A Temporary Immersion System Improves Regeneration of In Vitro Irradiated Recalcitrant Indica Rice (Oryza sativa L.) Embryogenic Calli
Abstract
1. Introduction
2. Results
2.1. Molecular Markers Used for the Identification of the Rice Cultivars
2.2. Embryogenic Calli Induction
2.3. Regeneration on Semisolid Medium
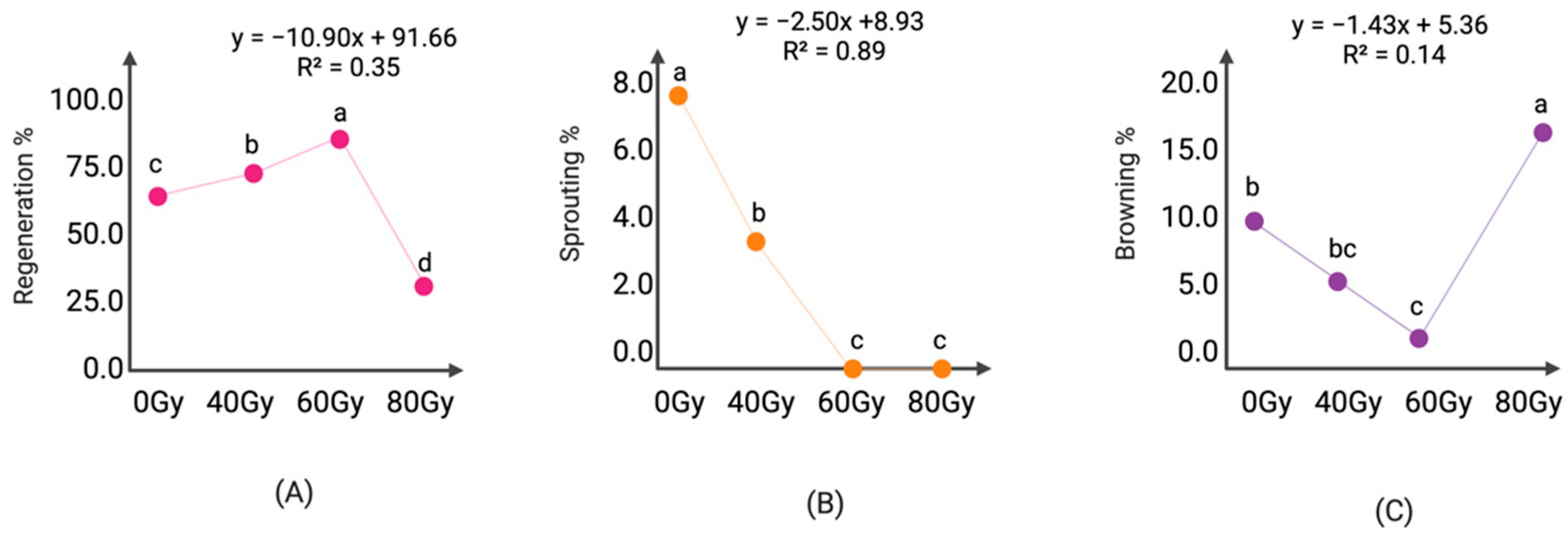
2.4. Gamma Radiation Mutagenesis
2.5. Regeneration in Recipient for Automated Temporary Immersion (RITA® Saint-Mathieu-de-Tréviers, France)
3. Discussion
4. Materials and Methods
4.1. Molecular Markers
4.2. Embryogenic Calli Induction
4.3. Regeneration on Semisolid Medium
4.4. Gamma Irradiation
4.5. Regeneration in Recipient for Automated Temporary Immersion (RITA®, Saint-Mathieu-de-Tréviers, France)
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ali, J.; Nicolas, K.L.C.; Akther, S.; Torabi, A.; Ebadi, A.A.; Marfori-Nazarea, C.M.; Mahender, A. Improved Anther Culture Media for Enhanced Callus Formation and Plant Regeneration in Rice (Oryza sativa L.). Plants 2021, 10, 839. [Google Scholar] [CrossRef]
- Wing, R.A.; Purugganan, M.D.; Zhang, Q. The Rice Genome Revolution: From an Ancient Grain to Green Super Rice. Nat. Rev. Genet. 2018, 19, 505–517. [Google Scholar] [CrossRef]
- Chen, E.; Huang, X.; Tian, Z.; Wing, R.A.; Han, B. The Genomics of Oryza Species Provides Insights into Rice Domestication and Heterosis. Annu. Rev. Plant Biol. 2019, 70, 639–665. [Google Scholar] [CrossRef]
- Kim, K.; Lee, S.C.; Lee, J.; Yu, Y.; Yang, K.; Choi, B.S.; Koh, H.J.; Waminal, N.E.; Choi, H.I.; Kim, N.H.; et al. Complete Chloroplast and Ribosomal Sequences for 30 Accessions Elucidate Evolution of Oryza AA Genome Species. Sci. Rep. 2015, 5, 15655. [Google Scholar] [CrossRef]
- Nadir, S.; Xiong, H.B.; Zhu, Q.; Zhang, X.L.; Xu, H.Y.; Li, J.; Dongchen, W.; Henry, D.; Guo, X.Q.; Khan, S.; et al. Weedy Rice in Sustainable Rice Production. A Review. Agron. Sustain. Dev. 2017, 37, 46. [Google Scholar] [CrossRef]
- Chauhan, B.S.; Jabran, K.; Mahajan, G. Rice Production Worldwide; Springer International Publishing: Cham, Switzerland, 2017; Volume 247. [Google Scholar] [CrossRef]
- Viana, V.E.; Pegoraro, C.; Busanello, C.; Costa de Oliveira, A. Mutagenesis in Rice: The Basis for Breeding a New Super Plant. Front. Plant Sci. 2019, 10, 1326. [Google Scholar] [CrossRef]
- Serrat, X.; Esteban, R.; Guibourt, N.; Moysset, L.; Nogués, S.; Lalanne, E. EMS Mutagenesis in Mature Seed-Derived Rice Calli as a New Method for Rapidly Obtaining TILLING Mutant Populations. Plant Methods 2014, 10, 5. [Google Scholar] [CrossRef]
- Soriano, J.D. Mutagenic Effects of Gamma Radiation on Rice. Bot. Gaz. 1961, 123, 57–63. [Google Scholar] [CrossRef]
- Romero, F.M.; Gatica-Arias, A. CRISPR/Cas9: Development and Application in Rice Breeding. Rice Sci. 2019, 26, 265–281. [Google Scholar] [CrossRef]
- Hernández-Soto, A.; Echeverría-Beirute, F.; Abdelnour-Esquivel, A.; Valdez-Melara, M.; Boch, J.; Gatica-Arias, A. Rice Breeding in the New Era: Comparison of Useful Agronomic Traits. Curr. Plant Biol. 2021, 27, 100211. [Google Scholar] [CrossRef]
- Oladosu, Y.; Rafii, M.Y.; Abdullah, N.; Hussin, G.; Ramli, A.; Rahim, H.A.; Miah, G.; Usman, M. Principle and Application of Plant Mutagenesis in Crop Improvement: A Review. Biotechnol. Biotechnol. Equip. 2016, 30, 1–16. [Google Scholar] [CrossRef]
- Zain, C.R.C.M.; Kadhimi, A.A.; Alhasnawi, A.N.; Isahak, A.; Mohamad, A.; Doni, F.; Yusoff, W.M.W. Enhancing of Drought-Tolerant Rice (Oryza sativa) Variety MRQ74 through Gamma Radiation and in vitro Pathway. Biotechnology 2016, 15, 125–134. [Google Scholar] [CrossRef][Green Version]
- Abdelnour-Esquivel, A.; Perez, J.; Rojas, M.; Vargas, W.; Gatica-Arias, A. Use of Gamma Radiation to Induce Mutations in Rice (Oryza sativa L.) and the Selection of Lines with Tolerance to Salinity and Drought. In Vitro Cell. Dev. Biol.—Plant 2020, 56, 88–97. [Google Scholar] [CrossRef]
- Fehér, A. Callus, Dedifferentiation, Totipotency, Somatic Embryogenesis: What These Terms Mean in the Era of Molecular Plant Biology? Front. Plant Sci. 2019, 10, 536. [Google Scholar] [CrossRef]
- Binte Mostafiz, S.; Wagiran, A. Efficient Callus Induction and Regeneration in Selected Indica Rice. Agronomy 2018, 8, 77. [Google Scholar] [CrossRef]
- Vega, R.; Vásquez, N.; Espinoza, A.M.; Gatica, A.M.; Valdez-Melara, M. Histology of Somatic Embryogenesis in Rice (Oryza sativa cv. 5272). Rev. Biol. Trop. 2009, 57 (Suppl. S1), 141–150. [Google Scholar] [CrossRef]
- Che Radziah, C.M.Z.; Naji Alhasnawi, A.; Kadhimi, A.A.; Isahak, A.; Mohamad, A.; Farshad Ashraf, M.; Doni, F.; Mohtar Wan Yusoff, W. Development of a Technique for Callus Induction and Plant Regeneration in Oryza sativa L. Var. MRQ74 and MR269. Adv. J. Food Sci. Technol. 2017, 13, 128–137. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 15, 474–497. [Google Scholar] [CrossRef]
- Valdez, M.; Muñoz, M.; Vega, J.R.; Espinoza, A.M. Plant Regeneration of Indica Rice (Oryza sativa) Cultivars from Mature Embryo-Derived Calli. Rev. Biol. Trop. 1997, 44–45, 13–21. [Google Scholar] [CrossRef]
- Pawar, B.; Kale, P.; Bahurupe, J.; Jadhav, A.; Kale, A.; Pawar, S. Proline and Glutamine Improve in vitro Callus Induction and Subsequent Shooting in Rice. Rice Sci. 2015, 22, 283–289. [Google Scholar] [CrossRef]
- Abiri, R.; Maziah, M.; Shaharuddin, N.A.; Yusof, Z.N.B.; Atabaki, N.; Hanafi, M.M.; Sahebi, M.; Azizi, P.; Kalhori, N.; Valdiani, A. Enhancing Somatic Embryogenesis of Malaysian Rice Cultivar MR219 Using Adjuvant Materials in a High-Efficiency Protocol. Int. J. Environ. Sci. Technol. 2017, 14, 1091–1108. [Google Scholar] [CrossRef]
- Hussain, Z.; Khan, M.H.; Bano, R.; Rashid, H.; Chaudhry, Z. Protocol Optimization for Efficient Callus Induction and Regeneration in Three Pakistani Rice Cultivars. Pak. J. Bot. 2010, 42, 879–887. [Google Scholar]
- Ming, N.J.; Mostafiz, S.B.; Johon, N.S.; Zulkifli, N.S.A.; Wagiran, A. Combination of Plant Growth Regulators, Maltose, and Partial Desiccation Treatment Enhance Somatic Embryogenesis in Selected Malaysian Rice Cultivar. Plants 2019, 8, 144. [Google Scholar] [CrossRef]
- Kashtwari, M.; Mansoor, S.; Wani, A.A.; Najar, M.A.; Deshmukh, R.K.; Baloch, F.S.; Abidi, I.; Zargar, S.M. Random Mutagenesis in Vegetatively Propagated Crops: Opportunities, Challenges and Genome Editing Prospects. Mol. Biol. Rep. 2021, 1, 1–21. [Google Scholar] [CrossRef]
- Lee, S.T.; Huang, W.L. Cytokinin, Auxin, and Abscisic Acid Affects Sucrose Metabolism Conduce to de Novo Shoot Organogenesis in Rice (Oryza sativa L.) Callus. Bot. Stud. 2013, 54, 5. [Google Scholar] [CrossRef]
- Zhang, K.; Su, J.; Xu, M.; Zhou, Z.; Zhu, X.; Ma, X.; Hou, J.; Tan, L.; Zhu, Z.; Cai, H.; et al. A Common Wild Rice-Derived BOC1 Allele Reduces Callus Browning in Indica Rice Transformation. Nat. Commun. 2020, 11, 443. [Google Scholar] [CrossRef]
- Debnath, S.; Chandel, R.K.; Devi, K.; Khan, Z. Mechanism and Molecular Response of Induced Genotoxicity and Oxidative Stress in Plants. In Induced Genotoxicity and Oxidative Stress in Plants; Khan, Z., Ansari, M.Y.K., Shahwar, D., Eds.; Springer: Singapore, 2021. [Google Scholar] [CrossRef]
- Xie, X.; He, Z.; Chen, N.; Tang, Z.; Wang, Q.; Cai, Y. The Roles of Environmental Factors in Regulation of Oxidative Stress in Plant. BioMed Res. Int. 2019, 2019, 21–27. [Google Scholar] [CrossRef]
- IBM Corp. Released 2020. IBM SPSS Statistics for Windows; Version 27.0; IBM Corp: Armonk, NY, USA, 2020. [Google Scholar]
- Sudhakar, D.; Duc, L.T.; Bong, B.B.; Tinjuangjun, P.; Maqbool, S.B.; Valdez, M.; Jefferson, R.; Christou, P. An Efficient Rice Transformation System Utilizing Mature Seed-Derived Explants and a Portable, Inexpensive Particle Bombardment Device. Transgenic Res. 1998, 7, 289–294. [Google Scholar] [CrossRef]
- Schneider, C.; Rasband, W.; Eliceiri, K. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]

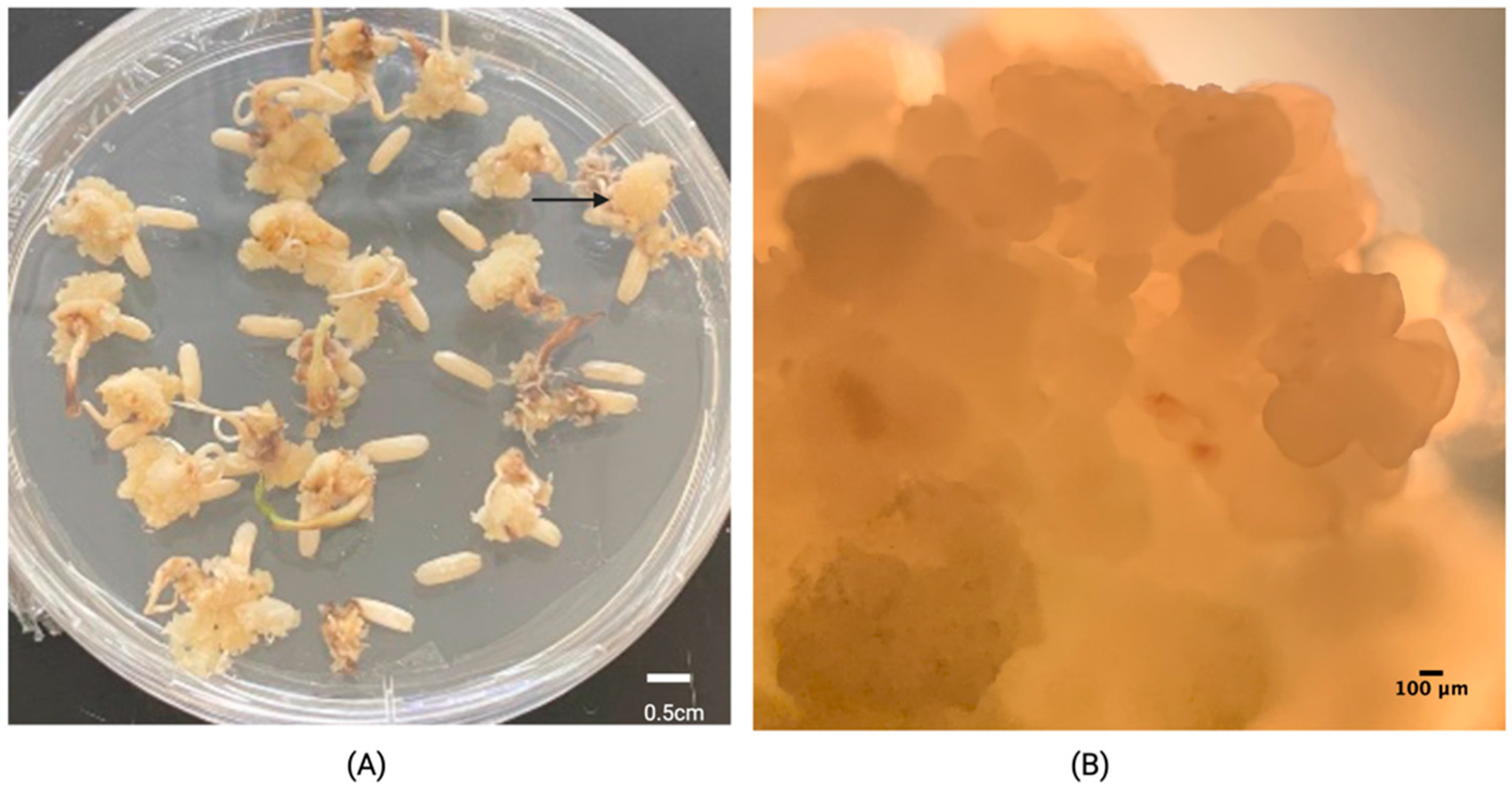



| Treatment 1 | id | Embryogenic Calli (%) | Browning Rate (%) |
|---|---|---|---|
| 2.5 mg L−1 2,4-D | i | 21.44 b | 2.00 b |
| 2.0 mg L−1 2,4-D | ii | 71.44 a | 3.55 a |
| 1.0 mg L−1 2,4-D + 1.0 mg L−1 BA | iii | 12.77 d | 0.66 c |
| 2.0 mg L−1 2,4-D + 1.0 mg L−1 BA | iv | 16.77 c | 0.21 c |
| 2.0 mg L−1 2,4-D + 0.25 mg L−1 TDZ | v | 23.00 b | 0.66 c |
| Induction Treatment 1 | Regeneration Treatment | Regeneration (%) | Sprouting (%) | Browning (%) |
|---|---|---|---|---|
| 2 mg 2,4-D | 0.5 mg NAA + 3 mg BA | 69.04 a | 7.14 ab | 9.52 d |
| 0.5 mg NAA + 0.5 mg TDZ | 38.09 c | 2.38 b | 61.90 a | |
| 0.5 mg NAA + 0.5 mg Kinetin | 47.61 b | 9.52 a | 23.80 c | |
| 0.5 mg NAA + 0.5 mg BA | 28.57 d | 2.38 b | 54.76 b | |
| 1 mg BA + 2 mg 2,4-D | 0.5 mg NAA + 3 mg BA | 28.29 b | 0 a | 58.43 b |
| 0.5 mg NAA + 0.25 mg TDZ | 58.82 a | 0 a | 100 a | |
| 0.5 mg NAA + 0.5 mg Kinetin | 9.22 c | 0 a | 56.81 b | |
| 0.5 mg NAA + 0.5 mg BA | 12.82 c | 0 a | 44.26 c | |
| 1 mg BA + 1 mg 2,4-D | 0.5 mg de NAA + 3 mg BA | 58.45 ab | 16.38 a | 18. 69 bc |
| 0.5 mg de NAA + 0.5 mg TDZ | 61.75 a | 10.71 ab | 27.93 a | |
| 0.5 mg de NAA + 0.5 mg Kinetin | 56.31 ab | 15.92 a | 20.01 ab | |
| 0.5 mg de NAA + 0.5 mg BA | 49.88 b | 4.16 b | 11.66 c | |
| 0.5 mg de NAA + 3 mg BA | 58.45 ab | 16.38 a | 18. 69 bc | |
| 2.5 mg 2,4-D | 0.5 mg NAA + 3 mg BA | 34.64 b | 0 b | 9.20 c |
| 0.5 mg NAA + 0.5 mg TDZ | 51.41 a | 9.61 a | 22.96 a | |
| 0.5 mg NAA + 0.5 mg Kinetin | 43.62 ab | 0 b | 15.73 b | |
| 0.5 mg NAA + 0.5 mg BA | 18.00 c | 0 b | 15.19 b | |
| 2 mg 2,4-D + 0.25 mg TDZ | 0.5 mg NAA + 3 mg BA | 77.27 a | 2.27 a | 96.59 a |
| 0.5 mg NAA + 0.5 mg TDZ | 50.25 b | 0 a | 86.36 b | |
| 0.5 mg NAA + 0.5 mg Kinetin | 73.86 a | 3.40 a | 82.95 b | |
| 0.5 mg NAA + 0.5 mg BA | 44.29 b | 0 a | 72.81 c |
| Lethal Gamma Rays Dose Model | Dose (Gy) | Lower Limit (Gy) | Upper Limit (Gy) |
|---|---|---|---|
| LD10 | 41.145 | 34.552 | 46.708 |
| LD20 | 64.799 | 60.083 | 69.20 |
| LD25 | 73.785 | 69.388 | 78.139 |
| LD30 | 81.855 | 77.507 | 86.403 |
| LD40 | 96.429 | 91.649 | 101.85 |
| LD50 | 110.050 | 104.435 | 116.720 |
| Immersion Time | Regeneration | Sprouting | Browning Rate |
|---|---|---|---|
| 60 s | 100.00 a | 25.00 a | 60.00 a |
| 120 s | 97.56 a | 31.71 a | 97.56 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Soto, A.; Pérez, J.; Fait-Zúñiga, R.; Rojas-Vásquez, R.; Gatica-Arias, A.; Vargas-Segura, W.; Abdelnour-Esquivel, A. A Temporary Immersion System Improves Regeneration of In Vitro Irradiated Recalcitrant Indica Rice (Oryza sativa L.) Embryogenic Calli. Plants 2022, 11, 375. https://doi.org/10.3390/plants11030375
Hernández-Soto A, Pérez J, Fait-Zúñiga R, Rojas-Vásquez R, Gatica-Arias A, Vargas-Segura W, Abdelnour-Esquivel A. A Temporary Immersion System Improves Regeneration of In Vitro Irradiated Recalcitrant Indica Rice (Oryza sativa L.) Embryogenic Calli. Plants. 2022; 11(3):375. https://doi.org/10.3390/plants11030375
Chicago/Turabian StyleHernández-Soto, Alejandro, Jason Pérez, Rebeca Fait-Zúñiga, Randall Rojas-Vásquez, Andrés Gatica-Arias, Walter Vargas-Segura, and Ana Abdelnour-Esquivel. 2022. "A Temporary Immersion System Improves Regeneration of In Vitro Irradiated Recalcitrant Indica Rice (Oryza sativa L.) Embryogenic Calli" Plants 11, no. 3: 375. https://doi.org/10.3390/plants11030375
APA StyleHernández-Soto, A., Pérez, J., Fait-Zúñiga, R., Rojas-Vásquez, R., Gatica-Arias, A., Vargas-Segura, W., & Abdelnour-Esquivel, A. (2022). A Temporary Immersion System Improves Regeneration of In Vitro Irradiated Recalcitrant Indica Rice (Oryza sativa L.) Embryogenic Calli. Plants, 11(3), 375. https://doi.org/10.3390/plants11030375






