Comparative Study of Phytochemistry, Antioxidant and Biological Activities of Berberis libanotica Fruit and Leaf Extracts
Abstract
1. Introduction
2. Results and Discussion
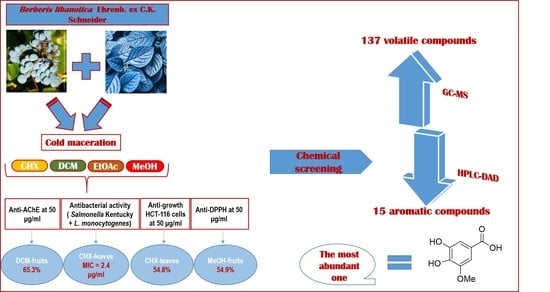
2.1. Plant Materials and Extraction Yields
2.2. Total Phenolic Content (TPC)
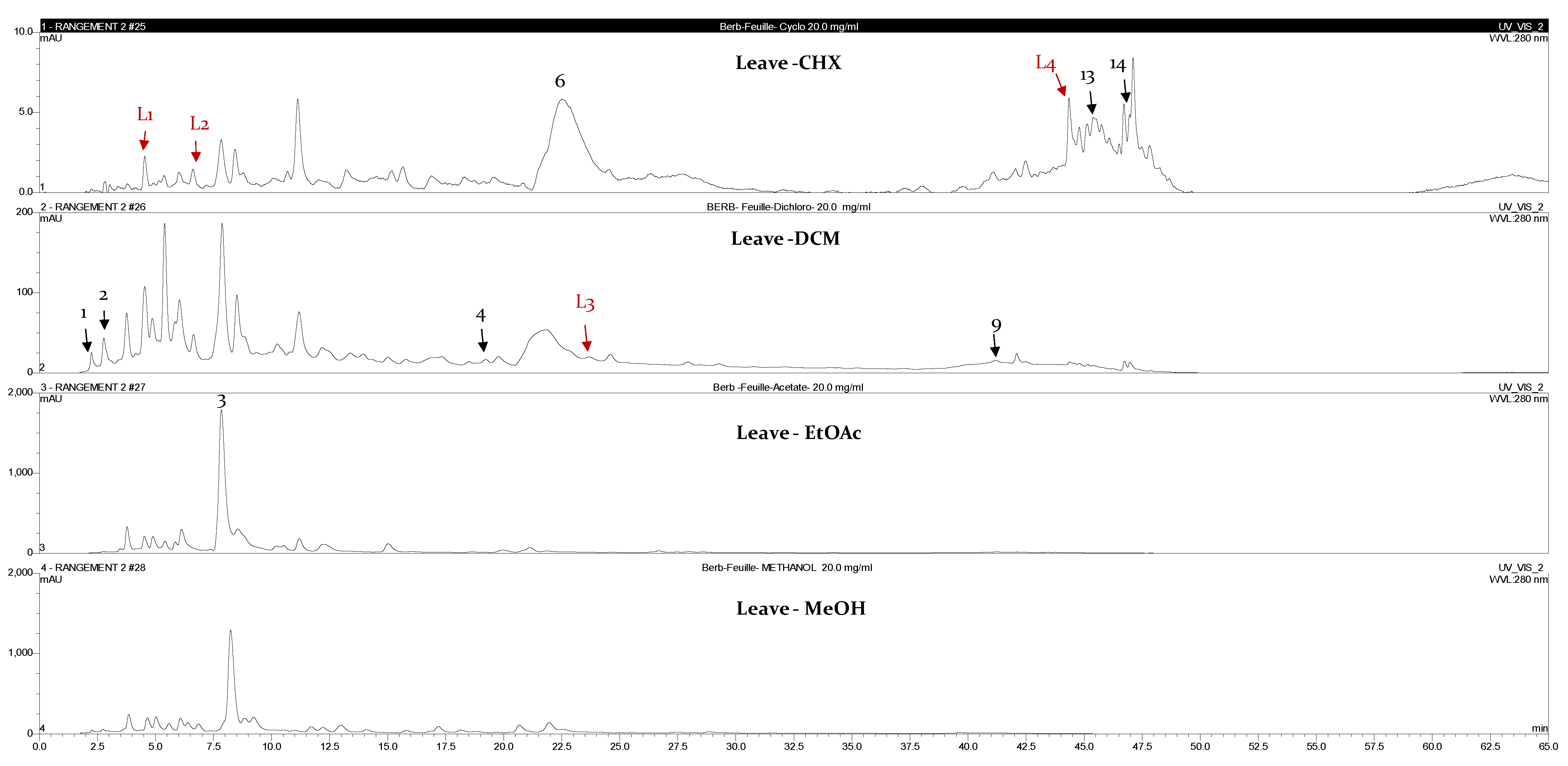
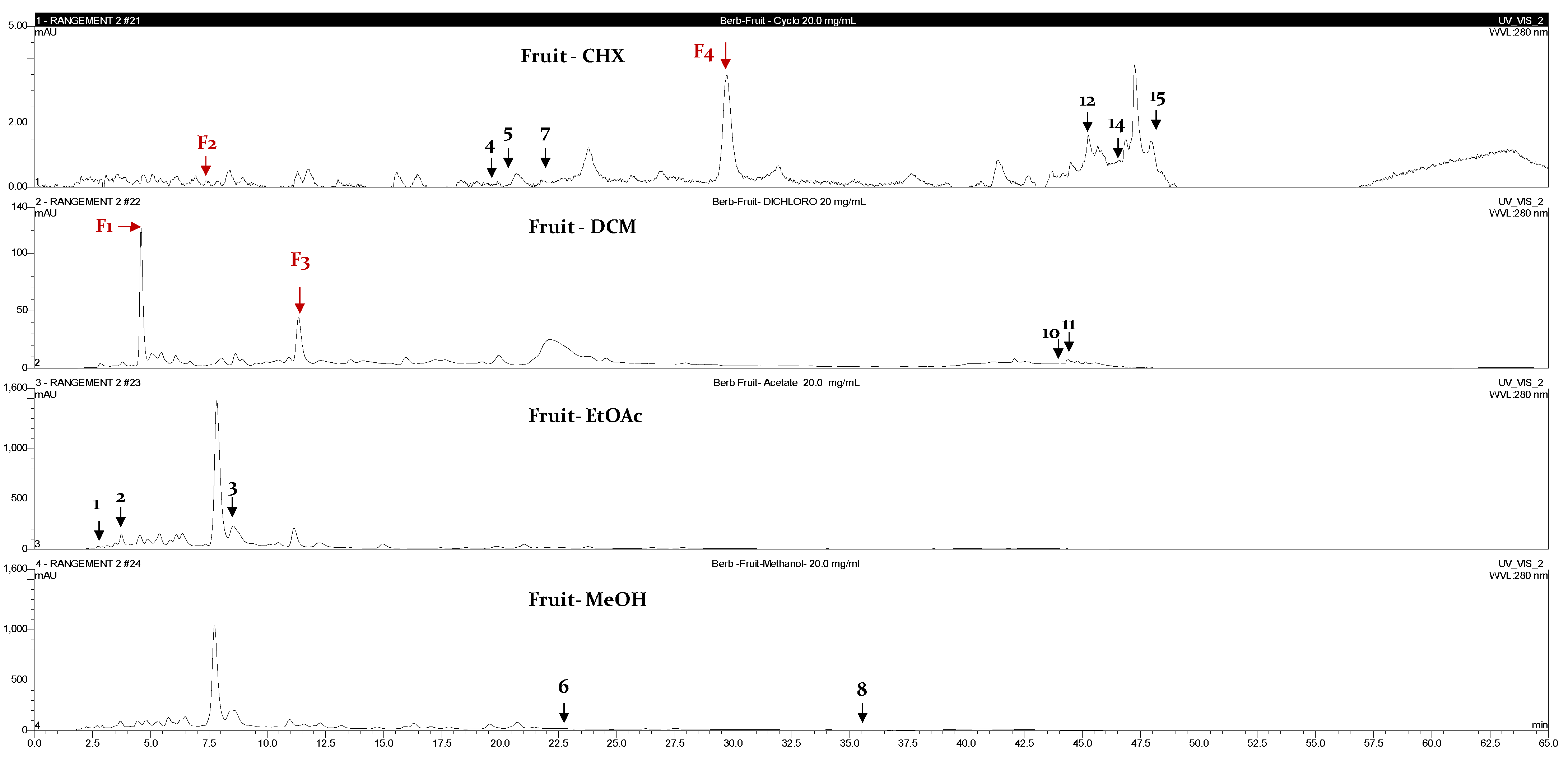
2.3. Chromatographic Fingerprint Analyses by High-Performance Liquid Chromatography Coupled with Diode Array Detector (HPLC-DAD)
2.4. Gas Chromatography Mass Spectrometry (GC-MS) Analysis
2.5. Antioxidant Capacity (DPPH Assay)
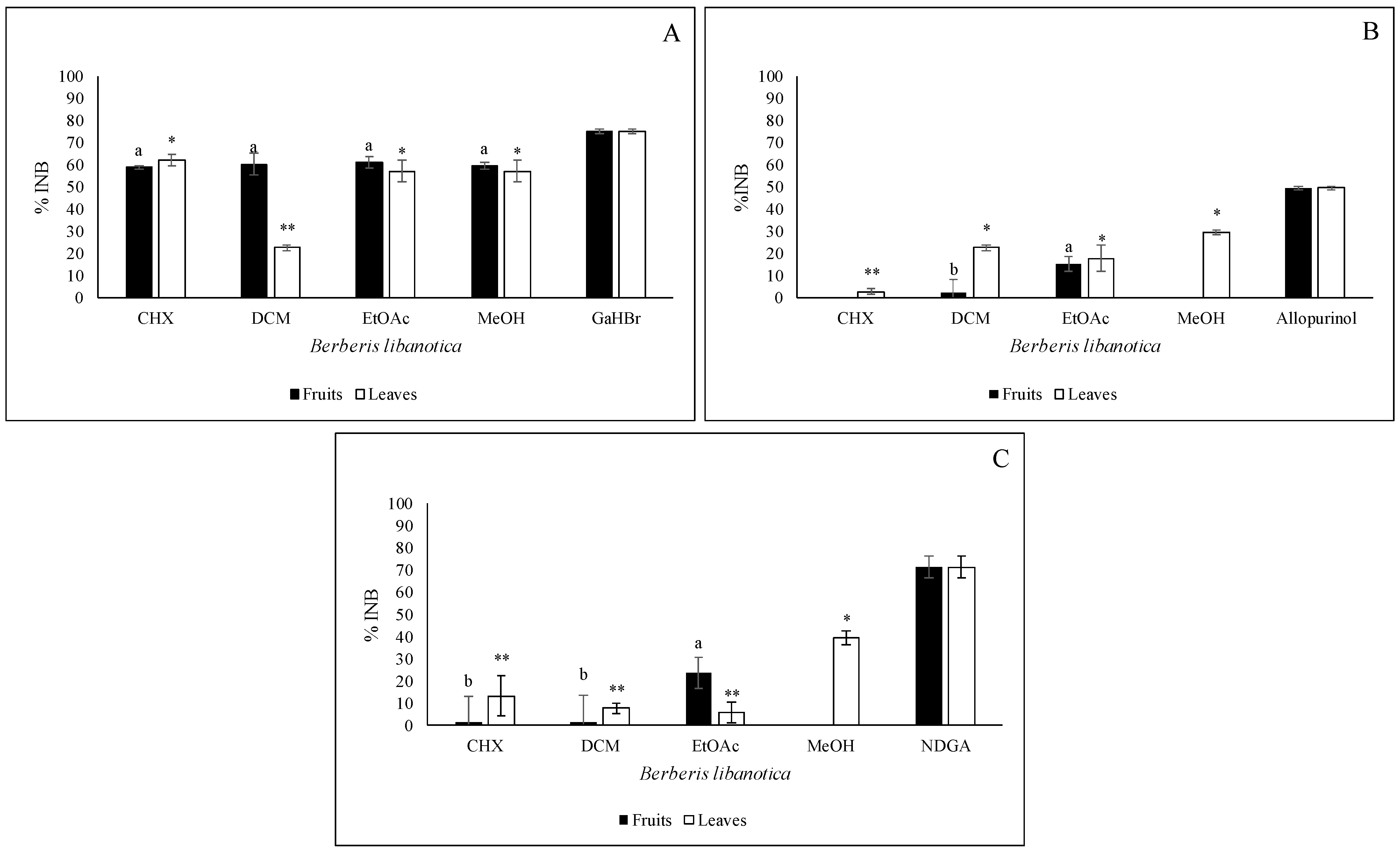
2.6. Anti-Acetylcholinesterase Activity
2.7. Anti-XOD Activity
2.8. Anti-Inflammatory Activity
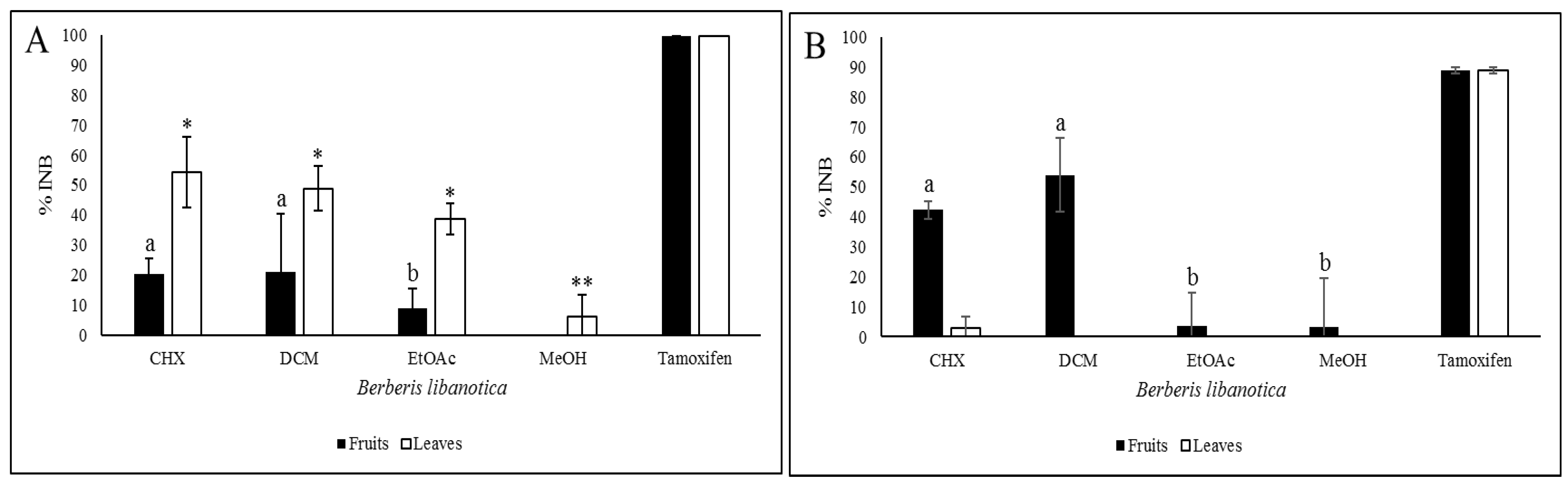
2.9. Anti-Proliferation Activity
2.10. Antibacterial Activity
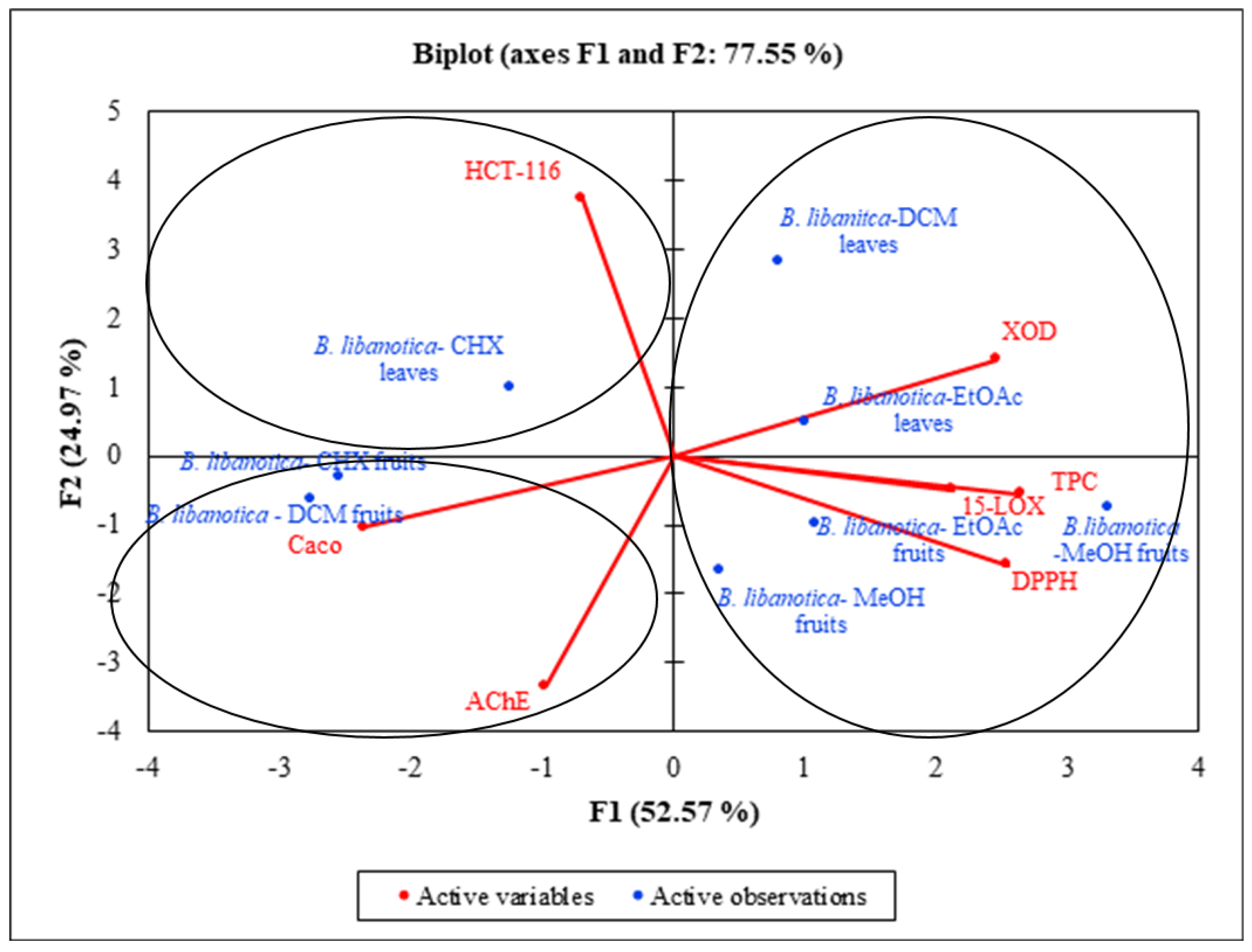
2.11. Statistical Analysis
3. Materials and Methods
3.1. Chemicals and Plant Materials
3.2. Preparation of the Extracts
3.3. Total Phenolic Content (TPC)
3.4. Chromatographic Fingerprint Analyses by High-Performance Liquid Chromatography Coupled with Diode Array Detector (HPLC-DAD)
3.5. Gas Chromatography Mass Spectrometry (GC-MS) Analysis
3.6. Antioxidant Capacity (DPPH Assay)
3.7. Anti-Acetylcholinesterase Activity
3.8. Anti-XOD Activity
3.9. Anti-Proliferation Activity
3.10. Anti-Inflammatory Activity
3.11. Antimicrobial Activity
3.12. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Salehi, B.; Selamoglu, Z.; Sener, B.; Kilic, M.; Jugran, A.K.; De Tommasi, N.; Sinisgalli, C.; Milella, L.; Rajkovic, J.; Morais-Braga, M.B.F.; et al. Berberis plants-drifting from farm to food applications, phytotherapy, and phytopharmacology. Foods 2019, 8, 522. [Google Scholar] [CrossRef] [PubMed]
- Belwal, T.; Bisht, A.; Devkota, H.P.; Ullah, H.; Khan, H.; Pandey, A.; Dutt, I.; Echeverría, J. Phytopharmacology and clinical updates of Berberis species against diabetes and other metabolic diseases. Front. Pharmacol. 2020, 11, 41. [Google Scholar] [CrossRef] [PubMed]
- Bonesi, M.; Loizzo, M.R.; Conforti, F.; Passalacqua, N.G.; Saab, A.; Menichini, F.; Tundis, R. Berberis aetnensis and B. libanotica: A comparative study on the chemical composition, inhibitory effect on key enzymes linked to Alzheimer’s disease and antioxidant activity. J. Pharm. Pharmacol. 2013, 65, 1726–1735. [Google Scholar] [CrossRef]
- Yoo, K.Y.; Hwang, I.K.; Lim, B.O.; Kang, T.C.; Kim, D.W.; Kim, S.M.; Won, M.H. Berberry extract reduces neuronal damage and N-methyl-D-aspartate receptor 1 immunoreactivity in the gerbil hippocampus after transient forebrain ischemia. Biol. Pharm. Bull. 2006, 29, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Boyer, L.; Garayev, E.E.; Bun, S.S.; Mabrouki, F.; Garayev, E.A.; Movsumov, I.S.; Habib, J.; Ollivier, E.; Elias, R. Biologically active compounds from Berberis libanotica. Chem. Nat. Compd. 2016, 52, 567–568. [Google Scholar] [CrossRef]
- Darwich, L.; El-Sawalhi, S.; Hamzeh, O.; El Beyrouthy, M.; Iriti, M.; Abdel-Massih, R.M. Antibacterial activity of two endemic Lebanese medicinal plants, Origanum libanoticum and Berberis libanotica, on human pathogenic bacteria. Plant Biosyst. 2021, 156, 1107–1116. [Google Scholar] [CrossRef]
- Diab, S.; Ftdanzi, C.; Léger, D.Y.; Ghezali, L.; Millot, M.; Martin, F.; Azar, R.; Esseily, F.; Saab, F.; Sol, V.; et al. Berberis libanotica extract targets NF-κB/COX-2, PI3K/Akt and mitochondrial/caspase signalling to induce human erythroleukemia cell apoptosis. Int. J. Oncol. 2015, 47, 220–230. [Google Scholar] [CrossRef]
- Esseily, F.; El Ezzy, M.; Ghali-Mustasib, H.; Safi, S.; Esseily, J.; Diab-Assaf, M.; Lampronti, I.; Saab, A. The ethanol fraction from the Stem of Berberis libanotica inhibits the viability of adult T cell leukemia. Miner. Biotecnol. 2012, 24, 129–133. [Google Scholar]
- Dawra, M.; Nehme, N.; El Rayess, Y.; El Beyrouthy, M.; Taillandier, P.; Bouajila, J. Folk medicinal applications, phytochemical composition and biological activities of some Lebanese endemic plants. S. Afr. J. Bot. 2022, 150, 511–527. [Google Scholar] [CrossRef]
- El Hosry, L.; Boyer, L.; Garayev, E.E.; Mabrouki, F.; Bun, S.S.; Debrauwer, L.; Cheble, L.; Elias, R. Chemical composition, antioxidant and cytotoxic activities of roots and fruits of Berberis libanotica. Nat. Prod. Commun. 2016, 11, 645–648. [Google Scholar] [CrossRef]
- Kostikova, A.V.; Veklich, N.T. HPLC analysis of phenolic compounds in leaves and inflorescences of Sorbaria pallasii. In Proceedings of the BIO Web of Conferences International Conferences “Plant Diversity: Status, Trends, Conservation Concept”, Novosibirsk, Russia, 30 September–3 October 2020; Volume 24, pp. 1–5. [Google Scholar] [CrossRef]
- Medina-Medrano, J.R.; Almaraz-Abarca, N.; Socorro González-Elizondo, M.; Uribe-Soto, J.N.; González-Valdez, L.S.; Herrera-Arrieta, Y. Phenolic constituents and antioxidant properties of five wild species of Physalis (Solanaceae). Bot. Stud. J. 2015, 56, 24. [Google Scholar] [CrossRef] [PubMed]
- Dubtsova, G.N.; Lomakin, A.A.; Azimkova, E.M.; Kosareva, K.V.; Dubtsov, G.G.; Kusova, I.U. Lipid composition of viburnum and barberry fruits. IOP Conf. Ser. Earth Environ. Sci. 2021, 640, 042002. [Google Scholar] [CrossRef]
- Koldaş, S.; Demirtas, I.; Ozen, T.; Demirci, M.A.; Behçet, L. Phytochemical screening, anticancer and antioxidant activities of Origanum vulgare L. ssp. viride (Boiss.) Hayek, a plant of traditional usage. J. Sci. Food Agric. 2015, 95, 786–798. [Google Scholar] [CrossRef]
- Asha, R.; Gayathri, D.V.; Abraham, A. Lupeol, a pentacyclic triterpenoid isolated from Vernonia cinerea attenuate selenite induced cataract formation in Sprague Dawley rat pups. Chem.-Biol. Interact. 2016, 245, 20–29. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Thakur, K.; Wei, C.K.; Wei, Z.J. Evaluation of inhibitory activity of natural plant polyphenols on Soybean lipoxygenase by UFLC-mass spectrometry. S. Afr. J. Bot. 2019, 120, 179–185. [Google Scholar] [CrossRef]
- Rzodkiewicz, P.; Gasinska, E.; Maslinski, S.; Bujalska-Zadrozny, M. Antinociceptive properties of esculetin in non-inflammatory and inflammatory models of pain in rats. Clin. Exp. Pharmacol. 2015, 42, 213–219. [Google Scholar] [CrossRef]
- Lee, D.G.; Park, J.H.; Yoo, K.H.; Chung, I.S.; Lee, Y.H.; Lee, J.K.; Lee, Y.H.; Han, J.K.; Cho, D.S.; Baek, N.I. 24-ethylcholesta-4,24(28)-dien-3,6-dione from Osmanthus fragrans var. aurantiacus flowers inhibits the growth of human colon cancer cell line, HCT-116. J. Appl. Biol. Chem. 2011, 54, 206–210. [Google Scholar] [CrossRef]
- Jabal, K.A.; Abdallah, H.M.; Mohamed, G.A.; Shehata, I.A.; Alfaifi, M.Y.; Elbehairi, S.E.I.; Abdulrahman, A.; Ibrahim, S.R.M. Perisomalien, a new cytotoxic scalarane sesterterpene from the fruits of Periploca somaliensis. Nat. Prod. Res. 2020, 34, 2167–2172. [Google Scholar] [CrossRef]
- Zhang, L.; Cai, Q.Y.A.N.; Liu, J.; Peng, J.U.N. Ursolic acid suppresses the invasive potential of colorectal cancer cells by regulating the TGF-β1/ZEB1 miR-200c signaling pathway. Oncol. Lett. 2019, 18, 274–3282. [Google Scholar] [CrossRef]
- Pejin, B.; Savic, A.; Sokovic, M.; Glamoclija, J.; Ciric, A.; Nikolic, M.; Mojovic, M. Further in vitro evaluation of antiradical and antimicrobial activities of phytol. Nat. Prod. Res. 2014, 28, 372–376. [Google Scholar] [CrossRef]
- Choi, U.; Lee, C.R. Distinct roles of outer membrane porins in antibiotic resistance and membrane integrity in Escherichia coli. Front. Microbiol. 2019, 10, 953. [Google Scholar] [CrossRef] [PubMed]
- He, C.L.; Fu, B.D.; Shen, H.Q.; Jiang, X.L.; Wei, X.B. Fumaric acid, an antibacterial component of Aloe vera L. Afr. J. Biotechnol. 2011, 10, 2973–2977. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, H.; Zhu, S.; Ma, C.; Wang, Z. Antibacterial activity and mechanism of action of chlorogenic acid. J. Food Sci. 2011, 76, 398–403. [Google Scholar] [CrossRef]
- Geng, Z.Z.; Zhang, J.J.; Lin, J.; Huang, M.Y.; An, L.K.; Zhang, H.; Bin, P.H.; Ye, C.; Chen, W.M. Novel cajaninstilbene acid derivatives as antibacterial agents. Eur. J. Med. Chem. 2015, 100, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Dawra, M.; Bouajila, J.; El Beyrouthy, M.; Abi Rizk, A.; Taillandier, P.; Nehme, N.; El Rayess, Y. Chemical Characterization and Antioxidant, Antibacterial, Antiacetylcholinesterase and Antiproliferation Properties of Salvia fruticosa Miller Extracts. Molecules 2023, 28, 2429. [Google Scholar] [CrossRef] [PubMed]
- Dawra, M.; El Rayess, Y.; El Beyrouthy, M.; Nehme, N.; El Hage, R.; Taillandier, P.; Bouajila, J. Biological activities and chemical characterization of the Lebanese endemic plant Origanum ehrenbergii Boiss. Flavour Fragr. J. 2021, 36, 339–351. [Google Scholar] [CrossRef]
- CLSI M100-S27; Performance Standards for Antimicrobial Susceptibility Testing: 27th Informational Supplement. CLSI: Wayne, PA, USA, 2017.

| Extraction Yields (dw %) | ||
|---|---|---|
| Leaves | Fruits | |
| CHX | 0.7 | 5.4 |
| DCM | 0.2 | 0.8 |
| EtOAc | 0.5 | 0.1 |
| MeOH | 16.8 | 15.6 |
| TPC (mg GAE/g of dw) | ||
|---|---|---|
| Leaves | Fruits | |
| CHX | 32.8 ± 1.6 c | 16.9 ± 4.3 +++ |
| DCM | 65.8 ± 1.8 b | 17.3 ± 0.7 +++ |
| EtOAc | 122.0 ± 1.5 a | 68.9 ± 2.5 ++ |
| MeOH | 132.7 ± 1.9 a | 94.8 ± 3.3 + |
| Concentration (mg/g of Extract) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Leaves | Fruits | ||||||||||||
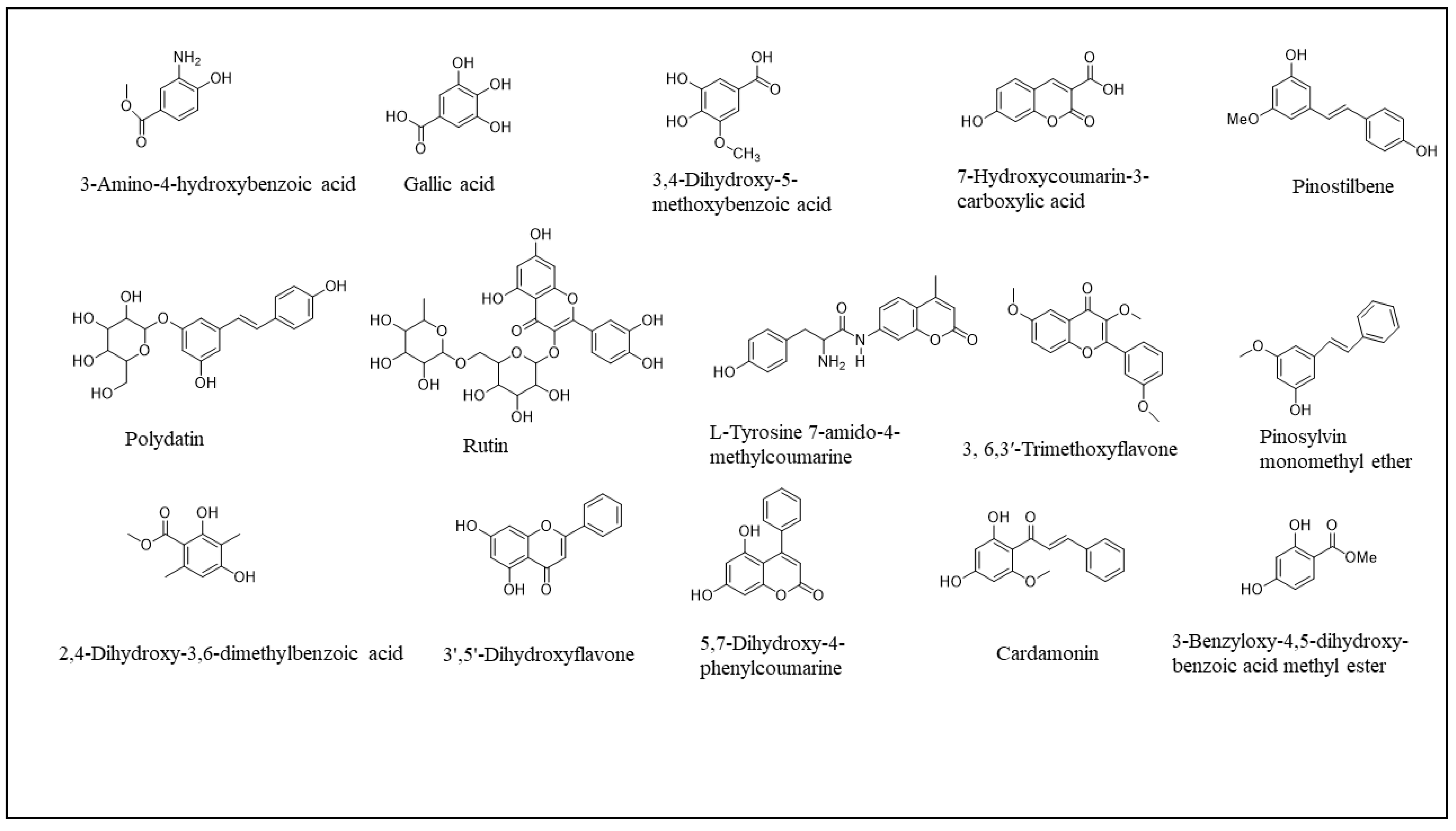
| № | tR (min) | λmax(nm) | Type of phenolic compound | Compound | Calibration curves | CHX | DCM | EtOAc | MeOH | CHX | DCM | EtOAc | MeOH |
| 1 | 2.2 | 281 | Derivative of the p-hydroxybenzoic acid | 3-Amino-4-hydroxybenzoic acid | y = 0.5959x + 0.4365 | 0.4 ± 0.0 | 1.3 ± 0.0 | 0.2 ± 0.0 | 1.2 ± 0.0 | ||||
| 2 | 3.4 | 269 | Phenolic acid | Gallic acid | y = 0.6442x − 0.4737 | 0.2 ± 0.0 | 0.2 ± 0.0 | ||||||
| 3 | 7.7 | 222 | Derivative of the p-hydroxybenzoic acid | 3,4-Dihydroxy-5-methoxybenzoic acid | y = 0.1682x − 0.047 | 0.2 ± 0.1 | 21.1 ± 1.9 | 239.3 ± 20.2 | 142.2 ± 0.0 | 164.4 ± 3.6 | 98.8 ± 4.3 | ||
| 4 | 19.1 | 265 | Derivative of methyl coumarin | L-Tyrosine 7-amido-4-methylcoumarine | y = 0.1483x − 0.2105 | 4.8 ± 0.6 | 0.2 ± 0.3 | ||||||
| 5 | 19.9 | 266 | Coumarin derivative | 7-Hydroxycoumarin-3-carboxylic acid | y = 0.0799x + 0.1586 | 1.8 ± 0.9 | 6.3 ± 0.7 | ||||||
| 6 | 22.6 | 230 | Flavonoid | Rutin | y = 0.1029x + 0.6179 | 4.0 ± 1.8 | 2.2 ± 1.7 | 0.3 ± 0.0 | |||||
| 7 | 23.3 | 267 | Stilbene | Polydatin | y = 0.0445x − 0.0083 | 0.5 ± 0.0 | |||||||
| 8 | 35.2 | 340 | Phenolic acid | 2,4-Dihydroxy-3,6-dimethylbenzoic acid | y = 0.1612x − 0.1498 | 0.1 ± 0.0 | |||||||
| 9 | 42.1 | 286 | Flavonoid | 3′,5′-Dihydroxyflavone | y = 0.1267x − 0.0317 | 4.7 ± 0.0 | 3.6 ± 0.0 | ||||||
| 10 | 43.4 | 281 | Derivative of phenyl coumarin | 5,7-Dihydroxy-4-phenylcoumarine | y = 0.1605x − 0.0115 | 0.5 ± 0.3 | |||||||
| 11 | 44.1 | 240 | Chalcone derivative | Cardamonin | y = 0.009x − 0.035 | 5.5 ± 3.9 | |||||||
| 12 | 44.6 | 240 | Stilbene | Pinostilbene | y = 0.041x + 0.0646 | 0.1 ± 0.0 | 1.0 ± 0.0 | ||||||
| 13 | 44.9 | 259 | Phenolic acid derivative | 3-Benzyloxy-4,5-dihydroxy-benzoic acid methyl ester | y = 0.0961x + 0.5481 | 0.1 ± 0.0 | |||||||
| 14 | 47.0 | 257 | Stilbene | Pinosylvin monomethyl ether | y = 0.1265x − 0.5347 | 1.1 ± 0.5 | 0.7 ± 0.1 | ||||||
| 15 | 47.9 | 262 | Methoxyphenol | 3, 6,3′-Trimethoxyflavone | y = 0.1017x + 0.1091 | 0.1 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.0 | |||||
| Compound Number | tR (min) | λmax (nm) | Suggested Chemical Class | Extract | References |
|---|---|---|---|---|---|
| Leaves | |||||
| L1 | 5 | 253 | p-hydroxybenzoic acid | CHX | [11] |
| L2 | 7.5 | 240 | Quercetin | CHX | [11] |
| L3 | 22.8 | 232 | Flavonol | DCM | [11] |
| L4 | 44.3 | 233 | Phenolic acid | CHX | [12] |
| Fruits | |||||
| F1 | 5 | 284 | p-hydroxybenzoic acid | DCM | [11] |
| F2 | 7.5 | 255 | Luteolin | CHX | [11] |
| F3 | 12.5 | 225 | Flavanone | DCM | [11] |
| F4 | 30 | 336 | Phenolic acid | CHX | [12] |
| B. libanotica—Before Derivatization | ||||||||
|---|---|---|---|---|---|---|---|---|
| Leaves | Fruits | |||||||
| № | RI | Compound | CHX | DCM | MeOH | CHX | DCM | MeOH |
| 1 | - | permethyl 99A | + | + | ||||
| 2 | - | 3-isopropylbenzaldehyde | + | |||||
| 3 | - | benzene, 2-methoxy-1,3,5-trimethyl- | + | |||||
| 4 | 1850 | neophytadiene | + | + | + | ++ | ||
| 5 | 1857 | hexahydrofarnesyl acetone | + | + | + | |||
| 6 | 1893 | 3,7,11,15-tetramethyl-2-hexadecen-1-ol | + | |||||
| 7 | 1930 | methyl palmitate | +++ | ++ | ||||
| 8 | 1937 | 7,9-di-tert-butyl-1-oxaspiro(4,5)deca-6,9-diene-2,8-dione | ++ | ++ | ++ | |||
| 9 | 1984 | palmitic acid | + | ++ | ++ | ++ | ||
| 10 | 2127 | phytol | + | + | + | + | ||
| 11 | 2132 | methyl stearate | + | + | ||||
| 12 | 2157 | linoleic acid | + | + | ++ | |||
| 13 | 2172 | oleic acid | + | +++ | + | |||
| 14 | 2372 | 4,8,12,16-tetramethylheptadecan-4-olide | + | + | ||||
| 15 | 2422 | phenol, 2,2′-methylenebis[6-(1,1-dimethylethyl)-4-methyl- | + | ++ | ++ | +++ | ||
| 16 | 2520 | 2-monopalmitin | +++ | ++ | +++ | ++ | ++ | |
| 17 | 2736 | α-monostearin | + | ++ | ||||
| 18 | 2878 | α-tocospiro A | + | + | + | + | ||
| 19 | 2900 | α-tocospiro B | + | + | + | + | ||
| 20 | 2902 | octacosane | +++ | +++ | +++ | |||
| 21 | 2914 | glaucine | + | ++ | ||||
| 22 | 2937 | heptacosane | + | |||||
| 23 | 2959 | 5-methylnonacosane | + | |||||
| 24 | 2950 | α-tocopherolquinone | +++ | |||||
| 25 | 2962 | vitamin E | ++ | ++ | ||||
| 26 | 3070 | β-sitosterol | + | +++ | +++ | |||
| 27 | 3079 | γ-sitosterol | +++ | |||||
| 28 | - | didehydroglaucine | + | ++ | ||||
| 29 | - | β-amyrin | ++ | + | ||||
| 30 | - | stigmasta-3,5-diene | + | |||||
| 31 | - | lupeol | + | +++ | + | |||
| B. libanotica—after derivatization | ||||||||
| Leaves | Fruits | |||||||
| № | tR (min) | Compounds | CHX | DCM | MeCN | CHX | DCM | MeCN |
| 1′ | 9.2 | 2,3-butanediol | + | |||||
| 2′ | 9.8 | carbamic acid | ++ | |||||
| 3′ | 9.8 | lactic acid | +++ | |||||
| 4′ | 10 | hexanoic acid | ++ | |||||
| 5′ | 10.3 | glycolic acid | +++ | |||||
| 6′ | 10.4 | 1-butoxy-2-propanol | + | |||||
| 7′ | 13.1 | hydracrylic acid | + | |||||
| 8′ | 21.6 | octanoic acid | + | |||||
| 9′ | 25.1 | glycerol | ++ | |||||
| 10′ | 27.2 | thymol | + | + | ||||
| 11′ | 29.2 | butanedioic acid | + | |||||
| 12′ | 29.4 | carvacrol | + | + | ||||
| 13′ | 31.3 | glyceric acid | + | |||||
| 14′ | 31.8 | pyrrole-2-carboxylic acid | + | |||||
| 15′ | 32.1 | nonaoic acid | + | |||||
| 16′ | 32.6 | fumaric acid | + | |||||
| 17′ | 33.2 | cuminol | + | |||||
| 18′ | 33.6 | 3-hydroxycaproic acid | + | |||||
| 19′ | 36.1 | 2-methyl-6-tertbutylphenol | + | |||||
| 20′ | 36.2 | malic acid | + | |||||
| 21′ | 36.2 | 4-methylbenzoic acid | + | |||||
| 22′ | 36.5 | 4-hydroxybutanoic acid | + | |||||
| 23′ | 36.8 | 2,4-di-tert-butylphenol | + | |||||
| 24′ | 37.2 | (L) erythronic acid | + | |||||
| 25′ | 37.3 | tert-butylhydroquinone | + | |||||
| 26′ | 37.5 | (D) erythronic acid | + | |||||
| 27′ | 37.7 | (L) xylose | + | |||||
| 28′ | 37.8 | (D) xylose | + | |||||
| 29′ | 37.9 | 3,4-dihydroxybenzaldehyde | ++ | |||||
| 30′ | 38.1 | 4-hydroxybenzoic acid | + | |||||
| 31′ | 38.3 | D-xylonic acid | + | |||||
| 32′ | 38.4 | (D)-ribonolactone | + | |||||
| 33′ | 38.44 | dodecanoic acid | + | + | ||||
| 34′ | 39.2 | homovanillyl alcohol | + | + | ||||
| 35′ | 39.3 | levoglucosan | + | |||||
| 36′ | 39.7 | (L)-ribonolactone | ++ | |||||
| 37′ | 39.9 | vanillic acid | + | |||||
| 38′ | 39.98 | 1-(3,4-dihydroxyphenyl) ethanone | + | |||||
| 39′ | 40.1 | loliolide | + | |||||
| 40′ | 40.2 | azealic acid | + | ++ | ||||
| 41′ | 40.4 | β-D-tagatopyranose | + | |||||
| 42′ | 40.5 | 2,5-furan diol | + | |||||
| 43′ | 40.5 | D-(-)-tagatofuranose | ++ | |||||
| 44′ | 40.6 | D-psicofuranose « isomer 2 » | + | |||||
| 45′ | 40.7 | D-(-)-fructofuranose « isomer 2 » | + | |||||
| 46′ | 40.76 | myristic acid | +++ | ++ | ||||
| 47′ | 40.8 | D-(-)-fructopyranose | ++ | |||||
| 48′ | 41.2 | quininic acid | + | |||||
| 49′ | 41.03 | methyl α-D-glucofuranoside | ++ | |||||
| 50′ | 41.4 | syringic acid | + | ++ | ||||
| 51′ | 41.5 | α-D-mannopyranose | + | |||||
| 52′ | 41.59 | talose | +++ | |||||
| 53′ | 41.7 | pentadecanoic acid | + | |||||
| 54′ | 42.1 | D-gluconic acid | +++ | |||||
| 55′ | 42.3 | L-gluconic acid | +++ | |||||
| 56′ | 42.4 | β-D-(+)-mannopyranose | + | |||||
| 57′ | 42.4 | α-linolenic acid | + | |||||
| 58′ | 42.5 | palmitelaidic acid | + | |||||
| 59′ | 42.4 | β-D-glucopyranose | +++ | |||||
| 60′ | 42.5 | α,β-glucooctanoic γ-lactone | + | |||||
| 61′ | 42.8 | glucaric acid | + | |||||
| 62′ | 43.2 | ferulic acid | + | |||||
| 63′ | 43.3 | esculetin | + | |||||
| 64′ | 43.6 | heptadecanoic acid | + | |||||
| 65′ | 43.7 | caffeic acid | +++ | |||||
| 66′ | 43.76 | 1-octadecanol | +++ | |||||
| 67′ | 44.2 | linoleic acid | +++ | +++ | ||||
| 68′ | 44.5 | stearic acid | +++ | + | ||||
| 69′ | 45.9 | 13-eicosenoic acid (Z) | + | + | ||||
| 70′ | 46.1 | arachidic acid | ++ | + | ||||
| 71′ | 46.9 | heneicosanoic acid | + | |||||
| 72′ | 47 | docosanol | +++ | |||||
| 73′ | 47.13 | 2-palmitoylglycerol | + | |||||
| 74′ | 47.17 | hernagine | + | |||||
| 75′ | 47.4 | 1-monopalmitine | +++ | +++ | ||||
| 76′ | 47.5 | 13-docosenoic acid (Z) | + | + | ||||
| 77′ | 47.6 | behenic acid | +++ | + | ||||
| 78′ | 47.7 | N-methylhernagine | +++ | |||||
| 79′ | 47.8 | (10E,15Z)-9,12,13-trihydroxy-10,15-octadecadienoic acid | + | |||||
| 80′ | 48.1 | sucrose | + | |||||
| 81′ | 48.37 | 2-monoolein | + | |||||
| 82′ | 48.38 | tricosanoic acid | + | |||||
| 83′ | 48.4 | tetracosanol | + | |||||
| 84′ | 48.5 | 2-monostearin | + | |||||
| 85′ | 48.6 | monolinolein | + | |||||
| 86′ | 48.66 | 1-linolenoylglycerol | + | |||||
| 87′ | 48.67 | 2-monolinolenin | + | |||||
| 88′ | 48.7 | glycerol monostearate | ++ | +++ | ||||
| 89′ | 49 | lignoceric acid | +++ | |||||
| 90′ | 49.6 | 2-phenylethyl β-D-glucopyranoside | +++ | |||||
| 91′ | 49.8 | 1-hexacosanol | + | |||||
| 92′ | 50.2 | γ-tocopherol | + | |||||
| 93′ | 50.4 | hexacosanoic acid | ++ | |||||
| 94′ | 50.7 | nonacosan-10-ol | +++ | |||||
| 95′ | 51.1 | cajaninstilbene acid | + | |||||
| 96′ | 51.13 | methyl chlorogenate | +++ | |||||
| 97′ | 51.14 | 1-octacosanol | ++ | |||||
| 98′ | 51.3 | chlorogenic acid | ++ | |||||
| 99′ | 51.8 | octacosanoate | ++ | |||||
| 100′ | 52.03 | hentriacontan-12-ol | + | |||||
| 101′ | 52.2 | campesterol | + | |||||
| 102′ | 52.4 | stigmasterol | + | |||||
| 103′ | 53.5 | α-amyrin | + | |||||
| 104′ | 52.7 | 1-triacontanol | + | |||||
| 105′ | 55.67 | oleanolic acid | + | |||||
| 106′ | 56.23 | Ursolic acid | ++ | |||||
| Bacterial Strains | MIC (μg/mL) | ||||
|---|---|---|---|---|---|
| CHX | DCM | EtOAc | MeOH | ||
| Salmonella enterica serovar Enteritidis | Gram (−) | 19.5 ± 0 | 39 ± 1.2 | - | - |
| Salmonella enterica serovar Kentucky | - | 39 ± 0 | - | - | |
| Salmonella enterica serovar Infantis | - | 19.5 ± 0 | - | - | |
| Escherichia coli ATCC 8739 | 9.7 ± 1.2 | - | - | - | |
| Listeria monocytogenes ATCC 19115 | Gram (+) | 19.5 ± 0 | 39 ± 0 | - | - |
| Listeria monocytogenes Fish filet | - | - | - | - | |
| Staphylococcus aureus ATCC 25923 | - | - | - | - | |
| Bacterial Strains | MIC (μg/mL) | ||||
|---|---|---|---|---|---|
| CHX | DCM | EtOAc | MeOH | ||
| Salmonella enterica serovar Enteritidis | Gram (−) | 9.7 ± 1.2 | 39 ± 0 | - | - |
| Salmonella enterica serovar Kentucky | 2.4 ± 1.2 | - | - | 625 ± 0 | |
| Salmonella enterica serovar Infantis | - | - | 156.2 ± 0 | 625 ± 0 | |
| Escherichia coli ATCC 8739 | 312.5 ± 1.2 | 625 ± 1.2 | 625 ± 0.4 | 625 ± 0 | |
| Listeria monocytogenes ATCC 19115 | Gram (+) | 2.4 ± 4.9 | - | 312.5 ± 0 | 625 ± 0 |
| Listeria monocytogenes Fish filet | 4.8 ± 1.2 | - | - | - | |
| Staphylococcus aureus ATCC 25923 | - | - | 78.1 ± 0 | - | |
| F1 | F2 | |
|---|---|---|
| TPC | 22.6 | 1.0 |
| DPPH | 20.8 | 8.0 |
| AChE | 3.0 | 35.8 |
| XOD | 19.4 | 6.2 |
| 15-LOX | 14.6 | 0.8 |
| HCT-116 | 1.5 | 44.7 |
| Caco-2 | 18.0 | 3.5 |
| F1 | F2 | |
|---|---|---|
| TPC | 0.9 | −0.1 |
| DPPH | 0.8 | −0.3 |
| AChE | −0.3 | −0.7 |
| XOD | 0.8 | 0.3 |
| 15-LOX | 0.7 | −0.1 |
| HCT-116 | −0.2 | 0.8 |
| Caco-2 | −0.8 | −0.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dawra, M.; Nehme, N.; El Beyrouthy, M.; Abi Rizk, A.; Taillandier, P.; Bouajila, J.; El Rayess, Y. Comparative Study of Phytochemistry, Antioxidant and Biological Activities of Berberis libanotica Fruit and Leaf Extracts. Plants 2023, 12, 2001. https://doi.org/10.3390/plants12102001
Dawra M, Nehme N, El Beyrouthy M, Abi Rizk A, Taillandier P, Bouajila J, El Rayess Y. Comparative Study of Phytochemistry, Antioxidant and Biological Activities of Berberis libanotica Fruit and Leaf Extracts. Plants. 2023; 12(10):2001. https://doi.org/10.3390/plants12102001
Chicago/Turabian StyleDawra, Michella, Nancy Nehme, Marc El Beyrouthy, Alain Abi Rizk, Patricia Taillandier, Jalloul Bouajila, and Youssef El Rayess. 2023. "Comparative Study of Phytochemistry, Antioxidant and Biological Activities of Berberis libanotica Fruit and Leaf Extracts" Plants 12, no. 10: 2001. https://doi.org/10.3390/plants12102001
APA StyleDawra, M., Nehme, N., El Beyrouthy, M., Abi Rizk, A., Taillandier, P., Bouajila, J., & El Rayess, Y. (2023). Comparative Study of Phytochemistry, Antioxidant and Biological Activities of Berberis libanotica Fruit and Leaf Extracts. Plants, 12(10), 2001. https://doi.org/10.3390/plants12102001