Salvia rosmarinus Spenn. (Lamiaceae) Hydroalcoholic Extract: Phytochemical Analysis, Antioxidant Activity and In Vitro Evaluation of Fatty Acid Accumulation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Extraction Procedure
2.3. HPLC Analysis
2.4. DPPH Assay: Radical Scavenging Activity
2.5. Evaluation of Radicals Scavenging via Electron Paramagnetic Spectroscopy (EPR)
2.6. Total Phenolic Content (TPC) Determination
2.7. Total Flavonoid Content (TFC) Determination
2.8. Cell Culture
2.9. Free Fatty Acid (FFA) Exposure and S. rosmarinus Spenn. Treatment
2.10. MTT Assay
2.11. Determination of Intracellular Total Fatty Acid Content
2.12. Fluorescence Image Acquisition
2.13. Statistical Analysis
3. Results
3.1. Phytochemical Characterization and Quantification via HPLC
3.2. In Vitro Antioxidant Activity of S. rosmarinus Spenn. Extracts
3.3. DPPH and Hydroxyl Radicals Scavenging Activity of S. rosmarinus Spenn. Extracts through EPR Spectroscopy
3.4. Characterization of Total Phenolic and Flavonoid Contents of S. rosmarinus Extracts
3.5. Antioxidant Activity Is Significantly Correlated with Total Phenolic and Flavonoid Contents
3.6. Treatment with Terpenoids-Rich or Polyphenols-Rich Salvia rosmarinus Extracts Does Not Affect Cell Viability
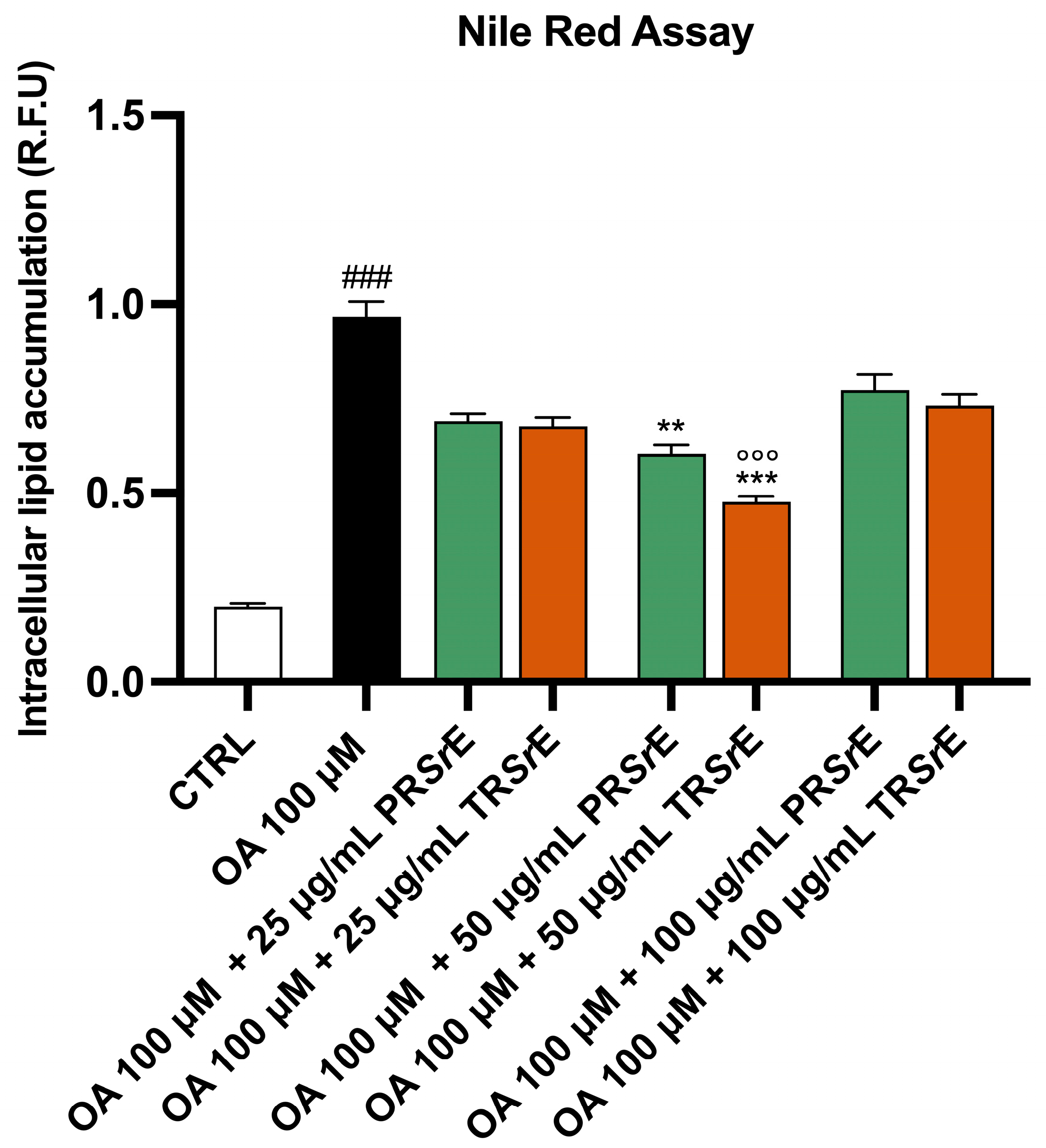
3.7. Salvia rosmarinus Spenn. Extracts Reduce Intracellular Lipid Accumulation
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- de Macedo, L.M.; dos Santos, É.M.; Militão, L.; Tundisi, L.L.; Ataide, J.A.; Souto, E.B.; Mazzola, P.G. Rosemary (Rosmarinus officinalis L.; syn Salvia rosmarinus Spenn.) and Its Topical Applications: A Review. Plants 2020, 9, 651. [Google Scholar] [CrossRef]
- Leporini, M.; Bonesi, M.; Loizzo, M.R.; Passalacqua, N.G.; Tundis, R. The Essential Oil of Salvia rosmarinus Spenn. from Italy as a Source of Health-Promoting Compounds: Chemical Profile and Antioxidant and Cholinesterase Inhibitory Activity. Plants 2020, 9, 798. [Google Scholar] [CrossRef]
- de Oliveira, J.R.; Camargo, S.E.A.; de Oliveira, L.D. Rosmarinus officinalis L. (rosemary) as therapeutic and prophylactic agent. J. Biomed. Sci. 2019, 26, 5. [Google Scholar] [CrossRef] [PubMed]
- Drew, B.T.; González-Gallegos, J.G.; Xiang, C.; Kriebel, R.; Drummond, C.P.; Walked, J.B.; Sytsma, K.J. Salvia united: The greatest good for the greatest number. Taxon 2017, 66, 133–145. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Carvalho-Costa, D.; Cavaleiro, C.; Costa, H.S.; Albuquerque, T.G.; Castilho, M.D.; Ramos, F.; Melo, N.R.; Sanches-Silva, A. A novel insight on an ancient aromatic plant: The rosemary (Rosmarinus officinalis L.). Trends Food Sci. Technol. 2015, 45, 355–368. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Munekata, P.E.S.; Pateiro, M.; Domínguez, R.; Alaghbari, M.A.; Tomasevic, I. Preservation of meat products with natural antioxidants from rosemary. IOP Conf. Ser. Earth Environ. Sci. 2021, 854, 012053. [Google Scholar] [CrossRef]
- Josipović, R.; Knežević, Z.M.; Frece, J.; Markov, K.; Kazazić, S.; Mrvčić, J. Improved Properties and Microbiological Safety of Novel Cottage Cheese Containing Spices. Food Technol. Biotechnol. 2015, 53, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Loi, S. De-orientalising Ritual Blood Calabria’s Vattienti, a Case Study. Ann. Ca’ Foscari Ser. Orient. 2018, 54, 461–490. [Google Scholar] [CrossRef]
- Sarkic, A.; Stappen, I. Essential Oils and Their Single Compounds in Cosmetics—A Critical Review. Cosmetics 2018, 5, 11. [Google Scholar] [CrossRef]
- Puerto, J. La Fuerza de Fierabrás. Medicina, Ciencia y Terapéutica en Tiempos del Quijote; Editorial Just in Time SL: Madrid, Spain, 2005. [Google Scholar]
- Colica, C.; Di Renzo, L.; Aiello, V.; De Lorenzo, A.; Abenavoli, L. Rosmarinic Acid as Potential Anti-Inflammatory Agent. Rev. Recent Clin. Trials 2018, 13, 240–242. [Google Scholar] [CrossRef]
- Ferreira, H.P. Assessment Report on Rosmarinus officinalis L., Aetheroleum and Rosmarinus officinalis L., Folium; Committee on Herbal Medicinal Products (HMPC): London, UK, 2010. [Google Scholar]
- Apostolides, N.A.; El Beyrouthy, M.; Dhifi, W.; Najm, S.; Cazier, F.; Najem, W.; Labaki, M.; AbouKaïs, A. Chemical Composition of Aerial Parts of Rosmarinus officinalis L. Essential Oil Growing Wild in Lebanon. J. Essent. Oil Bear. Plants 2013, 16, 274–282. [Google Scholar] [CrossRef]
- Saeidi, S.; Forgani, F.; Javadian, F.; Javadian, E. Effects of Rosmarinus officinalis Plant Extract on Trichomonas vaginalis Parasites and Candida albicans under Laboratory Conditions: An Experimental Study. Gene Cell Tissue 2019, 6, e92867. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Bonesi, M.; Passalacqua, N.G.; Saab, A.; Menichini, F.; Tundis, R. Antiproliferative activities on renal, prostate and melanoma cancer cell lines of Sarcopoterium spinosum aerial parts and its major constituent tormentic acid. Anti-Cancer Agents Med. Chem. 2013, 13, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Bai, N.; He, K.; Roller, M.; Lai, C.S.; Shao, X.; Pan, M.H.; Ho, C.T. Flavonoids and phenolic compounds from Rosmarinus officinalis. J. Agric. Food Chem. 2010, 58, 5363–5367. [Google Scholar] [CrossRef] [PubMed]
- Borrás-Linares, I.; Stojanović, Z.; Quirantes-Piné, R.; Arráez-Román, D.; Švarc-Gajić, J.; Fernández-Gutiérrez, A.; Segura-Carretero, A. Rosmarinus officinalis Leaves as a Natural Source of Bioactive Compounds. Int. J. Mol. Sci. 2014, 15, 20585–20606. [Google Scholar] [CrossRef]
- Rašković, A.; Milanović, I.; Pavlović, N.; Ćebović, T.; Vukmirović, S.; Mikov, M. Antioxidant activity of rosemary (Rosmarinus officinalis L.) essential oil and its hepatoprotective potential. BMC Complement. Altern. Med. 2014, 14, 225. [Google Scholar] [CrossRef] [PubMed]
- Ngo, S.N.T.; Williams, D.B.; Head, R.J. Rosemary and Cancer Prevention: Preclinical Perspectives. Crit. Rev. Food Sci. Nutr. 2011, 51, 946–954. [Google Scholar] [CrossRef]
- Morel, S.; Hugon, G.; Vitou, M.; Védère, M.; Fons, F.; Rapior, S.; Saint, N.; Carnac, G. A Bioassay-Guided Fractionation of Rosemary Leaf Extract Identifies Carnosol as a Major Hypertrophy Inducer in Human Skeletal Muscle Cells. Nutrients 2021, 13, 4190. [Google Scholar] [CrossRef]
- Soliman, G.A. Effect of Rosmarinus officinalis on Lipid Profile of Streptozotocin-induced Diabetic Rats. Egypt. J. Hosp. Med. 2013, 53, 809–815. [Google Scholar] [CrossRef]
- Wang, S.J.; Chen, Q.; Liu, M.Y.; Yu, H.Y.; Xu, J.Q.; Wu, J.Q.; Zhang, Y.; Wang, T. Regulation effects of rosemary (Rosmarinus officinalis Linn.) on hepatic lipid metabolism in OA induced NAFLD rats. Food Funct. 2019, 10, 7356–7365. [Google Scholar] [CrossRef]
- Musolino, V.; Gliozzi, M.; Scarano, F.; Bosco, F.; Scicchitano, M.; Nucera, S.; Carresi, C.; Ruga, S.; Zito, M.C.; Maiuolo, J.; et al. Bergamot Polyphenols Improve Dyslipidemia and Pathophysiological Features in a Mouse Model of Non-Alcoholic Fatty Liver Disease. Sci. Rep. 2020, 10, 2565. [Google Scholar] [CrossRef] [PubMed]
- Nucera, S.; Ruga, S.; Cardamone, A.; Coppoletta, A.R.; Guarnieri, L.; Zito, M.C.; Bosco, F.; Macrì, R.; Scarano, F.; Scicchitano, M.; et al. MAFLD progression contributes to altered thalamus metabolism and brain structure. Sci. Rep. 2022, 12, 1207. [Google Scholar] [CrossRef] [PubMed]
- Del Baño, M.J.; Castillo, J.; Benavente-García, O.; Lorente, J.; Martín-Gil, R.; Acevedo, C.; Alcaraz, M. Radioprotective-antimutagenic effects of rosemary phenolics against chromosomal damage induced in human lymphocytes by gamma-rays. J. Agric. Food Chem. 2006, 54, 2064–2068. [Google Scholar] [CrossRef]
- del Baño, M.J.; Lorente, J.; Castillo, J.; Benavente-García, O.; del Río, J.A.; Ortuño, A.; Quirin, K.W.; Gerard, D. Phenolic diterpenes, flavones, and rosmarinic acid distribution during the development of leaves, flowers, stems, and roots of Rosmarinus officinalis. Antioxidant activity. J. Agric. Food Chem. 2003, 51, 4247–4253. [Google Scholar] [CrossRef]
- Yesil-Celiktas, O.; Nartop, P.; Gurel, A.; Bedir, E.; Vardar-Sukan, F. Determination of phenolic content and antioxidant activity of extracts obtained from Rosmarinus officinalis’ calli. J. Plant Physiol. 2007, 164, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.W.; Lin, L.G.; Ye, W.C. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Nn, A. A Review on the Extraction Methods Use in Medicinal Plants, Principle, Strength and Limitation. Med. Aromat. Plants 2015, 4, 1–6. [Google Scholar]
- Ali, A.; Chua, B.L.; Chow, Y.H. An insight into the extraction and fractionation technologies of the essential oils and bioactive compounds in Rosmarinus officinalis L.: Past, present and future. TrAC Trends Anal. Chem. 2019, 118, 338–351. [Google Scholar] [CrossRef]
- Lešnik, S.; Furlan, V.; Bren, U. Rosemary (Rosmarinus officinalis L.): Extraction techniques, analytical methods and health-promoting biological effects. Phytochem. Rev. 2021, 20, 1273–1328. [Google Scholar] [CrossRef]
- Chemat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef] [PubMed]
- Musolino, V.; Macrì, R.; Cardamone, A.; Serra, M.; Coppoletta, A.R.; Tucci, L.; Maiuolo, J.; Lupia, C.; Scarano, F.; Carresi, C.; et al. Nocellara Del Belice (Olea europaea L. Cultivar): Leaf Extract Concentrated in Phenolic Compounds and Its Anti-Inflammatory and Radical Scavenging Activity. Plants 2022, 12, 27. [Google Scholar] [CrossRef] [PubMed]
- Irakli, M.; Chatzopoulou, P.; Ekateriniadou, L. Optimization of ultrasound-assisted extraction of phenolic compounds: Oleuropein, phenolic acids, phenolic alcohols and flavonoids from olive leaves and evaluation of its antioxidant activities. Ind. Crops Prod. 2018, 124, 382–388. [Google Scholar] [CrossRef]
- Erkan, N.; Ayranci, G.; Ayranci, E. Antioxidant activities of rosemary (Rosmarinus officinalis L.) extract, blackseed (Nigella sativa L.) essential oil, carnosic acid, rosmarinic acid and sesamol. Food Chem. 2008, 110, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Bernatoniene, J.; Cizauskaite, U.; Ivanauskas, L.; Jakstas, V.; Kalveniene, Z.; Kopustinskiene, D.M. Novel approaches to optimize extraction processes of ursolic, oleanolic and rosmarinic acids from Rosmarinus officinalis leaves. Ind. Crops Prod. 2016, 84, 72–79. [Google Scholar] [CrossRef]
- Giordano, A.; Morales-Tapia, P.; Moncada-Basualto, M.; Pozo-Martínez, J.; Olea-Azar, C.; Nesic, A.; Cabrera-Barjas, G. Polyphenolic Composition and Antioxidant Activity (ORAC, EPR and Cellular) of Different Extracts of Argylia radiata Vitroplants and Natural Roots. Molecules 2022, 27, 610. [Google Scholar] [CrossRef]
- Mollace, R.; Macrì, R.; Tavernese, A.; Gliozzi, M.; Musolino, V.; Carresi, C.; Maiuolo, J.; Fini, M.; Volterrani, M.; Mollace, V. Comparative Effect of Bergamot Polyphenolic Fraction and Red Yeast Rice Extract in Rats Fed a Hyperlipidemic Diet: Role of Antioxidant Properties and PCSK9 Expression. Nutrients 2022, 14, 477. [Google Scholar] [CrossRef]
- Lamponi, S.; Baratto, M.C.; Miraldi, E.; Baini, G.; Biagi, M. Chemical Profile, Antioxidant, Anti-Proliferative, Anticoagulant and Mutagenic Effects of a Hydroalcoholic Extract of Tuscan Rosmarinus officinalis. Plants 2021, 10, 97. [Google Scholar] [CrossRef]
- Araniti, F.; Marrelli, M.; Lupini, A.; Mercati, F.; Statti, G.A.; Abenavoli, M.R. Phytotoxic activity of Cachrys pungens Jan, a mediterranean species: Separation, identification and quantification of potential allelochemicals. Acta Physiol. Plant. 2014, 36, 1071–1083. [Google Scholar] [CrossRef]
- Kumar, P.; Nagarajan, A.; Uchil, P.D. Analysis of Cell Viability by the MTT Assay. Cold Spring Harb. Protoc. 2018, 6. [Google Scholar] [CrossRef]
- De Chiara, F.; Ferret-Miñana, A.; Fernández-Costa, J.M.; Senni, A.; Jalan, R.; Ramón-Azcón, J. Fatty Hepatocytes Induce Skeletal Muscle Atrophy In Vitro: A New 3D Platform to Study the Protective Effect of Albumin in Non-Alcoholic Fatty Liver. Biomedicines 2022, 10, 958. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, M.; Turnbull, T.; Sebastian, S.; Kempson, I. The MTT Assay: Utility, Limitations, Pitfalls, and Interpretation in Bulk and Single-Cell Analysis. Int. J. Mol. Sci. 2021, 22, 12827. [Google Scholar] [CrossRef] [PubMed]
- Kaczorová, D.; Karalija, E.; Dahija, S.; Bešta-Gajević, R.; Parić, A.; Ćavar Zeljković, S. Influence of Extraction Solvent on the Phenolic Profile and Bioactivity of Two Achillea Species. Molecules 2021, 26, 1601. [Google Scholar] [CrossRef] [PubMed]
- de AR Oliveira, G.; de Oliveira, A.E.; da Conceição, E.C.; Leles, M.I. Multiresponse optimization of an extraction procedure of carnosol and rosmarinic and carnosic acids from rosemary. Food Chem. 2016, 211, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Jacotet-Navarro, M.; Laguerre, M.; Fabiano-Tixier, A.S.; Tenon, M.; Feuillère, N.; Bily, A.; Chemat, F. What is the best ethanol-water ratio for the extraction of antioxidants from rosemary? Impact of the solvent on yield, composition, and activity of the extracts. Electrophoresis 2018, 39, 1946–1956. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, A.; Ponnuchamy, M.; Kumar, P.S.; Kapoor, A.; Vo, D.N.; Prabhakar, S. Techniques and modeling of polyphenol extraction from food: A review. Environ. Chem. Lett. 2021, 19, 3409–3443. [Google Scholar] [CrossRef] [PubMed]
- Paniwnyk, L.; Cai, H.; Albu, S.; Mason, T.J.; Cole, R. The enhancement and scale up of the extraction of anti-oxidants from Rosmarinus officinalis using ultrasound. Ultrason. Sonochem. 2009, 16, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Plaskova, A.; Mlcek, J. New insights of the application of water or ethanol-water plant extract rich in active compounds in food. Front. Nutr. 2023, 10, 1118761. [Google Scholar] [CrossRef]
- Arceusz, A.; Wesolowski, M.; Konieczynski, P. Methods for extraction and determination of phenolic acids in medicinal plants: A review. Nat. Prod. Commun. 2013, 8, 1821–1829. [Google Scholar] [CrossRef]
- Nguyen-Kim, M.; Truong, Q.; Nguyen, M.; Cao-Thi, B.; Tong, T.; Dao, T.; Tran, T.; Van Tan, L.; Le, X. Optimized extraction of polyphenols from leaves of Rosemary (Rosmarinus officinalis L.) grown in Lam Dong province, Vietnam, and evaluation of their antioxidant capacity. Open Chem. 2021, 19, 1043–1051. [Google Scholar] [CrossRef]
- Mwithiga, G.; Maina, S.; Gitari, J.; Muturi, P. Rosemary (Rosmarinus officinalis L.) growth rate, oil yield and oil quality under differing soil amendments. Heliyon 2022, 8, e09277. [Google Scholar] [CrossRef] [PubMed]
- Al-Fraihat, A.H.; Al-Dalain, S.Y.; Zatimeh, A.A.; Haddad, M.A. Enhancing Rosemary (Rosmarinus officinalis, L.) Growth and Volatile Oil Constituents Grown under Soil Salinity Stress by Some Amino Acids. Horticulturae 2023, 9, 252. [Google Scholar] [CrossRef]
- Rathore, S.; Mukhia, S.; Kapoor, S.; Bhatt, V.; Kumar, R.; Kumar, R. Seasonal variability in essential oil composition and biological activity of Rosmarinus officinalis L. accessions in the western Himalaya. Sci. Rep. 2022, 12, 3305. [Google Scholar] [CrossRef] [PubMed]
- Oualdi, I.; Brahmi, F.; Mokhtari, O.; Abdellaoui, S.; Tahani, A.; Oussaid, A. Rosmarinus officinalis from Morocco, Italy and France: Insight into chemical compositions and biological properties. Mater. Today Proc. 2021, 45, 7706–7710. [Google Scholar] [CrossRef]
- Pontillo, A.R.N.; Papakosta-Tsigkri, L.; Lymperopoulou, T.; Mamma, D.; Kekos, D.; Detsi, A. Conventional and Enzyme-Assisted Extraction of Rosemary Leaves (Rosmarinus officinalis L.): Toward a Greener Approach to High Added-Value Extracts. Appl. Sci. 2021, 11, 3724. [Google Scholar] [CrossRef]
- Zeroual, A.; Sakar, E.H.; Mahjoubi, F.; Chaouch, M.; Chaqroune, A.; Taleb, M. Effects of extraction technique and solvent on phytochemicals, antioxidant, and antimicrobial activities of cultivated and wild rosemary (Rosmarinus officinalis L.) from Taounate region. Biointerface Res. Appl. Chem. 2020, 12, 8441–8452. [Google Scholar]
- Butu, M.; Rodino, S.; Golea, D.A.; Butu, A. Screening of the antioxidant capacity and total phenolic content of three native plants. Dig. J. Nanomater. Biostruct. (DJNB) 2014, 9, 337–345. [Google Scholar]
- Mena, P.; Cirlini, M.; Tassotti, M.; Herrlinger, K.A.; Dall’Asta, C.; Del Rio, D. Phytochemical Profiling of Flavonoids, Phenolic Acids, Terpenoids, and Volatile Fraction of a Rosemary (Rosmarinus officinalis L.) Extract. Molecules 2016, 21, 1576. [Google Scholar] [CrossRef]
- Marrelli, M.; Amodeo, V.; Puntillo, D.; Statti, G.; Conforti, F. In vitro antioxidant and anti-denaturation effects of Buglossoides purpurocaerulea (L.) I. M. Johnst. fruit extract. Nat. Prod. Res. 2023, 37, 1012–1015. [Google Scholar] [CrossRef]
- Song, H.M.; Li, X.; Liu, Y.Y.; Lu, W.P.; Cui, Z.H.; Zhou, L.; Yao, D.; Zhang, H.M. Carnosic acid protects mice from high-fat diet-induced NAFLD by regulating MARCKS. Int. J. Mol. Med. 2018, 42, 193–207. [Google Scholar] [CrossRef] [PubMed]
- Habtemariam, S. Anti-Inflammatory Therapeutic Mechanisms of Natural Products: Insight from Rosemary Diterpenes, Carnosic Acid and Carnosol. Biomedicines 2023, 11, 545. [Google Scholar] [CrossRef]
- Nassir, F. NAFLD: Mechanisms, Treatments, and Biomarkers. Biomolecules 2022, 12, 824. [Google Scholar] [CrossRef] [PubMed]
- Yao, P.; Liu, Y. Terpenoids: Natural Compounds for Non-Alcoholic Fatty Liver Disease (NAFLD) Therapy. Molecules 2022, 28, 272. [Google Scholar] [CrossRef] [PubMed]
- Rau, O.; Wurglics, M.; Dingermann, T.; Abdel-Tawab, M.; Schubert-Zsilavecz, M. Screening of herbal extracts for activation of the human peroxisome proliferator-activated receptor. Die Pharm. 2006, 61, 952–956. [Google Scholar]
- Tu, Z.; Moss-Pierce, T.; Ford, P.; Jiang, T.A. Rosemary (Rosmarinus officinalis L.) extract regulates glucose and lipid metabolism by activating AMPK and PPAR pathways in HepG2 cells. J. Agric. Food Chem. 2013, 61, 2803–2810. [Google Scholar] [CrossRef]
- Timm, K.N.; Tyler, D.J. The Role of AMPK Activation for Cardioprotection in Doxorubicin-Induced Cardiotoxicity. Cardiovasc. Drugs Ther. 2020, 34, 255–269. [Google Scholar] [CrossRef]
- Finck, B.N.; Kelly, D.P. Peroxisome proliferator-activated receptor gamma coactivator-1 (PGC-1) regulatory cascade in cardiac physiology and disease. Circulation 2007, 115, 2540–2548. [Google Scholar] [CrossRef]
- Cordero-Herrera, I.; Kozyra, M.; Zhuge, Z.; McCann Haworth, S.; Moretti, C.; Peleli, M.; Caldeira-Dias, M.; Jahandideh, A.; Huirong, H.; Cruz, J.d.C.; et al. AMP-activated protein kinase activation and NADPH oxidase inhibition by inorganic nitrate and nitrite prevent liver steatosis. Proc. Natl. Acad. Sci. USA 2019, 116, 217–226. [Google Scholar] [CrossRef]
- Scicchitano, M.; Carresi, C.; Nucera, S.; Ruga, S.; Maiuolo, J.; Macrì, R.; Scarano, F.; Bosco, F.; Mollace, R.; Cardamone, A.; et al. Icariin Protects H9c2 Rat Cardiomyoblasts from Doxorubicin-Induced Cardiotoxicity: Role of Caveolin-1 Upregulation and Enhanced Autophagic Response. Nutrients 2021, 13, 4070. [Google Scholar] [CrossRef]
- Carresi, C.; Musolino, V.; Gliozzi, M.; Maiuolo, J.; Mollace, R.; Nucera, S.; Maretta, A.; Sergi, D.; Muscoli, S.; Gratteri, S.; et al. Anti-oxidant effect of bergamot polyphenolic fraction counteracts doxorubicin-induced cardiomyopathy: Role of autophagy and c-kitposCD45negCD31negcardiac stem cell activation. J. Mol. Cell. Cardiol. 2018, 119, 10–18. [Google Scholar] [CrossRef] [PubMed]




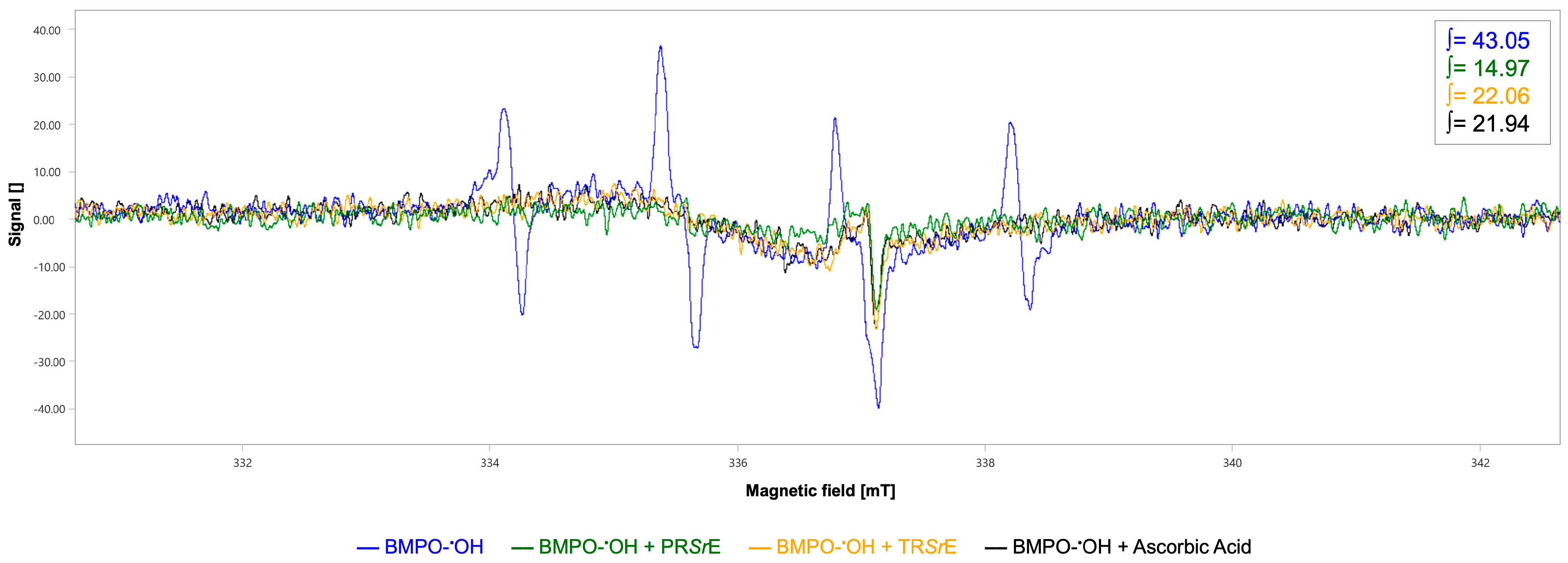




| Antioxidant Activity | Pearson’s r | p | |
|---|---|---|---|
| TPC | IC50, DPPH-EPR, BMPO-•OH | −0.975 | <0.001 |
| TFC | IC50, DPPH-EPR, BMPO-•OH | −0.993 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Musolino, V.; Macrì, R.; Cardamone, A.; Tucci, L.; Serra, M.; Lupia, C.; Maurotti, S.; Mare, R.; Nucera, S.; Guarnieri, L.; et al. Salvia rosmarinus Spenn. (Lamiaceae) Hydroalcoholic Extract: Phytochemical Analysis, Antioxidant Activity and In Vitro Evaluation of Fatty Acid Accumulation. Plants 2023, 12, 3306. https://doi.org/10.3390/plants12183306
Musolino V, Macrì R, Cardamone A, Tucci L, Serra M, Lupia C, Maurotti S, Mare R, Nucera S, Guarnieri L, et al. Salvia rosmarinus Spenn. (Lamiaceae) Hydroalcoholic Extract: Phytochemical Analysis, Antioxidant Activity and In Vitro Evaluation of Fatty Acid Accumulation. Plants. 2023; 12(18):3306. https://doi.org/10.3390/plants12183306
Chicago/Turabian StyleMusolino, Vincenzo, Roberta Macrì, Antonio Cardamone, Luigi Tucci, Maria Serra, Carmine Lupia, Samantha Maurotti, Rosario Mare, Saverio Nucera, Lorenza Guarnieri, and et al. 2023. "Salvia rosmarinus Spenn. (Lamiaceae) Hydroalcoholic Extract: Phytochemical Analysis, Antioxidant Activity and In Vitro Evaluation of Fatty Acid Accumulation" Plants 12, no. 18: 3306. https://doi.org/10.3390/plants12183306
APA StyleMusolino, V., Macrì, R., Cardamone, A., Tucci, L., Serra, M., Lupia, C., Maurotti, S., Mare, R., Nucera, S., Guarnieri, L., Marrelli, M., Coppoletta, A. R., Carresi, C., Gliozzi, M., & Mollace, V. (2023). Salvia rosmarinus Spenn. (Lamiaceae) Hydroalcoholic Extract: Phytochemical Analysis, Antioxidant Activity and In Vitro Evaluation of Fatty Acid Accumulation. Plants, 12(18), 3306. https://doi.org/10.3390/plants12183306










