Effects of Chili Straw Biochar on Alfalfa (Medicago sativa L.) Seed Germination and Seedling Growth on Electrolytic Manganese Residue
Abstract
1. Introduction
2. Results
2.1. Effects of Chili Straw Biochar on Alfalfa Germination on EMR
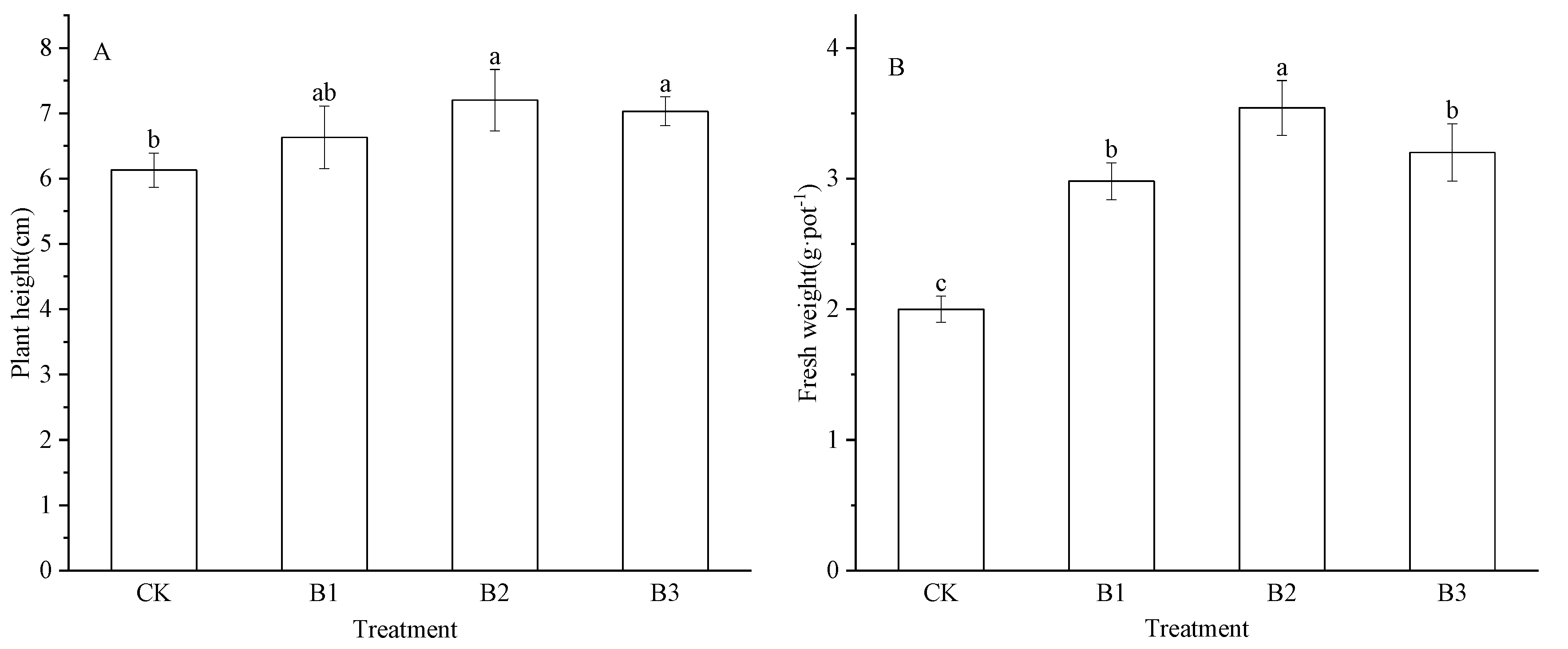
2.2. Effects of Chili Straw Biochar on Alfalfa Seedling Growth on EMR
2.3. Effects of Chili Straw Biochar on Physiological Characteristics of Alfalfa Seedlings Cultivated on EMR
2.4. Effects of Chili Straw Biochar on the Fundamental Properties of EMR
2.5. Correlation Analysis Between EMR and Alfalfa Parameters
3. Discussion
3.1. Mechanisms Underlying the Impact of Chili Straw Biochar on the Chemical Properties and Manganese Availability of EMR
3.2. Mechanisms Underlying the Impact of Chili Straw Biochar on Alfalfa Seed Germination in EMR
3.3. Mechanisms Underlying the Impact of Chili Straw Biochar on Alfalfa Seedling Growth in EMR
4. Materials and Methods
4.1. Test Materials
4.2. Experimental Design
4.3. Measurement Items and Methods
4.3.1. Seed GI
4.3.2. Physiological Indicators of Seedlings
4.3.3. Related Indicators of EMR
4.4. Data Processing
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Mn | Manganese |
| EMR | Electrolytic manganese residue |
| MDA | Malondialdehyde |
| AMn | Available Mn |
| GR | Germination rate |
| GE | Germination energy |
| GI | Germination index |
| OM | Organic matter |
| AN | Alkali-hydrolyzable nitrogen |
| AP | Available phosphorus |
| AK | Available potassium |
References
- Wu, T.; Ma, B.; An, Y.; Chen, Y.; Wang, C. Improvement of manganese electrolytic process and secondary resources recovery of manganese: A review. Process Saf. Environ. Prot. 2024, 186, 895–909. [Google Scholar] [CrossRef]
- Wu, Z.; Feng, Z.; Pu, S.; Zeng, C.; Zhao, Y.; Chen, C.; Song, H.; Feng, X. Mechanical properties and environmental characteristics of the synergistic preparation of cementitious materials using electrolytic manganese residue, steel slag, and blast furnace slag. Constr. Build. Mater. 2024, 411, 134480. [Google Scholar] [CrossRef]
- Wang, F.; Long, G.; Zhou, J.L. Enhanced green remediation and refinement disposal of electrolytic manganese residue using air-jet milling and horizontal-shaking leaching. J. Hazard. Mater. 2024, 465, 133419. [Google Scholar] [CrossRef]
- Li, W.; Jin, H.; Xie, H.; Wang, M.; Wang, X. Electrolytic manganese residue and red mud co-treatment: Synthesizing zeolite x and adsorbing leaching solution from electrolytic manganese residue. Sep. Purif. Technol. 2025, 356, 129795. [Google Scholar] [CrossRef]
- Shu, J.; Lin, F.; Chen, M.; Li, B.; Wei, L.; Wang, J.; Luo, Z.; Wang, R. An innovative method to enhance manganese and ammonia nitrogen leaching from electrolytic manganese residue by surfactant and anode iron plate. Hydrometallurgy 2020, 193, 105311. [Google Scholar] [CrossRef]
- Benidire, L.; Madline, A.; Pereira, S.I.A.; Castro, P.M.L.; Boularbah, A. Synergistic effect of organo-mineral amendments and plant growth-promoting rhizobacteria (PGPR) on the establishment of vegetation cover and amelioration of mine tailings. Chemosphere 2021, 262, 127803. [Google Scholar] [CrossRef]
- Bateman, A.M.; Erickson, T.E.; Merritt, D.J. Inorganic soil amendments alter seedling performance of native plant species in post-mining arid zone rehabilitation. J. Environ. Manag. 2019, 241, 179–186. [Google Scholar] [CrossRef]
- Nandillon, R.; Lebrun, M.; Miard, F.; Gaillard, M.; Sabatier, S.; Villar, M.; Bourgerie, S.; Morabito, D. Capability of amendments (biochar, compost and garden soil) added to a mining technosol contaminated by Pb and as to allow poplar seed (Populus nigra L.) germination. Environ. Monit. Assess. 2019, 191, 465. [Google Scholar] [CrossRef]
- Rosas-Ramírez, M.; Tovar-Sánchez, E.; Rodríguez-Solís, A.; Flores-Trujillo, K.; Castrejón-Godínez, M.L.; Mussali-Galante, P. Assisted phytoremediation between biochar and Crotalaria pumila to phytostabilize heavy metals in mine tailings. Plants 2024, 13, 2516. [Google Scholar] [CrossRef]
- Gao, Y.; Wu, P.; Jeyakumar, P.; Bolan, N.; Wang, H.; Gao, B.; Wang, S.; Wang, B. Biochar as a potential strategy for remediation of contaminated mining soils: Mechanisms, applications, and future perspectives. J. Environ. Manag. 2022, 313, 114973. [Google Scholar] [CrossRef]
- Williams, J.M.; Thomas, S.C. Effects of high-carbon wood ash biochar on volunteer vegetation establishment and community composition on metal mine tailings. Restor. Ecol. 2023, 31, 31. [Google Scholar] [CrossRef]
- Jia, J.; Dai, H.; Wei, S.; Xue, J.; Skuza, L.; Sun, Q.; Li, R. Toxicity of emerging contaminant antibiotics in soil to Capsicum annuum L. growth and their effects on it accumulating copper. Plant Physiol. Biochem. 2023, 196, 661–667. [Google Scholar] [CrossRef]
- Wang, X.L.; Feng, X.; Ma, Y. Activated carbon from chili straw: K2CO3 activation mechanism, adsorption of dyes, and thermal regeneration. Biomass Convers. Biorefinery 2024, 14, 19563–19580. [Google Scholar] [CrossRef]
- Zhang, X.; Yu, Z.; Lu, X.; Ma, X. Catalytic co-pyrolysis of microwave pretreated chili straw and polypropylene to produce hydrocarbons-rich bio-oil. Bioresour. Technol. 2021, 319, 124191. [Google Scholar] [CrossRef]
- Wei, P.; Chen, G.; Zhi, F.; Zhang, A.; Deng, H.; Wen, X.; Wang, F.; Yu, C. Investigation of pyrolysis and combustion characteristics of chili straw waste with different O2/N2 ratios and heating rates. Thermochim. Acta 2024, 742, 179875. [Google Scholar] [CrossRef]
- Mcdonald, I.; Baral, R.; Min, D. Effects of alfalfa and alfalfa-grass mixtures with nitrogen fertilization on dry matter yield and forage nutritive value. J. Anim. Sci. Technol. 2021, 63, 305–318. [Google Scholar] [CrossRef]
- Rezaeian, M.; Tohidi Moghadam, M.; Kiaei, M.M.; Mahmuod Zadeh, H. The effect of heavy metals on the nutritional value of alfalfa: Comparison of nutrients and heavy metals of alfalfa (Medicago sativa) in industrial and non-industrial areas. Toxicol. Res. 2020, 36, 183–193. [Google Scholar] [CrossRef]
- Ren, H.; Xiao, W.; Zhao, Y. Examining the effect of spontaneous combustion on vegetation restoration at coal waste dumps after reclamation: Taking Medicago sativa L. (alfalfa) as an indicator. Sci. Total Environ. 2023, 901, 165668. [Google Scholar] [CrossRef]
- Hayes, R.C.; Conyers, M.K.; Li, G.D.; Poile, G.J.; Price, A.; McVittie, B.J.; Gardner, M.J.; Sandral, G.A.; McCormick, J.I. Spatial and temporal variation in soil Mn concentrations and the impact of manganese toxicity on lucerne and subterranean clover seedlings. Crop Pasture Sci. 2012, 63, 875–885. [Google Scholar] [CrossRef]
- Gherardi, M.J.; Rengel, Z. Genotypes of lucerne (Medicago sativa L.) show differential tolerance to manganese deficiency and toxicity when grown in bauxite residue sand. Plant Soil 2003, 249, 287–296. [Google Scholar] [CrossRef]
- Deng, R.; Lan, Z.; Xiao, K.; Shang, X.; Fang, S. Biochar additions improve soil nutrient availability and biomass production of Cyclocarya paliurus via regulating soil bacterial community and pH value on the acidic soil. J. Soil Sci. Plant Nutr. 2025. [Google Scholar] [CrossRef]
- Bolan, N.; Sarmah, A.K.; Bordoloi, S.; Bolan, S.; Padhye, L.P.; Van Zwieten, L.; Sooriyakumar, P.; Khan, B.A.; Ahmad, M.; Solaiman, Z.M. Soil acidification and the liming potential of biochar. Environ. Pollut. 2023, 317, 120632. [Google Scholar] [CrossRef]
- Nath, H.; Sarkar, B.; Mitra, S.; Bhaladhare, S. Biochar from biomass: A review on biochar preparation its modification and impact on soil including soil microbiology. Geomicrobiol. J. 2022, 39, 373–388. [Google Scholar] [CrossRef]
- Wang, S.B.; Gao, P.L.; Zhang, Q.W. Application of biochar and organic fertilizer to saline-alkali soil in the yellow river delta: Effects on soil water, salinity, nutrients, and maize yield. Soil Use Manag. 2022, 38, 1679–1692. [Google Scholar] [CrossRef]
- Gao, S.; Hoffman-Krull, K.; Bidwell, A.L.; DeLuca, T.H. Locally produced wood biochar increases nutrient retention and availability in agricultural soils of the San Juan Islands, USA. Agric. Ecosyst. Environ. 2016, 233, 43–54. [Google Scholar] [CrossRef]
- Zhang, L.X.; Chang, L.; Liu, H.; de Jesús Puy Alquiza, M.; Li, Y. Biochar application to soils can regulate soil phosphorus availability: A review. Biochar 2025, 7, 13. [Google Scholar] [CrossRef]
- Zhang, H.-Q.; Qin, Y.; Li, Z.-Z.; Song, Z.-Z. Mixed application of biochar, maize straw, and nitrogen can improve organic carbon fractions and available nutrients of a sandy soil. Arid Land Res. Manag. 2023, 37, 115–133. [Google Scholar] [CrossRef]
- Penn, C.J.; Camberato, J.J. A critical review on soil chemical processes that control how soil pH affects phosphorus availability to plants. Agriculture 2019, 9, 120. [Google Scholar] [CrossRef]
- Igalavithana, A.D.; Lee, S.E.; Lee, Y.H.; Tsang, D.C.; Rinklebe, J.; Kwon, E.E.; Ok, Y.S. Heavy metal immobilization and microbial community abundance by vegetable waste and pine cone biochar of agricultural soils. Chemosphere 2017, 174, 593–603. [Google Scholar] [CrossRef]
- Hu, L.; Yang, Y.; Liu, X.H.; Li, S.; Li, K.; Deng, H. Effects of bagasse biochar application on soil organic carbon fixation in manganese-contaminated sugarcane fields. Chem. Biol. Technol. Agric. 2023, 10, 46. [Google Scholar] [CrossRef]
- Grygo-Szymanko, E.; Tobiasz, A.; Walas, S. Speciation analysis and fractionation of manganese: A review. TrAC Trends Anal. Chem. 2016, 80, 112–124. [Google Scholar] [CrossRef]
- Ameen, F.; Al-Homaidan, A.A. Treatment of heavy metal–polluted C sludge using biochar amendments and vermistabilization. Environ. Monit. Assess. 2022, 194, 861. [Google Scholar] [CrossRef]
- Gascó, G.; Cely, P.; Paz-Ferreiro, J.; Plaza, C.; Méndez, A. Relation between biochar properties and effects on seed germination and plant development. Biol. Agric. Hortic. 2016, 32, 237–247. [Google Scholar] [CrossRef]
- Simiele, M.; Argentino, O.; Baronti, S.; Scippa, G.S.; Chiatante, D.; Terzaghi, M.; Montagnoli, A. Biochar enhances plant growth, fruit yield, and antioxidant content of cherry tomato (Solanum lycopersicum L.) in a soilless substrate. Agriculture 2022, 12, 1135. [Google Scholar] [CrossRef]
- Li, L.; Cui, S.; Qian, D.; Lyu, S.; Liu, W.; Botella, J.R.; Li, H.; Burritt, D.J.; Tran, L.S.P.; Li, W. PDC1 is activated by ABF4 and inhibits seed germination by promoting ROS accumulation in Arabidopsis. Environ. Exp. Bot. 2023, 206, 105188. [Google Scholar] [CrossRef]
- Wang, R.; Fu, W.; Wang, J.; Zhu, L.; Wang, L.; Wang, J.; Ahmad, Z. Application of rice grain husk derived biochar in ameliorating toxicity impacts of Cu and Zn on growth, physiology and enzymatic functioning of wheat seedlings. Bull Environ. Contam. Toxicol. 2019, 103, 636–641. [Google Scholar] [CrossRef] [PubMed]
- Mokaram-Kashtiban, S.; Hosseini, S.M.; Kouchaksaraei, M.T.; Younesi, H. Biochar improves the morphological, physiological and biochemical properties of white willow seedlings in heavy metal-contaminated soil. Arch Biol Sci. 2019, 71, 281–291. [Google Scholar] [CrossRef]
- Qian, L.; Mei, C.; Li, T.; Luo, W.; Liu, W.; Chen, M.; Yang, X.; Li, X.; Cheng, B.; Ma, H. A versatile biochar fertilizer used for adsorption of heavy metals and enhancement of plant growth in metal contaminated soil. Environ. Technol. Innovation. 2024, 36, 103743. [Google Scholar] [CrossRef]
- Zahra, A.; Kayani, S.; Shahzad, A.; Sert, T.D.; Ozcelik, H.; Qin, M.; Naeem, M.; M Billah, M. Wood biochar induced metal tolerance in Maize (Zea mays L.) plants under heavy metal stress. Environ. Res. 2024, 262, 119940. [Google Scholar] [CrossRef] [PubMed]
- Kalaji, H.M.; Jajoo, A.; Oukarroum, A.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Łukasik, I.; Goltsev, V.; Ladle, R.J. Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol. Plant. 2016, 38, 102. [Google Scholar] [CrossRef]
- Soares, C.; Carvalho, M.E.A.; Azevedo, R.A.; Fidalgo, F. Plants facing oxidative challenges—A little help from the antioxidant networks. Environ. Exp. Bot. 2019, 161, 4–25. [Google Scholar] [CrossRef]
- Su, J.; Liu, Y.; Han, F.; Gao, F.; Gan, F.; Huang, K.; Li, Z. ROS, an important plant growth regulator in root growth and development: Functional genes and mechanism. Biology 2024, 13, 1033. [Google Scholar] [CrossRef]
- Ai, Y.; Wang, Y.; Song, L.; Hong, W.; Zhang, Z.; Li, X.; Zhou, S.; Zhou, J. Effects of biochar on the physiology and heavy metal enrichment of Vetiveria zizanioides in contaminated soil in mining areas. J. Hazard. Mater. 2023, 448, 130965. [Google Scholar] [CrossRef]
- Adams, J.M.; Turner, L.B.; Toop, T.A.; Kirby, M.E.; Rolin, C.; Judd, E.; Inkster, R.; McEvoy, L.; Mirza, W.M.; Theodorou, M.K.; et al. Evaluation of pyrolysis chars derived from marine macroalgae silage as soil amendments. Glob. Change Biol. 2020, 12, 706–727. [Google Scholar] [CrossRef]
- Arshad, M.; Khan, A.H.A.; Hussain, I.; Anees, M.; Iqbal, M.; Soja, G.; Linde, C.; Yousaf, S. The Reduction of chromium (VI) phytotoxicity and phytoavailability to wheat (Triticum aestivum L.) using biochar and bacteria. Appl. Soil Ecol. 2017, 114, 90–98. [Google Scholar] [CrossRef]
- Chen, G.; Wang, J.; He, G.; Li, S.; Li, X.; Tao, X.; Liang, S.; Deng, F.; Zeng, F.; Chen, Z.-H. Physiological and transcriptomic evidence of antioxidative system and ion transport in chromium detoxification in germinating seedlings of soybean. Environ. Pollut. 2023, 320, 121047. [Google Scholar] [CrossRef]
- Ge, S.; Zhu, Z.; Peng, L.; Chen, Q.; Jiang, Y. Soil nutrient status and leaf nutrient diagnosis in the main apple producing regions in China. Hortic. Plant J. 2018, 4, 89–93. [Google Scholar] [CrossRef]
- Asensio, V.; Abreu, C.H.; da Silva, F.C.; Chitolina, J.C. Evaluation of chemical extractants to assess metals phytoavailability in Brazilian municipal solid waste composts. Environ. Pollut. 2018, 243, 235–1241. [Google Scholar] [CrossRef] [PubMed]



| Treatment | GR (%) | GE (%) | GI |
|---|---|---|---|
| CK | 60.94 ± 2.58 a | 43.44 ± 2.77 c | 15.74 ± 0.71 b |
| B1 | 57.81 ± 4.25 a | 47.81 ± 2.77 b | 16.76 ± 1.19 b |
| B2 | 61.88 ± 3.75 a | 55.63 ± 2.98 a | 19.86 ± 1.35 a |
| B3 | 60.00 ± 4.45 a | 51.88 ± 2.17 ab | 17.13 ± 0.93 b |
| Treatment | Chlorophyll a (mg·g−1) | Chlorophyll b (mg·g−1) | Total Chlorophyll (mg·g−1) |
|---|---|---|---|
| CK | 1.46 ± 0.10 c | 0.57 ± 0.02 b | 2.03 ± 0.08 c |
| B1 | 1.82 ± 0.10 b | 0.82 ± 0.04 a | 2.64 ± 0.08 b |
| B2 | 2.02 ± 0.16 a | 0.87 ± 0.06 a | 2.89 ± 0.19 a |
| B3 | 1.94 ± 0.09 ab | 0.83 ± 0.03 a | 2.77 ± 0.10 ab |
| Treatment | pH | OM (g·kg−1) | AN (mg·kg−1) | AP (mg·kg−1) | AK (mg·kg−1) |
|---|---|---|---|---|---|
| CK | 7.32 ± 0.03 d | 9.12 ± 0.51 d | 19.18 ± 0.89 d | 6.19 ± 0.28 b | 357.18 ± 22.07 a |
| B1 | 7.58 ± 0.07 c | 10.51 ± 0.40 c | 21.71 ± 1.79 c | 6.99 ± 0.31 a | 349.33 ± 11.28 a |
| B2 | 7.80 ± 0.03 b | 12.14 ± 0.86 b | 26.27 ± 1.06 a | 6.94 ± 0.19 a | 364.79 ± 12.25 a |
| B3 | 8.02 ± 0.12 a | 13.34 ± 0.57 a | 23.79 ± 0.99 b | 6.38 ± 0.23 b | 339.88 ± 23.98 a |
| Sample | pH | Organic Matter (g·kg−1) | Total Nitrogen (g·kg−1) | Alkaline Nitrogen (mg·kg−1) | Available Phosphorus (mg·kg−1) | Available Potassium (mg·kg−1) | Total Mn (mg·kg−1) | Available Mn (mg·kg−1) |
|---|---|---|---|---|---|---|---|---|
| EMR | 7.46 | 5.17 | 0.45 | 18.96 | 6.83 | 385.49 | 41,052.67 | 644.84 |
| Chili Straw Biochar | 9.45 | 267.30 | 7.32 | - | - | - | 113.29 | 24.08 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, Y.; Xiang, Y.; Ren, J. Effects of Chili Straw Biochar on Alfalfa (Medicago sativa L.) Seed Germination and Seedling Growth on Electrolytic Manganese Residue. Plants 2025, 14, 2635. https://doi.org/10.3390/plants14172635
Luo Y, Xiang Y, Ren J. Effects of Chili Straw Biochar on Alfalfa (Medicago sativa L.) Seed Germination and Seedling Growth on Electrolytic Manganese Residue. Plants. 2025; 14(17):2635. https://doi.org/10.3390/plants14172635
Chicago/Turabian StyleLuo, Yang, Yangzhou Xiang, and Jun Ren. 2025. "Effects of Chili Straw Biochar on Alfalfa (Medicago sativa L.) Seed Germination and Seedling Growth on Electrolytic Manganese Residue" Plants 14, no. 17: 2635. https://doi.org/10.3390/plants14172635
APA StyleLuo, Y., Xiang, Y., & Ren, J. (2025). Effects of Chili Straw Biochar on Alfalfa (Medicago sativa L.) Seed Germination and Seedling Growth on Electrolytic Manganese Residue. Plants, 14(17), 2635. https://doi.org/10.3390/plants14172635







