Biological Traits and Genetic Relationships Amongst Cultivars of Three Species of Tagetes (Asteraceae)
Abstract
1. Introduction
2. Results
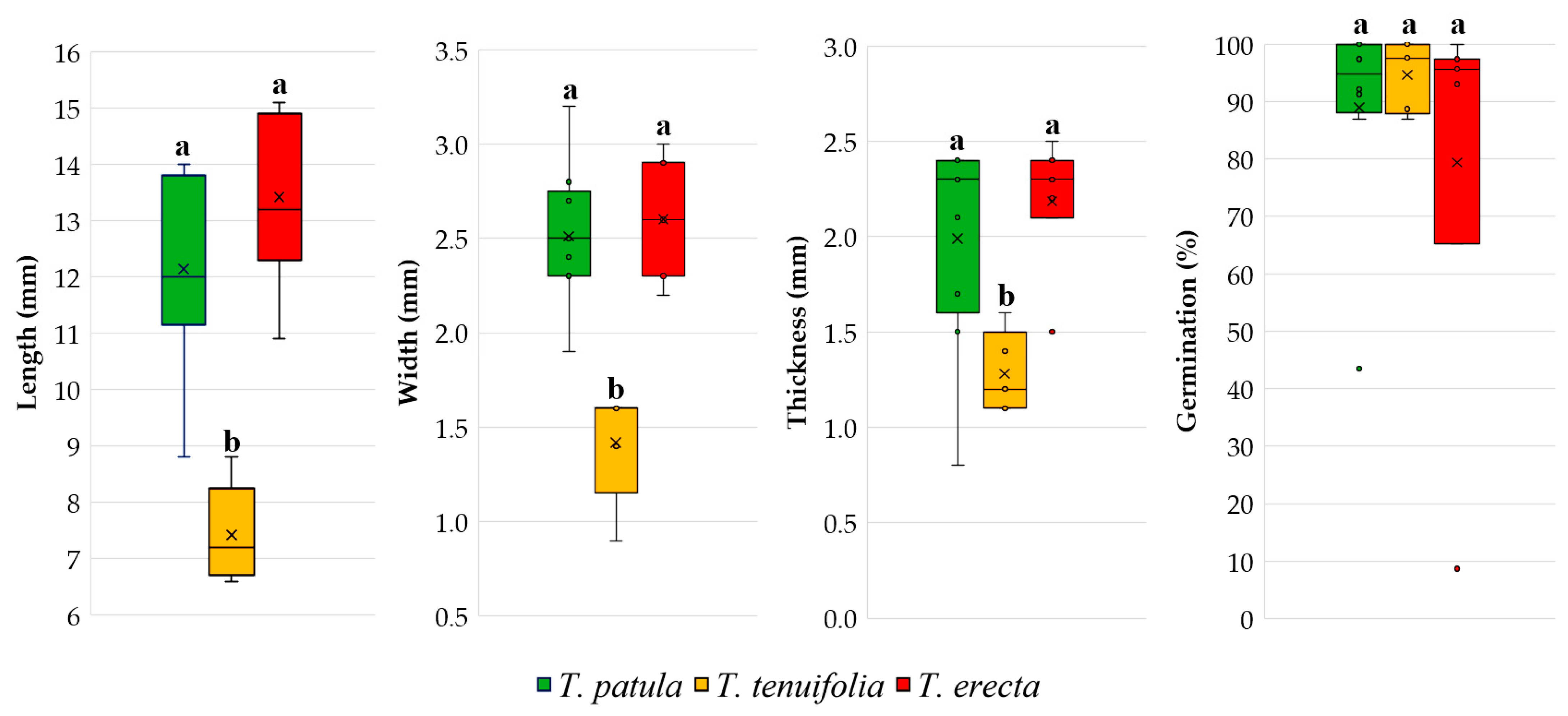
2.1. Seed Characteristics
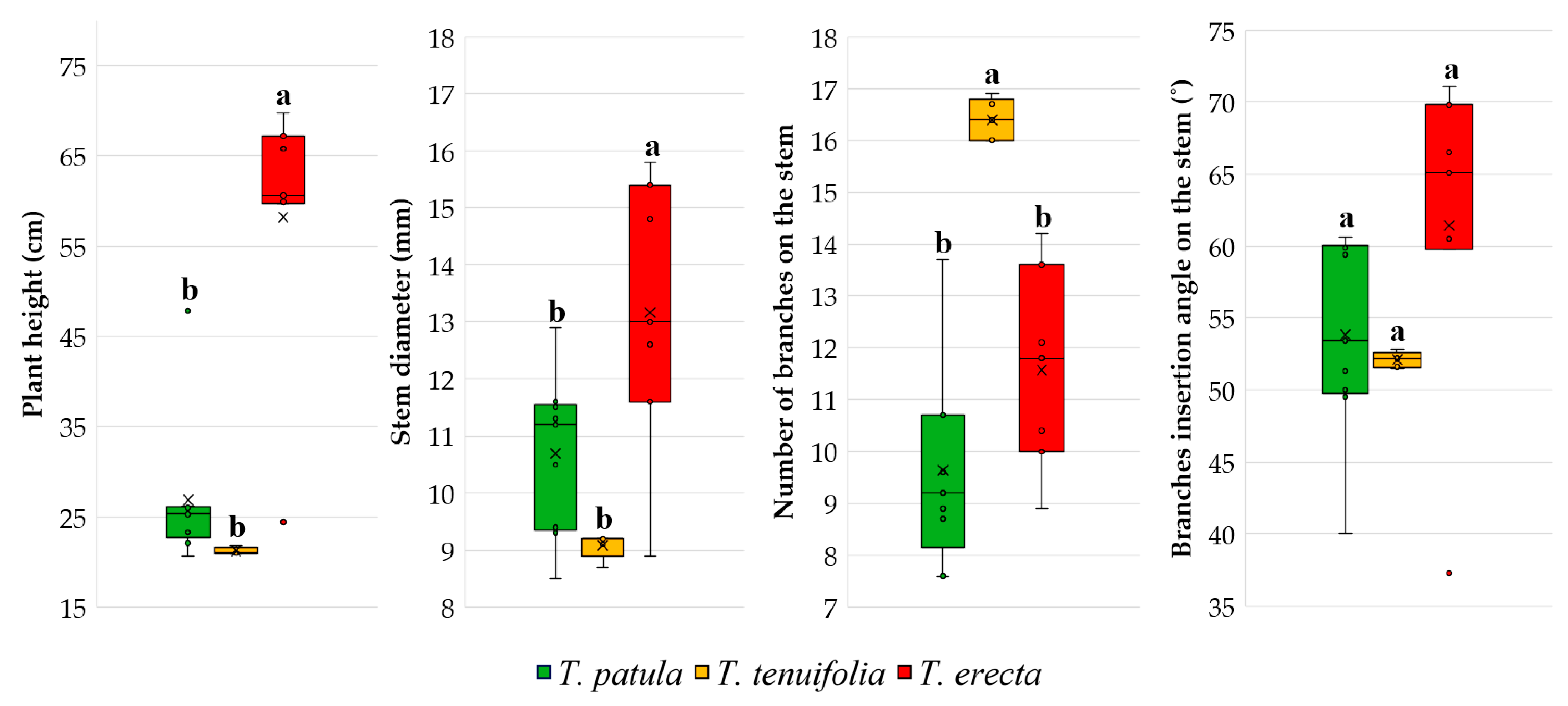
2.2. Plant Growth Traits
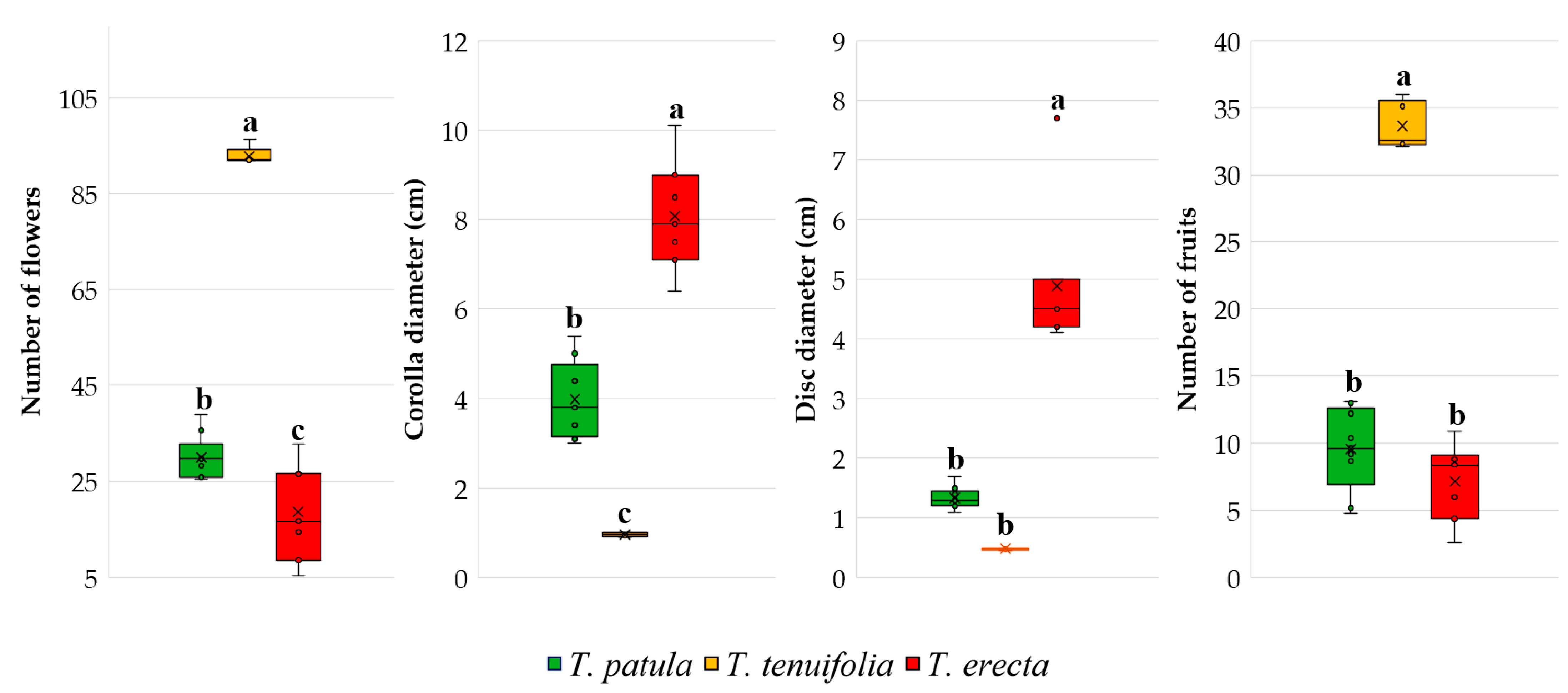
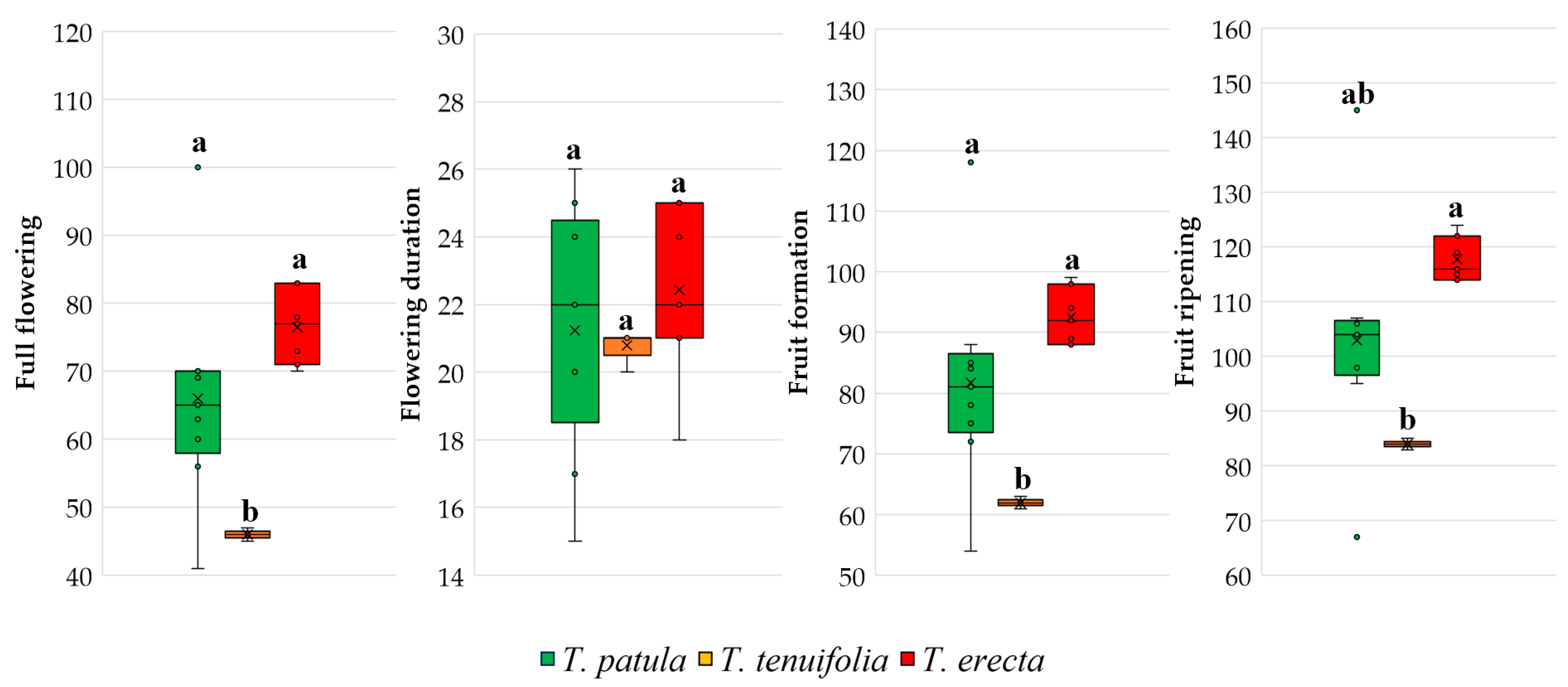
2.3. Main Traits Related to Flowers and Fruits
2.4. Phenotypic Correlations between the Analyzed Traits
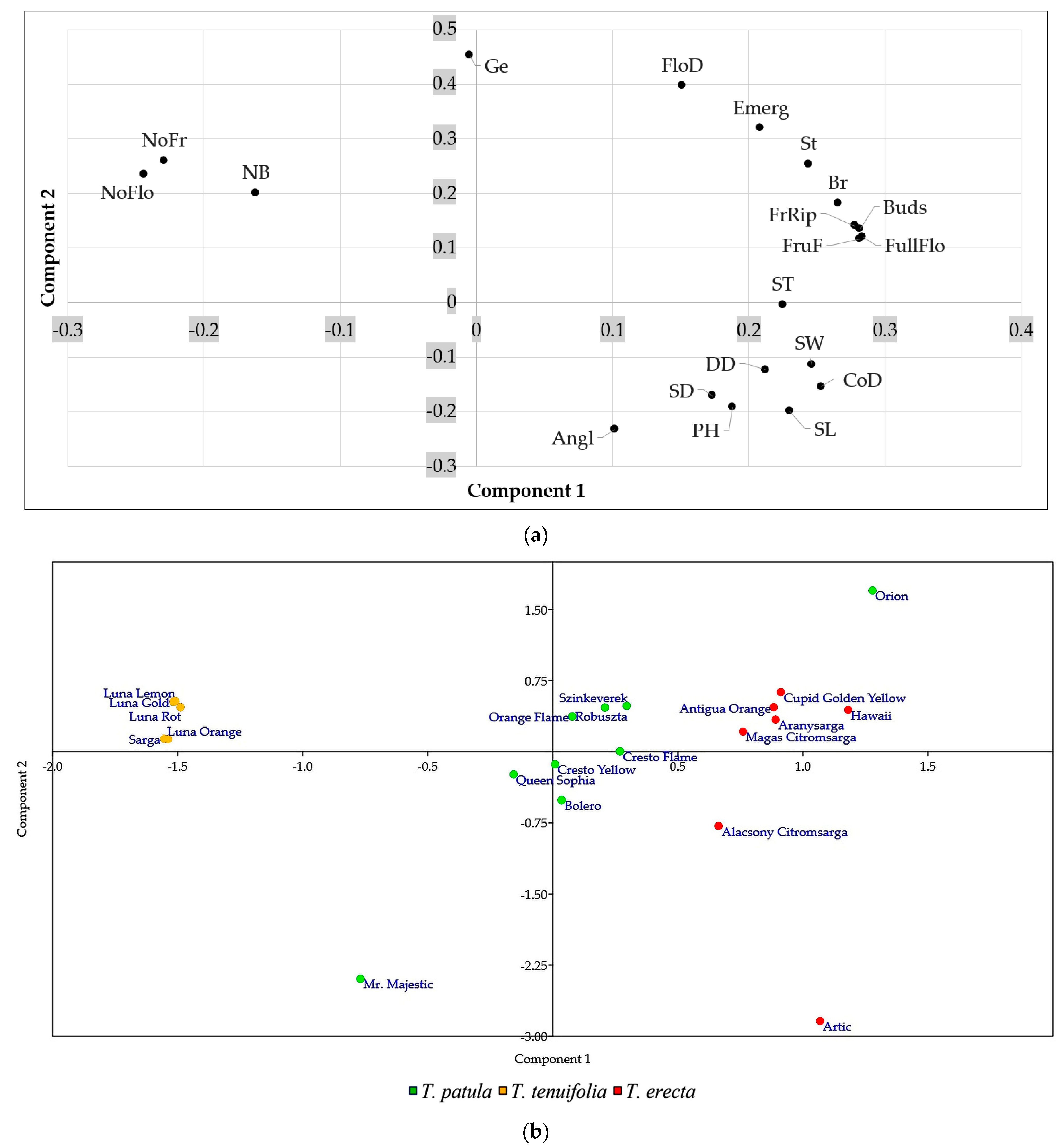
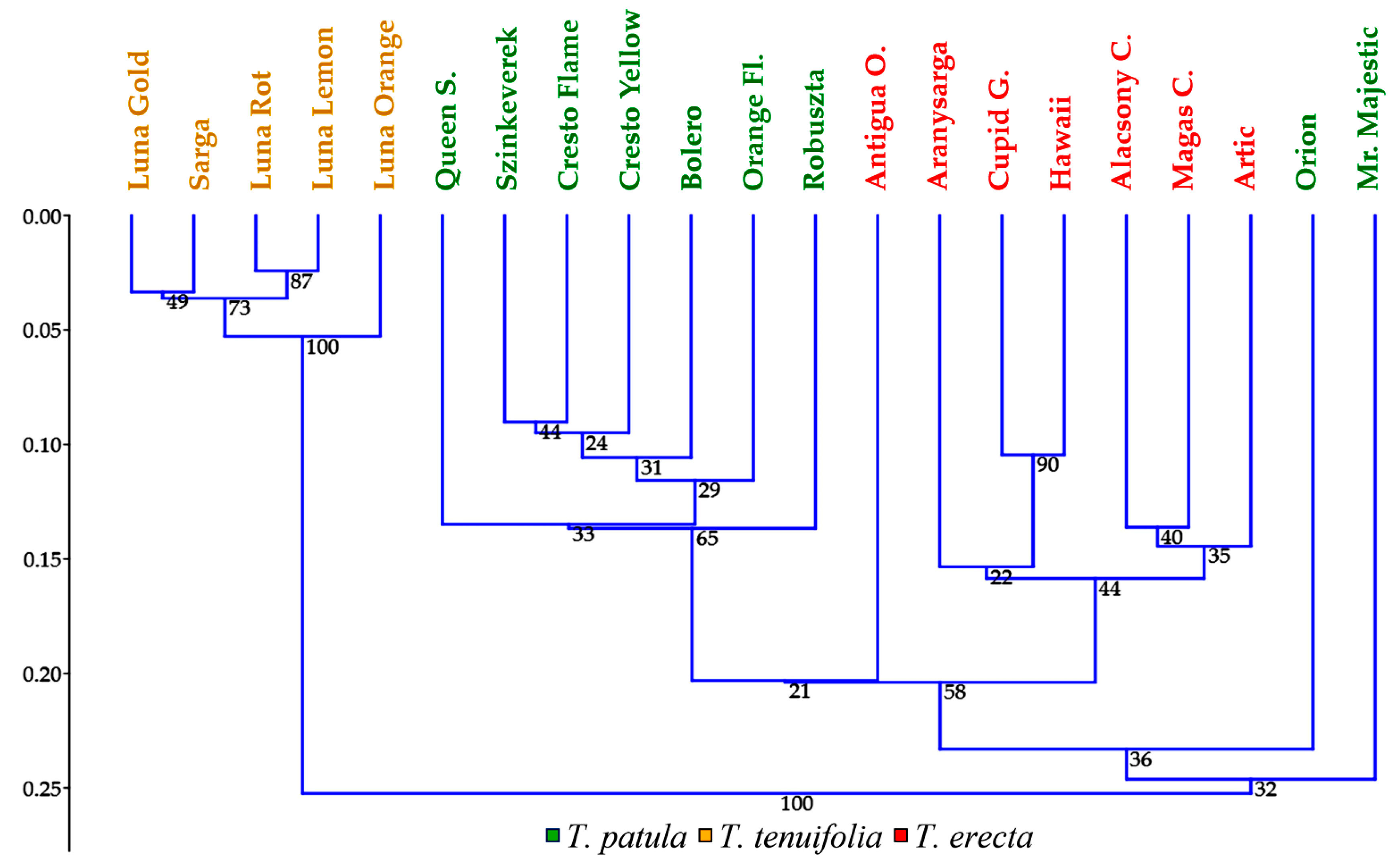
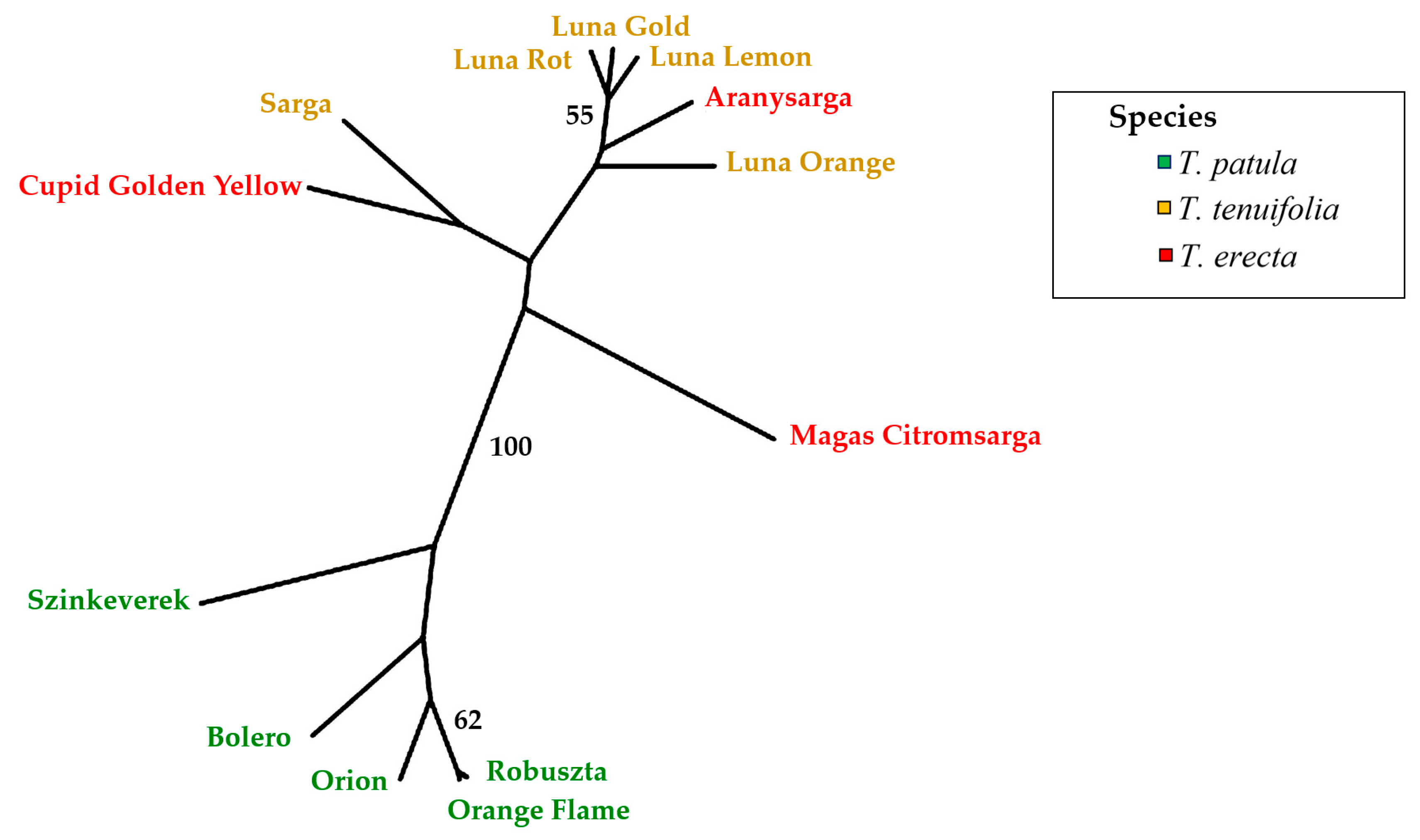
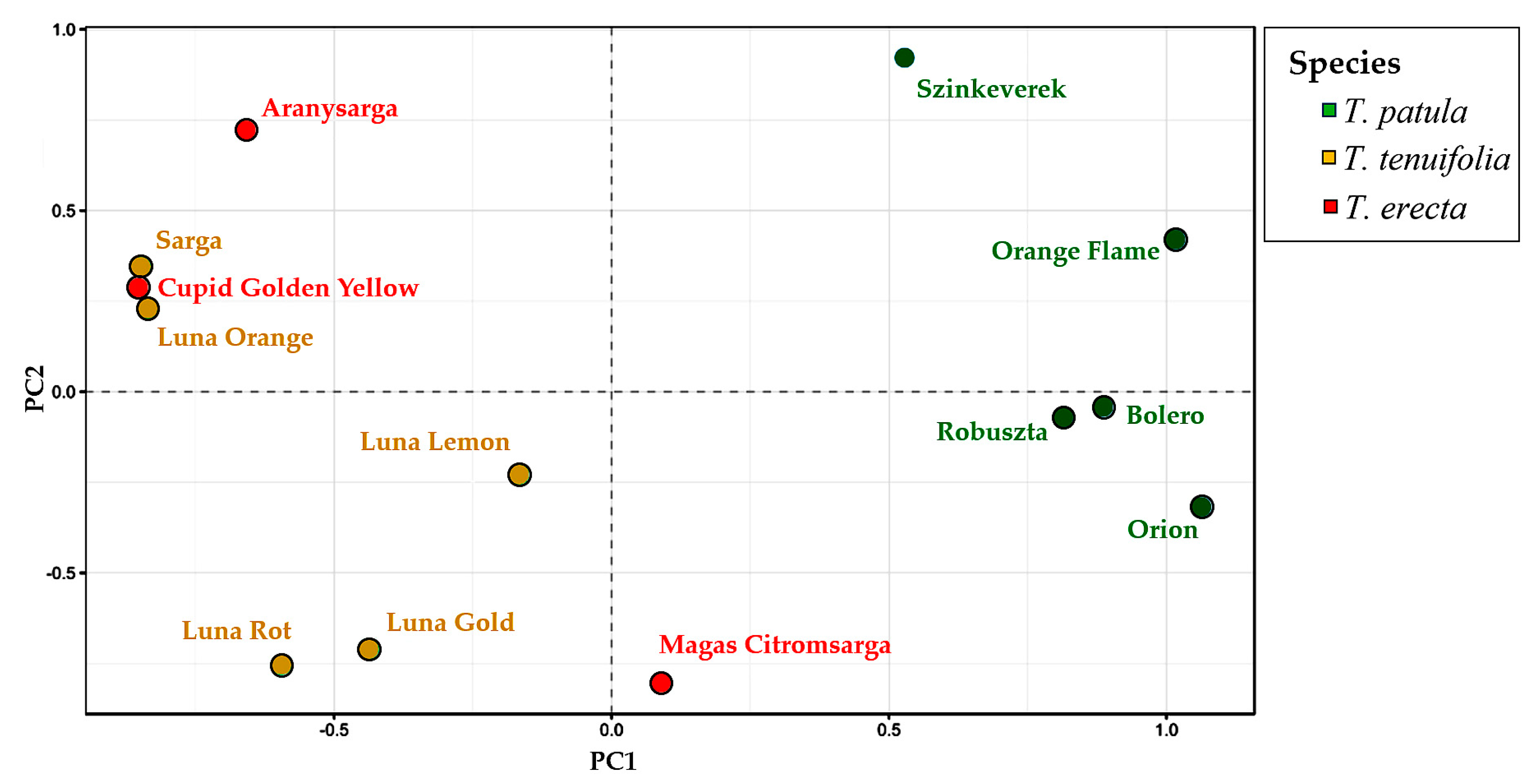
2.5. Multivariate Analysis of Traits (Principal Component Analysis) and Genetic Variation
3. Discussion
3.1. Morphological Traits of the Seeds and Germination
3.2. Vegetative and Reproductive Traits and Phenophases
3.3. Usefulness of the Statistical and Molecular Methods Used in the Evaluation and Selection of Genotypes
4. Materials and Methods
4.1. Plant Material
4.2. Analysis of Seed Characteristics and Seed Germination
4.3. Description of the Study Site, Phenological Studies and Morphologic Traits Analysed
4.4. Genetic Diversity Analysis
4.5. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Soule, J. Infrageneric systematics of tagetes. In Proceedings of the International Compositae Conference, Compositae: Systematics, Kew, UK, 24 July–5 August 1994; pp. 435–443. [Google Scholar]
- Salehi, B.; Valussi, M.; Morais-Braga, M.F.; Carneiro, J.N.; Leal, A.L.; Coutinho, H.D.; Vitalini, S.; Kręgiel, D.; Antolak, H.; Sharifi-Rad, M.; et al. Tagetes spp. Essential oils and other extracts: Chemical characterization and biological activity. Molecules 2018, 23, 2847. [Google Scholar] [CrossRef] [PubMed]
- Priyanka, D.; Shalini, T.; Verma, N.K. A brief study of marigold (Tagetes species): A review. Int. Res. J. Pharm. 2013, 4, 43–48. [Google Scholar]
- Vasudevan, P.; Kashyap, S.; Sharma, S. Tagetes: A multipurpose plant. Bioresour. Technol. 1997, 62, 29–35. [Google Scholar] [CrossRef]
- Singh, P.; Krishna, A.; Kumar, V.; Krishna, S.; Singh, K.; Gupta, M.; Singh, S. Chemistry and biology of industrial crop Tagetes species: A review. J. Essent. Oil Res. 2016, 28, 1–14. [Google Scholar] [CrossRef]
- Mir, R.A.; Ahanger, M.A.; Agarwal, R.M. Marigold: From mandap to medicine and from ornamentation to remediation. Am. J. Plant Sci. 2019, 10, 309–338. [Google Scholar] [CrossRef]
- Santos, P.C.; Santos, V.H.M.; Mecina, G.F.; Andrade, A.R.; Fegueiredo, P.A.; Moraes, V.M.O.; Silva, L.P.; Silva, R.M.G. Phytotoxicity of Tagetes erecta L. and Tagetes patula L. on plant germination and growth. S. Afr. J. Bot. 2015, 100, 114–121. [Google Scholar] [CrossRef]
- Tapia-Vázquez, I.; Montoya-Martínez, A.C.; De los Santos-Villalobos, S.; Ek-Ramos, M.J.; Montesinos-Matías, R.; Martínez-Anaya, C. Root-knot nematodes (Meloidogyne spp.) a threat to agriculture in Mexico: Biology, current control strategies, and perspectives. World J. Microb. Biotechnol. 2022, 38, 26. [Google Scholar] [CrossRef]
- Serrato-Cruz, M.Á.; Díaz-Cedillo, F.; Barajas-Pérez, J.S. Composición del aceite esencial en germoplasma de Tagetes filifolia Lag. de la región centro-sur de México. Agrociencia 2008, 42, 277–285. [Google Scholar]
- Cicevan, R.; Al-Hassan, M.; Sestras, A.; Boscaiu, M.; Zaharia, A.; Vicente, O.; Sestras, R. Comparative analysis of osmotic and ionic stress effects on seed germination in Tagetes (Asteraceae) cultivars. Propag. Ornam. Plants 2015, 15, 63–72. [Google Scholar]
- Cicevan, R.; Al Hassan, M.; Sestras, A.F.; Prohens, J.; Vicente, O.; Sestras, R.E.; Boscaiu, M. Screening for drought tolerance in cultivars of the ornamental genus Tagetes (Asteraceae). PeerJ 2016, 4, e2133. [Google Scholar] [CrossRef]
- Burlec, A.F.; Pecio, Ł.; Kozachok, S.; Mircea, C.; Corciovă, A.; Vereștiuc, L.; Cioancă, O.; Oleszek, W.; Hăncianu, M. Phytochemical profile, antioxidant activity, and cytotoxicity assessment of Tagetes erecta L. flowers. Molecules 2021, 26, 1201. [Google Scholar] [CrossRef] [PubMed]
- Pérez Gutierrez, R.; Hernández Luna, H.; Hernández Garrido, S. Antioxidant activity of Tagetes erecta essential oil. J. Chil. Chem. Soc. 2006, 51, 883–886. [Google Scholar] [CrossRef]
- Dharmagadda, V.; Naik, S.; Mittal, P.; Vasudevan, P. Larvicidal activity of Tagetes patula essential oil against three mosquito species. Bioresour. Technol. 2005, 96, 1235–1240. [Google Scholar] [CrossRef] [PubMed]
- Laosinwattana, C.; Wichittrakarn, P.; Teerarak, M. Chemical composition and herbicidal action of essential oil from Tagetes erecta L. leaves. Ind. Crops Prod. 2018, 126, 129–134. [Google Scholar] [CrossRef]
- Sing, Y.; Gupta, A.; Kannojia, P. Tagetes erecta (Marigold)—A review on its phytochemical and medicinal properties. Curr. Med. Drugs Res. 2020, 4, 1–6. [Google Scholar]
- Zhang, H.; Xin, H.; Cong, R.; Li, Z.; Song, L.; Wei, Z. Cross compatibility analysis to identify suitable parents of Tagetes erecta and T. patula for heterotic hybrid breeding. Not. Bot. Horti Agrobot. Cluj-Napoca 2019, 47, 676–682. [Google Scholar] [CrossRef]
- Adams, D.W. Restoring American Gardens: An Encyclopedia of Heirloom Ornamental Plants, 1640–1940; Timber Press: Portland, OR, USA, 2004. [Google Scholar]
- Alvarado-Sansininea, J.J.; Sánchez-Sánchez, L.; López-Muñoz, H.; Escobar, M.L.; Flores-Guzmán, F.; Tavera-Hernández, R.; Jiménez-Estrada, M. Quercetagetin and patuletin: Antiproliferative, necrotic and apoptotic activity in tumor cell lines. Cancer Res. 2018, 23, 2579. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Sandoval, J. Tagetes erecta (Mexican marigold). Invasive Species Compendium; CABI: Wallingford, UK, 2018. [Google Scholar]
- Serrato-Cruz, M.A. Colecta, Caracterizacion y Aprovechamiento de Tagetes erecta L. Como Ornamental. Avances. Metodologia de Investigacion.; Departamento de Fitotecnia, Universidad Autonoma Chapingo: Texcoco, Mexico, 2004. [Google Scholar]
- Sachin, T.M.; Homraj, S. A review of marigold’s beneficial aspects. Pharma Innov. J. 2021, 10, 422–427. [Google Scholar]
- Heuze, V.; Tran, G.; Hassoun, P.; Lebas, F. Mexican marigold (Tagetes erecta); Feedipedia, a programme by INRAE, CIRAD, AFZ and FAO: Rome, Italy, 2017. [Google Scholar]
- Saxena, P.; Sonwani, S.; Gupta, A.K. Assessment of air pollution tolerance index (APTI) for selected ornamental plants in urban metropolis of Northern India. Asian J. Chem. 2021, 33, 2150–2156. [Google Scholar] [CrossRef]
- Sîrbu, C.; Oprea, A.; Eliáš, P.J.; Ferus, P. New contribution to the study of alien Flora in Romania. J. Plant Develop. 2011, 18, 121–134. [Google Scholar]
- Schiavinato, D.J.; Gutierrez, D.G.; Bartoli, A. Typifications and nomenclatural clarifications in South American Tagetes (Asteraceae, Tageteae). Phytotaxa 2017, 326, 175–188. [Google Scholar] [CrossRef]
- Gilman, F.E. Tagetes patula French Marigold; Environmental Horticulture, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida: Gainesville, FL, USA, 2011; Volume FPS-571. [Google Scholar]
- Lim, T.K. Tagetes tenuifolia. In Edible Medicinal and Non-Medicinal Plants: Volume 7, Flowers; Lim, T.K., Ed.; Springer: Dordrecht, The Netherlands, 2014; pp. 469–472. [Google Scholar]
- Bosma, T.L.; Conway, K.E.; Dole, J.M.; Maness, N.O. Sowing dates and priming influence African marigold field emergence. HortTechnology 2003, 13, 487–493. [Google Scholar]
- Ilbi, H.; Powell, A.A.; Alan, O. Single radicle emergence count for predicting vigour of marigold (Tagetes spp.) seed lots. Seed Sci. Technol. 2020, 48, 381–389. [Google Scholar] [CrossRef]
- Sestras, R.E.; Pamfil, D.; Ardelean, M.; Botez, C.; Sestras, A.F.; Mitre, I.; Dan, C.; Mihalte, L. Use of phenotypic and MAS selection based on bulk segregant analysis to reveal the genetic variability induced by artificial hybridization in apple. Not. Bot. Horti Agrobot. Cluj-Napoca 2009, 37, 273–277. [Google Scholar] [CrossRef]
- Gheorghe, R.N.; Popescu, C.F.; Pamfil, D.; Ciocirlan, C.N.; Sestras, R. Genetic diversity of some Romanian grapevine cultivars as revealed by microsatellite markers. Rom. Biotechnol. Lett. 2010, 15, 26–31. [Google Scholar]
- Konyeme, T.E.; Agogbua, J.U.; Adewale, D.B.; Ogazie, C. Intraspecific diversity of Vigna subterranea accessions obtained from multinational sources. Not. Sci. Biol. 2021, 13, 10829. [Google Scholar] [CrossRef]
- Todea Morar, I.M.; Rensen, S.; Vilanova, S.; Boscaiu, M.; Holonec, L.; Sestras, A.F.; Vicente, O.; Prohens, J.; Sestras, R.E.; Plazas, M. Genetic relationships and reproductive traits of Romanian populations of silver fir (Abies alba): Implications for the sustainable management of local populations. Sustainability 2020, 12, 4199. [Google Scholar] [CrossRef]
- Kovács, Z.; Csergő, A.M.; Csontos, P.; Höhn, M. Ex situ conservation in botanical gardens–Challenges and scientific potential preserving plant biodiversity. Not. Bot. Horti Agrobot. Cluj-Napoca 2021, 49, 12334. [Google Scholar] [CrossRef]
- Santhosh, N.; Tejaswini, S.K.; Seetharamu, G.K. Genetic diversity for morphological characters and biochemical components in African marigold. Int. J. Chem. Stud. 2018, 6, 624–627. [Google Scholar]
- Kumar, A.; Pratap, B.; Gautam, D.K.; Yadav, V.; Gangadhara, K.; Beer, K.; Singh, A.K.; Kumar, V. Variability, heritability and genetic advance studies in French marigold (Tagetes patula L.). J. Pharmacogn. Phytochem. 2019, 8, 1046–1048. [Google Scholar]
- Modi, P.; Jain, D.; Kachhwaha, S.; Kothari, S.L. Analysis of genetic diversity among Tagetes patula L. cultivars based on RAPD markers. Indian J. Hortic. 2013, 70, 549–554. [Google Scholar]
- Shahzadi, I.; Ahmad, R.; Waheed, U.; Shah, M.M. Genetic diversity analysis of Tagetes species using PCR based molecular markers. Pak. J. Bot. 2016, 48, 1593–1599. [Google Scholar]
- Whankaew, S.; Hasthanasombut, S.; Thaikert, R.; Tappiban, P.; Smith, D.R.; Triwitayakorn, K. Molecular characterization and genetic relationship of marigolds (Tagetes spp.) based on simple sequence repeat markers. Plant Genet. Resour. 2014, 12, 317–322. [Google Scholar] [CrossRef]
- Datta, S.K. Breeding of ornamentals: Success and technological status. Nucleus 2021. [Google Scholar] [CrossRef]
- Singh, D.; Vishwakarma, A. Heterosis breeding in marigold: An efficient tool towards crop improvement. Adv. Res. Agri. Vet. Sci. 2020, 1, 32−50. [Google Scholar]
- Patel, M.A.; Chawla, S.L.; Chavan, S.K.; Shah, H.P.; Patil Sudha, D. Genetic variability, heritability and genetic advance studies in marigold (Tagetes spp.) under the South Gujarat region. Electron. J. Plant Breed. 2019, 10, 272–276. [Google Scholar] [CrossRef]
- Love, J.; Graham, S.W.; Irwin, J.A.; Ashton, P.A.; Bretagnolle, F.; Abbott, R.J. Self-pollination, style length development and seed set in self-compatible Asteraceae: Evidence from Senecio vulgaris L. Plant Ecol. Divers. 2016, 9, 371–379. [Google Scholar] [CrossRef]
- Nei, M. Estimation of average heterozygosity and genetic distance from a small number of individuals. Genetics 1978, 89, 583–590. [Google Scholar] [CrossRef]
- Ördögh, M. The effect of different substrate on the morphological characteristics of Hungarian Tagetes patula cultivars. Acta Biol. Marisiensis 2021, 4, 73–82. [Google Scholar] [CrossRef]
- Afzal, I.; Qasim, M.; Basra, S.; Shahid, M. Does halopriming improve germination and seedling vigour in marigold (Tagetes spp.)? Seed Sci.Technol. 2009, 37, 436–445. [Google Scholar] [CrossRef]
- Mukhtar, K.; Afzal, I.; Qasim, M.; Basra, S.M.A.; Shahid, M. Does priming promote germination and early stand establishment of French marigold (Tagetes patula L.) seeds by inducing physiological and biochemical changes? Acta Sci. Pol. Hortorum Cultus 2013, 12, 13–21. [Google Scholar]
- Afzal, I.; Rahim, A.; Qasim, M.; Younis, A.; Nawaz, A.; Bakhtavar, M. Inducing salt tolerance in French marigold (Tagetes patula) through seed priming. Acta Sci. Pol. Hortorum Cultus 2017, 16, 109–118. [Google Scholar] [CrossRef][Green Version]
- Wilczek, A.M.; Burghardt, L.T.; Cobb, A.R.; Cooper, M.D.; Welch, S.M.; Schmitt, J. Genetic and physiological bases for phenological responses to current and predicted climates. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 3129–3147. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.T.; Inouye, D.W.; McKinney, A.M.; Colautti, R.I.; Mitchell-Olds, T. Phenotypic plasticity and adaptive evolution contribute to advancing flowering phenology in response to climate change. Proc. Biol. Sci. 2012, 279, 3843–3852. [Google Scholar] [CrossRef] [PubMed]
- Neri, F.C.S.; Usberti, J.A.; Usberti, R.; Paiva, P.D.D.O. Comparações entre características morfo-agronômicas entre cultivares de Tagetes patula L. Rev. Bras. Hortic. Ornam. 2012, 18, 85. [Google Scholar] [CrossRef][Green Version]
- Ahmad, I.; Asif, M.U.; Amjad, A.; Ahmad, S. Fertilization enhances growth, yield, and xanthophyll contents of marigold. Turk. J. Agric. For. 2011, 35, 641–648. [Google Scholar]
- Aslam, A.; Zaman, F.; Qasim, M.; Ziaf, K. Impact of nitrogen and potash on rowth, flower and seed yield of African marigold (Tagetes erecta L.). Sci. Agric. 2016, 14, 266–269. [Google Scholar] [CrossRef]
- Sardoei, A.S.; Shahdadneghad, M.; Bahmanyar, M.A. Effects of topping and foliar application of gibberellic acid on some growth and flowering characteristics of African marigold. Flowers Ornam. Plants 2020, 5, 99–108. [Google Scholar]
- Williamson, D.F.; Parker, R.A.; Kendrick, J.S. The box plot: A simple visual method to interpret data. Ann. Intern. Med. 1989, 110, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, J.; Wang, Y.; Chen, H.; Xu, Y.; Yang, X.; Zhang, H.; Guan, J.; Wang, J. Selection of example varieties used in the DUS test guideline of Tagetes L. Agric. Sci. Technol. 2012, 13, 2110. [Google Scholar]
- Patel, M.A.; Chawla, S.L.; Chauhan, D.A.; Patil, S.; Chhatrola, H.N. Character association and path analysis studies in mar-igold (Tagetes spp.) under the South Gujarat region. J. Pharmacogn. Phytochem. 2018, 7, 3576–3580. [Google Scholar]
- Usha Bharathi, T.; Jawaharlal, M. Evaluation of African marigold (Tagetes erecta L.) genotypes for growth and flower yield under coimbatore conditions. Trends Biosci. 2014, 7, 2197–2201. [Google Scholar]
- Usha Bharathi, T.; Jawaharlal, M. Genetic divergence of African marigold (Tagetes erecta L.). Trends Biosci. 2014, 7, 2233–2236. [Google Scholar]
- Genotypes, L.J.; Ponnuswami, V. Correlation studies in Tagetes erecta L. Pharma Innov. J. 2019, 8, 1210–1212. [Google Scholar]
- Singh, K.P.N.; Bharadwaj, C.; Prasad, K.V.; Raju, D.V.S. Studies on character association and path analysis of quantitative traits among parental lines of marigold (Tagetes erecta and T. patula) and their interspecific F1 hybrids. Indian J. Hortic. 2009, 66, 348–352. [Google Scholar]
- Karuppaiah, P.; Kumar, P.S. Correlation and path analysis in African marigold (Tagetes erecta L.). Electron. J. Plant Breed. 2010, 1, 217–220. [Google Scholar]
- Panwar, S.; Singh, K.P.; Janakiram, T.; Banyal, N. Genetic variability, heritability and genetic advance in African marigold (Tagetes erecta L.) genotypes. Progress. Hortic. 2013, 45, 135–140. [Google Scholar]
- Patel, M.A.; Chawla, S.L.; Patel, A.I.; Shah, H.P.; Bhatt, D.S. Genetic divergence studies in marigold (Tagetes spp.). J. Pharmacogn. Phytochem. 2018, 7, 3572–3575. [Google Scholar]
- Sapna, P.; Singh, K.P.; Namita; Janakiram, T.; Sonah, H.; Sharma, T.R. DNA fingerprinting in African marigold (Tagetes erecta L.) genotypes using ISSR and URP markers. Indian J. Hortic. 2018, 75, 105–110. [Google Scholar]
- Puskás, M.; Höfer, M.; Sestraş, R.E.; Peil, A.; Sestraş, A.F.; Hanke, M.-V.; Flachowsky, H. Molecular and flow cytometric evaluation of pear (Pyrus L.) genetic resources of the German and Romanian national fruit collections. Genet. Resour. Crop Evol. 2016, 63, 1023–1033. [Google Scholar] [CrossRef]
- Zhang, H.; Cong, R.; Wang, M.; Dong, A.; Xin, H.; Yi, M.; Guo, H. Development of SSR molecular markers based on transcriptome sequencing of Tagetes erecta. Acta Hortic. Sin. 2018, 45, 159–167. [Google Scholar]
- ISTA. International Rules for Seed Testing; International Seed Testing Association: Basserdorf, Switzerland, 2017. [Google Scholar]
- Matei, I.; Pacurar, I.; Rosca, S.; Bilasco, S.; Sestras, P.; Rusu, T.; Jude, E.T.; Tăut, F.D. Land use favourability assessment based on soil characteristics and anthropic pollution. Case study Somesul Mic Valley Corridor, Romania. Agronomy 2020, 10, 1245. [Google Scholar] [CrossRef]
- Denny, E.G.; Gerst, K.L.; Miller-Rushing, A.J.; Tierney, G.L.; Crimmins, T.M.; Enquist, C.A.F.; Guertin, P.; Rosemartin, A.H.; Schwartz, M.D.; Thomas, K.A.; et al. Standardized phenology monitoring methods to track plant and animal activity for science and resource management applications. Int. J. Biometeorol. 2014, 58, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem. Bull. 1987, 19, 11–15. [Google Scholar]
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1989. [Google Scholar]
- Liu, K.; Muse, S. PowerMaker: An integrated analysis environment for genetic maker analysis. Bioinformatics (Oxf. Engl.) 2005, 21, 2128–2129. [Google Scholar] [CrossRef] [PubMed]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 4–9. [Google Scholar]


| No | Genotype | Seed Length | Seed Width | Seed Thickness | Germination | |||
|---|---|---|---|---|---|---|---|---|
| (mm) | CV% | (mm) | CV% | (mm) | CV% | (%) | ||
| T. patula | ||||||||
| 1 | Bolero | 8.8 e | 22.3 | 2.8 ab | 16.3 | 2.4 a | 12.6 | 78.3 d |
| 2 | Orange Flame | 11.3 cd | 2.8 | 2.7 ab | 21.4 | 2.3 ab | 20.1 | 100.0 a |
| 3 | Szinkeverek | 11.5 bcd | 4.1 | 2.4 bc | 10.2 | 2.3 ab | 9.7 | 97.4 ab |
| 4 | Robuszta | 13.7 abc | 2.5 | 2.5 abc | 9.6 | 1.7 ab | 8.0 | 100.0 a |
| 5 | Orion | 14.0 a | 17.5 | 3.2 a | 13.6 | 2.4 a | 8.6 | 100.0 a |
| 6 | Mr. Majestic | 13.0 abcd | 2.7 | 2.3 bc | 7.8 | 0.8 c | 14.9 | 43.5 e |
| 7 | Cresto Flame | 12.0 abcd | 1.6 | 2.5 abc | 7.6 | 2.4 a | 4.7 | 87.0 c |
| 8 | Cresto Yellow | 11.0 de | 2.2 | 1.9 c | 9.9 | 1.5 bc | 7.7 | 91.3 bc |
| 9 | Queen Sophia | 13.9 ab | 1.1 | 2.3 bc | 4.7 | 2.1 ab | 7.8 | 92.2 bc |
| T. tenuifolia | ||||||||
| 10 | Luna Gold | 7.7 ab | 1.2 | 1.6 a | 1.4 | 1.4 ab | 4.2 | 100.0 a |
| 11 | Luna Orange | 8.8 a | 0.8 | 0.9 b | 1.1 | 1.1 b | 6.3 | 88.7 bc |
| 12 | Luna Rot | 6.8 b | 1.0 | 1.6 a | 1.2 | 1.2 ab | 5.9 | 97.6 ab |
| 13 | Luna Lemon | 6.6 b | 1.7 | 1.4 ab | 1.1 | 1.1 b | 11.0 | 100.0 a |
| 14 | Sarga | 7.2 ab | 2.2 | 1.6 a | 1.6 | 1.6 a | 2.8 | 87.0 c |
| T. erecta | ||||||||
| 15 | Aranysarga | 10.9 c | 1.3 | 2.6 ab | 2.5 | 1.5 d | 5.8 | 93.0 a |
| 16 | Alacsony Citromsarga | 14.5 a | 0.9 | 3.0 a | 4.5 | 2.4 ab | 4.5 | 65.2 b |
| 17 | Magas Citromsarga | 12.3 bc | 1.1 | 2.2 b | 4.8 | 2.1 c | 7.4 | 100.0 a |
| 18 | Cupid Golden Yellow | 14.9 a | 1.2 | 2.9 a | 5.3 | 2.5 a | 4.0 | 97.4 a |
| 19 | Antigua Orange | 15.1 a | 1.1 | 2.6 ab | 4.3 | 2.2 bc | 5.5 | 95.7 a |
| 20 | Artic | 13.0 ab | 0.9 | 2.6 ab | 5.2 | 2.3 abc | 7.3 | 8.7 c |
| 21 | Hawaii | 13.2 ab | 1.6 | 2.3 b | 6.9 | 2.3 abc | 4.9 | 95.7 a |
| No | Genotype | Plant Height | Stem Diameter | No. Branches/Stem | Angle of Branch Insertion | ||||
|---|---|---|---|---|---|---|---|---|---|
| (cm) | CV% | (mm) | CV% | (n) | CV% | Degree | CV% | ||
| T. patula | |||||||||
| 1 | Bolero | 25.4 b | 11.3 | 10.5 bc | 0.8 | 8.7 b | 1.7 | 49.5 c | 2.7 |
| 2 | Orange Flame | 23.3 b | 10.5 | 8.5 d | 1.0 | 10.7 ab | 10.5 | 60.2 ab | 1.8 |
| 3 | Szinkeverek | 26.2 b | 6.3 | 11.5 ab | 4.0 | 8.9 b | 15.4 | 40.0 d | 4.7 |
| 4 | Robuszta | 22.1 b | 13.1 | 12.9 a | 3.2 | 13.7 a | 13.3 | 59.4 ab | 1.3 |
| 5 | Orion | 26.0 b | 6.4 | 9.4 cd | 2.7 | 7.6 b | 27.4 | 59.9 ab | 1.4 |
| 6 | Mr. Majestic | 20.7 b | 11.6 | 9.3 cd | 4.7 | 7.6 b | 11.7 | 60.6 a | 2.3 |
| 7 | Cresto Flame | 25.3 b | 16.2 | 11.3 b | 3.3 | 9.2 b | 9.0 | 50.0 c | 1.6 |
| 8 | Cresto Yellow | 25.4 b | 5.6 | 11.6 ab | 4.4 | 9.6 b | 11.8 | 51.3 c | 0.9 |
| 9 | Queen Sophia | 47.8 a | 3.1 | 11.2 b | 5.7 | 10.7 ab | 18.8 | 53.4 bc | 2.7 |
| T. tenuifolia | |||||||||
| 10 | Luna Gold | 21.8 a | 5.0 | 8.7 b | 8.2 | 16.0 b | 8.8 | 52.4 ab | 3.9 |
| 11 | Luna Orange | 21.0 b | 6.3 | 9.1 ab | 10.1 | 16.4 ab | 7.5 | 52.2 ab | 3.9 |
| 12 | Luna Rot | 21.1 b | 3.7 | 9.2 a | 7.7 | 16.9 a | 5.5 | 52.8 a | 3.0 |
| 13 | Luna Lemon | 21.3 ab | 4.7 | 9.2 a | 6.1 | 16.7 ab | 14.1 | 51.5 b | 2.7 |
| 14 | Sarga | 21.0 b | 5.8 | 9.2 a | 7.3 | 16.0 b | 8.8 | 51.6 b | 2.0 |
| T. erecta | |||||||||
| 15 | Aranysarga | 67.2 ab | 6.8 | 14.8 ab | 9.5 | 13.6 a | 9.8 | 66.5 ab | 3.1 |
| 16 | Alacsony Citromsarga | 69.8 a | 4.8 | 15.8 a | 6.6 | 14.2 a | 11.0 | 71.1 a | 1.3 |
| 17 | Magas Citromsarga | 59.7 b | 3.0 | 15.4 ab | 4.0 | 10.4 bc | 11.8 | 69.8 a | 1.2 |
| 18 | Cupid Golden Yellow | 59.9 ab | 2.4 | 12.6 abc | 3.4 | 8.9 c | 10.4 | 59.8 bc | 1.4 |
| 19 | Antigua Orange | 24.4 c | 4.6 | 8.9 c | 3.9 | 11.8 ab | 13.3 | 37.3 c | 1.8 |
| 20 | Artic | 60.6 ab | 2.2 | 11.6 bc | 4.2 | 10.0 bc | 14.4 | 65.1 ab | 1.9 |
| 21 | Hawaii | 65.8 ab | 3.7 | 13.0 ab | 2.1 | 12.1 ab | 22.0 | 60.5 bc | 1.8 |
| No | Genotype | No. Flowers | Corolla Diameter | Disc Diameter | No. Fruits | ||||
|---|---|---|---|---|---|---|---|---|---|
| (no) | CV% | (cm) | CV% | (cm) | CV% | (no) | CV% | ||
| T. patula | |||||||||
| 1 | Bolero | 28.3 bc | 7.1 | 3.4 d | 15.8 | 1.2 b | 12.6 | 5.2 cd | 20.9 |
| 2 | Orange Flame | 29.7 bc | 5.3 | 5.0 ab | 5.5 | 1.5 ab | 6.6 | 13.1 a | 9.7 |
| 3 | Szinkeverek | 25.4 c | 10.0 | 3.8 cd | 7.3 | 1.2 b | 20.1 | 13.0 a | 14.9 |
| 4 | Robuszta | 30.0 abc | 7.6 | 3.0 d | 6.6 | 1.3 ab | 22.7 | 4.8 d | 29.2 |
| 5 | Orion | 29.7 bc | 5.6 | 5.4 a | 4.6 | 1.7 a | 9.1 | 12.2 ab | 9.8 |
| 6 | Mr. Majestic | 35.7 ab | 3.7 | 3.1 d | 5.1 | 1.1 b | 14.4 | 10.4 ab | 18.0 |
| 7 | Cresto Flame | 25.9 c | 4.5 | 4.5 abc | 3.6 | 1.3 ab | 13.3 | 8.7 bcd | 18.2 |
| 8 | Cresto Yellow | 25.9 c | 3.0 | 4.4 bc | 4.4 | 1.3 ab | 9.9 | 9.2 abc | 25.3 |
| 9 | Queen Sophia | 38.9 a | 4.0 | 3.2 d | 6.6 | 1.4 ab | 8.9 | 9.6 abc | 18.2 |
| T. tenuifolia | |||||||||
| 10 | Luna Gold | 91.9 b | 2.8 | 0.9 b | 20.1 | 0.5 a | 18.9 | 35.1 ab | 13.3 |
| 11 | Luna Orange | 92.1 ab | 1.1 | 1.0 a | 13.5 | 0.5 a | 17.3 | 36.0 a | 9.0 |
| 12 | Luna Rot | 92.2 ab | 2.2 | 1.0 a | 15.0 | 0.5 a | 17.4 | 32.6 ab | 6.2 |
| 13 | Luna Lemon | 92.2 ab | 1.5 | 1.0 a | 16.0 | 0.5 a | 18.9 | 32.3 b | 7.4 |
| 14 | Sarga | 96.4 a | 3.8 | 1.0 a | 16.4 | 0.5 a | 16.2 | 32.1 b | 7.7 |
| T. erecta | |||||||||
| 15 | Aranysarga | 26.6 ab | 12.8 | 8.5 ab | 2.5 | 4.5 b | 3.3 | 8.4 abc | 28.5 |
| 16 | Alacsony Citromsarga | 32.7 a | 4.8 | 9.0 ab | 4.4 | 4.2 b | 6.6 | 10.9 a | 17.5 |
| 17 | Magas Citromsarga | 16.7 abc | 6.0 | 7.9 bc | 4.3 | 5.0 ab | 5.1 | 8.8 abc | 11.1 |
| 18 | Cupid Golden Yellow | 26.6 ab | 4.7 | 6.4 c | 1.9 | 4.1 b | 3.4 | 9.1 ab | 13.9 |
| 19 | Antigua Orange | 5.3 c | 16.2 | 10.1 a | 2.0 | 7.7 a | 2.7 | 2.6 d | 28.4 |
| 20 | Artic | 8.6 bc | 15.6 | 7.1 bc | 2.6 | 4.2 b | 2.5 | 4.4 cd | 32.0 |
| 21 | Hawaii | 14.4 abc | 9.9 | 7.5 bc | 1.9 | 4.5 b | 2.5 | 6.0bcd | 36.3 |
| No | Genotype | Phenological Phases (in Days) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Seedling Emergence | Stem Formation | Branching | Budding | Full Flowering | Flowering Duration | Fruit Formation | Fruit Ripening | ||
| T. patula | |||||||||
| 1 | Bolero | 14 ab | 24 b | 33 b | 45 b | 63 b | 24 ab | 78 b | 98 bc |
| 2 | Orange Flame | 16 ab | 29 ab | 36 ab | 46 b | 65 b | 17 cd | 81 ab | 106 abc |
| 3 | Szinkeverek | 16 ab | 30 ab | 40 ab | 55 ab | 70 ab | 25 a | 88 ab | 106 abc |
| 4 | Robuszta | 16 ab | 30 ab | 40 ab | 53 ab | 70 ab | 22 ab | 84 ab | 104 bc |
| 5 | Orion | 27 a | 45 a | 60 a | 77 a | 100 a | 26 a | 11 8a | 145 a |
| 6 | Mr. Majestic | 10 b | 19 b | 25 b | 33 b | 41 b | 15 d | 54 b | 67 c |
| 7 | Cresto Flame | 15 ab | 25 b | 38 ab | 51 ab | 69 ab | 20 bc | 85 ab | 107 ab |
| 8 | Cresto Yellow | 14 ab | 24 b | 37 ab | 50 ab | 60 b | 22 ab | 75 b | 95 bc |
| 9 | Queen Sophia | 14 ab | 23 b | 29 b | 40 b | 56 b | 20 bc | 72 b | 98 bc |
| T. tenuifolia | |||||||||
| 10 | Luna Gold | 14 ab | 24 ab | 29 ab | 36 ab | 46 ab | 21 a | 62 ab | 84 ab |
| 11 | Luna Orange | 14 ab | 22 b | 28 b | 37 a | 45 b | 21 a | 61 b | 83 b |
| 12 | Luna Rot | 13 b | 25 a | 30 a | 35 b | 47 a | 21 a | 63 a | 85 a |
| 13 | Luna Lemon | 15 a | 25 a | 29 ab | 36 ab | 46 ab | 21 a | 62 ab | 84 ab |
| 14 | Sarga | 14 ab | 24 ab | 29 ab | 36 ab | 46 ab | 20 b | 62 ab | 84 ab |
| T. erecta | |||||||||
| 15 | Aranysarga | 24 a | 39 a | 51 a | 65 a | 77 ab | 22 ab | 92 abc | 116 b |
| 16 | Alacsony Citromsarga | 16 ab | 31 b | 42 ab | 57 bc | 71 b | 22 ab | 88 c | 114 b |
| 17 | Magas Citromsarga | 14 b | 29 b | 40 b | 56 c | 70 b | 21 bc | 88 c | 114 b |
| 18 | Cupid Golden Yellow | 24 a | 34 ab | 44 ab | 61 ab | 83 a | 24 ab | 98 ab | 122 a |
| 19 | Antigua Orange | 16 ab | 31 b | 40 b | 61 ab | 78 ab | 25 a | 94 abc | 119 ab |
| 20 | Artic | 14 b | 29 b | 40 b | 57 bc | 73 b | 18 c | 89 bc | 115 b |
| 21 | Hawaii | 19 ab | 34 ab | 46 ab | 64 a | 83 a | 25 a | 99 a | 124 a |
| Correlated Traits | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 Seed length | 0.000 | 0.005 | 0.412 | 0.026 | 0.039 | 0.003 | 0.231 | 0.000 | 0.000 | 0.002 | 0.000 | 0.102 | 0.034 | 0.009 | 0.001 | 0.000 | 0.406 | 0.001 | 0.001 | |
| 2 Seed width | 0.746 | 0.000 | 0.452 | 0.059 | 0.073 | 0.001 | 0.183 | 0.000 | 0.001 | 0.027 | 0.000 | 0.016 | 0.003 | 0.001 | 0.000 | 0.000 | 0.255 | 0.000 | 0.000 | |
| 3 Seed thickness | 0.586 | 0.757 | 0.992 | 0.050 | 0.116 | 0.012 | 0.754 | 0.001 | 0.004 | 0.033 | 0.002 | 0.056 | 0.018 | 0.005 | 0.001 | 0.000 | 0.044 | 0.000 | 0.000 | |
| 4 Germination | −0.189 | −0.174 | 0.002 | 0.278 | 0.762 | 0.276 | 0.167 | 0.306 | 0.470 | 0.583 | 0.275 | 0.139 | 0.332 | 0.550 | 0.768 | 0.676 | 0.020 | 0.624 | 0.590 | |
| 5 Plant height | 0.486 | 0.418 | 0.434 | −0.248 | 0.000 | 0.598 | 0.001 | 0.028 | 0.000 | 0.001 | 0.040 | 0.089 | 0.041 | 0.027 | 0.013 | 0.023 | 0.547 | 0.023 | 0.012 | |
| 6 Stem diameter | 0.452 | 0.400 | 0.354 | −0.070 | 0.817 | 0.564 | 0.003 | 0.020 | 0.003 | 0.021 | 0.017 | 0.241 | 0.098 | 0.029 | 0.018 | 0.042 | 0.362 | 0.044 | 0.044 | |
| 7 No. branches/stem | −0.619 | −0.690 | −0.535 | 0.249 | −0.122 | −0.133 | 0.887 | 0.000 | 0.082 | 0.380 | 0.000 | 0.369 | 0.331 | 0.093 | 0.039 | 0.018 | 0.768 | 0.023 | 0.060 | |
| 8 Branches insertion | 0.273 | 0.302 | 0.073 | −0.313 | 0.673 | 0.617 | −0.033 | 0.377 | 0.108 | 0.316 | 0.368 | 0.300 | 0.160 | 0.142 | 0.233 | 0.306 | 0.189 | 0.325 | 0.250 | |
| 9 No. flowers | −0.822 | −0.797 | −0.670 | 0.235 | −0.478 | −0.504 | 0.769 | −0.203 | 0.000 | 0.001 | 0.000 | 0.200 | 0.042 | 0.004 | 0.000 | 0.000 | 0.351 | 0.000 | 0.001 | |
| 10 Corolla diameter | 0.735 | 0.685 | 0.597 | −0.167 | 0.708 | 0.614 | −0.389 | 0.361 | −0.810 | 0.000 | 0.000 | 0.052 | 0.004 | 0.001 | 0.000 | 0.000 | 0.195 | 0.000 | 0.000 | |
| 11 Disc diameter | 0.636 | 0.483 | 0.466 | −0.127 | 0.657 | 0.499 | −0.202 | 0.230 | −0.664 | 0.928 | 0.004 | 0.142 | 0.022 | 0.015 | 0.001 | 0.003 | 0.162 | 0.003 | 0.002 | |
| 12 No. fruits | −0.810 | −0.813 | −0.627 | 0.250 | −0.450 | −0.513 | 0.746 | −0.207 | 0.972 | −0.745 | −0.602 | 0.272 | 0.086 | 0.013 | 0.001 | 0.001 | 0.429 | 0.001 | 0.004 | |
| 13 Emergence | 0.367 | 0.520 | 0.423 | 0.334 | 0.380 | 0.267 | −0.207 | 0.238 | −0.291 | 0.429 | 0.332 | −0.251 | 0.000 | 0.000 | 0.000 | 0.000 | 0.004 | 0.000 | 0.000 | |
| 14 Stem formation | 0.465 | 0.623 | 0.509 | 0.222 | 0.450 | 0.370 | −0.223 | 0.318 | −0.448 | 0.606 | 0.497 | −0.383 | 0.919 | 0.000 | 0.000 | 0.000 | 0.004 | 0.000 | 0.000 | |
| 15 Branching | 0.556 | 0.687 | 0.585 | 0.138 | 0.483 | 0.476 | −0.376 | 0.332 | −0.599 | 0.680 | 0.521 | −0.533 | 0.866 | 0.961 | 0.000 | 0.000 | 0.004 | 0.000 | 0.000 | |
| 16 Budding | 0.675 | 0.724 | 0.670 | 0.068 | 0.535 | 0.511 | −0.454 | 0.272 | −0.723 | 0.787 | 0.659 | −0.652 | 0.793 | 0.909 | 0.972 | 0.000 | 0.003 | 0.000 | 0.000 | |
| 17 Full flowering | 0.697 | 0.786 | 0.767 | 0.097 | 0.495 | 0.447 | −0.510 | 0.235 | −0.739 | 0.747 | 0.617 | −0.680 | 0.793 | 0.893 | 0.948 | 0.979 | 0.003 | 0.000 | 0.000 | |
| 18 Flowering duration | 0.191 | 0.260 | 0.443 | 0.502 | 0.139 | 0.209 | −0.069 | −0.299 | −0.214 | 0.295 | 0.317 | −0.182 | 0.601 | 0.599 | 0.597 | 0.619 | 0.616 | 0.002 | 0.003 | |
| 19 Fruit formation | 0.678 | 0.769 | 0.779 | 0.114 | 0.493 | 0.444 | −0.494 | 0.226 | −0.718 | 0.741 | 0.611 | −0.650 | 0.788 | 0.896 | 0.948 | 0.978 | 0.997 | 0.630 | 0.000 | |
| 20 Fruit ripening | 0.652 | 0.732 | 0.790 | 0.125 | 0.537 | 0.444 | −0.417 | 0.262 | −0.672 | 0.748 | 0.633 | −0.600 | 0.784 | 0.894 | 0.931 | 0.960 | 0.982 | 0.609 | 0.989 |
| Name | Sequences (5′–3′) | Repeat Motif | NG | Na | He | Ho | PIC | |
|---|---|---|---|---|---|---|---|---|
| TE11 | F | CGCCTAATTTGTTGCATGG | (AG)19 | 3 | 2 | 0.20 | 0.08 | 0.18 |
| R | ATGTCACCGCCAAAGGATT | |||||||
| TE35 | F | ACCCTCCTTGACCCTGTTG | (CT)15 | 2 | 2 | 0.47 | 0.00 | 0.36 |
| R | GGTGTTGTTGCTGCTTGCT | |||||||
| TE38 | F | CGGAAACGAACGAGTGAAGT | (TA)7 | 4 | 3 | 0.38 | 0.09 | 0.34 |
| R | GCGTATAAAATCCCTGCCCT | |||||||
| TE59 | F | CCGGTTTGTGAAATCTGAAG | (AG)9 | 6 | 6 | 0.66 | 0.23 | 0.60 |
| R | CACAGCTAAACTCACGCACA | |||||||
| TE70 | F | AATCAGCCATTATCAACCCT | (AG)24 | 3 | 3 | 0.52 | 0.00 | 0.44 |
| R | CAACTCATGTTTCACCCAAA | |||||||
| TE78 | F | GAGAGGATCTGGTGGGATGA | (CT)7 | 4 | 5 | 0.53 | 0.20 | 0.41 |
| R | GGTGGCTCCAAACTACCAAG | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cicevan, R.; Sestras, A.F.; Plazas, M.; Boscaiu, M.; Vilanova, S.; Gramazio, P.; Vicente, O.; Prohens, J.; Sestras, R.E. Biological Traits and Genetic Relationships Amongst Cultivars of Three Species of Tagetes (Asteraceae). Plants 2022, 11, 760. https://doi.org/10.3390/plants11060760
Cicevan R, Sestras AF, Plazas M, Boscaiu M, Vilanova S, Gramazio P, Vicente O, Prohens J, Sestras RE. Biological Traits and Genetic Relationships Amongst Cultivars of Three Species of Tagetes (Asteraceae). Plants. 2022; 11(6):760. https://doi.org/10.3390/plants11060760
Chicago/Turabian StyleCicevan, Raluca, Adriana F. Sestras, Mariola Plazas, Monica Boscaiu, Santiago Vilanova, Pietro Gramazio, Oscar Vicente, Jaime Prohens, and Radu E. Sestras. 2022. "Biological Traits and Genetic Relationships Amongst Cultivars of Three Species of Tagetes (Asteraceae)" Plants 11, no. 6: 760. https://doi.org/10.3390/plants11060760
APA StyleCicevan, R., Sestras, A. F., Plazas, M., Boscaiu, M., Vilanova, S., Gramazio, P., Vicente, O., Prohens, J., & Sestras, R. E. (2022). Biological Traits and Genetic Relationships Amongst Cultivars of Three Species of Tagetes (Asteraceae). Plants, 11(6), 760. https://doi.org/10.3390/plants11060760














