1.91 ± 0.37 a | 39.47 ± 12.79 a |
| Cultivars | Ci (ppm) | E (mmol·m−2·s−1) | Gs (mmol·m−2·s−1) | Pn (μmol·m−2·s−1) | WUE (%) |
|---|---|---|---|---|---|
| Pawnee | 156.50 ± 28.37 a | 2.60 ± 0.80 b | 281.50 ± 53.03 a | 11.63 ± 1.85 a | 5.25 ± 1.69 a |
| Mandan | 176.00 ± 29.58 a | 3.37 ± 0.58 ab | 241.67 ± 13.92 b | 12.45 ± 1.26 a | 3.77 ± 0.65 b |
| Nacono | 182.33 ± 40.72 a | 3.50 ± 0.60 a | 251.00 ± 16.61 ab | 11.15 ± 1.37 a | 3.22 ± 0.37 b |
| Caddo | 166.67 ± 49.34 a | 3.45 ± 0.59 a | 250.50 ± 16.50 ab | 11.90 ± 1.37 a | 3.50 ± 0.48 b |
| Creek | 159.00 ± 17.99 a | 2.80 ± 0.67 ab | 236.83 ± 14.47 b | 11.28 ± 1.21 a | 4.16 ± 0.72 ab |
| Cultivars | Fo | Fm | Fo′ | Fm′ | Fv/Fo | Fv/Fm | qP | ΦPSII | NPQ |
|---|---|---|---|---|---|---|---|---|---|
| Pawnee | 281.44 ± 40.84 a | 1633.67 ± 196.80 a | 298.22 ± 11.03 bc | 1110.56 ± 33.39 abc | 4.82 ± 0.18 a | 0.83 ± 0.01 a | 0.98 ± 0.05 a | 0.73 ± 0.01 a | 0.47 ± 0.18 a |
| Mandan | 300.89 ± 41.49 a | 1678.67 ± 205.92 a | 307.11 ± 16.81 ab | 1130.56 ± 81.14 ab | 4.59 ± 0.23 ab | 0.82 ± 0.01 ab | 1.00 ± 0.05 a | 0.73 ± 0.01 a | 0.49 ± 0.21 a |
| Nacono | 302.67 ± 24.41 a | 1644.33 ± 205.33 a | 320.89 ± 15.82 a | 1167.44 ± 94.84 a | 4.43 ± 0.43 b | 0.81 ± 0.02 bc | 0.98 ± 0.04 a | 0.73 ± 0.02 a | 0.43 ± 0.28 a |
| Caddo | 283.78 ± 42.34 a | 1512.11 ± 211.50 a | 308.56 ± 18.61 ab | 1036.33 ± 80.03 c | 4.34 ± 0.30 b | 0.81 ± 0.01 c | 0.98 ± 0.07 a | 0.70 ± 0.02 b | 0.48 ± 0.29 a |
| Creek | 279.33 ± 37.77 a | 1596.47 ± 201.89 a | 289.11 ± 10.33 c | 1056.44 ± 83.43 bc | 4.44 ± 0.23 b | 0.82 ± 0.01 bc | 0.99 ± 0.05 a | 0.73 ± 0.02 a | 0.44 ± 0.24 a |
| Principal Component | Eigenvalue | Variance Contribution Rate/% | Cumulative Contribution Rate/% | Factor Weight |
|---|---|---|---|---|
| F1 | 9.380 | 16.750 | 16.750 | 0.180 |
| F2 | 8.532 | 15.236 | 31.986 | 0.163 |
| F3 | 5.292 | 9.449 | 41.435 | 0.101 |
| F4 | 4.712 | 8.414 | 49.849 | 0.090 |
| F5 | 4.207 | 7.513 | 57.363 | 0.081 |
| F6 | 3.753 | 6.702 | 64.064 | 0.072 |
| F7 | 3.378 | 6.032 | 70.096 | 0.065 |
| F8 | 2.461 | 4.394 | 74.490 | 0.047 |
| F9 | 2.395 | 4.277 | 78.767 | 0.046 |
| F10 | 1.779 | 3.177 | 81.945 | 0.034 |
| F11 | 1.484 | 2.650 | 84.595 | 0.028 |
| F12 | 1.347 | 2.405 | 86.999 | 0.026 |
| F13 | 1.248 | 2.228 | 89.227 | 0.024 |
| F14 | 1.147 | 2.049 | 91.276 | 0.022 |
| F15 | 1.080 | 1.928 | 93.204 | 0.021 |
| Cultivars | Scores | Ranking |
|---|---|---|
| Pawnee | −3.848 | 5 |
| Mandan | 2.667 | 1 |
| Nacono | −0.536 | 3 |
| Caddo | −0.566 | 4 |
| Creek | 2.283 | 2 |
| Soil Indicators | PH | SOC | TN | TP | TK | Olsen-P | Ex-K | Ex-Ca |
|---|---|---|---|---|---|---|---|---|
| Values | 6.92 | 102.4 g·kg−1 | 9.5 g·kg−1 | 3.3 g·kg−1 | 5.13 g·kg−1 | 8.02 mg·kg−1 | 86.74 mg·kg−1 | 800.56 mg·kg−1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, J.; Zhao, J.; Lu, L.; Tan, P.; Zhu, K.; Peng, F. Comprehensive Evaluation of Five Pecan (Carya illinoinensis) Cultivars at Seedling Stage Based on Principal Component Analysis. Plants 2025, 14, 2705. https://doi.org/10.3390/plants14172705
Zhu J, Zhao J, Lu L, Tan P, Zhu K, Peng F. Comprehensive Evaluation of Five Pecan (Carya illinoinensis) Cultivars at Seedling Stage Based on Principal Component Analysis. Plants. 2025; 14(17):2705. https://doi.org/10.3390/plants14172705
Chicago/Turabian StyleZhu, Jiaju, Juan Zhao, Longtao Lu, Pengpeng Tan, Kaikai Zhu, and Fangren Peng. 2025. "Comprehensive Evaluation of Five Pecan (Carya illinoinensis) Cultivars at Seedling Stage Based on Principal Component Analysis" Plants 14, no. 17: 2705. https://doi.org/10.3390/plants14172705
APA StyleZhu, J., Zhao, J., Lu, L., Tan, P., Zhu, K., & Peng, F. (2025). Comprehensive Evaluation of Five Pecan (Carya illinoinensis) Cultivars at Seedling Stage Based on Principal Component Analysis. Plants, 14(17), 2705. https://doi.org/10.3390/plants14172705







