Impact of Climate Change on Regulation of Genes Involved in Sex Determination and Fruit Production in Cucumber
Abstract
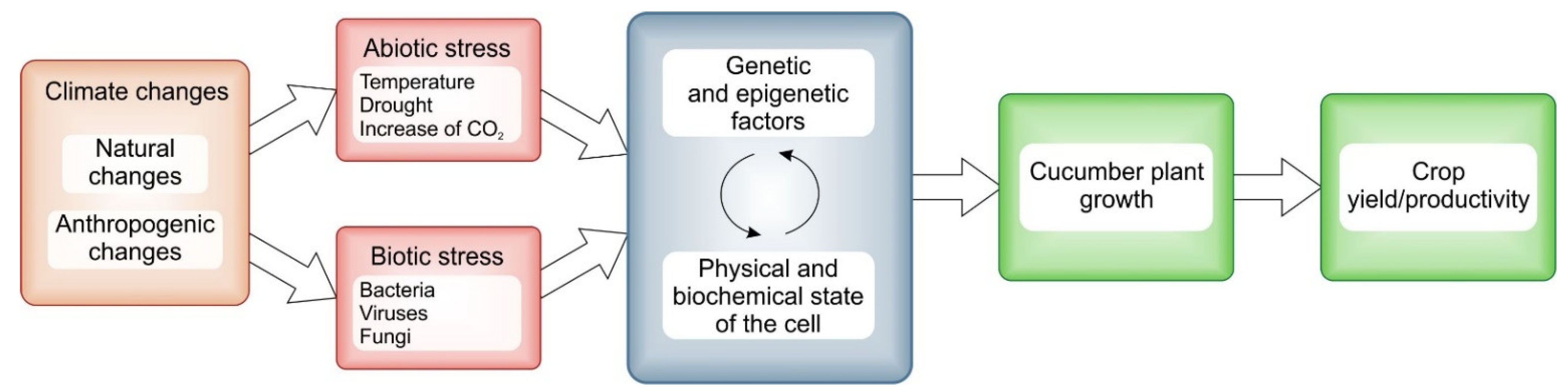
1. Introduction
2. Cucumber Growth, Flower and Fruit Development under the Impact of Climatic Changes
2.1. Basics of Cucumbers
2.2. Climatic Impact
2.2.1. Effect of Temperature Increase on Cucumber
2.2.2. Impact of Drought on Cucumber
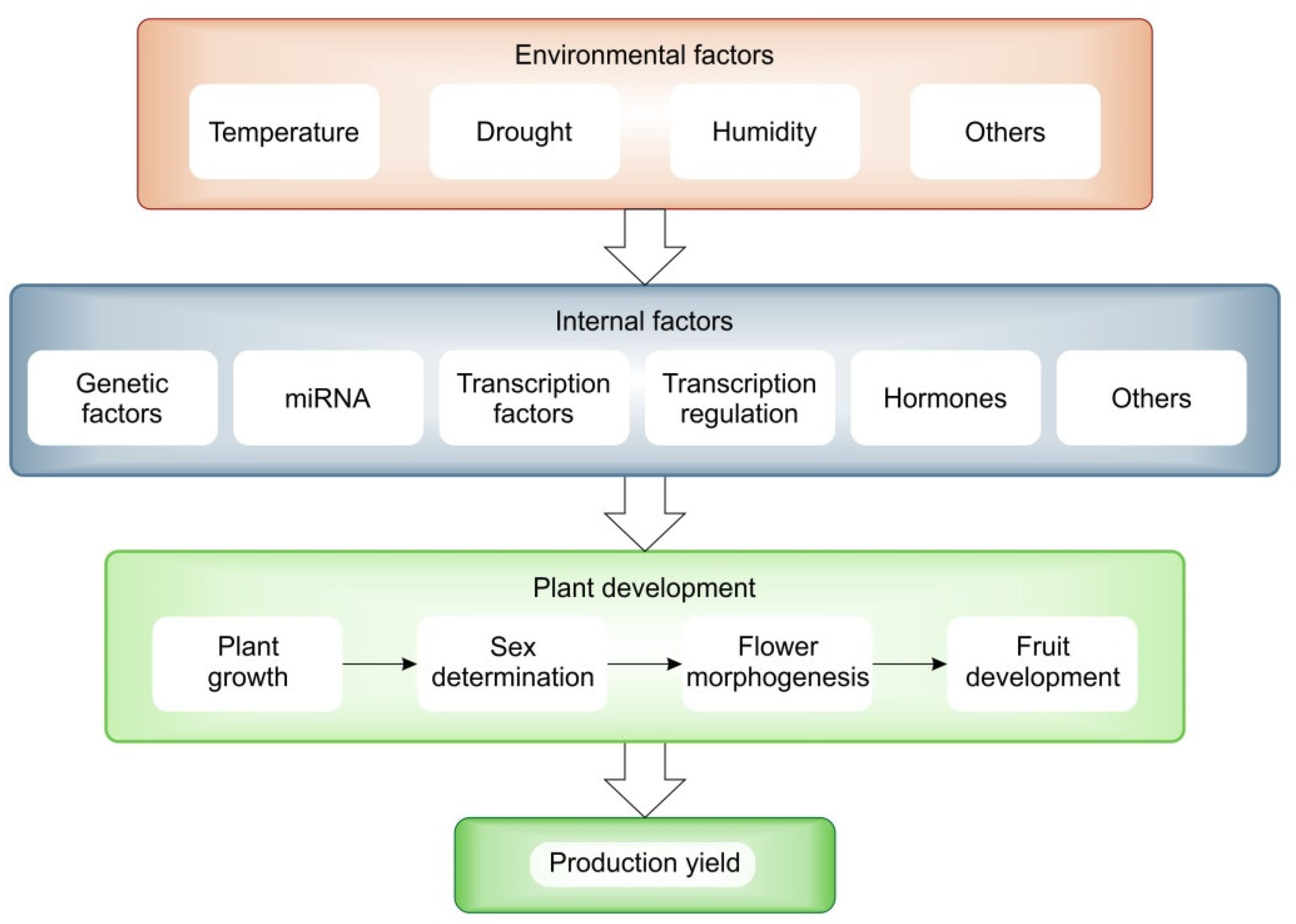
3. Molecular Regulation of Sex Determination, Flower and Fruit Development
3.1. Sex Determination and Flower Morphogenesis Processes
3.2. Factors Regulating Sex Determination
3.3. Climate Effect on Flowering
3.4. Fruit Development and Regulators of this Process
3.5. Regulation of mRNA and miRNA during Growth and Development by Climatic Factors
4. Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Das, A.; Singh, S.; Islam, Z.; Munshi, A.D.; Behera, T.K.; Dutta, S.; Weng, Y.; Dey, S.S. Current progress in genetic and ge-nomics-aided breeding for stress resistance in cucumber (Cucumis sativus L.). Sci. Hortic. 2022, 300, 111059. [Google Scholar] [CrossRef]
- Leisner, C.P. Review: Climate change impacts on food security-focus on perennial cropping systems and nutritional value. Plant Sci. 2020, 293, 110412. [Google Scholar] [CrossRef] [PubMed]
- Dombrovsky, A.; Tran-Nguyen, L.T.T.; Jones, R.A.C. Cucumber green mottle mosaic virus: Rapidly increasing global distribu-tion, etiology, epidemiology, and Management. Annu. Rev. Phytopathol. 2017, 55, 231–256. [Google Scholar] [CrossRef] [PubMed]
- Martín-Hernández, A.M.; Picó, B. Natural Resistances to Viruses in Cucurbits. Agronomy 2020, 11, 23. [Google Scholar] [CrossRef]
- Trebicki, P. Climate change and plant virus epidemiology. Virus Res. 2020, 286, 198059. [Google Scholar] [CrossRef]
- Jeffrey, C. An outline classification of the cucurbitaceae. In Biology and Utilization of the Cucurbitaceae; Cornell University Press: Ithaca, NY, USA, 2019; pp. 449–464. [Google Scholar]
- Pitrat, M.; Chauvet, M.; Foury, C. Diversity, history and production of cultivated cucurbits. Int. Symp. Cucurbits 1997, 492, 21–28. [Google Scholar] [CrossRef]
- Wubs, A.M.; Ma, Y.; Heuvelink, E.; Marcelis, L.F.M. Genetic differences in fruit-set patterns are determined by differences in fruit sink strength and a source: Sink threshold for fruit set. Ann. Bot. 2009, 104, 957–964. [Google Scholar] [CrossRef]
- Kahlen, K.; Stützel, H. Estimation of Geometric Attributes and Masses of Individual Cucumber Organs Using Three-dimensional Digitizing and Allometric Relationships. J. Am. Soc. Hortic. Sci. 2007, 132, 439–446. [Google Scholar] [CrossRef]
- Li, H.; Liu, S.S.; Yi, C.Y.; Wang, F.; Zhou, J.; Xia, X.J.; Shi, K.; Zhou, Y.H.; Yu, J.Q. Hydrogen peroxide mediates abscisic acid-induced HSP 70 accumulation and heat tolerance in grafted cucumber plants. Plant Cell Environ. 2014, 37, 2768–2780. [Google Scholar] [CrossRef]
- Ding, X.; Jiang, Y.; He, L.; Zhou, Q.; Yu, J.; Hui, D.; Huang, D. Exogenous glutathione improves high root-zone temperature tolerance by modulating photosynthesis, antioxidant and osmolytes systems in cucumber seedlings. Sci. Rep. 2016, 6, 35424. [Google Scholar] [CrossRef]
- Singh, M.C.; Singh, J.P.; Pandey, S.K.; Mahay, D.; Srivastava, V. Factors Affecting the Performance of Greenhouse Cucumber Cultivation-A Review. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 2304–2323. [Google Scholar] [CrossRef]
- Khurana, A.J.; Singh, M.B. Optimum temperature for the germination of seed. J. Appl. Ecol. 2001, 6, 71–78. [Google Scholar]
- EL-Aidy, F. Effect of plastic tunnel size on production of cucumber in Delta of egypt. Appl. Ecol. Environ. Res. 2007, 5, 11–24. [Google Scholar] [CrossRef]
- Olechowska, E.; Słomnicka, R.; Kaźmińska, K.; Olczak-Woltman, H.; Bartoszewski, G. The genetic basis of cold tolerance in cucumber (Cucumis sativus L.)—The latest developments and perspectives. J. Appl. Genet. 2022, 63, 597–608. [Google Scholar] [CrossRef]
- Onovo, J.A. Survey of Disease Incidence and Severity of Cucurbitaceous Crops in the Southeast, Annual Cropping Scheme Report; Vegetable Research Programme National Horticultural Research Institute: Mbato, Ivory Coast, 1992; 47p. [Google Scholar]
- Umeh, O.A.; Ojiako, F.O. Limitations of cucumber (Cucumis sativus L.) production for nutrition security in Southeast Nigeria. Int. J. Agric. Rural Dev. 2018, 21, 3437–3443. [Google Scholar]
- Martínez, C.; Jamilena, M. To be a male or a female flower, a question of ethylene in cucurbits. Curr. Opin. Plant Biol. 2021, 59, 101981. [Google Scholar] [CrossRef]
- Malepszy, S.; Niemirowicz-Szczytt, K. Sex determination in cucumber (Cucumis sativus) as a model system for molecular biology. Plant Sci. 1991, 80, 39–47. [Google Scholar] [CrossRef]
- Li, H.; Wang, F.; Chen, X.-J.; Shi, K.; Xia, X.-J.; Considine, M.J.; Yu, J.-Q.; Zhou, Y.-H. The sub/supra-optimal temperature-induced inhibition of photosynthesis and oxidative damage in cucumber leaves are alleviated by grafting onto figleaf gourd/luffa rootstocks. Physiol. Plant. 2014, 152, 571–584. [Google Scholar] [CrossRef]
- Pawełkowicz, M.E.; Skarzyńska, A.; Pląder, W.; Przybecki, Z. Genetic and molecular bases of cucumber (Cucumis sativus L.) sex determination. Mol. Breed. 2019, 39, 50. [Google Scholar] [CrossRef]
- Solomon, S.; Qin, D.; Manning, M.; Averyt, K.; Marquis, M.; Tignor, M.M. Climate Change 2007-The Physical Science Basis: Working Group I Contribution to the Fourth Assessment Report of the IPCC; Cambridge University Press: Cambridge, UK, 2007; Volume 4. [Google Scholar]
- Apata, T.G.; Samuel, K.; Adeola, A. Analysis of climate change perception and adaptation among arable food crop farmers in South Western Nigeria. In Proceedings of the International Association of Agricultural Economists’ 2009 Conference, Beijing, China, 16–22 August 2009; pp. 16–22. [Google Scholar]
- Ozor, N. Understanding climate change: Implications for Nigeria Agriculture, Policy and Extension. In Proceedings of the National Conference on Climate Change and the Nigeria Environment, Enugu, Nigeria, 29 June–2 July 2009. [Google Scholar]
- Backlund, P. Effects of Climate Change on Agriculture, Land Resources, Water Resources, and Biodiversity in the United States; DIANE Publishing: New York, NY, USA, 2009. [Google Scholar]
- Henson, R. The Rough Guide to Climate Change, 2nd ed.; The Science, The Solutions; Rough Guides Ltd.: London, UK, 2008; p. 384. [Google Scholar]
- Sage, R.F.; Kubien, D.S. The temperature response of C3 and C4photosynthesis. Plant Cell Environ. 2007, 30, 1086–1106. [Google Scholar] [CrossRef]
- Moore, C.E.; Meacham-Hensold, K.; Lemonnier, P.; Slattery, R.A.; Benjamin, C.; Bernacchi, C.J.; Lawson, T.; Cavanagh, A.P. The effect of increasing temperature on crop photosynthesis: From enzymes to ecosystems. J. Exp. Bot. 2021, 72, 2822–2844. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, J.; Farquhar, G.D. Effects of rising temperatures and [CO2] on the physiology of tropical forest trees. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 1811–1817. [Google Scholar] [CrossRef] [PubMed]
- Hatfield, J.L.; Prueger, J.H. Temperature extremes: Effect on plant growth and development. Weather. Clim. Extremes 2015, 10, 4–10. [Google Scholar] [CrossRef]
- Morison, J.I.L.; Lawlor, D.W. Interactions between increasing CO2 concentration and temperature on plant growth. Plant Cell Environ. 1999, 22, 659–682. [Google Scholar] [CrossRef]
- Naraghi, M.; Lotfi, M. Effect of different levels of shading on yield and fruit quality of cucumber (Cucumis sativus). IV Int. Symp. Cucurbits 2009, 871, 385–388. [Google Scholar] [CrossRef]
- Meng, L.; Qin, Z.; Li, S. Effect of high temperature on yield and quality of different cucumber cultivars. J. Am. Soc. Hortic. Sci. 2004, 6, 4–6. [Google Scholar]
- Kumar, S.N.; Aggarwal, P.K.; Rani, S.; Jain, S.; Saxena, R.; Chauhan, N. Impact of climate change on crop productivity in Western Ghats, coastal and northern regions of India. Curr. Sci. 2011, 101, 332–341. [Google Scholar]
- Ertan, S.K. Modelling the effect of temperature on seed germination in some cucurbits. Afr. J. Biotechnol. 2010, 9, 1343–1353. [Google Scholar] [CrossRef]
- Chen, L.; Yun, M.; Cao, Z.; Liang, Z.; Liu, W.; Wang, M.; Yan, J.; Yang, S.; He, X.; Jiang, B.; et al. Phenotypic Characteristics and Transcriptome of Cucumber Male Flower Development Under Heat Stress. Front. Plant Sci. 2021, 12, 758976. [Google Scholar] [CrossRef]
- Miao, M.M.; Lis, J. Effect of high temperature treatment at seedling stage on senescence, sexual differentiation and hormone contents of cucumber. Plant Physiol. Comm. Chin. Acad. Agric. Sci. 2001, 37, 195–198. [Google Scholar]
- Miao, M.M.; Cao, B.S. The relationship between heat injury and polyamines or proline contents during anther development and pollen germination in cucumber. Acta Hort. Sin. Chin. 2002, 29, 233–237. [Google Scholar]
- Barnabás, B.; Jäger, K.; Fehér, A. The effect of drought and heat stress on reproductive processes in cereals. Plant Cell Environ. 2008, 31, 11–38. [Google Scholar] [CrossRef]
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S.; et al. Crop Production under Drought and Heat Stress: Plant Responses and Management Options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef]
- Kaur, G.; Asthir, B. Molecular responses to drought stress in plants. Biol. Plant. 2017, 61, 201–209. [Google Scholar] [CrossRef]
- Mariani, L.; Ferrante, A. Agronomic management for enhancing plant tolerance to abiotic stresses—Drought, salinity, hypoxia, and lodging. Horticulturae 2017, 3, 52. [Google Scholar] [CrossRef]
- Shafiq, S.; Akram, N.A.; Ashraf, M.; García-Caparrós, P.; Ali, O.M.; Latef, A.A. Influence of glycine betaine (natural and synthetic) on growth, metabolism and yield production of drought-stressed maize (Zea mays L.) plants. Plants 2021, 10, 2540. [Google Scholar] [CrossRef]
- Alam, A.; Ullah, H.; Thuenprom, N.; Tisarum, R.; Cha-Um, S.; Datta, A. Seed priming with salicylic acid enhances growth, physiological traits, fruit yield, and quality parameters of cantaloupe under water-deficit stress. S. Afr. J. Bot. 2022, 150, 1–12. [Google Scholar] [CrossRef]
- Eid, M.A.M.; El-Hady, M.A.A.; Abdelkader, M.A.; Abd-Elkrem, Y.M.; El-Gabry, Y.A.; El-Temsah, M.E.; El-Areed, S.R.M.; Rady, M.M.; Alamer, K.H.; Alqubaie, A.I.; et al. Response in Physiological Traits and Antioxidant Capacity of Two Cotton Cultivars under Water Limitations. Agronomy 2022, 12, 803. [Google Scholar] [CrossRef]
- Farouk, S.; Al-Huqail, A.A. Sodium nitroprusside application regulates antioxidant capacity, improves phytopharmaceutical production and essential oil yield of marjoram herb under drought. Ind. Crop. Prod. 2020, 158, 113034. [Google Scholar] [CrossRef]
- Farouk, S.; Al-Ghamdi, A.A.M. Sodium nitroprusside application enhances drought tolerance in marjoram herb by promoting chlorophyll biosynthesis and enhancing osmotic adjustment capacity. Arab. J. Geosci. 2021, 14, 1–13. [Google Scholar] [CrossRef]
- Shemi, R.; Wang, R.; Gheith, E.-S.M.S.; Hussain, H.A.; Hussain, S.; Irfan, M.; Cholidah, L.; Zhang, K.; Zhang, S.; Wang, L. Effects of salicylic acid, zinc and glycine betaine on morpho-physiological growth and yield of maize under drought stress. Sci. Rep. 2021, 11, 3195. [Google Scholar] [CrossRef] [PubMed]
- Anjum, F.; Yaseen, M.; Rasul, E.; Wahid, A.; Anjum, S. Water stress in barley (Hordeum vulgare L.). II. Effect on chemical composition and chlorophyll contents. Pak. J. Agric. Sci. 2003, 40, 45–49. [Google Scholar]
- Ashraf, M.; Shahbaz, M.; Ali, Q. Drought-induced modulation in growth and mineral nutrients in canola (Brassica napus L.). Pak. J. Bot. 2013, 45, 93–98. [Google Scholar]
- Bañon, S.; Ochoa, J.; Franco, J.A.; Alarcón, J.J.; Sánchez-Blanco, M.J. Hardening of oleander seedlings by deficit irrigation and low air humidity. Environ. Exp. Bot. 2006, 56, 36–43. [Google Scholar] [CrossRef]
- Blum, A. Osmotic adjustment is a prime drought stress adaptive engine in support of plant production. Plant Cell Environ. 2016, 40, 4–10. [Google Scholar] [CrossRef]
- Lawlor, D.W.; Cornic, G. Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant Cell Environ. 2002, 25, 275–294. [Google Scholar] [CrossRef]
- González-Villagra, J.; Rodrigues-Salvador, A.; Nunes-Nesi, A.; Cohen, J.D.; Reyes-Díaz, M.M. Age-related mechanism and its relationship with secondary metabolism and abscisic acid in Aristotelia chilensis plants subjected to drought stress. Plant Physiol. Biochem. 2018, 124, 136–145. [Google Scholar] [CrossRef]
- Liu, B.B.; Li, M.; Li, Q.M.; Cui, Q.Q.; Zhang, W.D.; Ai, X.Z.; Bi, H.G. Combined effects of elevated CO2 concentration and drought stress on photosynthetic performance and leaf structure of cucumber (Cucumis sativus L.) seedlings. Photosynthetica 2018, 56, 942–952. [Google Scholar] [CrossRef]
- Farag, M.I.; Behera, T.K.; Munshi, A.D.; Bharadwaj, C.; Jat, G.S.; Khanna, M.; Chinnusamy, V. Physiological analysis of drought tolerance of cucumber (Cucumis sativus) genotypes. Indian J. Agric. Sci. 2019, 89, 1445–1450. [Google Scholar] [CrossRef]
- Metwaly, E.-S.E.; Al-Yasi, H.M.; Ali, E.F.; Farouk, H.A.; Farouk, S. Deteriorating Harmful Effects of Drought in Cucumber by Spraying Glycinebetaine. Agriculture 2022, 12, 2166. [Google Scholar] [CrossRef]
- Lu, Y.; Li, Y.; Zhang, J.; Xiao, Y.; Yue, Y.; Duan, L.; Zhang, M.; Li, Z. Overexpression of Arabidopsis Molybdenum Cofactor Sulfurase Gene Confers Drought Tolerance in Maize (Zea mays L.). PLoS ONE 2013, 8, e52126. [Google Scholar] [CrossRef]
- He, J.-D.; Zou, Y.-N.; Wu, Q.-S.; Kuča, K. Mycorrhizas enhance drought tolerance of trifoliate orange by enhancing activities and gene expression of antioxidant enzymes. Sci. Hortic. 2020, 262, 108745. [Google Scholar] [CrossRef]
- Takahashi, H.; Saito, T.; Suge, H. Intergeneric Translocation of Floral Stimulus across a Graft in Monoecious Cucurbitaceae with Special Reference to the Sex Expression of Flowers. Plant Cell Physiol. 1982, 23, 1–9. [Google Scholar] [CrossRef]
- Rudich, J.; Halevy, A.H.; Kedar, N. Increase in femaleness of three cucurbits by treatment with Ethrel, an ethylene releasing compound. Planta 1969, 86, 69–76. [Google Scholar] [CrossRef]
- Yin, T.; Quinn, J.A. Tests of a mechanistic model of one hormone regulating both sexes in Cucumis sativus (Cucurbitaceae). Am. J. Bot. 1995, 82, 1537–1546. [Google Scholar] [CrossRef]
- Trebitsh, T.; Rudich, J.; Riov, J. Auxin, biosynthesis of ethylene and sex expression in cucumber (Cucumis sativus). Plant Growth Regul. 1987, 5, 105–113. [Google Scholar] [CrossRef]
- Manzano, S.; Martínez, C.; Megías, Z.; Gómez, P.; Garrido, D.; Jamilena, M. The role of ethylene and brassinosteroids in the control of sex expression and flower development in Cucurbita pepo. Plant Growth Regul. 2011, 65, 213–221. [Google Scholar] [CrossRef]
- Adams, D.O.; Yang, S.F. Ethylene biosynthesis: Identification of 1-aminocyclopropane-1-carboxylic acid as an intermediate in the conversion of methionine to ethylene. Proc. Natl. Acad. Sci. USA 1979, 76, 170–174. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, S. Ethylene Biosynthesis and Regulation in Plants. In Ethylene in Plants; Wen, C.-K., Ed.; Springer: Dordrecht, The Netherlands, 2014; pp. 1–25. ISBN 978-94-017-9483-1. [Google Scholar]
- Chang, C. Q&A: How do plants respond to ethylene and what is its importance? BMC Biol. 2016, 14, 7. [Google Scholar]
- Trebitsh, T.; Staub, J.E.; O’Neill, S.D. Identification of a 1-Aminocyclopropane-1-Carboxylic Acid Synthase Gene Linked to the Female (F) Locus That Enhances Female Sex Expression in Cucumber. Plant Physiol. 1997, 113, 987–995. [Google Scholar] [CrossRef]
- Boualem, A.; Troadec, C.; Kovalski, I.; Sari, M.-A.; Perl-Treves, R.; Bendahmane, A. A Conserved Ethylene Biosynthesis Enzyme Leads to Andromonoecy in Two Cucumis Species. PLoS ONE 2009, 4, e6144. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Huang, S.; Liu, S.; Pan, J.; Zhang, Z.; Tao, Q.; Shi, Q.; Jia, Z.; Zhang, W.; Chen, H.; et al. Molecular Isolation of the M Gene Suggests That a Conserved-Residue Conversion Induces the Formation of Bisexual Flowers in Cucumber Plants. Genetics 2009, 182, 1381–1385. [Google Scholar] [CrossRef] [PubMed]
- Kubicki, B. Investigations of sex determination in cucumber (Cucumis sativus L.). VI. Androecism. Genet. Pol. 1969, 10, 88–99. [Google Scholar]
- Kubicki, B. Investigations on sex determination in cucumber (Cucumis sativus L.). VII. Andromonoecism and hermaphroditism. Genet. Pol. 1969, 10, 101–120. [Google Scholar]
- Boualem, A.; Troadec, C.; Camps, C.; Lemhemdi, A.; Morin, H.; Sari, M.-A.; Fraenkel-Zagouri, R.; Kovalski, I.; Dogimont, C.; Perl-Treves, R.; et al. A cucurbit androecy gene reveals how unisexual flowers develop and dioecy emerges. Science 2015, 350, 688–691. [Google Scholar] [CrossRef]
- Chen, H.; Sun, J.; Li, S.; Cui, Q.; Zhang, H.; Xin, F.; Wang, H.; Lin, T.; Gao, D.; Wang, S.; et al. An ACC Oxidase Gene Essential for Cucumber Carpel Development. Mol. Plant 2016, 9, 1315–1327. [Google Scholar] [CrossRef]
- Li, D.; Sheng, Y.; Niu, H.; Li, Z. Gene Interactions Regulating Sex Determination in Cucurbits. Front. Plant Sci. 2019, 10, 1231. [Google Scholar] [CrossRef]
- Robinson, R.W.; Munger, H.M.; Whitaker, T.W.; Bohn, G.W. Genes of the Cucurbitaceae1. Hortscience 1976, 11, 554–568. [Google Scholar] [CrossRef]
- Mibus, H.; Tatlioglu, T. Molecular characterization and isolation of the F/f gene for femaleness in cucumber (Cucumis sativus L.). Theor. Appl. Genet. 2004, 109, 1669–1676. [Google Scholar] [CrossRef]
- More, T.A.; Munger, H.M. Gynoecious sex expression and stability in cucumber (Cucumis sativus L.). Euphytica 1986, 35, 899–903. [Google Scholar] [CrossRef]
- Zhang, H.; Li, S.; Yang, L.; Cai, G.; Chen, H.; Gao, D.; Lin, T.; Cui, Q.; Wang, D.; Li, Z.; et al. Gain-of-function of the 1-aminocyclopropane-1-carboxylate synthase gene ACS1G induces female flower development in cucumber gynoecy. Plant Cell 2020, 33, 306–321. [Google Scholar] [CrossRef]
- Martin, A.; Troadec, C.; Boualem, A.; Rajab, M.; Fernandez, R.; Morin, H.; Pitrat, M.; Dogimont, C.; Bendahmane, A. A transposon-induced epigenetic change leads to sex determination in melon. Nature 2009, 461, 1135–1138. [Google Scholar] [CrossRef]
- Hu, B.; Li, D.; Liu, X.; Qi, J.; Gao, D.; Zhao, S.; Huang, S.; Sun, J.; Yang, L. Engineering Non-transgenic Gynoecious Cucumber Using an Improved Transformation Protocol and Optimized CRISPR/Cas9 System. Mol. Plant 2017, 10, 1575–1578. [Google Scholar] [CrossRef]
- Yamasaki, S.; Yamakuchi, R.; Yamanaka, S.; Manabe, K. Potential Involvement of Cell Cycle-Related Genes in the Arrest of Stamen Development of Female Flowers During Sex Expression in Cucumber (Cucumis sativus L.). Environ. Control. Biol. 2017, 55, 105–112. [Google Scholar] [CrossRef]
- Wang, R.; Lin, Y.; Jin, Q.; Yao, C.; Zhong, Y.; Wu, T. RNA-Seq analysis of gynoecious and weak female cucumber revealing the cell cycle pathway may regulate sex determination in cucumber. Gene 2019, 687, 289–297. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, M.; Shang, J.; Liu, Z.; Weng, Y.; Yue, H.; Li, Y.; Chen, P. A 5.5-KB LTR-retrotransposon insertion inside phy-tochrome B gene (csphyb) results in long hypocotyl and early flowering in cucumber (Cucumis sativus L.). Theor. Appl. Genet. 2023, 136, 68. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, H.; Quan, R.; Wang, X.-C.; Huang, R. Transcriptional Regulation of the Ethylene Response Factor LeERF2 in the Expression of Ethylene Biosynthesis Genes Controls Ethylene Production in Tomato and Tobacco. Plant Physiol. 2009, 150, 365–377. [Google Scholar] [CrossRef]
- Klee, H.J.; Giovannoni, J.J. Genetics and Control of Tomato Fruit Ripening and Quality Attributes. Annu. Rev. Genet. 2011, 45, 41–59. [Google Scholar] [CrossRef]
- Liu, M.; Pirrello, J.; Chervin, C.; Roustan, J.-P.; Bouzayen, M. Ethylene control of fruit ripening: Revisiting the complex network of transcriptional regulation. Plant Physiol. 2015, 169, 2380–2390. [Google Scholar] [CrossRef]
- Tao, Q.; Niu, H.; Wang, Z.; Zhang, W.; Wang, H.; Wang, S.; Zhang, X.; Li, Z. Ethylene responsive factor ERF110 mediates ethylene-regulated transcription of a sex determination-related orthologous gene in two Cucumis species. J. Exp. Bot. 2018, 69, 2953–2965. [Google Scholar] [CrossRef]
- Pan, J.; Wang, G.; Wen, H.; Du, H.; Lian, H.; He, H.; Pan, J.; Cai, R. Differential Gene Expression Caused by the F and M Loci Provides Insight Into Ethylene-Mediated Female Flower Differentiation in Cucumber. Front. Plant Sci. 2018, 9, 1091. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-H.; Li, F.; Duan, Q.-H.; Han, T.; Xu, Z.-H.; Bai, S.-N. Ethylene perception is involved in female cucumber flower development. Plant J. 2010, 61, 862–872. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.-N.; Xu, Z.-H. Unisexual cucumber flowers, sex and sex differentiation. Int. Rev. Cell Mol. Biol. 2013, 304, 1–55. [Google Scholar] [PubMed]
- García, A.; Aguado, E.; Parra, G.; Manzano, S.; Martínez, C.; Megías, Z.; Cebrián, G.; Romero, J.; Beltrán, S.; Garrido, D.; et al. Phenomic and Genomic Characterization of a Mutant Platform in Cucurbita pepo. Front. Plant Sci. 2018, 9, 1049. [Google Scholar] [CrossRef] [PubMed]
- García, A.; Aguado, E.; Martínez, C.; Loska, D.; Beltrán, S.; Valenzuela, J.L.; Garrido, D.; Jamilena, M. The ethylene receptors CpETR1A and CpETR2B cooperate in the control of sex determination in Cucurbita pepo. J. Exp. Bot. 2019, 71, 154–167. [Google Scholar] [CrossRef]
- Gu, H.T.; Wang, D.H.; Li, X.; He, C.X.; Xu, Z.H.; Bai, S.N. Characterization of an ethylene-inducible, calcium-dependent nu-clease that is differentially expressed in cucumber flower development. New Phytol. 2011, 192, 590–600. [Google Scholar] [CrossRef]
- Wu, T.; Qin, Z.; Zhou, X.; Feng, Z.; Du, Y. Transcriptome profile analysis of floral sex determination in cucumber. J. Plant Physiol. 2010, 167, 905–913. [Google Scholar] [CrossRef]
- Niu, H.; Wang, H.; Zhao, B.; He, J.; Yang, L.; Ma, X.; Cao, J.; Li, Z.; Shen, J. Exogenous auxin-induced enhancer of shoot re-generation 2 (ESR2) enhances femaleness of cucumber by activating the csacs2 gene. Hortic. Res. 2022, 9, uhab085. [Google Scholar] [CrossRef]
- Gu, R.; Liu, X.; Zhao, W.; Yan, S.; Sun, L.; Wu, B.; Zhang, X. Functional Characterization of the Promoter and Second Intron of CUM1 During Flower Development in Cucumber (Cucumis sativus L.). Hortic. Plant J. 2018, 4, 103–110. [Google Scholar] [CrossRef]
- Yan, S.; Che, G.; Ding, L.; Chen, Z.; Liu, X.; Wang, H.; Zhao, W.; Ning, K.; Zhao, J.; Tesfamichael, K.; et al. Different cucumber CsYUC genes regulate response to abiotic stresses and flower development. Sci. Rep. 2016, 6, 20760. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Liu, B.; Wang, W.; Liu, X.; Chen, C.; Liu, X.; Yang, S.; Ren, H. A GAMYB homologue CsGAMYB1 regulates sex expression of cucumber via an ethylene-independent pathway. J. Exp. Bot. 2014, 65, 3201–3213. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, B.; Yang, S.; An, J.; Chen, C.; Zhang, X.; Ren, H. A cucumber della homolog csgaip may inhibit staminate de-velopment through transcriptional repression of B class floral homeotic genes. PLoS ONE 2014, 9, e91804. [Google Scholar]
- Zhang, Y.; Zhao, G.; Li, Y.; Mo, N.; Zhang, J.; Liang, Y. Transcriptomic Analysis Implies That GA Regulates Sex Expression via Ethylene-Dependent and Ethylene-Independent Pathways in Cucumber (Cucumis sativus L.). Front. Plant Sci. 2017, 8, 10. [Google Scholar] [CrossRef]
- Ferrero, L.V.; Viola, I.L.; Ariel, F.D.; Gonzalez, D.H. Class I TCP Transcription Factors Target the Gibberellin Biosynthesis Gene GA20ox1 and the Growth-Promoting Genes HBI1 and PRE6 during Thermomorphogenic Growth in Arabidopsis. Plant Cell Physiol. 2019, 60, 1633–1645. [Google Scholar] [CrossRef]
- Fukuda, M.; Matsuo, S.; Kikuchi, K.; Mitsuhashi, W.; Toyomasu, T.; Honda, I. The endogenous level of GA1 is upregulated by high temperature during stem elongation in lettuce through LsGA3ox1 expression. J. Plant Physiol. 2009, 166, 2077–2084. [Google Scholar] [CrossRef]
- Papadopoulou, E.; Grumet, R. Brassinosteriod-induced Femaleness in Cucumber and Relationship to Ethylene Production. Hortscience 2005, 40, 1763–1767. [Google Scholar] [CrossRef]
- Pawełkowicz, M.; Osipowski, P.; Wojcieszek, M.; Woycicki, R.; Witkowicz, J.; Hincha, D.; Przybecki, Z. Corrected version Identification and characterization of genes connected with flower morphogenesis in cucumber. BioTechnologia 2012, 3, 123–134. [Google Scholar] [CrossRef]
- Pawełkowicz, M.E.; Wojcieszek, M.; Osipowski, P.; Krzywkowski, T.; Pląder, W.; Przybecki, Z. Identification and bioinformatics comparison of two novel phosphatases in monoecious and gynoecious cucumber lines. Photonics Appl. Astron Commun. Ind. High-Energy Phys. Exp. 2016, 10031, 756–766. [Google Scholar]
- León, P. Sugar and hormone connections. Trends Plant Sci. 2003, 8, 110–116. [Google Scholar] [CrossRef]
- Rolland, F.; Baena-Gonzalez, E.; Sheen, J. Sugar sensing and signaling in plants: Conserved and novel mechanisms. Annu. Rev. Plant Biol. 2006, 57, 675–709. [Google Scholar] [CrossRef]
- Turek, S.; Aparna; Skarzyńska, A.; Pląder, W.; Pawełkowicz, M. Understanding transcription factors and how they affect processes in cucumber sex determination. Metabolites 2023, 13, 740. [Google Scholar] [CrossRef]
- Cai, Y.; Bartholomew, E.S.; Dong, M.; Zhai, X.; Yin, S.; Zhang, Y.; Feng, Z.; Wu, L.; Liu, W.; Shan, N.; et al. The HD-ZIP IV transcription factor GL2-LIKE regulates male flowering time and fertility in cucumber. J. Exp. Bot. 2020, 71, 5425–5437. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ning, K.; Che, G.; Yan, S.; Han, L.; Gu, R.; Li, Z.; Weng, Y.; Zhang, X. Csspl functions as an adaptor between HD-ZIP III and CsWUS transcription factors regulating anther and ovule development in Cucumis sativus (cucumber). Plant J. 2018, 94, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Oda, S.; Kaneko, F.; Yano, K.; Fujioka, T.; Masuko, H.; Park, J.-I.; Kikuchi, S.; Hamada, K.; Endo, M.; Nagano, K.; et al. Mor-phological and gene expression analysis under cool temperature conditions in rice anther development. Genes Genet. Syst. 2010, 85, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Borghi, M.; De Souza, L.P.; Yoshida, T.; Fernie, A.R. Flowers and climate change: A metabolic perspective. New Phytol. 2019, 224, 1425–1441. [Google Scholar] [CrossRef]
- Lohani, N.; Singh, M.B.; Bhalla, P.L. High temperature susceptibility of sexual reproduction in crop plants. J. Exp. Bot. 2019, 71, 555–568. [Google Scholar] [CrossRef]
- Antunes, M.D.C.; Sfakiotakis, E.M. Effect of high temperature stress on ethylene biosynthesis, respiration and ripening of ‘Hayward’ kiwifruit. Postharvest Biol. Technol. 2000, 20, 251–259. [Google Scholar] [CrossRef]
- Biggs, M.S.; Woodson, W.R.; Handa, A.K. Biochemical basis of high-temperature inhibition of ethylene biosynthesis in ripening tomato fruits. Physiol. Plant. 1988, 72, 572–578. [Google Scholar] [CrossRef]
- Zhao, X.-C.; Schaller, G.E. Effect of salt and osmotic stress upon expression of the ethylene receptor ETR1 in Arabidopsis thaliana. FEBS Lett. 2004, 562, 189–192. [Google Scholar] [CrossRef]
- Young, T.E.; Meeley, R.B.; Gallie, D.R. ACC synthase expression regulates leaf performance and drought tolerance in maize. Plant J. 2004, 40, 813–825. [Google Scholar] [CrossRef]
- Jegadeesan, S.; Chaturvedi, P.; Ghatak, A.; Pressman, E.; Meir, S.; Faigenboim, A.; Rutley, N.; Beery, A.; Harel, A.; Weckwerth, W.; et al. Proteomics of Heat-Stress and Ethylene-Mediated Thermotolerance Mechanisms in Tomato Pollen Grains. Front. Plant Sci. 2018, 9, 1558. [Google Scholar] [CrossRef]
- Apelbaum, A.; Yang, S.F. Biosynthesis of stress ethylene induced by water deficit. Plant Physiol. 1981, 68, 594–596. [Google Scholar] [CrossRef]
- Jin, Y.; Pan, W.; Zheng, X.; Cheng, X.; Liu, M.; Ma, H.; Ge, X. OsERF101, an ERF family transcription factor, regulates drought stress response in reproductive tissues. Plant Mol. Biol. 2018, 98, 51–65. [Google Scholar] [CrossRef]
- Wan, L.; Zhang, J.; Zhang, H.; Zhang, Z.; Quan, R.; Zhou, S.; Huang, R. Transcriptional Activation of OsDERF1 in OsERF3 and OsAP2-39 Negatively Modulates Ethylene Synthesis and Drought Tolerance in Rice. PLoS ONE 2011, 6, e25216. [Google Scholar] [CrossRef]
- Yu, Y.; Yang, D.; Zhou, S.; Gu, J.; Wang, F.; Dong, J.; Huang, R. The ethylene response factor OSERF109 negatively affects ethylene biosynthesis and drought tolerance in Rice. Protoplasma 2016, 254, 401–408. [Google Scholar] [CrossRef]
- Trujillo, L.E.; Sotolongo, M.; Menéndez, C.; Ochogavía, M.E.; Coll, Y.; Hernández, I.; Borrás-Hidalgo, O.; Thomma, B.P.; Vera, P.; Hernández, L. Soderf3, a novel sugarcane ethylene responsive factor (ERF), enhances salt and drought tolerance when overexpressed in tobacco plants. Plant Cell Physiol. 2008, 49, 512–525. [Google Scholar] [CrossRef]
- Liang, S.; Xiong, W.; Yin, C.; Xie, X.; Jin, Y.-J.; Zhang, S.; Yang, B.; Ye, G.; Chen, S.; Luan, W.-J. Overexpression of OsARD1 Improves Submergence, Drought, and Salt Tolerances of Seedling Through the Enhancement of Ethylene Synthesis in Rice. Front. Plant Sci. 2019, 10, 1088. [Google Scholar] [CrossRef]
- Linsley-Noakes, G.C.; Allan, P. Effects of winter temperatures on flower development in two clones of kiwifruit (Actinidia deliciosa (A. Chev.) C.F. Liang et A.R. Ferguson). Sci. Hortic. 1987, 33, 249–260. [Google Scholar] [CrossRef]
- Ding, X.; Guo, Q.; Li, Q.; Gai, J.; Yang, S. Comparative Transcriptomics Analysis and Functional Study Reveal Important Role of High-Temperature Stress Response Gene GmHSFA2 During Flower Bud Development of CMS-Based F1 in Soybean. Front. Plant Sci. 2020, 11, 600217. [Google Scholar] [CrossRef]
- Dias, A.S.; Barreiro, M.G.; Campos, P.S.; Ramalho, J.C.; Lidon, F.C. Wheat Cellular Membrane Thermotolerance Under Heat Stress. J. Agron. Crop. Sci. 2010, 196, 100–108. [Google Scholar] [CrossRef]
- Djanaguiraman, M.; Prasad, P.V.V. Ethylene production under high temperature stress causes premature leaf senescence in soybean. Funct. Plant Biol. 2010, 37, 1071–1084. [Google Scholar] [CrossRef]
- Irigoyen, J.J.; Emerich, D.W.; Sánchez-Díaz, M. Alfalfa leaf senescence induced by drought stress: Photosynthesis, hydrogen peroxide metabolism, lipid peroxidation and ethylene evolution. Physiol. Plant. 1992, 84, 67–72. [Google Scholar] [CrossRef]
- McMichael, B.L.; Jordan, W.R.; Powell, R.D. An Effect of Water Stress on Ethylene Production by Intact Cotton Petioles. Plant Physiol. 1972, 49, 658–660. [Google Scholar] [CrossRef] [PubMed]
- Arraes, F.B.M.; Beneventi, M.A.; Lisei de Sa, M.E.; Paixao, J.F.; Albuquerque, E.V.; Marin, S.R.; Purgatto, E.; Nepomuceno, A.L.; Grossi-de-Sa, M.F. Implications of ethylene biosynthesis and signaling in soybean drought stress tolerance. BMC Plant Biol. 2015, 15, 213. [Google Scholar] [CrossRef] [PubMed]
- Spikman, G. Development and ethylene production of buds and florets of cut freesia inflorescences as influenced by silver thiosulphate, aminoethoxyvinylglycine and sucrose. Sci. Hortic. 1989, 39, 73–81. [Google Scholar] [CrossRef]
- Dar, R.A.; Nisar, S.; Tahir, I. Ethylene: A key player in ethylene sensitive flower senescence: A review. Sci. Hortic. 2021, 290, 110491. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, B.; Wang, S.; Lin, T.; Yang, L.; Zhao, Z.; Zhang, Z.; Huang, S.; Yang, X. Genome-wide Target Mapping Shows Histone Deacetylase Complex1 Regulates Cell Proliferation in Cucumber Fruit. Plant Physiol. 2020, 182, 167–184. [Google Scholar] [CrossRef]
- Xin, T.; Zhang, Z.; Li, S.; Zhang, S.; Li, Q.; Zhang, Z.-H.; Huang, S.; Yang, X. Genetic Regulation of Ethylene Dosage for Cucumber Fruit Elongation. Plant Cell 2019, 31, 1063–1076. [Google Scholar] [CrossRef]
- Zhao, J.; Jiang, L.; Che, G.; Pan, Y.; Li, Y.; Hou, Y.; Zhao, W.; Zhong, Y.; Ding, L.; Yan, S.; et al. A Functional Allele of CsFUL1 Regulates Fruit Length through Repressing CsSUP and Inhibiting Auxin Transport in Cucumber. Plant Cell 2019, 31, 1289–1307. [Google Scholar] [CrossRef]
- Zhou, G.; Chen, C.; Liu, X.; Yang, K.; Wang, C.; Lu, X.; Tian, Y.; Chen, H. The formation of hollow trait in cucumber (Cucumis sativus L.) fruit is controlled by CS ALMT2. Int. J. Mol. Sci. 2022, 23, 6173. [Google Scholar] [CrossRef]
- Martínez, C.; Manzano, S.; Megías, Z.; Garrido, D.; Picó, B.; Jamilena, M. Involvement of ethylene biosynthesis and signalling in fruit set and early fruit development in zucchini squash (Cucurbita pepo L.). BMC Plant Biol. 2013, 13, 139. [Google Scholar] [CrossRef]
- Shinozaki, Y.; Nicolas, P.; Fernandez-Pozo, N.; Ma, Q.; Evanich, D.J.; Shi, Y.; Xu, Y.; Zheng, Y.; Snyder, S.I.; Martin, L.B.B.; et al. High-resolution spatiotemporal transcriptome mapping of tomato fruit development and ripening. Nat. Commun. 2018, 9, 364. [Google Scholar] [CrossRef]
- Shnaider, Y.; Mitra, D.; Miller, G.; Baniel, A.; Doniger, T.; Kuhalskaya, A.; Scossa, F.; Fernie, A.R.; Brotman, Y.; Perl-Treves, R. Cucumber ovaries inhibited by dominant fruit express a dynamic developmental program, distinct from either senes-cence-determined or fruit-setting ovaries. Plant J. 2018, 96, 651–669. [Google Scholar] [CrossRef]
- Liu, B.; Liu, X.; Yang, S.; Chen, C.; Xue, S.; Cai, Y.; Wang, D.; Yin, S.; Gai, X.; Ren, H. Silencing of the gibberellin receptor homolog, csgid1a, affects locule formation in cucumber (Cucumis sativus) fruit. New Phytol. 2016, 210, 551–563. [Google Scholar] [CrossRef]
- Li, S.; Wang, C.; Zhou, X.; Liu, D.; Liu, C.; Luan, J.; Qin, Z.; Xin, M. The curvature of cucumber fruits is associated with spatial variation in auxin accumulation and expression of a YUCCA biosynthesis gene. Hortic. Res. 2020, 7, 135. [Google Scholar] [CrossRef]
- Wei, K.; Chen, J.; Wang, Y.; Chen, Y.; Chen, S.; Lin, Y.; Pan, S.; Zhong, X.; Xie, D. Genome-wide analysis of Bzip-encoding genes in maize. DNA Res. 2012, 19, 463–476. [Google Scholar] [CrossRef]
- Baloglu, M.C.; Eldem, V.; Hajyzadeh, M.; Unver, T. Genome-Wide Analysis of the bZIP Transcription Factors in Cucumber. PLoS ONE 2014, 9, e96014. [Google Scholar] [CrossRef]
- Li, D.; Fu, F.; Zhang, H.; Song, F. Genome-wide systematic characterization of the bZIP transcriptional factor family in tomato (Solanum lycopersicum L.). BMC Genom. 2015, 16, 771. [Google Scholar] [CrossRef]
- Yang, S.; Cai, Y.; Liu, X.; Dong, M.; Zhang, Y.; Chen, S.; Zhang, W.; Li, Y.; Tang, M.; Zhai, X.; et al. A CsMYB6-CsTRY module regulates fruit trichome initiation in cucumber. J. Exp. Bot. 2018, 69, 1887–1902. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, C.; Duan, L.; Luan, Q.; Li, J.; Yang, A.; Qi, X.; Ren, Z. csmyb60 is a key regulator of flavonols and proantho-cyanidans that determine the colour of fruit spines in cucumber. J. Exp. Bot. 2018, 70, 69–84. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, J.; Yang, Y.; Luo, J.; Zheng, X.; Wen, C.; Xu, Y. Transcription Factor CsWIN1 Regulates Pericarp Wax Biosynthesis in Cucumber Grafted on Pumpkin. Front. Plant Sci. 2019, 10, 1564. [Google Scholar] [CrossRef]
- Che, G.; Pan, Y.; Liu, X.; Li, M.; Zhao, J.; Yan, S.; He, Y.; Wang, Z.; Cheng, Z.; Song, W.; et al. Natural variation in CRABS CLAW contributes to fruit length divergence in cucumber. Plant Cell 2022, 35, 738–755. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, H.E.; Song, H.; Staub, J.E.; Simon, P.W. Inheritance of beta-carotene-associated flesh color in cucumber (Cucumis sativus L.) fruit. Euphytica 2010, 171, 301–311. [Google Scholar] [CrossRef]
- Lu, H.W.; Miao, H.; Tian, G.L.; Wehner, T.C.; Gu, X.F.; Zhang, S.P. Molecular mapping and candidate gene analysis for yellow fruit flesh in cucumber. Mol. Breed. 2015, 35, 64. [Google Scholar] [CrossRef]
- Bo, K.; Wei, S.; Wang, W.; Miao, H.; Dong, S.; Zhang, S.; Gu, X. QTL mapping and genome-wide association study reveal two novel loci associated with green flesh color in cucumber. BMC Plant Biol. 2019, 19, 243. [Google Scholar] [CrossRef] [PubMed]
- Rice, C.A.; Rymal, K.S.; Chambliss, O.L.; Johnson, F.A. Chromatographic and mass spectral analysis of cucurbitacins of three Cucumis sativus cultivars. J. Agric. Food Chem. 1981, 29, 194–196. [Google Scholar] [CrossRef]
- Balkema-Boomstra, A.G.; Zijlstra, S.; Verstappen, F.W.A.; Inggamer, H.; Mercke, P.E.; Jongsma, M.A.; Bouwmeester, H.J. Role of Cucurbitacin C in Resistance to Spider Mite (Tetranychus urticae) in Cucumber (Cucumis sativus L.). J. Chem. Ecol. 2003, 29, 225–235. [Google Scholar] [CrossRef]
- Shang, Y.; Ma, Y.; Zhou, Y.; Zhang, H.; Duan, L.; Chen, H.; Zeng, J.; Zhou, Q.; Wang, S.; Gu, W.; et al. Biosynthesis, regulation, and domestication of bitterness in cucumber. Science 2014, 346, 1084–1088. [Google Scholar] [CrossRef]
- Liu, Z.; Li, Y.; Cao, C.; Liang, S.; Ma, Y.; Liu, X.; Pei, Y. The role of H2S in low temperature-induced cucurbitacin C increases in cucumber. Plant Mol. Biol. 2019, 99, 535–544. [Google Scholar] [CrossRef]
- Xin, M.; Wang, L.; Liu, Y.; Feng, Z.; Zhou, X.; Qin, Z. Transcriptome profiling of cucumber genome expression in response to long-term low nitrogen stress. Acta Physiol. Plant. 2017, 39, 130. [Google Scholar] [CrossRef]
- Wang, M.; Jiang, B.; Peng, Q.; Liu, W.; He, X.; Liang, Z.; Lin, Y. Transcriptome Analyses in Different Cucumber Cultivars Provide Novel Insights into Drought Stress Responses. Int. J. Mol. Sci. 2018, 19, 2067. [Google Scholar] [CrossRef]
- Wang, M.; Chen, L.; Liang, Z.; He, X.; Liu, W.; Jiang, B.; Yan, J.; Sun, P.; Cao, Z.; Peng, Q.; et al. Metabolome and transcriptome analyses reveal chlorophyll and anthocyanin metabolism pathway associated with cucumber fruit skin color. BMC Plant Biol. 2020, 20, 386. [Google Scholar] [CrossRef]
- Liu, H.-C.; Liao, H.-T.; Charng, Y.-Y. The role of class A1 heat shock factors (HSFA1s) in response to heat and other stresses in Arabidopsis. Plant Cell Environ. 2011, 34, 738–751. [Google Scholar] [CrossRef]
- He, X.; Guo, S.; Wang, Y.; Wang, L.; Shu, S.; Sun, J. Systematic identification and analysis of heat-stress-responsive lncrnas, circrnas and mirnas with associated co-expression and Cerna networks in cucumber (Cucumis sativus L.). Physiol. Plant. 2019, 168, 736–754. [Google Scholar] [CrossRef]
- Ara, N.; Nakkanong, K.; Lv, W.; Yang, J.; Hu, Z.; Zhang, M. Antioxidant enzymatic activities and gene expression associated with heat tolerance in the stems and roots of two cucurbit species (“Cucurbita maxima” and “Cucurbita moschata”) and their interspecific inbred line “Maxchata”. Int. J. Mol. Sci. 2013, 14, 24008–24028. [Google Scholar] [CrossRef]
- Rehman, S.; Rashid, A.; Manzoor, M.A.; Li, L.; Sun, W.; Riaz, M.W.; Li, D.; Zhuge, Q. Genome-Wide Evolution and Comparative Analysis of Superoxide Dismutase Gene Family in Cucurbitaceae and Expression Analysis of Lagenaria siceraria Under Multiple Abiotic Stresses. Front. Genet. 2022, 12, 784878. [Google Scholar] [CrossRef]
- Yu, B.; Yan, S.; Zhou, H.; Dong, R.; Lei, J.; Chen, C.; Cao, B. Overexpression of CsCaM3 Improves High Temperature Tolerance in Cucumber. Front. Plant Sci. 2018, 9, 797. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, T.; Liu, Y.; Li, Y.; Wang, M.; Zhu, B.; Liao, D.; Yun, T.; Huang, W.; Zhang, W.; et al. Pumpkin (Cucurbita moschata) HSP20 gene family identification and expression under heat stress. Front. Genet. 2021, 12, 753953. [Google Scholar] [CrossRef]
- He, Y.; Fan, M.; Sun, Y.; Li, L. Genome-wide analysis of watermelon Hsp20s and their expression profiles and subcellular lo-cations under stresses. Int. J. Mol. Sci. 2018, 20, 12. [Google Scholar] [CrossRef]
- Wang, M.; Zhou, S.; Lu, J.; Xu, A.; Huang, Y.; Bie, Z.; Cheng, F. CmRCC1 Gene From Pumpkin Confers Cold Tolerance in Tobacco by Modulating Root Architecture and Photosynthetic Activity. Front. Plant Sci. 2021, 12, 2828. [Google Scholar] [CrossRef]
- Huang, J.; Hai, Z.; Wang, R.; Yu, Y.; Chen, X.; Liang, W.; Wang, H. Genome-wide analysis of HSP20 gene family and expression patterns under heat stress in cucumber (Cucumis sativus L.). Front. Plant Sci. 2022, 13, 968418. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Zhang, S.; Wei, S.; Liu, Y.; Li, C.; Bo, K.; Miao, H.; Gu, X.; Zhang, S. Identification of Quantitative Trait Loci Controlling High-Temperature Tolerance in Cucumber (Cucumis sativus L.) Seedlings. Plants 2020, 9, 1155. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Dong, S.; Wei, S.; Wang, W.; Miao, H.; Bo, K.; Gu, X.; Zhang, S. QTL mapping of heat tolerance in cucumber (Cucumis sativus L.) at adult stage. Plants 2021, 10, 324. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Huang, L.; Jiang, Q.; Zhu, C. MicroRNAs as Important Regulators of Heat Stress Responses in Plants. J. Agric. Food Chem. 2020, 68, 11320–11326. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, W.; Wang, M.; Zhang, H.-Y.; Liu, J.-H. The MIR396B of poncirus trifoliate functions in cold tolerance by regulating ACC oxidase gene expression and modulating ethylene–polyamine homeostasis. Plant Cell Physiol. 2016, 57, 1865–1878. [Google Scholar] [CrossRef]
- Pan, C.; Ye, L.; Zheng, Y.; Wang, Y.; Yang, D.; Liu, X.; Chen, L.; Zhang, Y.; Fei, Z.; Lu, G. Identification and expression profiling of microRNAs involved in the stigma exsertion under high-temperature stress in tomato. BMC Genom. 2017, 18, 843. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhang, S.; Chen, F.; Liu, B.; Wu, L.; Li, F.; Zhang, J.; Bao, M.; Liu, G. Genome-wide identification and characterization of the SBP-box gene family in Petunia. BMC Genom. 2018, 19, 193. [Google Scholar] [CrossRef]
- Ma, Y.; Guo, J.W.; Bade, R.; Men, Z.H.; Hasi, A. Genome-wide identification and phylogenetic analysis of the SBP-box gene family in melons. Genet. Mol. Res. 2014, 13, 8794–8806. [Google Scholar] [CrossRef]
- Kazan, K.; Lyons, R. The link between flowering time and stress tolerance. J. Exp. Bot. 2015, 67, 47–60. [Google Scholar] [CrossRef]
- May, P.; Liao, W.; Wu, Y.; Shuai, B.; Richard McCombie, W.; Zhang, M.Q.; Liu, Q.A. The effects of carbon dioxide and tem-perature on microrna expression in Arabidopsis development. Nat. Commun. 2013, 4, 2145. [Google Scholar] [CrossRef]
- Huo, H.; Wei, S.; Bradford, K.J. DELAY OF GERMINATION1 (DOG1) regulates both seed dormancy and flowering time through microRNA pathways. Proc. Natl. Acad. Sci. USA 2016, 113, E2199–E2206. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, F.; Cao, H.; Peng, H.; Ni, Z.; Sun, Q.; Yao, Y. TamiR159 Directed Wheat TaGAMYB Cleavage and Its Involvement in Anther Development and Heat Response. PLoS ONE 2012, 7, e48445. [Google Scholar] [CrossRef]
- Yang, X.; Wang, K.; Ge, L.; Chen, X.; Zhang, L.; Song, X. Transcription factor TaGAMYB from wheat (Triticum aestivum L.) regulates flowering time and fertility in transgenic Arabidopsis thaliana. Planta 2023, 257, 16. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; Wang, Z.; Guo, X.; Wang, F.; Xia, X.-J.; Zhou, J.; Shi, K.; Yu, J.-Q.; Zhou, Y.-H. Microarray and genetic analysis reveals that Csa-miR159b plays a critical role in abscisic acid-mediated heat tolerance in grafted cucumber plants. Plant Cell Environ. 2016, 39, 1790–1804. [Google Scholar] [CrossRef]
- Zhang, X.; Lai, Y.; Zhang, W.; Ahmad, J.; Qiu, Y.; Zhang, X.; Duan, M.; Liu, T.; Song, J.; Wang, H.; et al. MicroRNAs and their targets in cucumber shoot apices in response to temperature and photoperiod. BMC Genom. 2018, 19, 819. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aparna; Skarzyńska, A.; Pląder, W.; Pawełkowicz, M. Impact of Climate Change on Regulation of Genes Involved in Sex Determination and Fruit Production in Cucumber. Plants 2023, 12, 2651. https://doi.org/10.3390/plants12142651
Aparna, Skarzyńska A, Pląder W, Pawełkowicz M. Impact of Climate Change on Regulation of Genes Involved in Sex Determination and Fruit Production in Cucumber. Plants. 2023; 12(14):2651. https://doi.org/10.3390/plants12142651
Chicago/Turabian StyleAparna, Agnieszka Skarzyńska, Wojciech Pląder, and Magdalena Pawełkowicz. 2023. "Impact of Climate Change on Regulation of Genes Involved in Sex Determination and Fruit Production in Cucumber" Plants 12, no. 14: 2651. https://doi.org/10.3390/plants12142651
APA StyleAparna, Skarzyńska, A., Pląder, W., & Pawełkowicz, M. (2023). Impact of Climate Change on Regulation of Genes Involved in Sex Determination and Fruit Production in Cucumber. Plants, 12(14), 2651. https://doi.org/10.3390/plants12142651





