Overcoming the Challenges of Phytochemicals in Triple Negative Breast Cancer Therapy: The Path Forward
Abstract
1. Introduction
2. Risk Factors for Triple Negative Breast Cancer (TNBC)
2.1. Hereditary Factors
2.2. Environmental Risk Factors
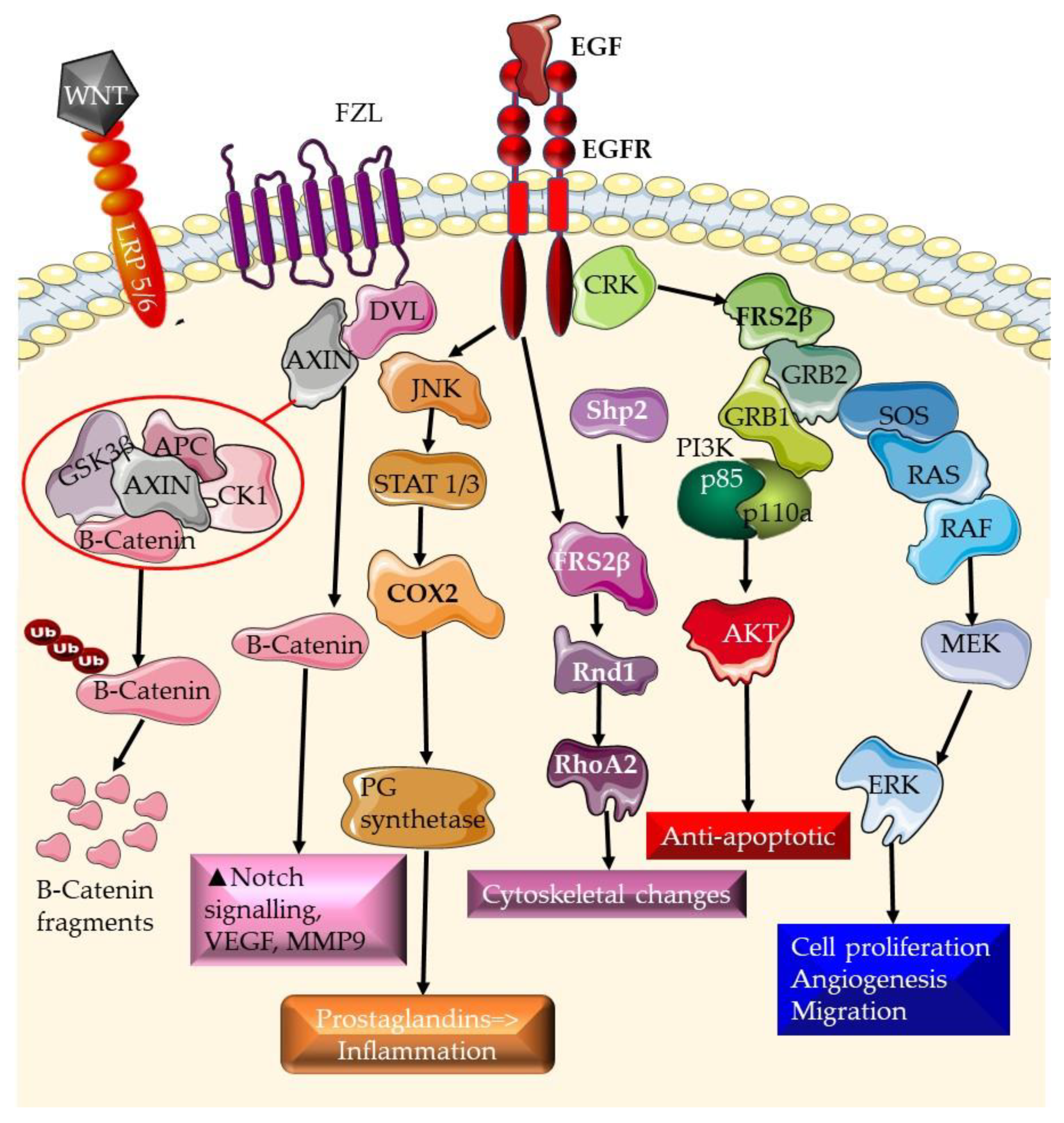
3. Triple Negative Breast Cancer (TNBC) Signaling Pathways
3.1. EGFR Pathway
3.2. Wnt/β-Catenin Pathway
3.3. Targeting Pathways Involved in Tissue Remodeling, Angiogenesis, and Metastasis in TNBC
3.4. Phytochemicals Serving as Antioxidants: Cancer Prevention
3.5. Induction of Apoptosis
4. Current Treatments for Triple Negative Breast Cancer: An Overview
5. Phytochemicals: An Emerging Therapeutic Option for TNBC
| Compound | Origin | Formula | Ref |
|---|---|---|---|
| Catechin | |||
| Epigallocatechin-3-O-gallate | Green Tea | C22H18O11 | [158] |
| Capsaicinoid | |||
| Capsaicin | Chili Peppers | C18H27NO3 | [162,163,164] |
| Dithiocarbamic Ester and an Indole Phytoalexin | |||
| Brassinin | Vegetables | C11H12N2S2 | [149] |
| Flavanoid | |||
| Hesperidin | Citrus Fruit | C28H34O15 | [156] |
| Hesperetin | C16H14O6 | ||
| Naringenin | C15H12O5 | ||
| Naringin | C27H32O14 | ||
| Nobiletin | C21H22O8 | ||
| Quercetin | Onion | C15H10O7 | [165,166] |
| Tangeretin | Citrus Fruit | C20H20O7 | [156] |
| Isoquinoline Aalkaloid) | |||
| Berberine | Berries | C20H18NO4 | [154] |
| emodin (1,3,8-trihydroxy-6-methylanthraquinone). | Rhubarb, Buckthorn, Japanese Knotweed | C15H10O5 | [155] |
| Phenols | |||
| Curcumin | Curcuma Longa | C21H20O6 | [143] |
| Stillbenoid | |||
| Resveratrol | Red Grapes | C14H12O3 | [152,153] |

6. Phytochemicals for TNBC: Limitations and Future Directions
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Carneiro, M.M. Reflections on Pink October. Women Health 2021, 61, 915–916. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Wang, P.; He, S.; Zhu, J.; Shi, Y.; Wang, J. Progress and prospect of immunotherapy for triple-negative breast cancer. Front. Oncol. 2022, 12, 919072. [Google Scholar] [CrossRef] [PubMed]
- Parkin, D.M.; Fernández, L.M. Use of statistics to assess the global burden of breast cancer. Breast J. 2006, 12, S70–S80. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.; Morgan, E.; Rumgay, H.; Mafra, A.; Singh, D.; Laversanne, M.; Vignat, J.; Gralow, J.R.; Cardoso, F.; Siesling, S. Current and future burden of breast cancer: Global statistics for 2020 and 2040. Breast 2022, 66, 15–23. [Google Scholar] [CrossRef]
- Cao, W.; Chen, H.-D.; Yu, Y.-W.; Li, N.; Chen, W.-Q. Changing profiles of cancer burden worldwide and in China: A secondary analysis of the global cancer statistics 2020. Chin. Med. J. 2021, 134, 783–791. [Google Scholar] [CrossRef]
- Kumar, P.; Aggarwal, R. An overview of triple-negative breast cancer. Arch. Gynecol. Obstet. 2016, 293, 247–269. [Google Scholar] [CrossRef]
- Boyle, P. Triple-negative breast cancer: Epidemiological considerations and recommendations. Ann. Oncol. 2012, 23, vi7–vi12. [Google Scholar] [CrossRef]
- Anders, C.; Carey, L.A. Understanding and treating triple-negative breast cancer. Oncology 2008, 22, 1233. [Google Scholar]
- Bosch, A.; Eroles, P.; Zaragoza, R.; Viña, J.R.; Lluch, A. Triple-negative breast cancer: Molecular features, pathogenesis, treatment and current lines of research. Cancer Treat. Rev. 2010, 36, 206–215. [Google Scholar] [CrossRef]
- Yi, D.; Xu, L.; Luo, J.; You, X.; Huang, T.; Zi, Y.; Li, X.; Wang, R.; Zhong, Z.; Tang, X. Germline TP53 and MSH6 mutations implicated in sporadic triple-negative breast cancer (TNBC): A preliminary study. Hum. Genom. 2019, 13, 4. [Google Scholar] [CrossRef]
- Dietze, E.C.; Sistrunk, C.; Miranda-Carboni, G.; O’regan, R.; Seewaldt, V.L. Triple-negative breast cancer in African-American women: Disparities versus biology. Nat. Rev. Cancer 2015, 15, 248–254. [Google Scholar] [CrossRef]
- Churpek, J.E.; Walsh, T.; Zheng, Y.; Moton, Z.; Thornton, A.M.; Lee, M.K.; Casadei, S.; Watts, A.; Neistadt, B.; Churpek, M.M. Inherited predisposition to breast cancer among African American women. Breast Cancer Res. Treat. 2015, 149, 31–39. [Google Scholar] [CrossRef]
- Jung, S.Y.; Kim, H.Y.; Nam, B.H.; Min, S.Y.; Lee, S.J.; Park, C.; Kwon, Y.; Kim, E.A.; Ko, K.L.; Shin, K.H.; et al. Worse prognosis of metaplastic breast cancer patients than other patients with triple-negative breast cancer. Breast Cancer Res. Treat. 2010, 120, 627–637. [Google Scholar] [CrossRef]
- Lara-Medina, F.; Pérez-Sánchez, V.; Saavedra-Pérez, D.; Blake-Cerda, M.; Arce, C.; Motola-Kuba, D.; Villarreal-Garza, C.; González-Angulo, A.M.; Bargalló, E.; Aguilar, J.L. Triple-negative breast cancer in Hispanic patients: High prevalence, poor prognosis, and association with menopausal status, body mass index, and parity. Cancer 2011, 117, 3658–3669. [Google Scholar] [CrossRef]
- Andre, F.; Zielinski, C. Optimal strategies for the treatment of metastatic triple-negative breast cancer with currently approved agents. Ann. Oncol. 2012, 23, vi46–vi51. [Google Scholar] [CrossRef]
- Isakoff, S.J. Triple negative breast cancer: Role of specific chemotherapy agents. Cancer J. 2010, 16, 53. [Google Scholar] [CrossRef]
- Joensuu, H.; Gligorov, J. Adjuvant treatments for triple-negative breast cancers. Ann. Oncol. 2012, 23, vi40–vi45. [Google Scholar] [CrossRef]
- Al-Mahmood, S.; Sapiezynski, J.; Garbuzenko, O.B.; Minko, T. Metastatic and triple-negative breast cancer: Challenges and treatment options. Drug Deliv. Transl. Res. 2018, 8, 1483–1507. [Google Scholar] [CrossRef]
- Yin, L.; Duan, J.-J.; Bian, X.-W.; Yu, S.-c. Triple-negative breast cancer molecular subtyping and treatment progress. Breast Cancer Res. 2020, 22, 61. [Google Scholar] [CrossRef]
- Robertson, L.; Hanson, H.; Seal, S.; Warren-Perry, M.; Hughes, D.; Howell, I.; Turnbull, C.; Houlston, R.; Shanley, S.; Butler, S. BRCA1 testing should be offered to individuals with triple-negative breast cancer diagnosed below 50 years. Br. J. Cancer 2012, 106, 1234–1238. [Google Scholar] [CrossRef]
- Tun, N.; Villani, G.; Ong, K.; Yoe, L.; Bo, Z. Risk of having BRCA1 mutation in high-risk women with triple-negative breast cancer: A meta-analysis. Clin. Genet. 2014, 85, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Mahfoudh, W.; Bettaieb, I.; Ghedira, R.; Snoussi, K.; Bouzid, N.; Klayech, Z.; Gabbouj, S.; Remadi, Y.; Hassen, E.; Bouaouina, N. Contribution of BRCA1 5382insC mutation in triple negative breast cancer in Tunisia. J. Transl. Med. 2019, 17, 123. [Google Scholar] [CrossRef] [PubMed]
- Sporikova, Z.; Koudelakova, V.; Trojanec, R.; Hajduch, M. Genetic markers in triple-negative breast cancer. Clin. Breast Cancer 2018, 18, e841–e850. [Google Scholar] [CrossRef] [PubMed]
- Kriegsmann, M.; Endris, V.; Wolf, T.; Pfarr, N.; Stenzinger, A.; Loibl, S.; Denkert, C.; Schneeweiss, A.; Budczies, J.; Sinn, P. Mutational profiles in triple-negative breast cancer defined by ultradeep multigene sequencing show high rates of PI3K pathway alterations and clinically relevant entity subgroup specific differences. Oncotarget 2014, 5, 9952. [Google Scholar] [CrossRef]
- Ciuro, J.; Beyer, A.; Fritzler, J.; Jackson, N.; Ahsan, S. Health care disparities and demand for expanding hereditary breast cancer screening guidelines in African Americans. Clin. Breast Cancer 2021, 21, e220–e227. [Google Scholar] [CrossRef]
- Lee, E.; McKean-Cowdin, R.; Ma, H.; Spicer, D.V.; Van Den Berg, D.; Bernstein, L.; Ursin, G. Characteristics of triple-negative breast cancer in patients with a BRCA1 mutation: Results from a population-based study of young women. J. Clin. Oncol. 2011, 29, 4373. [Google Scholar] [CrossRef]
- Siddharth, S.; Sharma, D. Racial disparity and triple-negative breast cancer in African-American women: A multifaceted affair between obesity, biology, and socioeconomic determinants. Cancers 2018, 10, 514. [Google Scholar] [CrossRef]
- Newman, L.A.; Kaljee, L.M. Health disparities and triple-negative breast cancer in African American women: A review. JAMA Surg. 2017, 152, 485–493. [Google Scholar] [CrossRef]
- Liedtke, C.; Rody, A.; Gluz, O.; Baumann, K.; Beyer, D.; Kohls, E.-B.; Lausen, K.; Hanker, L.; Holtrich, U.; Becker, S. The prognostic impact of age in different molecular subtypes of breast cancer. Breast Cancer Res. Treat. 2015, 152, 667–673. [Google Scholar] [CrossRef]
- Trevino, K.M.; Stern, A.; Prigerson, H.G. Adapting psychosocial interventions for older adults with cancer: A case example of Managing Anxiety from Cancer (MAC). J. Geriatr. Oncol. 2020, 11, 1319–1323. [Google Scholar] [CrossRef]
- Picon-Ruiz, M.; Morata-Tarifa, C.; Valle-Goffin, J.J.; Friedman, E.R.; Slingerland, J.M. Obesity and adverse breast cancer risk and outcome: Mechanistic insights and strategies for intervention. CA A Cancer J. Clin. 2017, 67, 378–397. [Google Scholar] [CrossRef]
- Rose, D.P.; Gracheck, P.J.; Vona-Davis, L. The interactions of obesity, inflammation and insulin resistance in breast cancer. Cancers 2015, 7, 2147–2168. [Google Scholar] [CrossRef]
- Salaudeen, A.L.; Banerjee, S. Network medicine-based analysis of association between gynecological cancers and metabolic and hormonal disorders. Appl. Biochem. Biotechnol. 2021, 194, 323–338. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Piperigkou, Z.; Karamanos, N.K.; Zolota, V. Altered Adipokine Expression in Tumor Microenvironment Promotes Development of Triple Negative Breast Cancer. Cancers 2022, 14, 4139. [Google Scholar] [CrossRef]
- Student, S.; Hejmo, T.; Poterała-Hejmo, A.; Leśniak, A.; Bułdak, R. Anti-androgen hormonal therapy for cancer and other diseases. Eur. J. Pharmacol. 2020, 866, 172783. [Google Scholar] [CrossRef]
- Jain, V.; Kumar, H.; Anod, H.V.; Chand, P.; Gupta, N.V.; Dey, S.; Kesharwani, S.S. A review of nanotechnology-based approaches for breast cancer and triple-negative breast cancer. J. Control. Release 2020, 326, 628–647. [Google Scholar] [CrossRef]
- Masuda, H.; Zhang, D.; Bartholomeusz, C.; Doihara, H.; Hortobagyi, G.N.; Ueno, N.T. Role of epidermal growth factor receptor in breast cancer. Breast Cancer Res. Treat. 2012, 136, 331–345. [Google Scholar] [CrossRef]
- You, K.S.; Yi, Y.W.; Cho, J.; Park, J.-S.; Seong, Y.-S. Potentiating therapeutic effects of epidermal growth factor receptor inhibition in triple-negative breast cancer. Pharmaceuticals 2021, 14, 589. [Google Scholar] [CrossRef]
- Khan, M.A.; Jain, V.K.; Rizwanullah, M.; Ahmad, J.; Jain, K. PI3K/AKT/mTOR pathway inhibitors in triple-negative breast cancer: A review on drug discovery and future challenges. Drug Discov. Today 2019, 24, 2181–2191. [Google Scholar] [CrossRef]
- Costa, R.L.; Han, H.S.; Gradishar, W.J. Targeting the PI3K/AKT/mTOR pathway in triple-negative breast cancer: A review. Breast Cancer Res. Treat. 2018, 169, 397–406. [Google Scholar] [CrossRef]
- Wang, X.; Song, H.; Fang, L.; Wu, T. EIF4A3-mediated circPRKCI expression promotes triple-negative breast cancer progression by regulating WBP2 and PI3K/AKT signaling pathway. Cell Death Discov. 2022, 8, 92. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Richmond, A.; Yan, C. Immunomodulatory Properties of PI3K/AKT/mTOR and MAPK/MEK/ERK Inhibition Augment Response to Immune Checkpoint Blockade in Melanoma and Triple-Negative Breast Cancer. Int. J. Mol. Sci. 2022, 23, 7353. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lu, Q.; Li, N.; Xu, M.; Miyamoto, T.; Liu, J. Sulforaphane suppresses metastasis of triple-negative breast cancer cells by targeting the RAF/MEK/ERK pathway. NPJ Breast Cancer 2022, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Koni, M.; Pinnarò, V.; Brizzi, M.F. The Wnt signalling pathway: A tailored target in cancer. Int. J. Mol. Sci. 2020, 21, 7697. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, N.; Panda, C.K. Wnt/β-catenin signaling pathway as chemotherapeutic target in breast cancer: An update on pros and cons. Clin. Breast Cancer 2020, 20, 361–370. [Google Scholar] [CrossRef]
- Mosalpuria, K.; Hall, C.; Krishnamurthy, S.; Lodhi, A.; Hallman, D.M.; Baraniuk, M.S.; Bhattacharyya, A.; Lucci, A. Cyclooxygenase-2 expression in non-metastatic triple-negative breast cancer patients. Mol. Clin. Oncol. 2014, 2, 845–850. [Google Scholar] [CrossRef]
- Alikanoglu, A.S.; Yildirim, M.; Suren, D.; Yildiz, M.; Kaya, V.; Dilli, U.D.; Sezer, C. Expression of cyclooxygenase-2 and Bcl-2 in breast cancer and their relationship with triple-negative disease. J. Balk. Union Oncol. 2014, 19, 430–434. [Google Scholar]
- Cháirez-Ramírez, M.H.; de la Cruz-López, K.G.; García-Carrancá, A. Polyphenols as Antitumor Agents Targeting Key Players in Cancer-Driving Signaling Pathways. Front. Pharmacol. 2021, 12, 710304. [Google Scholar] [CrossRef]
- Bublil, E.M.; Yarden, Y. The EGF receptor family: Spearheading a merger of signaling and therapeutics. Curr. Opin. Cell Biol. 2007, 19, 124–134. [Google Scholar] [CrossRef]
- Scaltriti, M.; Baselga, J. The epidermal growth factor receptor pathway: A model for targeted therapy. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2006, 12, 5268–5272. [Google Scholar] [CrossRef]
- Wieduwilt, M.J.; Moasser, M.M. The epidermal growth factor receptor family: Biology driving targeted therapeutics. Cell. Mol. Life Sci. CMLS 2008, 65, 1566–1584. [Google Scholar] [CrossRef]
- Wee, P.; Wang, Z. Epidermal growth factor receptor cell proliferation signaling pathways. Cancers 2017, 9, 52. [Google Scholar] [CrossRef]
- Koustas, E.; Karamouzis, M.V.; Mihailidou, C.; Schizas, D.; Papavassiliou, A.G. Co-targeting of EGFR and autophagy signaling is an emerging treatment strategy in metastatic colorectal cancer. Cancer Lett. 2017, 396, 94–102. [Google Scholar] [CrossRef]
- Cordover, E.; Minden, A. Signaling pathways downstream to receptor tyrosine kinases: Targets for cancer treatment. J. Cancer Metastasis Treat. 2020, 6, 45. [Google Scholar] [CrossRef]
- Vara, J.Á.F.; Casado, E.; de Castro, J.; Cejas, P.; Belda-Iniesta, C.; González-Barón, M. PI3K/Akt signalling pathway and cancer. Cancer Treat. Rev. 2004, 30, 193–204. [Google Scholar] [CrossRef]
- Zhang, X.; Jin, B.; Huang, C. The PI3K/Akt pathway and its downstream transcriptional factors as targets for chemoprevention. Curr. Cancer Drug Targets 2007, 7, 305–316. [Google Scholar] [CrossRef]
- Kolch, W. Meaningful relationships: The regulation of the Ras/Raf/MEK/ERK pathway by protein interactions. Biochem. J. 2000, 351, 289–305. [Google Scholar] [CrossRef]
- Chang, F.; Steelman, L.S.; Shelton, J.G.; Lee, J.T.; Navolanic, P.M.; Blalock, W.L.; Franklin, R.; McCubrey, J.A. Regulation of cell cycle progression and apoptosis by the Ras/Raf/MEK/ERK pathway. Int. J. Oncol. 2003, 22, 469–480. [Google Scholar]
- Basu, N.; Narad, P.; Guptasarma, M.L.; Tandon, C.; Das, B.C.; Tandon, S. Computational and In Vitro Approaches to Elucidate the Anti-cancer Effects of Arnica montana in Hormone-Dependent Breast Cancer. Homeopath. J. Fac. Homeopath. 2022, 111, 288–300. [Google Scholar] [CrossRef]
- Acharya, R.; Chacko, S.; Bose, P.; Lapenna, A.; Pattanayak, S.P. Structure Based Multitargeted Molecular Docking Analysis of Selected Furanocoumarins against Breast Cancer. Sci. Rep. 2019, 9, 15743. [Google Scholar] [CrossRef]
- Mahnashi, M.H.; Alqahtani, Y.S.; Alyami, B.A.; Alqarni, A.O.; Ullah, F.; Wadood, A.; Sadiq, A.; Shareef, A.; Ayaz, M. Cytotoxicity, anti-angiogenic, anti-tumor and molecular docking studies on phytochemicals isolated from Polygonum hydropiper L. BMC Complement. Med. Ther. 2021, 21, 239. [Google Scholar] [CrossRef] [PubMed]
- Shilpa, G.; Lakshmi, S.; Jamsheena, V.; Lankalapalli, R.S.; Prakash, V.; Anbumani, S.; Priya, S. Studies on the mode of action of synthetic diindolylmethane derivatives against triple negative breast cancer cells. Basic Clin. Pharmacol. Toxicol. 2022, 131, 224–240. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, M.; Huang, K.L.; Tang, C.K. 3,3′-Diindolylmethane (DIM) inhibits the growth and invasion of drug-resistant human cancer cells expressing EGFR mutants. Cancer Lett. 2010, 295, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Nasry, W.H.S.; Rodriguez-Lecompte, J.C.; Martin, C.K. Role of COX-2/PGE2 mediated inflammation in oral squamous cell carcinoma. Cancers 2018, 10, 348. [Google Scholar] [CrossRef]
- Sarkar, F.H.; Adsule, S.; Li, Y.; Padhye, S. Back to the future: COX-2 inhibitors for chemoprevention and cancer therapy. Mini Rev. Med. Chem. 2007, 7, 599–608. [Google Scholar] [CrossRef]
- Singh, T.; Katiyar, S.K. Honokiol, a phytochemical from Magnolia spp., inhibits breast cancer cell migration by targeting nitric oxide and cyclooxygenase-2. Int. J. Oncol. 2011, 38, 769–776. [Google Scholar] [CrossRef]
- Islam, M.A.; Zilani, M.N.H.; Biswas, P.; Khan, D.A.; Rahman, M.H.; Nahid, R.; Nahar, N.; Samad, A.; Ahammad, F.; Hasan, M.N. Evaluation of in vitro and in silico anti-inflammatory potential of some selected medicinal plants of Bangladesh against cyclooxygenase-II enzyme. J. Ethnopharmacol. 2022, 285, 114900. [Google Scholar] [CrossRef]
- Linus, L.O.; Wang, S.L.; Shi, N.; Hanson, C.; Lu, Y.T.; Alolga, R.N.; Liu, Q.; Njokuocha, R.C.; Qi, L.W. The new plant Parinari kerstingii Engl.: Toxicity studies and anti-inflammatory properties. J. Ethnopharmacol. 2018, 220, 26–34. [Google Scholar] [CrossRef]
- Galasso, S.; Pacifico, S.; Kretschmer, N.; Pan, S.P.; Marciano, S.; Piccolella, S.; Monaco, P.; Bauer, R. Influence of seasonal variation on Thymus longicaulis C. Presl chemical composition and its antioxidant and anti-inflammatory properties. Phytochemistry 2014, 107, 80–90. [Google Scholar] [CrossRef]
- Teo, J.-L.; Kahn, M. The Wnt signaling pathway in cellular proliferation and differentiation: A tale of two coactivators. Adv. Drug Deliv. Rev. 2010, 62, 1149–1155. [Google Scholar] [CrossRef]
- Logan, C.Y.; Nusse, R. The Wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol. 2004, 20, 781–810. [Google Scholar] [CrossRef]
- Reya, T.; Clevers, H. Wnt signalling in stem cells and cancer. Nature 2005, 434, 843–850. [Google Scholar] [CrossRef]
- Nusse, R. Wnt signaling in disease and in development. Cell Res. 2005, 15, 28–32. [Google Scholar] [CrossRef]
- Chien, A.J.; Conrad, W.H.; Moon, R.T. A Wnt survival guide: From flies to human disease. J. Investig. Dermatol. 2009, 129, 1614–1627. [Google Scholar] [CrossRef]
- Van Amerongen, R.; Mikels, A.; Nusse, R. Alternative wnt signaling is initiated by distinct receptors. Sci. Signal. 2008, 1, re9. [Google Scholar] [CrossRef]
- Behrens, J.; Von Kries, J.P.; Kühl, M.; Bruhn, L.; Wedlich, D.; Grosschedl, R.; Birchmeier, W. Functional interaction of β-catenin with the transcription factor LEF-1. Nature 1996, 382, 638–642. [Google Scholar] [CrossRef]
- Molenaar, M.; Van De Wetering, M.; Oosterwegel, M.; Peterson-Maduro, J.; Godsave, S.; Korinek, V.; Roose, J.; Destrée, O.; Clevers, H. XTcf-3 transcription factor mediates β-catenin-induced axis formation in Xenopus embryos. Cell 1996, 86, 391–399. [Google Scholar] [CrossRef]
- Manandhar, S.; Kabekkodu, S.P.; Pai, K.S.R. Aberrant canonical Wnt signaling: Phytochemical based modulation. Phytomedicine Int. J. Phytother. Phytopharm. 2020, 76, 153243. [Google Scholar] [CrossRef]
- Bae, S.-Y.; Kim, H.J.; Lee, K.-J.; Lee, K. Translationally controlled tumor protein induces epithelial to mesenchymal transition and promotes cell migration, invasion and metastasis. Sci. Rep. 2015, 5, 8061. [Google Scholar] [CrossRef]
- Lemieux, É.; Bergeron, S.; Durand, V.; Asselin, C.; Saucier, C.; Rivard, N. Constitutively active MEK1 is sufficient to induce epithelial-to-mesenchymal transition in intestinal epithelial cells and to promote tumor invasion and metastasis. Int. J. Cancer 2009, 125, 1575–1586. [Google Scholar] [CrossRef]
- Pearson, G.W. Control of invasion by epithelial-to-mesenchymal transition programs during metastasis. J. Clin. Med. 2019, 8, 646. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Sinha, M.; Luxon, B.A.; Bresnick, A.R.; O’Connor, K.L. Integrin α6β4 controls the expression of genes associated with cell motility, invasion, and metastasis, including S100A4/metastasin. J. Biol. Chem. 2009, 284, 1484–1494. [Google Scholar] [CrossRef] [PubMed]
- Canel, M.; Serrels, A.; Frame, M.C.; Brunton, V.G. E-cadherin–integrin crosstalk in cancer invasion and metastasis. J. Cell Sci. 2013, 126, 393–401. [Google Scholar] [CrossRef]
- Stetler-Stevenson, W.G.; Liotta, L.A.; Kleiner, D.E., Jr. Extracellular matrix 6: Role of matrix metalloproteinases in tumor invasion and metastasis. FASEB J. 1993, 7, 1434–1441. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.D.; Haridas, N.; Patel, J.B.; Shah, F.D.; Shukla, S.N.; Shah, P.M.; Patel, P.S. Matrix metalloproteinases and their inhibitors: Correlation with invasion and metastasis in oral cancer. Indian J. Clin. Biochem. 2010, 25, 250–259. [Google Scholar] [CrossRef]
- Stuelten, C.H.; Parent, C.A.; Montell, D.J. Cell motility in cancer invasion and metastasis: Insights from simple model organisms. Nat. Rev. Cancer 2018, 18, 296–312. [Google Scholar] [CrossRef]
- Tanaka, T.; Bai, Z.; Srinoulprasert, Y.; Yang, B.; Hayasaka, H.; Miyasaka, M. Chemokines in tumor progression and metastasis. Cancer Sci. 2005, 96, 317–322. [Google Scholar] [CrossRef]
- Karnoub, A.E.; Weinberg, R.A. Chemokine networks and breast cancer metastasis. Breast Dis. 2007, 26, 75–85. [Google Scholar] [CrossRef]
- Pavlakovic, H.; Havers, W.; Schweigerer, L. Multiple angiogenesis stimulators in a single malignancy: Implications for anti-angiogenic tumour therapy. Angiogenesis 2001, 4, 259–262. [Google Scholar] [CrossRef]
- Ranieri, G.; Gasparini, G. Angiogenesis and angiogenesis inhibitors: A new potential anticancer therapeutic strategy. Curr. Drug Targets. Immune Endocr. Metab. Disord. 2001, 1, 241–253. [Google Scholar] [CrossRef]
- Napoleone, E.; di Santo, A.; Peri, G.; Mantovani, A.; de Gaetano, G.; Donati, M.B.; Lorenzet, R. The long pentraxin PTX3 up-regulates tissue factor in activated monocytes: Another link between inflammation and clotting activation. J. Leukoc. Biol. 2004, 76, 203–209. [Google Scholar] [CrossRef]
- Iruela-Arispe, M.L.; Dvorak, H.F. Angiogenesis: A dynamic balance of stimulators and inhibitors. Thromb. Haemost. 1997, 78, 672–677. [Google Scholar] [CrossRef]
- Mousa, L.; Salem, M.E.; Mikhail, S. Biomarkers of Angiogenesis in Colorectal Cancer. Biomark. Cancer 2015, 7, 13–19. [Google Scholar] [CrossRef]
- Ali, S.H.; O’Donnell, A.L.; Balu, D.; Pohl, M.B.; Seyler, M.J.; Mohamed, S.; Mousa, S.; Dandona, P. Estrogen receptor-alpha in the inhibition of cancer growth and angiogenesis. Cancer Res. 2000, 60, 7094–7098. [Google Scholar]
- Kerbel, R.S. Tumor angiogenesis. N. Engl. J. Med. 2008, 358, 2039–2049. [Google Scholar] [CrossRef]
- Christiansen, J.J.; Rajasekaran, A.K. Reassessing epithelial to mesenchymal transition as a prerequisite for carcinoma invasion and metastasis. Cancer Res. 2006, 66, 8319–8326. [Google Scholar] [CrossRef]
- Hseu, Y.-C.; Lin, Y.-C.; Rajendran, P.; Thigarajan, V.; Mathew, D.C.; Lin, K.-Y.; Way, T.-D.; Liao, J.-W.; Yang, H.-L. Antrodia salmonea suppresses invasion and metastasis in triple-negative breast cancer cells by reversing EMT through the NF-κB and Wnt/β-catenin signaling pathway. Food Chem. Toxicol. 2019, 124, 219–230. [Google Scholar] [CrossRef]
- Zhang, F.-L.; Cao, J.-L.; Xie, H.-Y.; Sun, R.; Yang, L.-F.; Shao, Z.-M.; Li, D.-Q. Cancer-associated MORC2-mutant M276I regulates an hnRNPM-mediated CD44 splicing switch to promote invasion and metastasis in triple-negative breast cancer. Cancer Res. 2018, 78, 5780–5792. [Google Scholar] [CrossRef]
- Menter, D.G.; DuBois, R.N. Prostaglandins in cancer cell adhesion, migration, and invasion. Int. J. Cell Biol. 2012, 2012, 723419. [Google Scholar] [CrossRef]
- Mythreye, K.; Blobe, G.C. Proteoglycan signaling co-receptors: Roles in cell adhesion, migration and invasion. Cell. Signal. 2009, 21, 1548–1558. [Google Scholar] [CrossRef]
- Hamidi, H.; Ivaska, J. Every step of the way: Integrins in cancer progression and metastasis. Nat. Rev. Cancer 2018, 18, 533–548. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, C.; Zhao, C.; Zhai, L.; Lv, S. Downregulation of β3 integrin by miR-30a-5p modulates cell adhesion and invasion by interrupting Erk/Ets-1 network in triple-negative breast cancer Corrigendum in/10.3892/ijo. 2021.5169. Int. J. Oncol. 2016, 48, 1155–1164. [Google Scholar] [CrossRef]
- Chen, J.s.; Wang, Q.; Fu, X.h.; Huang, X.H.; Chen, X.l.; Cao, L.q.; Chen, L.z.; Tan, H.x.; Li, W.; Bi, J. Involvement of PI3K/PTEN/AKT/mTOR pathway in invasion and metastasis in hepatocellular carcinoma: Association with MMP-9. Hepatol. Res. 2009, 39, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Westermarck, J.; Kähäri, V.M. Regulation of matrix metalloproteinase expression in tumor invasion. FASEB J. 1999, 13, 781–792. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Xiao, F.; Ding, P.; Qin, H.; Huang, R. Long noncoding RNA highly up-regulated in liver cancer predicts unfavorable outcome and regulates metastasis by MMPs in triple-negative breast cancer. Arch. Med. Res. 2016, 47, 446–453. [Google Scholar] [CrossRef]
- Zhao, S.; Ma, W.; Zhang, M.; Tang, D.; Shi, Q.; Xu, S.; Zhang, X.; Liu, Y.; Song, Y.; Liu, L. High expression of CD147 and MMP-9 is correlated with poor prognosis of triple-negative breast cancer (TNBC) patients. Med. Oncol. 2013, 30, 335. [Google Scholar] [CrossRef]
- Li, X.; Xiang, Y.; Li, F.; Yin, C.; Li, B.; Ke, X. WNT/β-catenin signaling pathway regulating T cell-inflammation in the tumor microenvironment. Front. Immunol. 2019, 10, 2293. [Google Scholar] [CrossRef]
- Chow, M.T.; Luster, A.D. Chemokines in cancer. Cancer Immunol. Res. 2014, 2, 1125–1131. [Google Scholar] [CrossRef]
- Kulbe, H.; Levinson, N.R.; Balkwill, F.; Wilson, J.L. The chemokine network in cancer-much more than directing cell movement. Int. J. Dev. Biol. 2004, 48, 489–496. [Google Scholar] [CrossRef]
- Billard, M.J.; Fitzhugh, D.J.; Parker, J.S.; Brozowski, J.M.; McGinnis, M.W.; Timoshchenko, R.G.; Serafin, D.S.; Lininger, R.; Klauber-Demore, N.; Sahagian, G. G protein coupled receptor kinase 3 regulates breast cancer migration, invasion, and metastasis. PLoS ONE 2016, 11, e0152856. [Google Scholar] [CrossRef]
- Liubomirski, Y.; Lerrer, S.; Meshel, T.; Rubinstein-Achiasaf, L.; Morein, D.; Wiemann, S.; Körner, C.; Ben-Baruch, A. Tumor-stroma-inflammation networks promote pro-metastatic chemokines and aggressiveness characteristics in triple-negative breast cancer. Front. Immunol. 2019, 10, 757. [Google Scholar] [CrossRef]
- Mukherji, B.; MacAlister, T.J.; Guha, A.; Gillies, C.G.; Jeffers, D.C.; Slocum, S.K. Spontaneous in vitro transformation of human fibroblasts. J. Natl. Cancer Inst. 1984, 73, 583–593. [Google Scholar]
- Dreher, D.; Junod, A.F. Role of oxygen free radicals in cancer development. Eur. J. Cancer 1996, 32a, 30–38. [Google Scholar] [CrossRef]
- Marshall, H.E.; Merchant, K.; Stamler, J.S. Nitrosation and oxidation in the regulation of gene expression. FASEB J. 2000, 14, 1889–1900. [Google Scholar] [CrossRef]
- Esposito, F.; Ammendola, R.; Faraonio, R.; Russo, T.; Cimino, F.J.N.r. Redox control of signal transduction, gene expression and cellular senescence. Neurochem. Res. 2004, 29, 617–628. [Google Scholar] [CrossRef]
- Srinivas, U.S.; Tan, B.W.Q.; Vellayappan, B.A.; Jeyasekharan, A.D. ROS and the DNA damage response in cancer. Redox Biol. 2019, 25, 101084. [Google Scholar] [CrossRef]
- Gupta, N.; Verma, K.; Nalla, S.; Kulshreshtha, A.; Lall, R.; Prasad, S. Free Radicals as a Double-Edged Sword: The Cancer Preventive and Therapeutic Roles of Curcumin. Molecules 2020, 25, 5390. [Google Scholar] [CrossRef]
- Dimitrios, B. Sources of natural phenolic antioxidants. Trends Food Sci. Technol. 2006, 17, 505–512. [Google Scholar] [CrossRef]
- Chirumbolo, S.; Bjørklund, G.; Lysiuk, R.; Vella, A.; Lenchyk, L.; Upyr, T. Targeting Cancer with Phytochemicals via Their Fine Tuning of the Cell Survival Signaling Pathways. Int. J. Mol. Sci. 2018, 19, 3568. [Google Scholar] [CrossRef]
- Albensi, B.C. What Is Nuclear Factor Kappa B (NF-κB) Doing in and to the Mitochondrion? Front. Cell Dev. Biol. 2019, 7, 154. [Google Scholar] [CrossRef]
- Mohan, S.; Abdelwahab, S.I.; Kamalidehghan, B.; Syam, S.; May, K.S.; Harmal, N.S.M.; Shafifiyaz, N.; Hadi, A.H.A.; Hashim, N.M.; Rahmani, M. Involvement of NF-κB and Bcl2/Bax signaling pathways in the apoptosis of MCF7 cells induced by a xanthone compound Pyranocycloartobiloxanthone A. Phytomedicine 2012, 19, 1007–1015. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Deng, X.; Yu, C.; Zhao, G.; Zhou, J.; Zhang, G.; Li, M.; Jiang, D.; Quan, Z.; Zhang, Y. Synergistic inhibitory effects of capsaicin combined with cisplatin on human osteosarcoma in culture and in xenografts. J. Exp. Clin. Cancer Res. CR 2018, 37, 251. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Colgate, E.C.; Miranda, C.L.; Stevens, J.F.; Bray, T.M.; Ho, E. Xanthohumol, a prenylflavonoid derived from hops induces apoptosis and inhibits NF-kappaB activation in prostate epithelial cells. Cancer Lett. 2007, 246, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.Y.; Yu, C.C.; Huang, C.C.; Liao, Y.W.; Hsieh, P.L.; Chu, P.M.; Yu, C.H.; Lin, S.S. Magnolol inhibits cancer stemness and IL-6/Stat3 signaling in oral carcinomas. J. Formos. Med. Assoc. Taiwan Yi Zhi 2022, 121, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Luo, J.; Luo, X.; Jia, W.; Fang, Z.; Yi, S.; Li, L. Morusin exerts anti-cancer activity in renal cell carcinoma by disturbing MAPK signaling pathways. Ann. Transl. Med. 2020, 8, 327. [Google Scholar] [CrossRef]
- Khwaza, V.; Oyedeji, O.O.; Aderibigbe, B.A. Ursolic Acid-Based Derivatives as Potential Anti-Cancer Agents: An Update. Int. J. Mol. Sci. 2020, 21, 5920. [Google Scholar] [CrossRef]
- Gambari, R.; Borgatti, M.; Lampronti, I.; Fabbri, E.; Brognara, E.; Bianchi, N.; Piccagli, L.; Yuen, M.C.; Kan, C.W.; Hau, D.K.; et al. Corilagin is a potent inhibitor of NF-kappaB activity and downregulates TNF-alpha induced expression of IL-8 gene in cystic fibrosis IB3-1 cells. Int. Immunopharmacol. 2012, 13, 308–315. [Google Scholar] [CrossRef]
- Parker, C.C.; Ampil, F.; Burton, G.; Li, B.D.; Chu, Q.D. Is breast conservation therapy a viable option for patients with triple-receptor negative breast cancer? Surgery 2010, 148, 386–391. [Google Scholar] [CrossRef]
- O’connell, M.J.; Martenson, J.A.; Wieand, H.S.; Krook, J.E.; Macdonald, J.S.; Haller, D.G.; Mayer, R.J.; Gunderson, L.L.; Rich, T.A. Improving adjuvant therapy for rectal cancer by combining protracted-infusion fluorouracil with radiation therapy after curative surgery. N. Engl. J. Med. 1994, 331, 502–507. [Google Scholar] [CrossRef]
- Norton, L. Theoretical concepts and the emerging role of taxanes in adjuvant therapy. Oncol. 2001, 6, 30–35. [Google Scholar] [CrossRef]
- Lynce, F.; Nunes, R. Role of platinums in triple-negative breast cancer. Curr. Oncol. Rep. 2021, 23, 50. [Google Scholar] [CrossRef]
- McClendon, A.K.; Dean, J.L.; Rivadeneira, D.B.; Yu, J.E.; Reed, C.A.; Gao, E.; Farber, J.L.; Force, T.; Koch, W.J.; Knudsen, E.S. CDK4/6 inhibition antagonizes the cytotoxic response to anthracycline therapy. Cell Cycle 2012, 11, 2747–2755. [Google Scholar] [CrossRef]
- Abbas, Z.; Rehman, S. An overview of cancer treatment modalities. Neoplasm 2018, 1, 139–157. [Google Scholar]
- Marra, A.; Viale, G.; Curigliano, G. Recent advances in triple negative breast cancer: The immunotherapy era. BMC Med. 2019, 17, 90. [Google Scholar] [CrossRef]
- Rupp, T.; Genest, L.; Babin, D.; Legrand, C.; Hunault, M.; Froget, G.; Castagné, V. Anti-CTLA-4 and anti-PD-1 immunotherapies repress tumor progression in preclinical breast and colon model with independent regulatory T cells response. Transl. Oncol. 2022, 20, 101405. [Google Scholar] [CrossRef]
- Singh, S.; Numan, A.; Maddiboyina, B.; Arora, S.; Riadi, Y.; Md, S.; Alhakamy, N.A.; Kesharwani, P. The emerging role of immune checkpoint inhibitors in the treatment of triple-negative breast cancer. Drug Discov. Today 2021, 26, 1721–1727. [Google Scholar] [CrossRef]
- Malhotra, J.; Jabbour, S.K.; Aisner, J. Current state of immunotherapy for non-small cell lung cancer. Transl. Lung Cancer Res. 2017, 6, 196. [Google Scholar] [CrossRef]
- Muthiah, I.; Rajendran, K.; Dhanaraj, P. In silico molecular docking and physicochemical property studies on effective phytochemicals targeting GPR116 for breast cancer treatment. Mol. Cell. Biochem. 2021, 476, 883–896. [Google Scholar] [CrossRef]
- Shukla, S.; Mehta, A. Anticancer potential of medicinal plants and their phytochemicals: A review. Braz. J. Bot. 2015, 38, 199–210. [Google Scholar] [CrossRef]
- Dandawate, P.R.; Subramaniam, D.; Jensen, R.A.; Anant, S. Targeting cancer stem cells and signaling pathways by phytochemicals: Novel approach for breast cancer therapy. In Proceedings of the Seminars in Cancer Biology; Academic Press: Cambridge, MA, USA, 2016; pp. 192–208. [Google Scholar]
- Wahle, K.W.; Brown, I.; Rotondo, D.; Heys, S.D. Plant phenolics in the prevention and treatment of cancer. Bio-Farms Nutraceuticals Funct. Food Saf. Control. Biosens. 2010, 698, 36–51. [Google Scholar]
- Farnood, P.R.; Pazhooh, R.D.; Asemi, Z.; Yousefi, B. Targeting Signaling Pathway by Curcumin in Osteosarcoma. Curr. Mol. Pharmacol. 2023, 16, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Ashrafizadeh, M.; Ahmadi, Z.; Mohamamdinejad, R.; Yaribeygi, H.; Serban, M.C.; Orafai, H.M.; Sahebkar, A. Curcumin Therapeutic Modulation of the Wnt Signaling Pathway. Curr. Pharm. Biotechnol. 2020, 21, 1006–1015. [Google Scholar] [CrossRef] [PubMed]
- Farghadani, R.; Naidu, R. Curcumin: Modulator of Key Molecular Signaling Pathways in Hormone-Independent Breast Cancer. Cancers 2021, 13, 3427. [Google Scholar] [CrossRef] [PubMed]
- Bimonte, S.; Barbieri, A.; Palma, G.; Rea, D.; Luciano, A.; D’Aiuto, M.; Arra, C.; Izzo, F. Dissecting the role of curcumin in tumour growth and angiogenesis in mouse model of human breast cancer. BioMed Res. Int. 2015, 2015, 878134. [Google Scholar] [CrossRef]
- Nagaraju, G.P.; Aliya, S.; Zafar, S.F.; Basha, R.; Diaz, R.; El-Rayes, B.F. The impact of curcumin on breast cancer. Integr. Biol. 2012, 4, 996–1007. [Google Scholar] [CrossRef]
- Labbozzetta, M.; Notarbartolo, M.; Poma, P.; Maurici, A.; Inguglia, L.; Marchetti, P.; Rizzi, M.; Baruchello, R.; Simoni, D.; D’Alessandro, N. Curcumin as a possible lead compound against hormone-independent, multidrug-resistant breast cancer. Ann. New York Acad. Sci. 2009, 1155, 278–283. [Google Scholar] [CrossRef]
- Gu, Y.; Becker, V.; Qiu, M.; Tang, T.; Ampofo, E.; Menger, M.D.; Laschke, M.W. Brassinin Promotes the Degradation of Tie2 and FGFR1 in Endothelial Cells and Inhibits Triple-Negative Breast Cancer Angiogenesis. Cancers 2022, 14, 3540. [Google Scholar] [CrossRef]
- Shukla, Y.; Singh, R. Resveratrol and cellular mechanisms of cancer prevention. Ann. N. Y. Acad. Sci. 2011, 1215, 1–8. [Google Scholar] [CrossRef]
- Nicolini, G.; Rigolio, R.; Miloso, M.; Bertelli, A.A.; Tredici, G. Anti-apoptotic effect of trans-resveratrol on paclitaxel-induced apoptosis in the human neuroblastoma SH-SY5Y cell line. Neurosci. Lett. 2001, 302, 41–44. [Google Scholar] [CrossRef]
- Kim, D.H.; Sung, B.; Kim, J.-A.; Kang, Y.J.; Hwang, S.Y.; Hwang, N.-L.; Suh, H.; Choi, Y.H.; Im, E.; Chung, H.Y. HS-1793, a resveratrol analogue, downregulates the expression of hypoxia-induced HIF-1 and VEGF and inhibits tumor growth of human breast cancer cells in a nude mouse xenograft model. Int. J. Oncol. 2017, 51, 715–723. [Google Scholar] [CrossRef]
- Gali-Muhtasib, H.; Hmadi, R.; Kareh, M.; Tohme, R.; Darwiche, N. Cell death mechanisms of plant-derived anticancer drugs: Beyond apoptosis. Apoptosis 2015, 20, 1531–1562. [Google Scholar] [CrossRef]
- Albring, K.F.; Weidemüller, J.; Mittag, S.; Weiske, J.; Friedrich, K.; Geroni, M.C.; Lombardi, P.; Huber, O. Berberine acts as a natural inhibitor of Wnt/β-catenin signaling--identification of more active 13-arylalkyl derivatives. BioFactors 2013, 39, 652–662. [Google Scholar] [CrossRef]
- Pooja, T.; Karunagaran, D. Emodin suppresses Wnt signaling in human colorectal cancer cells SW480 and SW620. Eur. J. Pharmacol. 2014, 742, 55–64. [Google Scholar] [CrossRef]
- Chen, H.; Liu, R.H. Potential Mechanisms of Action of Dietary Phytochemicals for Cancer Prevention by Targeting Cellular Signaling Transduction Pathways. J. Agric. Food Chem. 2018, 66, 3260–3276. [Google Scholar] [CrossRef]
- Gandhi, G.R.; Vasconcelos, A.B.S.; Wu, D.T.; Li, H.B.; Antony, P.J.; Li, H.; Geng, F.; Gurgel, R.Q.; Narain, N.; Gan, R.Y. Citrus Flavonoids as Promising Phytochemicals Targeting Diabetes and Related Complications: A Systematic Review of In Vitro and In Vivo Studies. Nutrients 2020, 12, 2907. [Google Scholar] [CrossRef]
- Wang, Y.-C.; Bachrach, U. The specific anti-cancer activity of green tea (−)-epigallocatechin-3-gallate (EGCG). Amino Acids 2002, 22, 131–143. [Google Scholar] [CrossRef]
- Braicu, C.; Gherman, C.D.; Irimie, A.; Berindan-Neagoe, I. Epigallocatechin-3-Gallate (EGCG) inhibits cell proliferation and migratory behaviour of triple negative breast cancer cells. J. Nanosci. Nanotechnol. 2013, 13, 632–637. [Google Scholar] [CrossRef]
- Bimonte, S.; Cascella, M.; Barbieri, A.; Arra, C.; Cuomo, A. Current shreds of evidence on the anticancer role of EGCG in triple negative breast cancer: An update of the current state of knowledge. Infect. Agents Cancer 2020, 15, 2. [Google Scholar] [CrossRef]
- Bimonte, S.; Cascella, M.; Barbieri, A.; Arra, C.; Cuomo, A. Shining a light on the effects of the combination of (–)-epigallocatechin-3-gallate and tapentadol on the growth of human triple-negative breast cancer cells. Vivo 2019, 33, 1463–1468. [Google Scholar] [CrossRef]
- Lu, M.; Chen, C.; Lan, Y.; Xiao, J.; Li, R.; Huang, J.; Huang, Q.; Cao, Y.; Ho, C.-T. Capsaicin—The major bioactive ingredient of chili peppers: Bio-efficacy and delivery systems. Food Funct. 2020, 11, 2848–2860. [Google Scholar] [CrossRef] [PubMed]
- Bencze, N.; Schvarcz, C.; Kriszta, G.; Danics, L.; Szőke, É.; Balogh, P.; Szállási, Á.; Hamar, P.; Helyes, Z.; Botz, B. Desensitization of capsaicin-sensitive afferents accelerates early tumor growth via increased vascular leakage in a murine model of triple negative breast cancer. Front. Oncol. 2021, 11, 685297. [Google Scholar] [CrossRef] [PubMed]
- Szallasi, A. Capsaicin and cancer: Guilty as charged or innocent until proven guilty? Temperature 2022, 10, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Umar, S.M.; Patra, S.; Kashyap, A.; Dev JR, A.; Kumar, L.; Prasad, C.P. Quercetin impairs HuR-driven progression and migration of triple negative breast cancer (TNBC) cells. Nutr. Cancer 2022, 74, 1497–1510. [Google Scholar] [CrossRef]
- Sultan, A.S.; Khalil, M.I.; Sami, B.M.; Alkhuriji, A.F.; Sadek, O. Quercetin induces apoptosis in triple-negative breast cancer cells via inhibiting fatty acid synthase and β-catenin. Int. J. Clin. Exp. Pathol. 2017, 10, 156–172. [Google Scholar]
- Mondal, P.; Natesh, J.; Penta, D.; Meeran, S.M. Progress and promises of epigenetic drugs and epigenetic diets in cancer prevention and therapy: A clinical update. In Proceedings of the Seminars in Cancer Biology; Academic Press: Cambridge, MA, USA, 2022; pp. 503–522. [Google Scholar]
- Braakhuis, A.J.; Campion, P.; Bishop, K.S. Reducing breast cancer recurrence: The role of dietary polyphenolics. Nutrients 2016, 8, 547. [Google Scholar] [CrossRef]
- Solanki, R.; Jodha, B.; Prabina, K.E.; Aggarwal, N.; Patel, S. Recent advances in phytochemical based nano-drug delivery systems to combat breast cancer: A review. J. Drug Deliv. Sci. Technol. 2022, 77, 103832. [Google Scholar] [CrossRef]
- Bhaskara, V.K.; Mittal, B.; Mysorekar, V.V.; Amaresh, N.; Simal-Gandara, J. Resveratrol, cancer and cancer stem cells: A review on past to future. Curr. Res. Food Sci. 2020, 3, 284–295. [Google Scholar] [CrossRef]
- Alissa, E.M. Medicinal herbs and therapeutic drugs interactions. Ther. Drug Monit. 2014, 36, 413–422. [Google Scholar] [CrossRef]
- Bandaranayake, W.M. Quality control, screening, toxicity, and regulation of herbal drugs. Mod. Phytomedicine Turn. Med. Plants Into Drugs 2006, 25–57. [Google Scholar]
- Colalto, C. Herbal interactions on absorption of drugs: Mechanisms of action and clinical risk assessment. Pharmacol. Res. 2010, 62, 207–227. [Google Scholar] [CrossRef]
- Liebelt, D.J.; Jordan, J.T.; Doherty, C.J. Only a matter of time: The impact of daily and seasonal rhythms on phytochemicals. Phytochem. Rev. 2019, 18, 1409–1433. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alaouna, M.; Penny, C.; Hull, R.; Molefi, T.; Chauke-Malinga, N.; Khanyile, R.; Makgoka, M.; Bida, M.; Dlamini, Z. Overcoming the Challenges of Phytochemicals in Triple Negative Breast Cancer Therapy: The Path Forward. Plants 2023, 12, 2350. https://doi.org/10.3390/plants12122350
Alaouna M, Penny C, Hull R, Molefi T, Chauke-Malinga N, Khanyile R, Makgoka M, Bida M, Dlamini Z. Overcoming the Challenges of Phytochemicals in Triple Negative Breast Cancer Therapy: The Path Forward. Plants. 2023; 12(12):2350. https://doi.org/10.3390/plants12122350
Chicago/Turabian StyleAlaouna, Mohammed, Clement Penny, Rodney Hull, Thulo Molefi, Nkhensani Chauke-Malinga, Richard Khanyile, Malose Makgoka, Meshack Bida, and Zodwa Dlamini. 2023. "Overcoming the Challenges of Phytochemicals in Triple Negative Breast Cancer Therapy: The Path Forward" Plants 12, no. 12: 2350. https://doi.org/10.3390/plants12122350
APA StyleAlaouna, M., Penny, C., Hull, R., Molefi, T., Chauke-Malinga, N., Khanyile, R., Makgoka, M., Bida, M., & Dlamini, Z. (2023). Overcoming the Challenges of Phytochemicals in Triple Negative Breast Cancer Therapy: The Path Forward. Plants, 12(12), 2350. https://doi.org/10.3390/plants12122350






