Effect of Anthocyanin-Enriched Brine on Nutritional, Functional and Sensory Properties of Pickled Baby Corn
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemical Composition of Baby Corn Samples
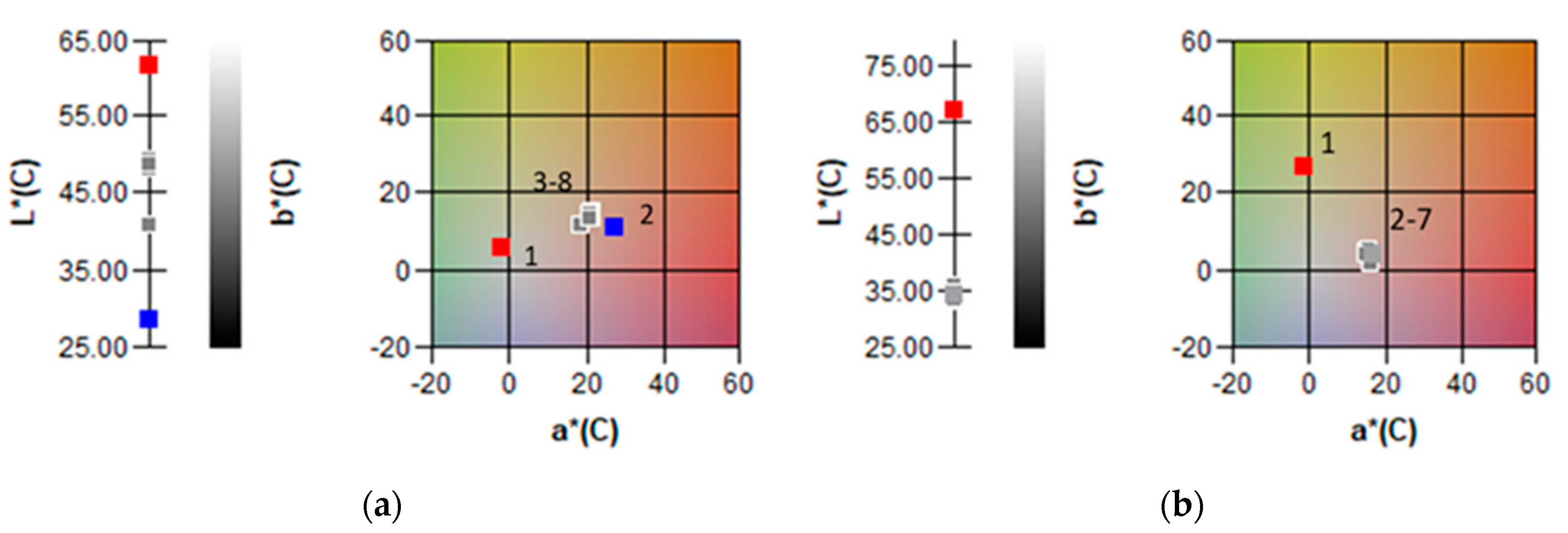
2.2. Phenolic Compound Content, the Antioxidant Capacity and the Colour of Baby Corn Samples
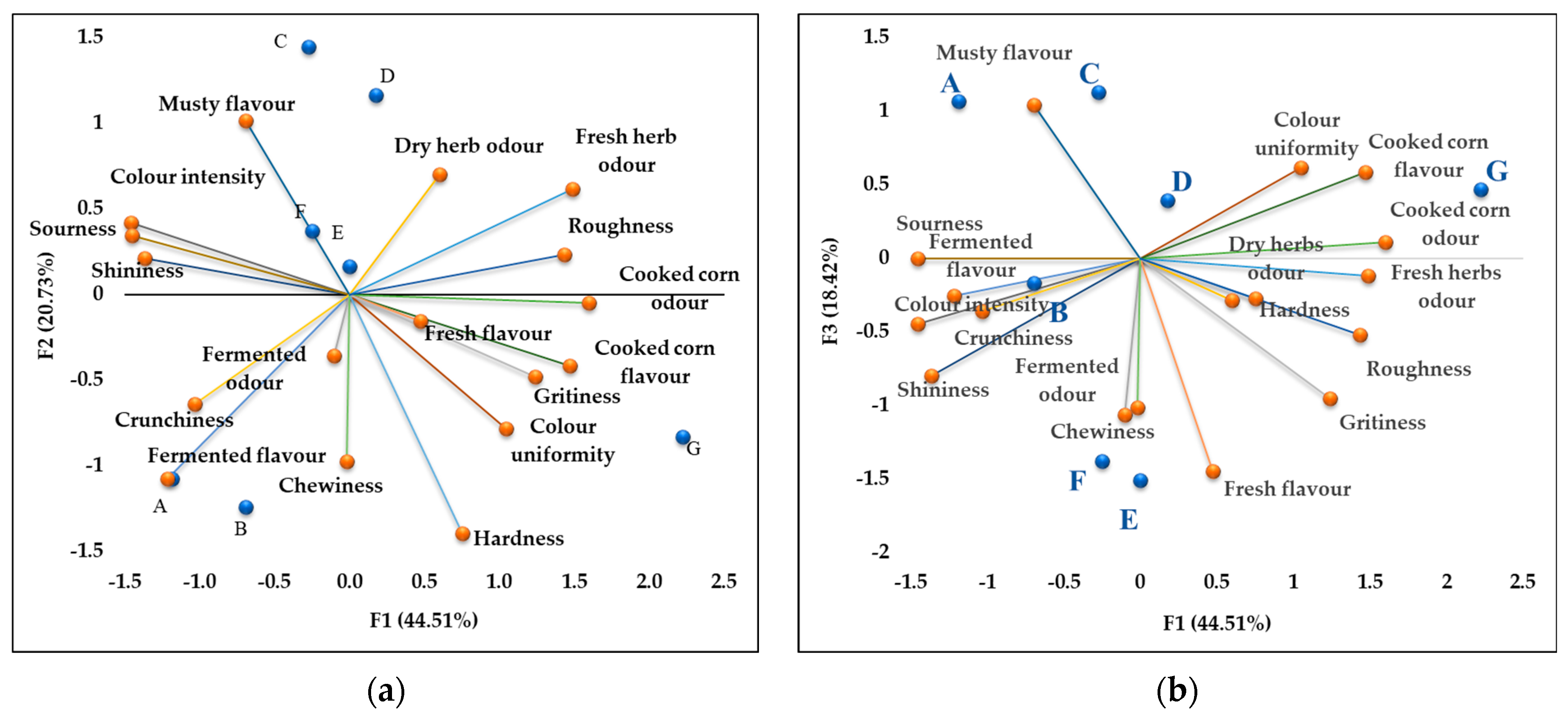
2.3. Sensory Properties of Baby Corn Samples
3. Materials and Methods
3.1. Plant Material
3.2. Technological Steps for Baby Corn Preparation
3.2.1. Anthocyanins Extraction from BSSC
3.2.2. Marinating Brine Preparation
3.2.3. Preparation of Baby Corn Products
3.3. Chemical Procedure
3.3.1. Analysis of Total Protein Content in Baby Corn
3.3.2. Analysis of Sugars Content in Baby Corn
3.3.3. Analysis of Dietary Fibres Content in Baby Corn
3.3.4. Extraction of Free Soluble Phenolic Compounds from Baby Corn
3.3.5. Analysis of Total Free Soluble Phenolic Content in Baby Corn (TPC)
3.3.6. Analysis of Total Flavonoid Content in Baby Corn (TFC)
3.3.7. Analysis of Total Anthocyanin Content in Baby Corn (TAC)
3.3.8. Analysis of Individual Anthocyanins in Baby Corn by HPLC
3.3.9. Analysis of Individual Phenolic Acids and Flavonoids in Baby Corn by HPLC
3.3.10. Analysis of the Total Antioxidant Capacity of Baby Corn (AC)
3.4. Measurement of Colour of Baby Corn
3.5. Baby Corn Product Sensory Evaluation
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Akram, A.; Parisa, Z. Chemical composition profile of canned and frozen sweet corn (Zea mays L.) in Iran. Orient. J. Chem. 2015, 31, 1065–1070. [Google Scholar] [CrossRef]
- Singh, M.K.; Singh, R.N.; Singh, S.P.; Yadav, M.K.; Singh, V.K. Integrated nutrient management for higher yield, quality and profitability of baby corn (Zea mays). Indian J. Agron. 2010, 55, 100–104. [Google Scholar]
- Das, A.K.; Singh, V. Antioxidative free and bound phenolic constituents in botanical fractions of Indian specialty corn (Zea mays L.). Food Chem. 2016, 201, 298–306. [Google Scholar] [CrossRef]
- Hong, H.T.; Netzel, M.E.; O’Hare, T.J. Anthocyanin composition and changes during kernel development in purple-pericarp supersweet sweetcorn. Food Chem. 2020, 315, 126284. [Google Scholar] [CrossRef]
- Nurul, S.R.; Asmah, R. Evaluation of antioxidant properties in fresh and pickled papaya. Int. Food Res. J. 2012, 19, 1117–1124. [Google Scholar]
- Featherstone, S. Canning of pickled products. In A Complete Course in Canning and Related Processes, 14th ed.; Woodhead Publishing: Sawston, UK, 2016; Volume 3, pp. 351–368. [Google Scholar] [CrossRef]
- Takebayashi, J.; Oki, T.; Watanabe, J.; Yamasaki, K.; Chen, J.; Sato-Furukawa, M. Hydrophilic antioxidant capacities of vegetables and fruits commonly consumed in Japan and estimated average daily intake of hydrophilic antioxidants from these foods. J. Food Compos. Anal. 2013, 29, 25–31. [Google Scholar] [CrossRef]
- Isabelle, M.; Lee, B.L.; Lim, M.T.; Koh, W.P.; Huang, D.; Ong, C.N. Antioxidant activity and profiles of common vegetables in Singapore. Food Chem. 2010, 120, 993–1003. [Google Scholar] [CrossRef]
- Feng, X.; Pan, L.; Wang, Q.; Liao, Z.; Wang, X.; Zhang, X.; Guo, W.; Hu, E.; Li, J.; Xu, J.; et al. Nutritional and physicochemical characteristics of purple sweet corn juice before and after boiling. PLoS ONE 2020, 15, e0233094. [Google Scholar] [CrossRef]
- Kaur, N.; Kaur, K.; Aggarwall, P. Parameter optimization and nutritional evaluation of naturally fermented baby corn pickle. Agric. Res. J. 2018, 55, 548–553. [Google Scholar] [CrossRef]
- Nikolić, V.; Žilić, S.; Simić, M.; Perić, V.; Srdić, J.; Vasić, M. Optimization of anthocyanins extraction process from black soybean seed coat for the preparation of corn-based functional food. J. Process. Energy Agric. 2022, 26, 19–22. [Google Scholar] [CrossRef]
- Tsuda, T.; Horio, F.; Uchida, K.; Aoki, H.; Osawa, T. Dietary cyanidin 3-O-beta-D-glucoside-rich purple corn color prevents obesity and ameliorates hyperglycemia in mice. J. Nutr. 2003, 133, 2125–2130. [Google Scholar] [CrossRef] [PubMed]
- Afzal, M.; Redha, A.; Al Hasan, R. Anthocyanins potentially contribute to defense against Alzheimer’s disease. Molecules 2019, 24, 4255. [Google Scholar] [CrossRef] [PubMed]
- Choung, M.G.; Baek, I.Y.; Kang, S.T.; Han, W.Y.; Shin, D.C.; Moon, H.P.; Kang, K.H. Isolation and determination of anthocyanins in seed coats of black soybean (Glycine max (L.) merr.). J. Agric. Food Chem. 2001, 49, 5848–5851. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.F.; Zhang, F.X.; Zhang, M.W.; Wei, Z.C.; Yang, C.Y.; Zhang, Y.; Chi, J.W. Phenolic composition and antioxidant activity in seed coats of 60 Chinese black soybean (Glycine max L. Merr.) varieties. J. Agric. Food Chem. 2011, 59, 5935–5944. [Google Scholar] [CrossRef]
- Žilić, S.; Dodig, D.; Vančetović, J.; Grčić, N.; Perić, V.; Titan, P.; Maksimović, V. Composition of anthocyanins in colored grains and the relationship of their non-acylated and acylated derivatives. Pol. J. Food Nutr. Sci. 2019, 69, 137–146. [Google Scholar] [CrossRef]
- Žilić, S.; Simić, M.; Belović, M.; Škrobot, D.; Srdić, J.; Perić, V. Chemical, rheological and sensory characteristics of sweet spreads made from by-products of soya bean and corn. Int. J. Food Sci. Technol. 2020, 55, 1559–1571. [Google Scholar] [CrossRef]
- Yang, Z.; Zhou, Y.; Xing, J.-J.; Guo, X.-N.; Zhu, K.-X. Effect of superheated steam treatment and extrusion on lipid stability of black soybean noodles during storage. Food Control 2022, 132, 108388. [Google Scholar] [CrossRef]
- Ma, M.; Xie, Y.; Wang, C. Effect of anthocyanin-rich extract from black soybean coat on wheat dough rheology and noodle texture. J. Food Process. Preserv. 2020, 45, e15007. [Google Scholar] [CrossRef]
- Hou, D.; Duan, W.; Xue, Y.; Yousaf, L.; Hu, J.; Shen, Q. Effects of superfine grinding and extrusion on dough mixing properties and noodle quality of black soybean flour. J. Food Meas. Charact. 2020, 14, 125–134. [Google Scholar] [CrossRef]
- Slavin, M.; Lu, Y.; Kaplan, N.; Yu, L. Effects of baking on cyanidin-3-glucoside content and antioxidant properties of black and yellow soybean crackers. Food Chem. 2013, 141, 1166–1174. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Su, H.-J.; Shiau, S.-Y. Rheological and textural characteristics of black soybean touhua (soft soybean curd) prepared with glucono-δ-lactone. Food Chem. 2009, 115, 585–591. [Google Scholar] [CrossRef]
- Yuan, G.; Jia, Y.; Pan, Y.; Li, W.; Wang, C.; Xu, L.; Wang, C.; Chen, H. Preparation and characterization of shrimp shell waste protein-based films modified with oolong tea, corn silk and black soybean seed coat extracts. Polym. Test. 2020, 81, 106235. [Google Scholar] [CrossRef]
- Wang, X.; Yong, H.; Gao, L.; Li, L.; Jin, M.; Liu, J. Preparation and characterization of antioxidant and PH-sensitive films based on chitosan and black soybean seed coat extract. Food Hydrocoll. 2019, 89, 56–66. [Google Scholar] [CrossRef]
- Hooda, S.; Kawatra, A. Nutritional evaluation of baby corn (Zea mays). Nutr. Food Sci. 2013, 43, 68–73. [Google Scholar] [CrossRef]
- Rekha, S.; Bindu, S. Sensory and nutritional evaluation of value added products prepared from baby corn. Asian J. Dairy Foods Home Sci. 2012, 31, 301–305. [Google Scholar]
- Watson, S.A. Corn: Chemistry and Technology. In Description, Development, Structure, and Composition of the Corn Kernel, 2nd ed.; White, P.J., Johnson, L.A., Eds.; American Association of Cereal Chemists: St. Paul, MN, USA, 2003; pp. 69–101. [Google Scholar]
- Nikolić, V.; Žilić, S.; Radosavljević, M.; Simić, M.; Filipović, M.; Čamdžija, Z.; Sečanski, M. Grain properties of new inbred lines in comparison with maize hybrids. J. Process. Energy Agric. 2020, 24, 3–4. [Google Scholar] [CrossRef]
- Mader, T.L.; Guyer, P.Q.; Stock, R. G74-100 Feeding High Moisture Corn; University of Nebraska: Lincoln, RI, USA, 1974; Extension 248. [Google Scholar]
- Nogoy, K.M.; Zhang, Y.; Lee, Y.H.; Li, X.Z.; Seong, H.A.; Choi, S.H. Nutrient composition and in vitro fermentability of corn grain and stover harvested at different periods in Goesan, a mountainous area. J. Anim. Sci. Technol. 2019, 61, 18–27. [Google Scholar] [CrossRef]
- Nguyen, P.M.; Tran, T.Y.N.; Mai, H.D.; An, T.M.L.; Tran, T.H.L. Technical Factors Affecting to Production of Pickled Baby Corn Canning. J. Pharm. Sci. Res. 2019, 11, 1431–1436. [Google Scholar]
- Azanza, F.; Klein, B.P.; Juvik, J.A. Sensory characterization of sweet corn lines differing in physical and chemical composition. J. Food Sci. 1996, 61, 253–257. [Google Scholar] [CrossRef]
- Özlem, A.; Gulcan, K.; Engin, K.; Zekiye Budak, B.; Kenan, S.; Yasemin, E.; Imren, K. Kernel Quality of Some Sweet Corn Varieties in Relation to Processing. Not. Bot. Horti. Agrobot. 2014, 42, 414–419. [Google Scholar] [CrossRef]
- Szymanek, M.; Tanaś, W.; Kassar, F.H. Kernel carbohydrates concentration in sugary-1, sugary enhanced and shrunken sweet corn kernels. Agric. Agric. Sci. Proc. 2015, 7, 260–264. [Google Scholar] [CrossRef]
- Goldfein, K.R.; Slavin, J.L. Why Sugar Is Added to Food: Food Science 101. Compr. Rev. Food Sci. Food Saf. 2015, 14, 644–656. [Google Scholar] [CrossRef]
- Ledenčan, T.; Horvat, D.; Špoljarić Marković, S.; Svečnjak, Z.; Jambrović, A.; Šimić, D. Effect of Harvest Date on Kernel Quality and Antioxidant Activity in su1 Sweet Corn Genotypes. Agronomy 2022, 12, 1224. [Google Scholar] [CrossRef]
- Zhu, S.; Mount, J.; Colins, J. Sugar and soluble solids changes in refrigerated sweet corn (Zea mays L). J. Food Sci. 1992, 57, 749–751. [Google Scholar] [CrossRef]
- Schwieterman, M.L.; Colquhoun, T.A.; Jaworski, E.A.; Bartoshuk, L.M.; Gilbert, J.L.; Tieman, D.M.; Odabasi, A.Z.; Moskowitz, H.R.; Klee, H.J.; Sims, C.A.; et al. Strawberry flavor: Diverse chemical compositions, a seasonal in-fluence, and effects on sensory perception. PLoS ONE 2014, 9, e88446. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, C.; Alaswad, A.; Benyounis, K.Y.; Olabi, A.G. Pretreatment techniques used in biogas production from grass. Renew. Sustain. Energy Rev. 2017, 68, 1193–1204. [Google Scholar] [CrossRef]
- Tezcan, E.; Atıcı, O.G. A new method for recovery of cellulose from lignocellulosic bio-waste: Pile processing. Waste Manag. 2017, 70, 181–188. [Google Scholar] [CrossRef]
- Ma, S.; Wang, H.; Li, J.; Fu, Y.; Zhu, W. Methane production performances of different compositions in lignocellulosic biomass through anaerobic digestion. Energy 2019, 189, 116190. [Google Scholar] [CrossRef]
- Radosavljević, M.; Milašinović-Šeremešić, M.; Terzić, D.; Jovanović, Ž.; Srdić, J.; Nikolić, V. Grain chemical composition of dents, popping maize and sweet maize genotypes. J. Process. Energy Agric. 2020, 24, 77–80. [Google Scholar] [CrossRef]
- Velmurugan, P.; Hur, H.; Balachandar, V.; Kamala-Kannan, S.; Lee, K.J.; Lee, S.M.; Chae, J.C.; Shea, P.J.; Oh, B.T. Monascus pigment production by solid-state fermentation with corn cob substrate. J. Biosci. Bioeng. 2011, 112, 590–594. [Google Scholar] [CrossRef]
- Tolera, A.; Berg, T.; Sundstol, F. The effect of variety on maize grain and crop residue yield and nutritive value of the stover. Anim. Feed Sci. Technol. 1999, 79, 165–177. [Google Scholar] [CrossRef]
- Yang, Z.; Cao, L.; Li, Y.; Zhang, M.; Zeng, F.; Yao, S. Effect of pH on hemicellulose extraction and physicochemical characteristics of solids during hydrothermal pretreatment of eucalyptus. BioResources 2020, 15, 6627–6635. [Google Scholar] [CrossRef]
- Messias, R.S.; Galli, V.; Silva, S.D.A.; Schirmer, M.A.; Rombaldi, C.V. Micronutrient and functional compounds biofortification of maize grains. Crit. Rev. Food Sci. Nutr. 2015, 55, 123–139. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Huang, L.; Deng, Y.; Chi, J.; Zhang, Y.; Wei, Z.; Zhang, M. Phenolic content and antioxidant activity of eight representative sweet corn varieties grown in South China. Int. J. Food Prop. 2017, 20, 3043–3055. [Google Scholar] [CrossRef]
- Song, W.; Derito, C.M.; Liu, M.K.; He, X.J.; Dong, M.; Liu, R.H. Cellular Antioxidant Activity of Common Vegetables. J. Agric. Food Chem. 2010, 58, 6621–6629. [Google Scholar] [CrossRef]
- Hu, X.; Liu, H.; Yu, Y.; Li, G.; Qi, X.; Li, Y.; Li, T.; Guo, X.; Liu, R.H. Accumulation of phenolics, antioxidant and antiproliferative activity of sweet corn (Zea mays L.) during kernel maturation. Int. J. Food Sci. Technol. 2021, 56, 2462–2470. [Google Scholar] [CrossRef]
- Dewanto, V.; Wu, X.; Liu, R.H. Processed sweet corn has higher antioxidant activity. J. Agric. Food Chem. 2002, 50, 4959–4964. [Google Scholar] [CrossRef]
- Montilla, E.C.; Hillebrand, S.; Antezana, A.; Winterhalter, P. Soluble and bound phenolic compounds in different Bolivian purple corn (Zea mays L.) cultivars. J. Agric. Food Chem. 2011, 59, 7068–7074. [Google Scholar] [CrossRef] [PubMed]
- Žilić, S.; Serpen, A.; Akillioglu, G.; Gökmen, V.; Vančetović, J. Phenolic compounds, carotenoids, anthocyanins, and antioxidant capacity of colored maize (Zea mays L.) kernels. J. Agric. Food Chem. 2012, 60, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Adom, K.K.; Liu, R.H. Antioxidant activity of grains. J. Agric. Food Chem. 2002, 50, 6182–6187. [Google Scholar] [CrossRef]
- Pedreschi, R.; Cisneros-Zevallos, L. Phenolic profiles of Andean purple corn (Zea mays L.). Food Chem. 2007, 100, 956–963. [Google Scholar] [CrossRef]
- De la Parra, C.; Serna Saldivar, S.O.; Liu, R.H. Effect of processing on the phytochemical profiles and antioxidant activity of corn for production of masa, tortillas, and tortilla chips. J. Agric. Food Chem. 2007, 55, 4177–4183. [Google Scholar] [CrossRef] [PubMed]
- Saulnier, L.; Thibault, J.F. Ferulic acid and diferulic acids as components of sugar-beet pectins and maize bran heteroxylans. J. Sci. Food Agric. 1999, 79, 396–402. [Google Scholar] [CrossRef]
- Mazza, G.; Miniati, E. Anthocyanins in Fruits, Vegetables and Grains, 1st ed.; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar] [CrossRef]
- Martinez-Zapata, M.J.; Vernooij, R.W.; Uriona Tuma, S.M.; Stein, A.T.; Moreno, R.M.; Vargas, E.; Capellà, D.; Bonfill Cosp, X. Phlebotonics for venous insufficiency. Cochrane Database Syst. Rev. 2016, 4, CD003229. [Google Scholar] [CrossRef]
- Morling, J.R.; Yeoh, S.E.; Kolbach, D.N. Rutosides for prevention of post-thrombotic syndrome. Cochrane Database Syst. Rev. 2018, 11, CD005626. [Google Scholar] [CrossRef]
- Kähkönen, M.P.; Heinonen, M. Antioxidant activity of anthocyanins and their aglycones. J. Agric. Food Chem. 2003, 51, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-J.; Hyun, J.-N.; Kim, J.-A.; Park, J.-C.; Kim, M.-Y.; Kim, J.-G.; Lee, S.-J.; Chung, S.-C.; Chung, I.-M. Relationship between phenolic compounds, anthocyanins content and antioxidant activity in colored barley germplasm. J. Agric. Food Chem. 2007, 55, 4802–4809. [Google Scholar] [CrossRef]
- Cabrita, L.; Fossen, T.; Andersen, Ø.M. Colour and stability of the six common anthocyanidin 3-glucosides in aqueous solutions. Food Chem. 2000, 68, 101–107. [Google Scholar] [CrossRef]
- Wahyuningsih, S.; Wulandari, L.; Wartono, M.W.; Munawaroh, H.; Ramelan, A.H. The Effect of pH and Color Stability of Anthocyanin on Food Colorant. IOP Conf. Ser. Mater. Sci. Eng. 2017, 193, 012047. [Google Scholar] [CrossRef]
- Heredia, F.J.; Francia-Aricha, E.M.; Rivas-Gonzalo, J.C.; Vicario, I.M.; Santos-Buelga, C. Chromatic characterization of anthocyanins from red grapes—I. PH effect. Food Chem. 1998, 63, 491–498. [Google Scholar] [CrossRef]
- Ananga, A.; Georgiev, V.; Ochieng, J.; Phills, B.; Tsolova, V. Production of Anthocyanins in Grape Cell Cultures: A Potential Source of Raw Material for Pharmaceutical, Food, and Cosmetic Industries. In The Mediterranean Genetic Code—Grapevine and Olive; Poljuha, D., Sladonja, B., Eds.; IntechOpen: London, UK, 2013; pp. 247–287. [Google Scholar] [CrossRef]
- Stintzing, C.F.; Stintzing, S.A.; Carle, R.; Frei, B.; Wrolstad, E.R. Color and antioxidant properties of cyanidin-based anthocyanin pigments. J. Agric. Food Chem. 2002, 50, 6172–6181. [Google Scholar] [CrossRef]
- Bąkowska-Barczak, A. Acylated anthocyanins as stable, natural food colorants. Pol. J. Food Nutr. Sci. 2005, 14, 107–116. [Google Scholar]
- Singh-Ackbarali, D.; Maharajm, R. Sensory evaluation as a tool in determining acceptability of innovative products developed by undergraduate students in food science and technology at the University of Trinidad and Tobago. J. Curric. Teach. 2014, 3, 10–27. [Google Scholar] [CrossRef]
- Simić, M.; Žilić, S.; Šimuruna, O.; Filipčev, B.; Škrobot, D.; Vančetović, J. Effects of anthocyanin-rich popping corn flour on the phenolic profile and the antioxidant capacity of mix-bread and its physical and sensory properties. Pol. J. Food Nutr. Sci. 2018, 68, 299–308. [Google Scholar] [CrossRef]
- Lesschaeve, I.; Noble, A.C. Polyphenols: Factors influencing their sensory properties and their effects on food and beverage preferences. Am. J. Clin. Nutr. 2005, 81, 330S–335S. [Google Scholar] [CrossRef]
- Van der Veer, O. The Human Intake of Salt and the Development of Low-Sodium and No-Salt-Added Processed Foods and Salt Substitutes. A Literature Review; Department of Human Nutrition, University of Agriculture: Wageningen, The Netherlands, 1985. [Google Scholar]
- Hutton, T. Sodium: Technological functions of salt in the manufacturing of food and drink products. Br. Food J. 2002, 104, 126–152. [Google Scholar] [CrossRef]
- Brady, M. Sodium Survey of the usage and functionality of salt as an ingredient in UK manufactured food products. Br. Food J. 2002, 104, 84–125. [Google Scholar] [CrossRef]
- Abdel-Aal, E.S.M.; Hucl, P. A rapid method for quantifying total anthocyanins in blue aleurone and purple pericarp wheats. Cereal Chem. 1999, 76, 350–354. [Google Scholar] [CrossRef]
- Official Gazette of SFRY. The Rule Book of Methods of Physical and Chemical Analysis for Quality Control of Grain, Milling and Bakery Products, Pasta and Frozen Dough; No. 74/88; Institution for Standardization: Belgrade, Serbia, 1988. [Google Scholar]
- Egan, H.; Kirk, R.; Sawyer, R. The Luff Schoorl method. Sugars and Preserves. In Pearson’s Chemical Analysis of Foods, 8th ed.; Churchill Livingstone: New York, NY, USA, 1981; pp. 152–153. [Google Scholar]
- Mertens, D.R. Critical conditions in determining detergent fibers. In Proceedings of the Forage Analysis Workshop, Denver, CO, USA, 16–17 September 1992; National Forage Testing Association: Omaha, NE, USA, 1992; pp. C1–C8. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Markham, K.R. Flavones, flavonols and their glycosides. In Methods in Plant Biochemistry; Harbone, J.B., Ed.; Academic Press: London, UK, 1993; pp. 197–235. [Google Scholar] [CrossRef]
- Serpen, A.; Gökmen, V.; Pellegrini, N.; Fogliano, V. Direct measurement of the total antioxidant capacity of cereal products. J. Cereal Sci. 2008, 48, 816–820. [Google Scholar] [CrossRef]
| Compounds | Control | Popping Corn | Semi Flint Corn | Sweet Corn | |||
|---|---|---|---|---|---|---|---|
| Salt | Salt + Sugar | Salt | Salt + Sugar | Salt | Salt + Sugar | ||
| Protein (%) | 17.86 ± 0.07 a | 17.21 ± 0.07 b | 16.61 ± 0.01 c | 14.61 ± 0.13 e | 12.89 ± 0.01 f | 17.08 ± 0.12 bc | 15.29 ± 0.29 d |
| Sugars | |||||||
| Total sugars (%) | 28.64 ± 0.72 a | 6.45 ± 0.18 e | 14.73 ± 0.06 b | 6.90 ± 0.76 de | 16.47 ± 0.11 a | 5.87 ± 0.22 e | 19.24 ± 0.37 a |
| Sucrose (%) | 17.79 ± 0.05 a | 0.62 ± 0.02 d | 3.45 ± 0.10 c | 1.80 ± 0.07 cd | 3.31 ± 0.13 c | 1.11 ± 0.08 d | 5.82 ± 0.17 b |
| Total reducing sugars (%) | 9.91 ± 0.07 c | 5.80 ± 0.25 d | 11.09 ± 0.11 b | 5.00 ± 0.15 e | 12.99 ± 0.42 a | 4.81 ± 0.16 e | 13.13 ± 0.16 a |
| Dietary fibres | |||||||
| NDF (%) | 46.21 ± 0.08 a | 34.80 ± 0.57 b | 32.09 ± 0.81 c | 31.95 ± 0.73 c | 26.69 ± 0.83 d | 31.00 ± 0.33 c | 26.46 ± 0.59 d |
| ADF (%) | 13.05 ± 0.21 a | 6.44 ± 0.26 b | 6.39 ± 0.28 b | 6.66 ± 0.05 b | 6.50 ± 0.04 b | 6.41 ± 0.03 b | 5.63 ± 0.33 b |
| ADL (%) | 6.46 ± 0.44 a | 2.46 ± 0.12 bc | 1.89 ± 0.06 c | 1.76 ± 0.08 c | 1.68 ± 0.47 c | 2.23 ± 0.05 bc | 3.07 ± 0.21 b |
| Hemicellulose (%) | 33.16 ± 0.28 a | 28.36 ± 0.31 b | 25.70 ± 0.53 c | 25.29 ± 0.68 c | 20.20 ± 0.80 c | 24.59 ± 0.30 bc | 20.33 ± 0.92 b |
| Cellulose (%) | 6.59 ± 0.64 a | 3.98 ± 0.38 bc | 4.50 ± 0.23 b | 4.90 ± 0.13 b | 4.50 ± 0.50 b | 4.19 ± 0.08 bc | 3.06 ± 0.11 c |
| pH | 5.09 a | 3.07 b | 3.10 b | 3.03 c | 2.98 d | 3.00 cd | 3.01 cd |
| Control | Popping Corn | Semi Flint Corn | Sweet Corn | ||||
|---|---|---|---|---|---|---|---|
| Salt | Salt + Sugar | Salt | Salt + Sugar | Salt | Salt + Sugar | ||
| Total-free soluble phenolic compounds | |||||||
| Total phenolics (mg CE/kg) | 3540.54 ± 34 e | 6534.21 ± 98 a | 6156.57 ± 100 ab | 5828.93 ± 24 bc | 5631.75 ± 42 c | 5466.68 ± 40 cd | 4813.93 ± 202 d |
| Total flavonoids (mg/kg) | 2.74 ± 0.01 f | 4.06 ± 0.04 a | 3.91 ± 0.03 b | 3.73 ± 0.01 c | 3.36 ± 0.01 d | 2.98 ± 0.04 e | 2.92 ± 0.03 e |
| Total anthocyanins (mg CGE/kg) | n.d. | 881.23 ± 8.4 a | 748.55 ± 8.8 b | 765.24 ± 7.7 b | 757.76 ± 12.9 b | 877.63 ± 36 a | 752.18 ± 15 b |
| Free soluble phenolic acids (μg/g) | |||||||
| 3,4-Dihydroxybenzoic acid | n.d. | 177.39 ± 2.64 a | 137.23 ± 6.5 b | 118.33 ± 1.09 c | 103.21 ± 0.83 d | 78.68 ± 1.91 e | 77.39 ± 4.4 e |
| Chlorogenic acid | 1195.75 ± 13.4 c | 1558.57 ± 50 a | 1478.46 ± 33 b | 675.41 ± 5.47 d | 542.91 ± 17.5 e | 15.20 ± 1.0 f | 16.02 ± 0.34 f |
| Caffeic acid | n.d. | 60.75 ± 2.08 a | 53.05 ± 0.68 b | n.d. | n.d. | n.d. | n.d. |
| p-Coumaric acid | 32.88 ± 1.65 a | 6.49 ± 0.27 b | 5.17 ± 0.29 bc | 5.19 ± 0.11 bc | 3.67 ± 0.0 c | 6.79 ± 0.65 b | 6.03 ± 0.21 bc |
| Ferulic acid | 232.53 ± 4.98 a | 151.97 ± 1.30 b | 84.09 ± 0.18 c | 45.03 ± 1.67 d | 34.90 ± 0.98 e | 52.84 ± 0.21 d | 47.55 ± 0.76 d |
| Vanillic acid | 66.94 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Syringic acid | 7.79 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| Flavonoids (μg/g) | |||||||
| Epicatechin | n.d. | 167.39 ± 1.31 a | 149.56 ± 0.45 b | 163.26 ± 5.95 a | 164.72 ± 2.11 a | 164.97 ± 0.98 a | 148.23 ± 0.14 b |
| Rutin | n.d. | 180.21 ± 2.06 a | 164.89 ± 3.32 b | 123.83 ± 1.00 cd | 118.15 ± 0.06 de | 125.93 ± 0.18 c | 113.62 ± 1.23 e |
| Anthocyanins (μg/g) | |||||||
| De-3-Glu | n.d. | 19.60 ± 0.17 b | 12.24 ± 0.17 c | n.d. | n.d. | 34.93 ± 1.39 a | 18.39 ± 1.73 b |
| Cy-3-Glu | n.d. | 233.11 ± 1.53 b | 188.39 ± 2.22 cd | 184.62 ± 1.20 d | 193.16 ± 0.04 c | 247.47 ± 2.08 a | 186.24 ± 0.17 d |
| Pg-3-Glu | n.d. | n.d. | n.d. | n.d. | n.d. | 17.61 ± 0.08 a | 12.50 ± 0.54 b |
| Antioxidant capacity (mmol Trolox Eq/kg) | 40.48 ± 0.47 e | 54.54 ± 0.31 a | 53.26 ± 0.65 ab | 48.79 ± 0.50 d | 51.83 ± 0.30 b | 51.19 ± 0.0 bc | 49.38 ± 0.95 cd |
| Control | Popping Corn | Semi Flint Corn | Sweet Corn | ||||
|---|---|---|---|---|---|---|---|
| Salt | Salt + Sugar | Salt | Salt + Sugar | Salt | Salt + Sugar | ||
| L* | 67.13 a | 33.55 de | 35.69 b | 33.77 d | 33.22 e | 34.81 c | 33.95 d |
| a* | −1.31 f | 16.26 b | 16 c | 15.20 e | 15.54 d | 15.17 e | 16.93 a |
| b* | 26.68 a | 1.50 e | 3.66 d | 4.63 c | 5.40 b | 3.68 d | 4.00 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simić, M.; Nikolić, V.; Škrobot, D.; Srdić, J.; Perić, V.; Despotović, S.; Žilić, S. Effect of Anthocyanin-Enriched Brine on Nutritional, Functional and Sensory Properties of Pickled Baby Corn. Plants 2023, 12, 1812. https://doi.org/10.3390/plants12091812
Simić M, Nikolić V, Škrobot D, Srdić J, Perić V, Despotović S, Žilić S. Effect of Anthocyanin-Enriched Brine on Nutritional, Functional and Sensory Properties of Pickled Baby Corn. Plants. 2023; 12(9):1812. https://doi.org/10.3390/plants12091812
Chicago/Turabian StyleSimić, Marijana, Valentina Nikolić, Dubravka Škrobot, Jelena Srdić, Vesna Perić, Saša Despotović, and Slađana Žilić. 2023. "Effect of Anthocyanin-Enriched Brine on Nutritional, Functional and Sensory Properties of Pickled Baby Corn" Plants 12, no. 9: 1812. https://doi.org/10.3390/plants12091812
APA StyleSimić, M., Nikolić, V., Škrobot, D., Srdić, J., Perić, V., Despotović, S., & Žilić, S. (2023). Effect of Anthocyanin-Enriched Brine on Nutritional, Functional and Sensory Properties of Pickled Baby Corn. Plants, 12(9), 1812. https://doi.org/10.3390/plants12091812








