Assessment of the Fertilization Capacity of the Aquaculture Sediment for Wheat Grass as Sustainable Alternative Use
Abstract
:1. Introduction
2. Results
2.1. Sediment Composition
2.2. Morphology of Wheat Grass and Yield of Wheat Grass Juice
2.3. Color of Wheat Grass
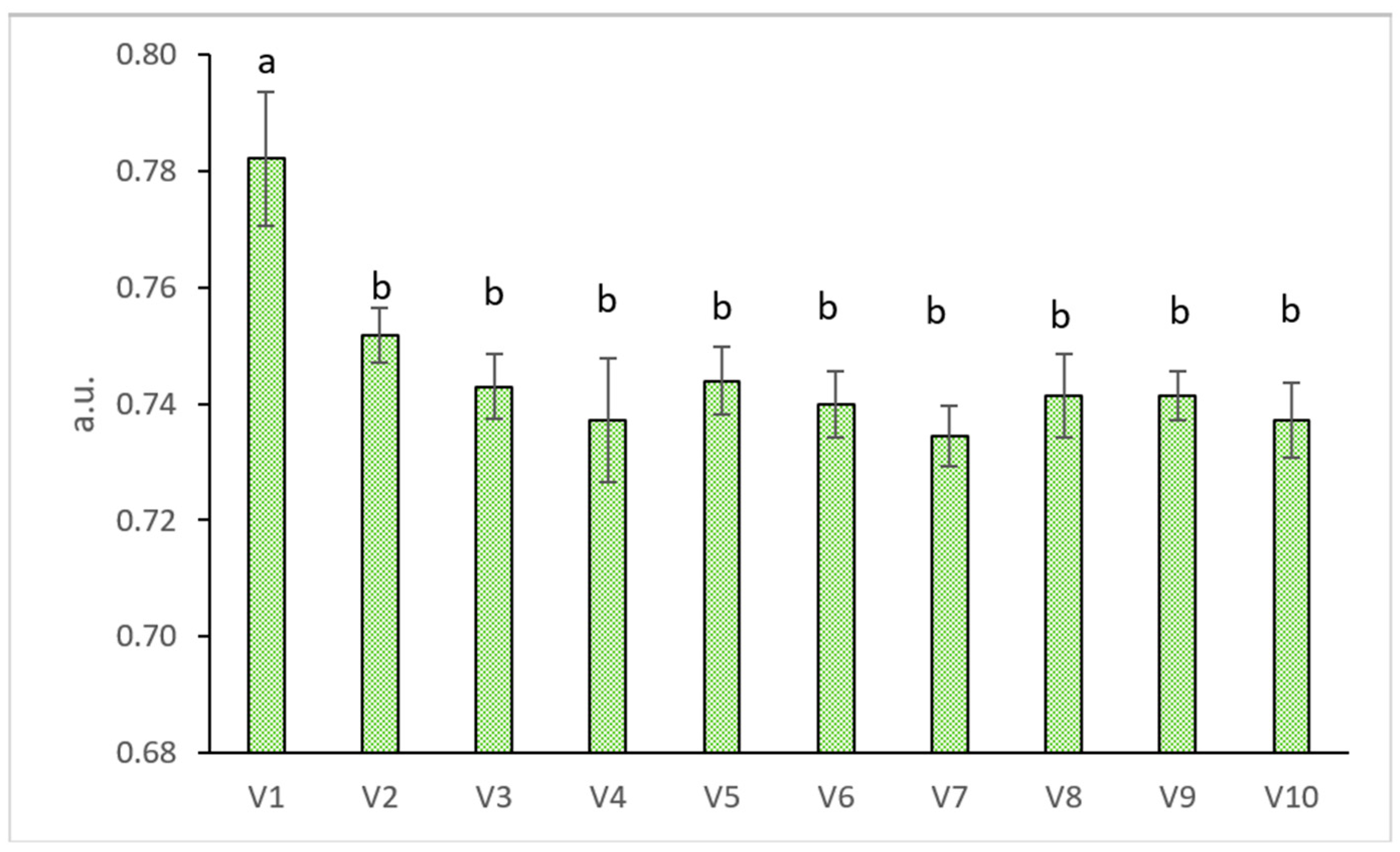
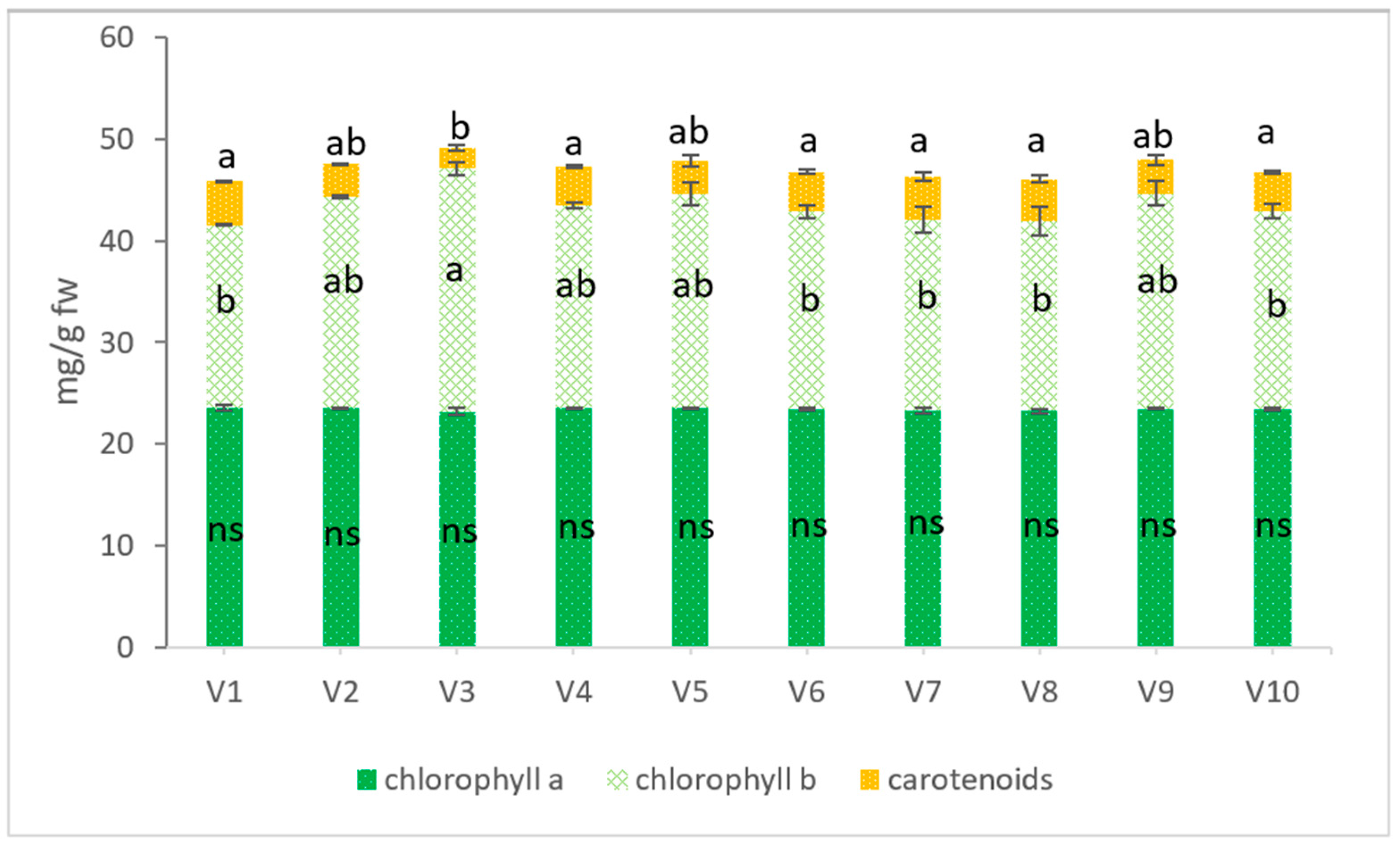
2.4. Physiology of Wheat Grass
2.5. Wheat Grass Juice Quality
2.6. Chemical Composition of Leachate
3. Discussion
4. Materials and Methods
4.1. Sediment Collection
4.2. Sediment Analysis
4.3. Elemental Analysis of Sediment
4.4. Microbiological Analysis of Sediment
4.5. Chemical Composition of Sediment Leachate
4.6. Cultivation of Wheat Grass
4.7. Fresh Yield and Morphological Parameters of Wheat Grass
4.8. Leaf Area of Wheat Grass
4.9. The Color of Wheatgrass
4.10. Total Chlorophyll Content of Wheat Grass
4.11. Gas Exchange Measurement of Wheat Grass
4.12. Chlorophyll Fluorescence of Wheat Grass
4.13. Wheat Grass Juice Extraction
4.14. Chlorophyll Content in Wheat Grass Juice
4.15. Total Phenolic, Total Flavonoid, and Antioxidant Activity of Wheat Grass Juice
4.16. Statistical Analyses of Data
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Dróżdż, D.; Malińska, K.; Mazurkiewicz, J.; Kacprzak, M.; Mrowiec, M.; Szczypiór, A.; Postawa, P.; Stachowiak, T. Fish pond sediment from aquaculture production—Current practices and the potential for nutrient recovery: A Review. Int. Agrophys. 2020, 34, 33–41. [Google Scholar] [CrossRef]
- Dróżdż, D.; Malińska, K.; Kacprzak, M.; Mrowiec, M.; Szczypiór, A.; Postawa, P.; Stachowiak, T. Potential of Fish Pond Sediments Composts as Organic Fertilizers. Waste Biomass Valoriz. 2020, 11, 5151–5163. [Google Scholar] [CrossRef] [Green Version]
- FAO. The State of World Fisheries and Aquaculture (SOFIA); FAO: Rome, Italy, 2020; ISBN 978-92-5-132692-3. [Google Scholar]
- Mizanur, R.; Yakupitiyage, A.; Ranamukhaarachchi, S.L. Agricultural use of fishpond sediment for environmental amelioration. Thammasat Int. J. Sci. Technol. 2004, 9, 1–12. [Google Scholar]
- Junejo, S.H.; Baig, J.A.; Kazi, T.G.; Afridi, H.I. Cadmium and Lead Hazardous Impact Assessment of Pond Fish Species. Biol. Trace Elem. Res. 2019, 191, 502–511. [Google Scholar] [CrossRef] [PubMed]
- Vaverková, M.D.; Elbl, J.; Koda, E.; Adamcová, D.; Bilgin, A.; Lukas, V.; Podlasek, A.; Kintl, A.; Wdowska, M.; Brtnický, M.; et al. Chemical Composition and Hazardous Effects of Leachate from the Active Municipal Solid Waste Landfill Surrounded by Farmlands. Sustainability 2020, 12, 4531. [Google Scholar] [CrossRef]
- Negi, P.; Mor, S.; Ravindra, K. Impact of landfill leachate on the groundwater quality in three cities of North India and health risk assessment. Environ. Dev. Sustain. 2020, 22, 1455–1474. [Google Scholar] [CrossRef]
- Burducea, M.; Lobiuc, A.; Asandulesa, M.; Zaltariov, M.-F.; Burducea, I.; Popescu, S.M.; Zheljazkov, V.D. Effects of Sewage Sludge Amendments on the Growth and Physiology of Sweet Basil. Agronomy 2019, 9, 548. [Google Scholar] [CrossRef] [Green Version]
- Marval, S.; Hejduk, T.; Zajíček, A. An Analysis of Sediment Quality from the Perspective of Land Use in the Catchment and Pond Management. Soil Sediment Contam. Int. J. 2020, 29, 397–420. [Google Scholar] [CrossRef]
- Brigham, R. Assessing the Effects of Lake Dredged Sediments on Soil Health: Agricultural and Environmental Implications on Midwest Ohio; Bowling Green State University: Bowling Green, OH, USA, 2020. [Google Scholar]
- Thi Da, C.; Anh Tu, P.; Livsey, J.; Tang, V.T.; Berg, H.; Manzoni, S. Improving Productivity in Integrated Fish-Vegetable Farming Systems with Recycled Fish Pond Sediments. Agronomy 2020, 10, 1025. [Google Scholar] [CrossRef]
- Haque, M.B.; Belton, B.; Alam, M.A.; Ahmed, A.G.; Alam, M.R. Reuse of fish pond sediments as fertilizer for fodder grass production in Bangladesh: Potential for sustainable intensification and improved nutrition. Agric. Ecosyst. Environ. 2016, 216, 226–236. [Google Scholar] [CrossRef]
- Neculita, M.; Moga, M.L. Analysis of Romanian fisheries and aquaculture in regional context. USV Ann. Econ. Public Adm. 2015, 15, 127–132. [Google Scholar]
- Bostock, J.; Lane, A.; Hough, C.; Yamamato, K. An assessment of the economic contribution of EU aquaculture production and the influence of policies for its sustainable development. Aquacult. Int. 2016, 24, 699–733. [Google Scholar] [CrossRef] [Green Version]
- National Strategy of the Fisheries Sector 2014–2020. Ministry of Environment and Climate Change, Ministry of Agriculture and Rural Development (Romanian). pp. 1–62. Available online: https://www.madr.ro/docs/fep/programare-2014-2020/Strategia-Nationala-a-Sectorului-Pescaresc-2014-2020-update-apr2014.pdf (accessed on 1 October 2021).
- Brodny, J.; Tutak, M. Analyzing Similarities between the European Union Countries in Terms of the Structure and Volume of Energy Production from Renewable Energy Sources. Energies 2020, 13, 913. [Google Scholar] [CrossRef] [Green Version]
- Jităreanu, A.; Caba, I.C.; Trifan, A.; Pădureanu, S.; Agoroaei, L. Triticum aestivum Assay—A Useful Tool for Environmental Monitoring and Toxicity Assessment. Not. Bot. Horti Agrobot. Cluj-Napoca 2019, 47, 1005–1018. [Google Scholar] [CrossRef] [Green Version]
- Dumitru, G.; Dirvariu, L.; Barbacariu, C.A.; Miron, I.; Sandu, I.; Todirascu Ciornea, E. The Effect of Wheatgrass Juice Administration on Physiological State and Oxidative Stress in Carp. Rev. Chim. 2018, 69, 4046–4051. [Google Scholar] [CrossRef]
- Fortună, M.E.; Vasilache, V.; Ignat, M.; Silion, M.; Vicol, T.; Patras, X.; Miron, I.; Lobiuc, A. Elemental and macromolecular modifications in Triticum aestivum L. plantlets under different cultivation conditions. PLoS ONE 2018, 13, e0202441. [Google Scholar] [CrossRef] [PubMed]
- Urbaniak, M.; Lee, S.; Takazawa, M.; Mierzejewska, E.; Baran, A.; Kannan, K. Effects of soil amendment with PCB-contaminated sediment on the growth of two cucurbit species. Environ. Sci. Pollut. Res. 2020, 27, 8872–8884. [Google Scholar] [CrossRef] [Green Version]
- Nakajima, T.; Hudson, M.J.; Uchiyama, J.; Makibayashi, K.; Zhang, J. Common carp aquaculture in Neolithic China dates back 8000 years. Nat. Ecol. Evol. 2019, 3, 1415–1418. [Google Scholar] [CrossRef]
- Wezel, A.; Robin, J.; Guerin, M.; Arthaud, F.; Vallod, D. Management effects on water quality, sediments and fish production in extensive fish ponds in the Dombes region, France. Limnologica 2013, 43, 210–218. [Google Scholar] [CrossRef]
- Avnimelech, Y.; Ritvo, G. Shrimp and fish pond soils: Processes and management. Aquaculture 2003, 220, 549–567. [Google Scholar] [CrossRef] [Green Version]
- Verdegem, M.C.J. Nutrient discharge from aquaculture operations in function of system design and production environment. Rev. Aquac. 2013, 5, 158–171. [Google Scholar] [CrossRef]
- Silburn, B.; Kröger, S.; Parker, E.R.; Sivyer, D.B.; Hicks, N.; Powell, C.F.; Johnson, M.; Greenwood, N. Benthic pH gradients across a range of shelf sea sediment types linked to sediment characteristics and seasonal variability. Biogeochemistry 2017, 135, 69–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harbi, A.H. Faecal coliforms in pond water, sediments and hybrid tilapia Oreochromis niloticus × Oreochromis aureus in Saudi Arabia. Aquac. Res. 2003, 34, 517–524. [Google Scholar] [CrossRef]
- Golubkina, N.; Kharchenko, V.; Moldovan, A.; Zayachkovsky, V.; Stepanov, V.; Pivovarov, V.; Sekara, A.; Tallarita, A.; Caruso, G. Nutritional Value of Apiaceae Seeds as Affected by 11 Species and 43 cultivars. Horticulturae 2021, 7, 57. [Google Scholar] [CrossRef]
- Tanase, C.; Coșarcă, S.; Muntean, D.-L. A Critical Review of Phenolic Compounds Extracted from the Bark of Woody Vascular Plants and Their Potential Biological Activity. Molecules 2019, 24, 1182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stoleru, V.; Slabu, C.; Vitanescu, M.; Peres, C.; Cojocaru, A.; Covasa, M.; Mihalache, G. Tolerance of Three Quinoa Cultivars (Chenopodium quinoa Willd.) to Salinity and Alkalinity Stress During Germination Stage. Agronomy 2019, 9, 287. [Google Scholar] [CrossRef] [Green Version]
- Burducea, M.; Zheljazkov, V.; Lobiuc, A.; Pintilie, C.; Vîrgolici, M.; Silion, M.; Asandulesa, M.; Burducea, I.; Zamfirache, M. Biosolids application improves mineral composition and phenolic profile of basil cultivated on eroded soil. Sci. Hortic. 2019, 249, 407–418. [Google Scholar] [CrossRef]
- Monostori, I.; Árendás, T.; Hoffman, B.; Galiba, G.; Gierczik, K.; Szira, F.; Vágújfalvi, A. Relationship between SPAD value and grain yield can be affected by cultivar, environment and soil nitrogen content in wheat. Euphytica 2016, 211, 103–112. [Google Scholar] [CrossRef] [Green Version]
- Young, A.J.; Lowe, G.L. Carotenoids—Antioxidant Properties. Antioxidants 2018, 7, 28. [Google Scholar] [CrossRef] [Green Version]
- Kuen-Song, L.; Ni-Bin, C.; Tien-Deng, C. Fine structure characterization of zero-valent iron nanoparticles for decontamination of nitrites and nitrates in wastewater and groundwater. Sci. Technol. Adv. Mater. 2008, 9, 025015. [Google Scholar] [CrossRef]
- Fernández-Nava, Y.; Marañón, E.; Soons, J.; Castrillón, L. Denitrification of wastewater containing high nitrate and calcium concentrations. Bioresour. Technol. 2008, 99, 7976–7981. [Google Scholar] [CrossRef] [PubMed]
- Lembi, C.A. 24—CONTROL OF NUISANCE ALGAE. In Aquatic Ecology, Freshwater Algae of North America; Wehr, J.D., Sheath, R.G., Eds.; Academic Press: Amsterdam, The Netherlands, 2003; pp. 805–834. [Google Scholar] [CrossRef]
- Florea, N.; Balaceanu, V.; Rauta, C.; Canarache, A. Soil studies development methodology. Incdpapm-ICPA 1987, 1, 20. [Google Scholar]
- Untea, A.; Criste, R.D.; Vladescu, L. Development and validation of a microwave digestion—FAAS procedure for Cu, Mn and Zn determination in liver. Rev. Chim. 2012, 63, 341–346. [Google Scholar]
- Teliban, G.-C.; Burducea, M.; Zheljazkov, V.D.; Dincheva, I.; Badjakov, I.; Munteanu, N.; Mihalache, G.; Cojocaru, A.; Popa, L.-D.; Stoleru, V. The Effect of Myco-Biocontrol Based Formulates on Yield, Physiology and Secondary Products of Organically Grown Basil. Agriculture 2021, 11, 180. [Google Scholar] [CrossRef]
- Burducea, M.; Zheljazkov, V.D.; Dincheva, I.; Lobiuc, A.; Teliban, G.C.; Stoleru, V.; Zamfirache, M.M. Fertilization modifies the essential oil and physiology of basil varieties. Ind. Crop. Prod. 2018, 121, 282–293. [Google Scholar] [CrossRef]
- Wellburn, A.R. The spectral determination of chlorophyll a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Lobiuc, A.; Vasilache, V.; Oroian, M.; Stoleru, T.; Burducea, M.; Pintilie, O.; Zamfirache, M.M. Blue and red LED illumination improves growth and bioactive compounds contents in acyanic and cyanic Ocimum basilicum L. Microgreens. Molecules 2017, 22, 2111. [Google Scholar] [CrossRef] [Green Version]
- Teliban, G.-C.; Stoleru, V.; Burducea, M.; Lobiuc, A.; Munteanu, N.; Popa, L.-D.; Caruso, G. Biochemical, Physiological and Yield Characteristics of Red Basil as Affected by Cultivar and Fertilization. Agriculture 2020, 10, 48. [Google Scholar] [CrossRef] [Green Version]
- Cojocaru, A.; Vlase, L.; Munteanu, N.; Stan, T.; Teliban, G.C.; Burducea, M.; Stoleru, V. Dynamic of Phenolic Compounds, Antioxidant Activity, and Yield of Rhubarb under Chemical, Organic and Biological Fertilization. Plants 2020, 9, 355. [Google Scholar] [CrossRef]
| Sampling Station | Collection Station 1 | Collection Station 2 | Collection Station 3 |
|---|---|---|---|
| pH | 8.06 a ± 0.05 | 7.68 b ± 0.05 | 7.67 b ± 0.04 |
| Humus% | 3.31 a,b ± 0.04 | 3.16 b ± 0.1 | 3.62 a ± 0.07 |
| Organic matter% | 6.43 a ± 0.01 | 5.57 c ± 0.02 | 6.26 b ± 0.03 |
| N total% | 0.16 b ± 0 | 0.16 b ± 0.00 | 0.19 a ± 0.00 |
| P mg/kg | 118.67 a ± 3.28 | 64.33 b ± 2.03 | 66.33 b ± 1.33 |
| K mg/kg | 276.33 a ± 2.6 | 235.67 c ± 1.76 | 250.67 b ± 2.03 |
| Cu mg/kg | 28.27 ns ± 0.05 | 27.29 ns ± 0.05 | 28.13 ns ± 0.05 |
| Fe mg/kg | 33,695.62 ns ± 0.05 | 33,083.57 ns ± 0.05 | 34,609.12 ns ± 0.05 |
| Mn mg/kg | 455.92 ns ± 0.05 | 448.52 ns ± 0.05 | 449.53 ns ± 0.05 |
| Zn mg/kg | 82.67 ns ± 0.05 | 81.41 ns ± 0.05 | 84.11 ns ± 0.05 |
| Pb mg/kg | 10.89 ns ± 0.05 | 10.58 ns ± 0.05 | 11.04 ns ± 0.05 |
| Cd mg/kg | bdl | bdl | bdl |
| Total coliforms cfu/g | 1020 | 1536 | 760 |
| E. coli cfu/g | nd | nd | nd |
| Treatment | Plant Height (cm) | Leaf Area (mm2) | Plant Biomass (g/Plant) | Wheat Grass Juice Yield (g/100 g) |
|---|---|---|---|---|
| V1 | 17.53 ns ± 0.61 | 135.61 ns ± 7.31 | 3.93 ns ± 0.19 | 58.00 b ± 1.16 |
| V2 | 18.06 ns ± 0.44 | 121.12 ns ± 10.88 | 3.39 ns ± 0.31 | 50.14 b,c,d ± 1.04 |
| V3 | 18.72 ns ± 0.48 | 128.32 ns ± 2.15 | 3.82 ns ± 0.01 | 48.34 c,d ± 0.88 |
| V4 | 19.22 ns ± 0.43 | 129.17 ns ± 2.34 | 3.97 ns ± 0.07 | 55.54 a,b,c ± 1.08 |
| V5 | 18.06 ns ± 0.57 | 116.93 ns ± 14.35 | 3.57 ns ± 0.42 | 50.00 b,c,d ± 1.16 |
| V6 | 18.06 ns ± 0.49 | 139.21 ns ± 3.19 | 4.26 ns ± 0.07 | 54.00 a,b,c ± 1.16 |
| V7 | 17.38 ns ± 0.42 | 116.44 ns ± 5.78 | 3.55 ns ± 0.20 | 56.80 a,b ± 1.34 |
| V8 | 19.03 ns ± 0.69 | 126.86 ns ± 12.02 | 3.83 ns ± 0.12 | 52.34 a,b,c,d ± 0.88 |
| V9 | 18.81 ns ± 0.56 | 110.26 ns ± 8.83 | 3.51 ns ± 0.27 | 56.34 a,b ± 1.46 |
| V10 | 16.94 ns ± 0.44 | 109.67 ns ± 4.10 | 3.47 ns ± 0.09 | 46.00 d ± 3.06 |
| Treatment | L | a | b |
|---|---|---|---|
| V1 | 37.81 ns ± 0.84 | −9.11 a,b ± 0.2 | 18.11 ns ± 0.87 |
| V2 | 34.75 ns ± 1.26 | −8.59 a,b ± 0.19 | 17.19 ns ± 0.44 |
| V3 | 36.62 ns ± 0.45 | −8.64 a,b ± 0.08 | 16.89 ns ± 0.28 |
| V4 | 38.56 ns ± 0.74 | −9.27 b ± 0.05 | 18.91 ns ± 0.09 |
| V5 | 37.33 ns ± 0.41 | −9.03 a,b ± 0.07 | 18.49 ns ± 0.27 |
| V6 | 38.47 ns ± 0.49 | −9.04 a,b ± 0.05 | 17.96 ns ± 0.39 |
| V7 | 35.06 ns ± 0.43 | −8.48 a ± 0.07 | 17.09 ns ± 0.19 |
| V8 | 36.7 ns ± 1.21 | −8.8 a,b ± 0.22 | 17.37 ns ± 0.93 |
| V9 | 36.86 ns ± 0.35 | −8.79 a,b ± 0.09 | 17.23 ns ± 0.13 |
| V10 | 35.97 ns ± 1.18 | −8.43 a ± 0.22 | 16.44 ns ± 0.95 |
| Treatment | Ci | E | Gs | A | CCI |
|---|---|---|---|---|---|
| V1 | 378.97 a ± 1.66 | 1.35 b,c ± 0.03 | 0.10 a,b,c ± 0.00 | 1.47 a ± 0.07 | 1.54 ns ± 0.14 |
| V2 | 373.13 a ± 1.46 | 1.11 b,c ± 0.02 | 0.07 b,c ± 0.00 | 1.29 a ± 0.06 | 1.53 ns ± 0.20 |
| V3 | 356.13 b,c ± 1.47 | 1.64 b ± 0.03 | 0.11 a,b ± 0.00 | 1.42 a ± 0.09 | 1.48 ns ± 0.09 |
| V4 | 371.27 a,b ± 1.07 | 1.47 b,c ± 0.02 | 0.09 a,b,c ± 0.00 | 1.54 a ± 0.05 | 1.83 ns ± 0.23 |
| V5 | 344.77 c ± 2.81 | 0.98 c ± 0.03 | 0.05 c,d ± 0.00 | 1.44 a ± 0.11 | 1.6 ns ± 0.17 |
| V6 | 349.9 c ± 2.18 | 2.19 a ± 0.18 | 0.13 a ± 0.03 | 1.66 a ± 0.11 | 1.57 ns ± 0.16 |
| V7 | 351.43 c ± 3.30 | 1.38 b,c ± 0.02 | 0.07 b,c ± 0.00 | 1.28 a ± 0.10 | 1.53 ns ± 0.14 |
| V8 | 344.13 c ± 3.14 | 1.58 b ± 0.30 | 0.05 c,d ± 0.00 | 1.36 a ± 0.14 | 1.43 ns ± 0.08 |
| V9 | 344.97 c ± 3.71 | 1.52 b,c ± 0.15 | 0.09 a,b,c ± 0.02 | 1.38 a ± 0.13 | 1.42 ns ± 0.08 |
| V10 | 316.63 d ± 8.27 | 0.36 d ± 0.01 | 0.02 d ± 0.00 | 0.82 b ± 0.09 | 1.4 ns ± 0.05 |
| Treatment | Total Phenols Galic Acid µg/mL | Total Flavonoids Quercitin µg/mL | Antioxidant Activity DPPH % Inhibition |
|---|---|---|---|
| V1 | 2.97 c ± 0.10 | 1.77 a,b ± 0.11 | 53.35 c ± 1.49 |
| V2 | 2.94 c,d ± 0.10 | 1.42 c ± 0.13 | 47.99 e ± 0.42 |
| V3 | 2.69 d,e ± 0.22 | 1.41 c ± 0.11 | 35.48 f ± 0.42 |
| V4 | 3.06 b,c ± 0.02 | 1.71 a,b ± 0.12 | 60.26 a,b ± 0.34 |
| V5 | 2.87 c,d ± 0.12 | 1.78 a,b ± 0.06 | 60.52 a,b ± 0.08 |
| V6 | 2.80 c,d,e ± 0.05 | 1.63 a,b,c ± 0.13 | 58.04 b ± 0.23 |
| V7 | 3.39 a ± 0.03 | 1.85 a ± 0.04 | 62.70 a ± 0.51 |
| V8 | 3.28 a,b ± 0.02 | 1.7 a,b ± 0.02 | 52.37 c,d ± 1.64 |
| V9 | 2.55 e ± 0.03 | 1.52 b,c ± 0.06 | 48.2 d,e ± 0.66 |
| V10 | 2.80 c,d,e ± 0.04 | 1.61 a,b,c ± 0.06 | 52.88 c ± 1.11 |
| Treatment | pH | NO3 (mg/L) | NO2 (mg/L) | NH3 (mg/L) |
|---|---|---|---|---|
| V1 | 7.90 b,c,d ± 0.06 | 20.20 c ± 1.33 | 0.27 d ± 0.01 | 0.03 b ± 0.00 |
| V2 | 7.97 b ± 0.03 | 19.03 d ± 1.48 | 1.19 b ± 0.00 | 0.04 a,b ± 0.00 |
| V3 | 7.93 b,c ± 0.03 | 8.77 e ± 0.03 | 0.73 c ± 0.00 | 0.02 b ± 0.00 |
| V4 | 7.97 b ± 0.03 | 8.77 e ± 0.03 | 0.28 d ± 0.01 | 0.03 b ± 0.00 |
| V5 | 7.97 b ± 0.03 | 36.63 b,c ± 1.48 | 1.19 b ± 0.00 | 0.05 a,b ± 0.00 |
| V6 | 7.73 c,d ± 0.03 | 35.2 b,c ± 2.54 | 1.17 b ± 0.02 | 0.05 a,b ± 0.00 |
| V7 | 7.77 c,d ± 0.03 | 52.77 a,b ± 4.38 | 1.33 a ± 0.00 | 0.05 a,b ± 0.00 |
| V8 | 8.37 a ± 0.03 | 52.47 a,b ± 8.97 | 1.37 a ± 0.02 | 0.14 a ± 0.06 |
| V9 | 7.77 c,d ± 0.03 | 65.97 a ± 4.38 | 1.15 b ± 0.02 | 0.05 a,b ± 0.00 |
| V10 | 7.87 b,c,d ± 0.03 | 33.67 c ± 2.92 | 1.35 a ± 0.02 | 0.02 b ± 0.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burducea, M.; Lobiuc, A.; Dirvariu, L.; Oprea, E.; Olaru, S.M.; Teliban, G.-C.; Stoleru, V.; Poghirc, V.A.; Cara, I.G.; Filip, M.; et al. Assessment of the Fertilization Capacity of the Aquaculture Sediment for Wheat Grass as Sustainable Alternative Use. Plants 2022, 11, 634. https://doi.org/10.3390/plants11050634
Burducea M, Lobiuc A, Dirvariu L, Oprea E, Olaru SM, Teliban G-C, Stoleru V, Poghirc VA, Cara IG, Filip M, et al. Assessment of the Fertilization Capacity of the Aquaculture Sediment for Wheat Grass as Sustainable Alternative Use. Plants. 2022; 11(5):634. https://doi.org/10.3390/plants11050634
Chicago/Turabian StyleBurducea, Marian, Andrei Lobiuc, Lenuta Dirvariu, Eugen Oprea, Stefan Mihaita Olaru, Gabriel-Ciprian Teliban, Vasile Stoleru, Vlad Andrei Poghirc, Irina Gabriela Cara, Manuela Filip, and et al. 2022. "Assessment of the Fertilization Capacity of the Aquaculture Sediment for Wheat Grass as Sustainable Alternative Use" Plants 11, no. 5: 634. https://doi.org/10.3390/plants11050634
APA StyleBurducea, M., Lobiuc, A., Dirvariu, L., Oprea, E., Olaru, S. M., Teliban, G.-C., Stoleru, V., Poghirc, V. A., Cara, I. G., Filip, M., Rusu, M., Zheljazkov, V. D., & Barbacariu, C.-A. (2022). Assessment of the Fertilization Capacity of the Aquaculture Sediment for Wheat Grass as Sustainable Alternative Use. Plants, 11(5), 634. https://doi.org/10.3390/plants11050634









