Nutrients, Phytic Acid and Bioactive Compounds in Marketable Pulses
Abstract
1. Introduction
2. Results and Discussion
2.1. Protein, Fat and Phytic Acid
2.2. Phenolic Profile
2.3. Multi-Mineral Profile
2.4. Cluster and Multivariate Analysis
3. Materials and Methods
3.1. Plant Material
3.2. Moisture, Protein and Fat Content
3.3. Phytic Acid
3.4. HPLC-MS/MS Analysis
3.5. ICP-MS Analysis
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization (FAO). Crops Statistics-Concepts, Definitions and Classifications. 2022. Available online: https://www.fao.org/economic/the-statistics-division-ess/methodology/methodology-systems/crops-statistics-concepts-definitions-and-classifications/en/ (accessed on 10 October 2022).
- Duc, G.; Agrama, H.; Bao, S.; Berger, J.; Bourion, V.; De Ron, A.M.; Gowda, C.L.L.; Mikic, A.; Millot, D.; Singh, K.B.; et al. Breeding annual grain legumes for sustainable agriculture: New methods to approach complex traits and target mew cultivar ideotypes. Crit. Rev. Plant Sci. 2015, 34, 381–411. [Google Scholar] [CrossRef]
- Röös, E.; Carlsson, G.; Ferawati, F.; Hefni, M.; Stephan, A.; Tidåker, P.; Witthöft, C. Less meat, more legumes: Prospects and challenges in the transition toward sustainable diets in Sweden. Renew. Agric. Food Syst. 2020, 35, 192–205. [Google Scholar] [CrossRef]
- Tiwari, B.; Narpinder, S. Pulse Chemistry and Technology; Royal Society of Chemistry: Cambridge, UK, 2012; pp. 1–301. [Google Scholar]
- Ferreira, H.; Vasconcelos, M.; Gil, A.M.; Pinto, E. Benefits of pulse consumption on metabolism and health: A systematic review of randomized controlled trials. Crit. Rev. Food Sci. Nutr. 2020, 61, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Singh, J.P.; Shevkani, K.; Singh, N.; Kaur, A. Bioactive constituents in pulses and their health benefits. J. Food Sci. Technol. 2017, 54, 858–870. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Basu, P.S. Non-nutritive bioactive compounds in pulses and their impact on human health: An overview. Food Nutr. Sci. 2012, 2012, 1664–1672. [Google Scholar] [CrossRef]
- Al Jitan, S.; Alkhoori, S.A.; Yousef, L.F. Phenolic acids from plants: Extraction and application to human health. Stud. Nat. Prod. Chem. 2018, 58, 389–417. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects–A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Wang, Z.; Li, X.; Zhen, S.; Li, X.; Wang, C.; Wang, Y. The important role of quinic acid in the formation of phenolic compounds from pyrolysis of chlorogenic acid. J. Therm. Anal. Calorim. 2013, 114, 1231–1238. [Google Scholar] [CrossRef]
- Singh, N. Pulses: An overview. J. Food Sci. Technol. 2017, 54, 853–857. [Google Scholar] [CrossRef]
- Arnoldi, A.; Zanoni, C.; Lammi, C.; Boschin, G. The role of grain legumes in the prevention of hypercholesterolemia and hypertension. Crit. Rev. Plant Sci. 2014, 34, 144–168. [Google Scholar] [CrossRef]
- Duranti, M. Grain legume proteins and nutraceutical properties. Fitoterapia 2006, 77, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Carbas, B.; Machado, N.; Pathania, S.; Brites, C.; Rosa, E.A.; Barros, A.I. Potential of legumes: Nutritional value, bioactive properties, innovative food products, and application of eco-friendly tools for their assessment. Food Rev. Int. 2021, 1–29. [Google Scholar] [CrossRef]
- Samtiya, M.; Aluko, R.E.; Dhewa, T. Plant food anti-nutritional factors and their reduction strategies: An overview. Food Prod. Process. Nutr. 2020, 2, 6. [Google Scholar] [CrossRef]
- Kaale, L.D.; Siddiq, M.; Hooper, S. Lentil (Lens culinaris Medik) as nutrient-rich and versatile food legume: A review. Legume Sci. 2022, e169. [Google Scholar] [CrossRef]
- Hall, C.; Hillen, C.; Garden Robinson, J. Composition, nutritional value, and health benefits of pulses. Cereal Chem. 2017, 94, 11–31. [Google Scholar] [CrossRef]
- Boeck, T.; Sahin, A.W.; Zannini, E.; Arendt, E.K. Nutritional properties and health aspects of pulses and their use in plant-based yogurt alternatives. Compr. Rev. Food Sci. Food Saf. 2021, 20, 3858–3880. [Google Scholar] [CrossRef]
- Kumar, Y.; Basu, S.; Goswami, D.; Devi, M.; Shivhare, U.S.; Vishwakarma, R.K. Anti-nutritional compounds in pulses: Implications and alleviation methods. Legume Sci. 2021, 4, e111. [Google Scholar] [CrossRef]
- Caprioli, G.; Giusti, F.; Ballini, R.; Sagratini, G.; Vila-Donat, P.; Vittori, S.; Fiorini, D. Lipid nutritional value of legumes: Evaluation of different extraction methods and determination of fatty acid composition. Food Chem. 2016, 192, 965–971. [Google Scholar] [CrossRef]
- Alandia, G.; Pulvento, C.; Sellami, M.H.; Hoidal, N.; Anemone, T.; Nigussie, E.; Agüero, J.J.; Lavini, A.; Jacobsen, S.E. Grain legumes may enhance high-quality food production in Europe. In Emerging Research in Alternative Crops; Hirich, A., Choukr-Allah, R., Ragab, R., Eds.; Springer: Cham, Switzerland, 2020; pp. 25–53. [Google Scholar]
- Walter, S.; Zehring, J.; Mink, K.; Quendt, U.; Zocher, K.; Rohn, S. Protein content of peas (Pisum sativum) and beans (Vicia faba)–Influence of cultivation conditions. J. Food Compos. Anal. 2022, 105, 104257. [Google Scholar] [CrossRef]
- Velicković, D.; Vučelić-Radović, B.; Blagojević, S.; Barać, M.; Stanojević, S.; Ljubicić, M. A modification of a method for phytic acid determination. J. Serbian Chem. Soc. 1999, 64, 303–310. Available online: https://aspace.agrif.bg.ac.rs/handle/123456789/158 (accessed on 10 October 2022).
- Nath, H.; Samtiya, M.; Dhewa, T. Beneficial attributes and adverse effects of major plant-based foods anti-nutrients on health: A review. Hum. Nutr. Metab. 2022, 28, 200147. [Google Scholar] [CrossRef]
- Upadhyay, J.; Tiwari, N.; Durgapal, S.; Jantwal, A.; Kumar, A. Phytic acid: As a natural antioxidant. In Antioxidants Effects in Health; Nabavi, S.M., Sanches Silva, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 437–450. [Google Scholar]
- Vasić, M.; Tepić, A.; Mihailović, V.; Mikić, A.; Gvozdanović-Varga, J.; Šumić, Z.; Todorović, V. Phytic acid content in different dry bean and faba bean landraces and cultivars. Rom. Agric. Res. 2012, 29, 79–85. Available online: https://fiver.ifvcns.rs/handle/123456789/1145 (accessed on 10 October 2022).
- Giusti, F.; Caprioli, G.; Ricciutelli, M.; Vittori, S.; Sagratini, G. Determination of fourteen polyphenols in pulses by high performance liquid chromatography-diode array detection (HPLC-DAD) and correlation study with antioxidant activity and colour. Food Chem. 2017, 221, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Caprioli, G.; Nzekoue, F.K.; Giusti, F.; Vittori, S.; Sagratini, G. Optimization of an extraction method for the simultaneous quantification of sixteen polyphenols in thirty-one pulse samples by using HPLC-MS/MS dynamic-MRM triple quadrupole. Food Chem. 2018, 266, 490–497. [Google Scholar] [CrossRef]
- Elessawy, F.M.; Bazghaleh, N.; Vandenberg, A.; Purves, R.W. Polyphenol profile comparisons of seed coats of five pulse crops using a semi-quantitative liquid chromatography-mass spectrometric method. Phytochem. Anal. 2020, 31, 458–471. [Google Scholar] [CrossRef]
- Magalhães, S.C.; Taveira, M.; Cabrita, A.R.; Fonseca, A.J.; Valentão, P.; Andrade, P.B. European marketable grain legume seeds: Further insight into phenolic compounds profiles. Food Chem. 2017, 215, 177–184. [Google Scholar] [CrossRef]
- Orčić, D.; Francišković, M.; Bekvalac, K.; Svirčev, E.; Beara, I.; Lesjak, M.; Mimica-Dukić, N. Quantitative determination of plant phenolics in Urtica dioica extracts by high-performance liquid chromatography coupled with tandem mass spectrometric detection. Food Chem. 2014, 143, 48–53. [Google Scholar] [CrossRef]
- Blandino, M.; Bresciani, A.; Locatelli, M.; Loscalzo, M.; Travaglia, F.; Vanara, F.; Marti, A. Pulse type and extrusion conditions affect phenolic profile and physical properties of extruded products. Food Chem. 2023, 403, 134369. [Google Scholar] [CrossRef]
- Elessawy, F.M.; Vandenberg, A.; El-Aneed, A.; Purves, R.W. An untargeted metabolomics approach for correlating pulse crop seed coat polyphenol profiles with antioxidant capacity and iron chelation ability. Molecules 2021, 26, 3833. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). DRV Finder, Dietary Reference Values for the EU-Minerals. 2022. Available online: https://multimedia.efsa.europa.eu/drvs/index.htm (accessed on 10 October 2022).
- Lopez-Martinez, L.X. Bioactive compounds of runner bean (Phaseolus coccineus L.). In Bioactive Compounds in Underutilized Vegetables and Legumes; Murthy, H.N., Paek, K.Y., Eds.; Springer: Cham, Switzerland, 2020; pp. 1–17. [Google Scholar]
- de Oliveira, A.P.; Mateó, B.D.S.O.; Fioroto, A.M.; de Oliveira, P.V.; Naozuka, J. Effect of cooking on the bioaccessibility of essential elements in different varieties of beans (Phaseolus vulgaris L.). J. Food Comp. Anal. 2018, 67, 135–140. [Google Scholar] [CrossRef]
- Guild, G.E.; Paltridge, N.G.; Andersson, M.S.; Stangoulis, J.C. An energy-dispersive X-ray fluorescence method for analysing Fe and Zn in common bean, maize and cowpea biofortification programs. Plant Soil 2017, 419, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Alvarado-López, A.N.; Gómez-Oliván, L.M.; Heredia, J.B.; Baeza-Jiménez, R.; Garcia-Galindo, H.S.; Lopez-Martinez, L.X. Nutritional and bioactive characteristics of Ayocote bean (Phaseolus coccienus L.): An underutilized legume harvested in Mexico. CYTA J. Food 2019, 17, 199–206. [Google Scholar] [CrossRef]
- Ciurescu, G.; Toncea, I.; Ropotă, M.; Hăbeanu, M. Seeds composition and their nutrients quality of some pea (Pisum sativum L.) and lentil (Lens culinaris Medik.) cultivars. Rom. Agric. Res. 2018, 35, 101–108. Available online: https://www.incda-fundulea.ro/rar.htm (accessed on 10 October 2022).
- Lizarazo, C.I.; Yli-Halla, M.; Stoddard, F.L. Pre-crop effects on the nutrient composition and utilization efficiency of faba bean (Vicia faba L.) and narrow-leafed lupin (Lupinus angustifolius L.). Nutr. Cycl. Agroecosys. 2015, 103, 311–327. [Google Scholar] [CrossRef]
- Khazaei, H.; Vandenberg, A. Seed mineral composition and protein content of faba beans (Vicia faba L.) with contrasting tannin contents. Agronomy 2020, 10, 511. [Google Scholar] [CrossRef]
- Farooq, M.; Hussain, M.; Usman, M.; Farooq, S.; Alghamdi, S.S.; Siddique, K.H. Impact of abiotic stresses on grain composition and quality in food legumes. J. Agric. Food Chem. 2018, 66, 8887–8897. [Google Scholar] [CrossRef]
- Türk, Z. Effects of different harvesting times on protein content and mineral nutrient values of some lentil (Lens culinaris) cultivars seeds. J. Plant Nutr. 2020, 43, 563–577. [Google Scholar] [CrossRef]
- Ereifej, K.I.; Al-Karaki, G.N.; Hammouri, M.K. Seed chemical composition of improved chickpea cultivars grown under semiarid Mediterranean conditions. Int. J. Food Prop. 2001, 4, 239–246. [Google Scholar] [CrossRef]
- Wang, N.; Daun, J.K. Effect of variety and crude protein content on nutrients and certain antinutrients in field peas (Pisum sativum). J. Sci. Food Agric. 2004, 84, 1021–1029. [Google Scholar] [CrossRef]
- Wang, N.; Daun, J.K. Effects of variety and crude protein content on nutrients and anti-nutrients in lentils (Lens culinaris). Food Chem. 2006, 95, 493–502. [Google Scholar] [CrossRef]
- Haug, W.; Lantzsch, H.J. Sensitive method for the rapid determination of phytate in cereals and cereal products. J. Sci. Food Agric. 1983, 34, 1423–1426. [Google Scholar] [CrossRef]
- Šibul, F.; Orčić, D.; Vasić, M.; Anačkov, G.; Nađpal, J.; Savić, A.; Mimica-Dukić, N. Phenolic profile, antioxidant and anti-inflammatory potential of herb and root extracts of seven selected legumes. Ind. Crops Prod. 2016, 83, 641–653. [Google Scholar] [CrossRef]
- Šibul, F.S.; Orčić, D.Z.; Svirčev, E.; Mimica-Dukić, N.M. Optimization of extraction conditions for secondary biomolecules from various plant species. Hem. Ind. 2016, 70, 473–483. [Google Scholar] [CrossRef]
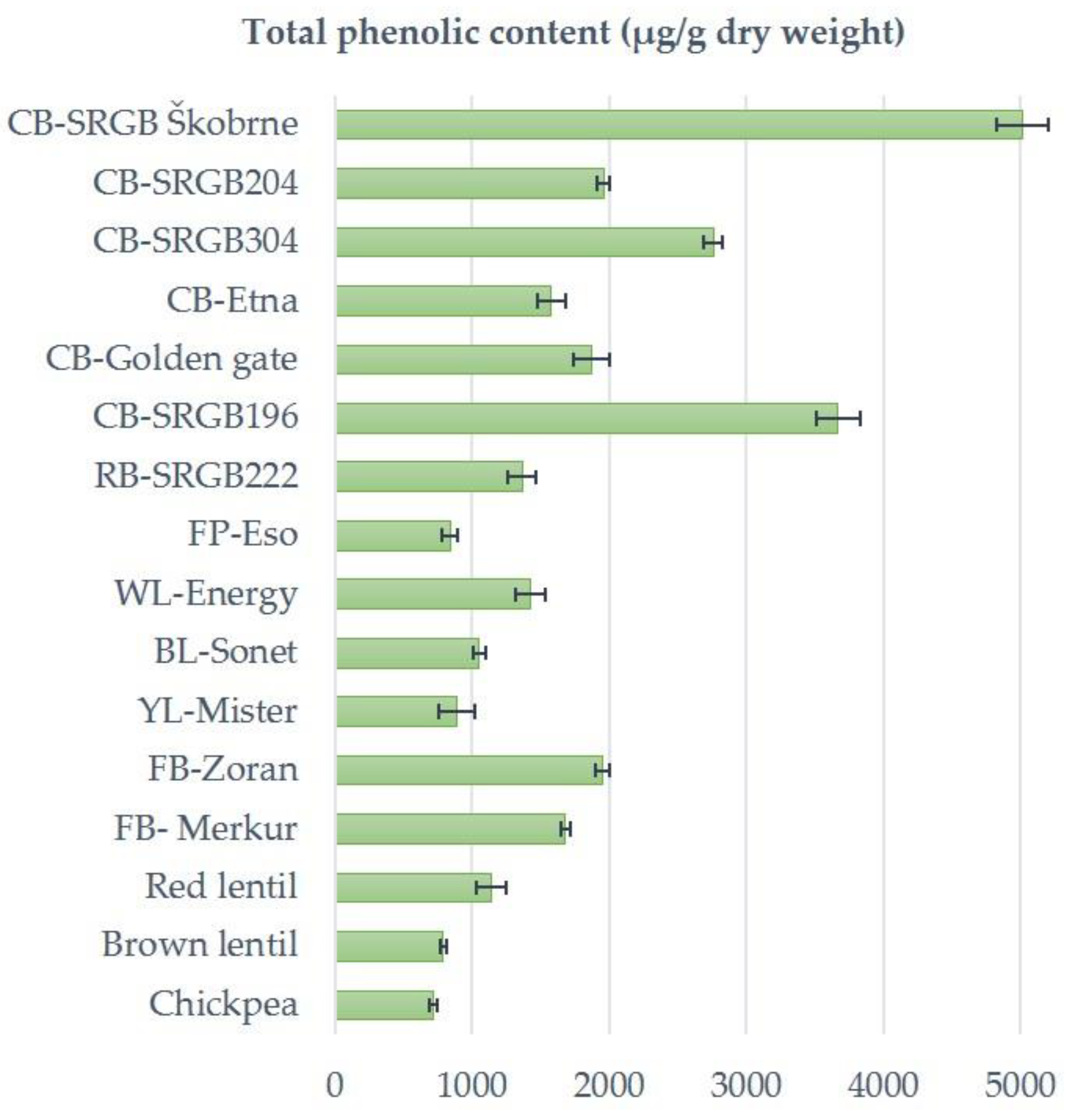

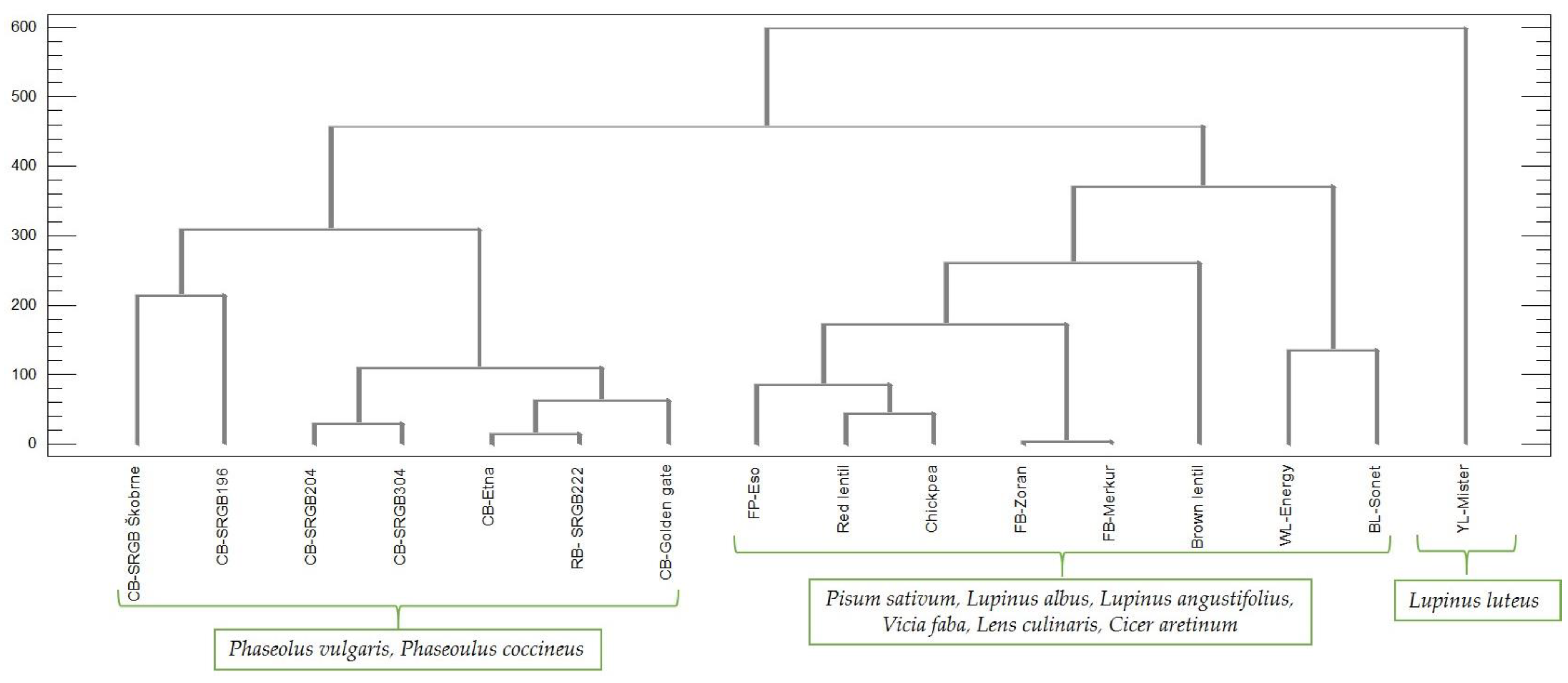
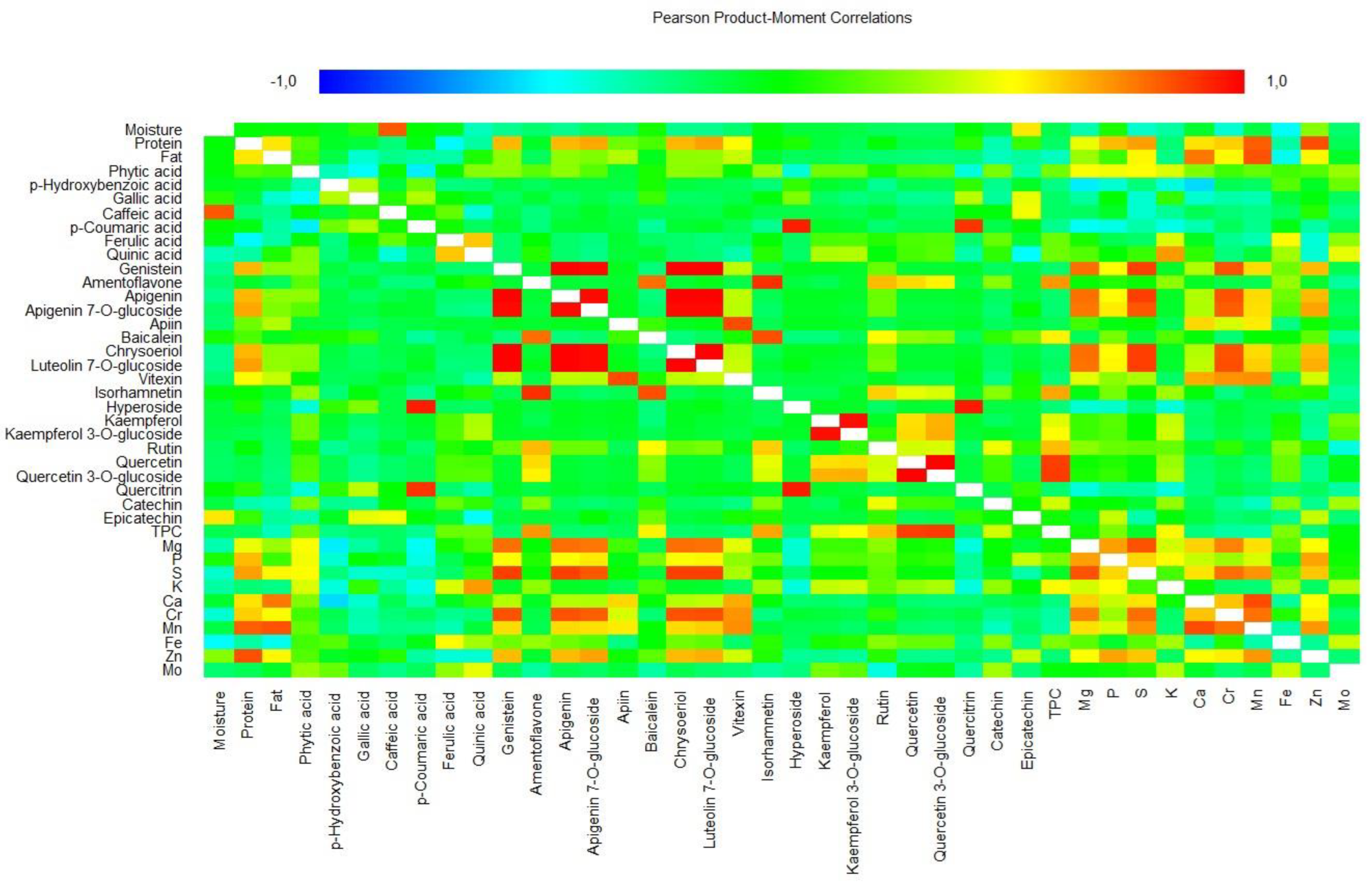
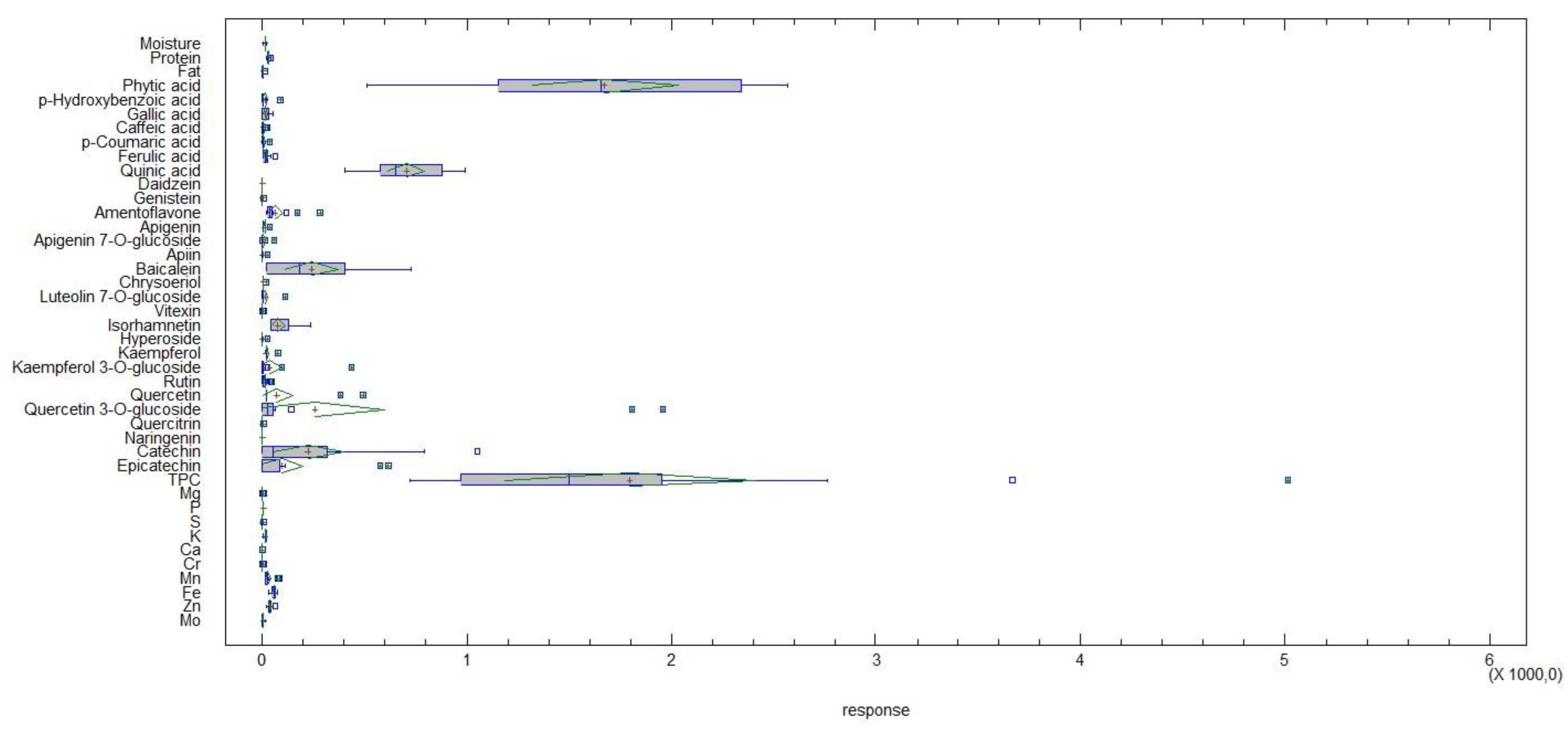
| Species | Common Name | Code/ Cultivar Name | Moisture (%) | Crude Protein (%) | Crude Fat (%) | Phytic Acid (mg/100 g DW) |
|---|---|---|---|---|---|---|
| Phaseolus vulgaris L. | Common bean | SRGB Škobrne | 9.0 | 23.9 | 1.0 | 1745 |
| SRGB204 | 8.9 | 24.0 | 0.6 | 2566 | ||
| SRGB304 | 9.1 | 22.1 | 1.4 | 1743 | ||
| Etna | 11.2 | 20.2 | 1.3 | 1401 | ||
| Golden gate | 9.1 | 24.3 | 0.8 | 2550 | ||
| SRGB196 | 9.4 | 23.0 | 0.9 | 2206 | ||
| Phaseolus coccineus L. | Runner bean | SRGB222 | 8.1 | 18.0 | 2.0 | 1115 |
| Pisum sativum L. subsp. arvense | Field pea | Eso | 15.6 | 18.5 | 1.6 | 2399 |
| Lupinus albus L. | White lupin | Energy | 13.5 | 38.4 | 8.5 | 2315 |
| Lupinus angustifolius L. | Blue lupin | Sonet | 8.4 | 31.7 | 5.4 | 1386 |
| Lupinus luteus L. | Yellow lupin | Mister | 7.8 | 43.1 | 4.8 | 2368 |
| Vicia faba L. var. minor | Faba bean | Zoran | 13.6 | 28.3 | 0.9 | 1564 |
| Merkur | 14.4 | 29.4 | 0.8 | 507 | ||
| Lens culinaris L. | Red lentil | unknown * | 9.7 | 25.9 | 1.2 | 1191 |
| Brown lentil | unknown * | 9.4 | 27.6 | 0.8 | 612 | |
| Cicer aretinum L. | Chickpea | unknown * | 9.2 | 19.1 | 5.4 | 1116 |
| Range | 7.8–15.6 | 16.7–43.1 | 0.6–8.5 | 507–2566 | ||
| Compound | Common Bean | Runner Bean | Field Pea | |||||
|---|---|---|---|---|---|---|---|---|
| SRGB Škobrne | SRGB204 | SRGB304 | Etna | Golden Gate | SRGB196 | SRGB222 | Eso | |
| p-Hydroxybenzoic acid | <0.16 ** | 2.98 (0.18) | <0.16 | <0.16 | <0.16 | 2.54 (0.15) | 5.88 (0.35) | 4.21 (0.25) |
| Gallic acid | <0.12 | 31.8 (2.9) | <0.12 | <0.12 | <0.12 | <0.12 | 49.5 (4.5) | <0.12 |
| Caffeic acid | <0.8 | <0.8 | <0.8 | 10.2 (0.7) | <0.8 | <0.8 | <0.8 | 24.8 (1.7) |
| p-Coumaric acid | 1.38 (0.12) | 1.63 (0.15) | 2.22 (0.20) | 10.90 (0.98) | 1.09 (0.10) | 3.17 (0.29) | 5.10 (0.46) | 4.86 (0.44) |
| Ferulic acid | 19.80 (0.02) | 20.50 (0.02) | 12.50 (0.01) | 65.40 (0.07) | 7.95 (0.01) | 24.40 (0.02) | 38.50 (0.04) | 16.80 (0.02) |
| Quinic acid | 646 (0.7) | 922 (0.9) | 767 (0.8) | 917 (0.9) | 709 (0.7) | 916 (0.9) | 992 (1.0) | 545 (0.6) |
| Genistein | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 |
| Amentoflavone | 282 (8.5) | 171 (5.1) | 118 (3.5) | 20.5 (0.6) | 19.8 (0.6) | 18.1 (0.5) | 21.6 (0.7) | 20.7 (0.6) |
| Apigenin | <8 | <8 | <8 | <8 | <8 | <8 | <8 | <8 |
| Apigenin 7-O-glucoside | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 |
| Apiin | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 |
| Baicalein | 724 (2.2) | 579 (1.7) | 518 (1.6) | <16 | <16 | <16 | <16 | <16 |
| Chrysoeriol | <4 | <4 | <4 | <4 | <4 | <4 | <4 | <4 |
| Luteolin 7-O-glucoside | 5.94 (0.18) | 4.96 (0.15) | 4.95 (0.15) | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 |
| Vitexin | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 |
| Isorhamnetin | 236 (14) | 174 (10) | 149 (9) | <40 | <40 | <40 | <40 | <40 |
| Hyperoside | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 |
| Kaempferol | <16 | <16 | <16 | <16 | <16 | 77.2 (5.4) | <16 | <16 |
| Kaempferol 3-O-glucoside | 16.2 (0.7) | <0.08 | 95.9 (3.8) | 8.62 (0.34) | <0.08 | 436 (17) | <0.08 | 2.85 (0.11) |
| Rutin | 40.1 (1.2) | 6.15 (0.18) | 46.3 (1.4) | 9.38 (0.28) | <2 | 7.82 (0.23) | 10.90 (0.33) | 6.87 (0.21) |
| Quercetin | 494 (1.5) | <16 | <16 | <16 | <16 | 379 (1.1) | <16 | <16 |
| Quercetin 3-O-glucoside | 1958 (59) | 44.6 (1.3) | 140 (4.2) | 31.0 (0.9) | <0.06 | 1804 (54) | 24.3 (0.7) | <0.06 |
| Quercitrin | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 |
| Catechin | 589 (6) | <0.4 | 793 (8) | 421 (4) | 1052 (11) | <0.4 | 216 (2) | 212 (2) |
| Epicatechin | <0.4 | <0.4 | 112.8 (1.3) | 83.4 (0.8) | 81.4 (0.8) | <0.4 | <0.4 | <0.4 |
| Compound | White Lupin | Blue Lupin | Yellow Lupin | Faba Bean | Red Lentil | Brown Lentil | Chickpea | |
|---|---|---|---|---|---|---|---|---|
| Energy | Sonet | Mister | Zoran | Merkur | ||||
| p-Hydroxybenzoic acid | <0.16 ** | <0.16 | <0.16 | 8.65 (0.52) | 7.55 (0.45) | 88.3 (5.3) | 17.7 (1.1) | 18.4 (1.1) |
| Gallic acid | <0.12 | <0.12 | <0.12 | 41.1 (3.7) | 34.9 (3.1) | 31.4 (2.8) | 31.4 (2.8) | <0.12 |
| Caffeic acid | <0.8 | <0.8 | <0.8 | 15.0 (1.1) | 10.9 (0.8) | <0.8 | <0.8 | <0.8 |
| p-Coumaric acid | <0.2 | 1.72 (0.15) | 2.55 (0.23) | 4.77 (0.43) | 6.00 (0.54) | 7.76 (0.70) | 35.80 (3.22) | <0.2 |
| Ferulic acid | 2.88 (0.003) | 9.31 (0.009) | 2.61 (0.003) | 10.60 (0.01) | 6.04 (0.006) | 4.87 (0.005) | 4.14 (0.004) | 2.61 (0.003) |
| Quinic acid | 844 (0.8) | 628 (0.6) | 569 (0.6) | 407 (0.4) | 525 (0.5) | 610 (0.6) | 574 (0.6) | 653 (0.7) |
| Genistein | <0.2 | <0.2 | 4.14 (0.29) | <0.2 | <0.2 | <0.2 | <0.2 | <0.2 |
| Amentoflavone | 50.6 (1.5) | 36.8 (1.1) | 34.1 (1.0) | 45.4 (1.4) | 40.3 (1.2) | 29.5 (0.9) | 27.9 (0.8) | 45.4 (1.4) |
| Apigenin | <8 | <8 | 35.9 (2.5) | <8 | <8 | <8 | <8 | <8 |
| Apigenin 7-O-glucoside | <0.2 | 1.51 (0.08) | 59.6 (3.0) | <0.2 | 13.0 (0.7) | <0.2 | <0.2 | <0.2 |
| Apiin | <0.06 | 20.1 (1.0) | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 |
| Baicalein | 399 (1.2) | 341 (1.0) | <16 | 405 (1.2) | 354 (1.1) | 368 (1.1) | <16 | <16 |
| Chrysoeriol | <4 | <4 | 17.2 (0.5) | <4 | <4 | <4 | <4 | <4 |
| Luteolin 7-O-glucoside | 4.95 (0.15) | 5.57 (0.17) | 113 (3.4) | 4.88 (0.15) | 4.76 (0.14) | <0.2 | 6.03 (0.18) | <0.2 |
| Vitexin | <0.2 | 5.25 (0.26) | 2.70 (0.14) | 1.88 (0.09) | <0.2 | <0.2 | <0.2 | <0.2 |
| Isorhamnetin | 124 (7) | <40 | <40 | 129 (8) | <40 | <40 | <40 | <40 |
| Hyperoside | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | <0.06 | 20.4 (1.2) | <0.06 |
| Kaempferol | <16 | <16 | <16 | <16 | <16 | <16 | <16 | <16 |
| Kaempferol 3-O-glucoside | <0.08 | <0.08 | <0.08 | 2.15 (0.09) | 1.54 (0.06) | <0.08 | <0.08 | <0.08 |
| Rutin | <2 | 3.38 (0.10) | 22.1 (0.7) | 10.3 (0.3) | 14.3 (0.4) | <2 | <2 | <2 |
| Quercetin | <16 | <16 | <16 | <16 | <16 | <16 | <16 | <16 |
| Quercetin 3-O-glucoside | <0.06 | <0.06 | 23.4 (0.7) | 65.0 (2.0) | 45.0 (1.4) | <0.06 | <0.06 | <0.06 |
| Quercitrin | <0.06 | <0.06 | <0.06 | 2.32 (0.14) | <0.06 | <0.06 | 7.04 (0.42) | <0.06 |
| Catechin | <0.4 | <0.4 | <0.4 | 184 (2) | 43.5 (0.4) | <0.4 | 66.1 (0.7) | <0.4 |
| Epicatechin | <0.4 | <0.4 | <0.4 | 613 (6) | 575 (6) | <0.4 | <0.4 | <0.4 |
| Common Name | Code/Cultivar Name | Macro-Minerals (g/kg DW) | Micro-Minerals (mg/kg DW) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mg | P | S | K | Ca | Cr | Mn | Fe | Zn | Mo | ||
| Common bean | SRGB Škobrne | 1.42 | 4.82 | 2.12 | 13.93 | 0.99 | 0.26 | 11.95 | 64.59 | 28.06 | 0.57 |
| SRGB204 | 1.64 | 5.90 | 2.28 | 19.05 | 1.18 | 0.18 | 12.35 | 69.91 | 27.94 | 3.19 | |
| SRGB304 | 1.55 | 5.00 | 2.16 | 14.41 | 0.94 | 0.33 | 10.97 | 57.75 | 28.93 | 0.85 | |
| Etna | 1.47 | 4.18 | 1.97 | 13.53 | 1.33 | 0.21 | 13.59 | 77.34 | 26.76 | 5.83 | |
| Golden gate | 1.67 | 5.87 | 2.38 | 16.25 | 1.18 | 0.19 | 11.86 | 58.74 | 27.63 | 8.46 | |
| SRGB196 | 1.73 | 5.87 | 2.20 | 17.84 | 0.81 | 0.21 | 9.45 | 56.92 | 20.32 | 4.69 | |
| Runner bean | SRGB222 | 1.76 | 4.65 | 1.99 | 17.77 | 1.12 | 0.29 | 12.65 | 52.70 | 23.02 | 2.43 |
| Field pea | Eso | 1.30 | 3.66 | 1.58 | 9.11 | 0.91 | 0.23 | 8.34 | 46.84 | 26.35 | 0.56 |
| White lupin | Energy | 1.46 | 6.19 | 2.88 | 12.32 | 1.98 | 0.21 | 72.79 | 30.39 | 52.88 | 3.11 |
| Blue lupin | Sonet | 1.75 | 4.89 | 2.23 | 11.05 | 2.27 | 1.69 | 79.54 | 43.93 | 34.87 | 1.72 |
| Yellow lupin | Mister | 2.93 | 7.47 | 4.69 | 12.08 | 1.83 | 2.88 | 82.92 | 64.87 | 63.27 | 1.41 |
| Faba bean | Zoran | 1.36 | 6.46 | 1.48 | 12.55 | 1.07 | 0.18 | 16.41 | 43.27 | 49.13 | 0.92 |
| Merkur | 1.40 | 6.19 | 1.64 | 12.67 | 1.02 | 0.16 | 16.79 | 46.88 | 44.55 | 0.97 | |
| Red lentil | not defined * | 0.76 | 3.72 | 1.82 | 9.22 | 0.24 | 0.15 | 13.65 | 65.89 | 33.00 | 5.26 |
| Brown lentil | not defined * | 0.78 | 3.14 | 1.57 | 7.75 | 0.78 | 0.26 | 9.62 | 47.32 | 19.62 | 0.54 |
| Chickpea | not defined * | 1.33 | 2.94 | 2.05 | 10.08 | 1.31 | 0.96 | 25.77 | 55.46 | 28.57 | 2.74 |
| Range | 0.76–2.93 | 2.94–7.47 | 1.48–4.69 | 7.75–19.05 | 0.24–2.27 | 0.15–2.88 | 8.34–82.92 | 30.39–77.34 | 19.62–63.27 | 0.54–8.46 | |
| Compound | Precursor Ion (m/z) | Product Ion (m/z) | Retention Time (min) |
|---|---|---|---|
| p-Hydroxybenzoic acid | 137 | 93 | 1.08 |
| Gallic acid | 169 | 125 | 0.58 |
| Caffeic acid | 179 | 135 | 1.18 |
| p-Coumaric acid | 163 | 119 | 1.69 |
| Ferulic acid | 193 | 134 | 1.90 |
| Quinic acid | 191 | 85 | 0.52 |
| Genistein | 269 | 133 | 4.12 |
| Amentoflavone | 537 | 375 | 5.78 |
| Apigenin | 269 | 117 | 4.71 |
| Apigenin 7-O-glucoside | 431 | 268 | 2.81 |
| Apiin | 563 | 269 | 2.60 |
| Baicalein | 445 | 269 | 3.40 |
| Chrysoeriol | 299 | 284 | 4.82 |
| Luteolin 7-O-glucoside | 447 | 285 | 2.13 |
| Vitexin | 431 | 311 | 1.90 |
| Isorhamnetin | 315 | 300 | 4.79 |
| Hyperoside | 463 | 300 | 2.16 |
| Kaempferol | 285 | 285 | 4.55 |
| Kaempferol 3-O-glucoside | 447 | 284 | 2.80 |
| Rutin | 609 | 300 | 2.33 |
| Quercetin | 301 | 151 | 3.74 |
| Quercetin 3-O-glucoside | 463 | 300 | 2.25 |
| Quercitrin | 447 | 300 | 2.75 |
| Catechin | 289 | 245 | 0.74 |
| Epicatechin | 289 | 245 | 0.95 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sinkovič, L.; Pipan, B.; Šibul, F.; Nemeš, I.; Tepić Horecki, A.; Meglič, V. Nutrients, Phytic Acid and Bioactive Compounds in Marketable Pulses. Plants 2023, 12, 170. https://doi.org/10.3390/plants12010170
Sinkovič L, Pipan B, Šibul F, Nemeš I, Tepić Horecki A, Meglič V. Nutrients, Phytic Acid and Bioactive Compounds in Marketable Pulses. Plants. 2023; 12(1):170. https://doi.org/10.3390/plants12010170
Chicago/Turabian StyleSinkovič, Lovro, Barbara Pipan, Filip Šibul, Ivana Nemeš, Aleksandra Tepić Horecki, and Vladimir Meglič. 2023. "Nutrients, Phytic Acid and Bioactive Compounds in Marketable Pulses" Plants 12, no. 1: 170. https://doi.org/10.3390/plants12010170
APA StyleSinkovič, L., Pipan, B., Šibul, F., Nemeš, I., Tepić Horecki, A., & Meglič, V. (2023). Nutrients, Phytic Acid and Bioactive Compounds in Marketable Pulses. Plants, 12(1), 170. https://doi.org/10.3390/plants12010170









